Optimization of optical properties of Ba0.2Sr0.8TiO3 thin films for a glucose sensor implementation
Abstract
The development of non-invasive blood glucose measuring devices continues to be developed. The photosensor is one of the instruments for detection the blood glucose, and the high performance of sensor is depending on the optical properties. Consequently, the observation about optical properties of Ba0.2Sr0.8TiO3 (Barium Strontium Titanate) is important. In this paper, a solution of Ba0.2Sr0.8TiO3 (Barium Strontium Titanate) with chlorophyll extract from spinach and papaya leaves has been deposited on the p-type Si (100) by spin coating technique. This research aims to find the effects of presence of chlorophyll in the Ba0.2Sr0.8TiO3 films according to their optical properties and phonon mode. The film fabricated consisting of BST, BST with 2.5% spinach chlorophyll, BST with 5% spinach chlorophyll, BST with 2.5% papaya chlorophyll, and BST with 5% papaya chlorophyll. Therefore, the characterization was using UV-Vis spectrophotometer and micro Raman spectroscopy. The result showed that the addition of papaya chlorophyll have the optimal character than spinach chlorophyll. BST with papaya chlorophyll showed a significant decrease in the energy gap. Also, it has a phonon mode with a Raman shift that supports the result.
1.Introduction
Globally 4.6 million cases of death occur each year due to diabetes. Children and adolescents died of insulin deficiency without being diagnosed [22]. Diabetes mellitus is a disease caused by increasing the blood glucose levels due to the disruption of the metabolic system, where the pancreatic organs are unable to produce the insulin hormone according to the body’s needs [19]. If a wound occurs, then the blood is difficult to stop so the wound becomes more severe and rot. Measurement of blood glucose levels by taking a blood sample is very risky if the scar does not dry out immediately. For this reason, the development of non-invasive blood glucose measuring devices continues to be developed. Non-invasive measurements on this device utilize optical phenomena in the form of light absorption at specific wavelengths of blood glucose (visible light 534 nm and infrared 939 to 2326 nm). The amount of this absorption depends on the concentration of glucose in the bloodstream [3].
In some countries, biosensor as a measurement of blood glucose continues to be developed. The photo-sensor is one of the tools used for detection of blood glucose levels. So, we need the instrument for measuring the intensity of the light captured after passing through the tip of the finger. Because one of the organs of the human body that is easily penetrated by light is the fingertips (Fig. 1) [2].
One of the materials that can be used as a sensor is Barium Strontium Titanate (BST) [15]. Irzaman et al., (2016) explained that the BST ferroelectric material can be used for light sensor applications and can be developed into solar cells. BST is a semiconductor material that has been researched in recent years for the various application, especially many types of sensors [8,12,17,18,24,38].
The performance of barium strontium titanate (Ba1-xSrxTiO3) thin film is excellent in applications of tunable microwave devices, fast nonlinear optics, and high-density gigabit dynamic random access memory [34]. The physical properties of BST films are strongly influenced by composition, dopants, deposition, internal stresses, electronic structures, and others. Currently, the development of BST sensor is continuously carried out to increase sensitivity or accuracy in the measurement process [8].
In Indonesia, the application of chlorophyll as an energy source is being pursued because chlorophyll is found in all photosynthetic plant so chlorophyll is easy to find out [6,36]. All of chlorophyll can fluorescence, if it gets irradiated with a particular spectrum of light (excitation spectrum), then the light it passes on (emission spectrum) is light on a different spectrum. For example, chlorophyll-a dissolved in acetone 85% has a maximum excitation between wavelengths of 430–450 nm (blue-purple) and will give a maximum emission between wavelengths of 650–675 nm (dark red) [40]. In material science, it is usually applied as an energy storage and detector material. For the example [18] use chlorophyll as Thiourea doping for the active potential of the photovoltaic cell layer [1].
In this paper, we compare the chlorophyll extracts taken from several plants. The leaves investigations were spinach and papaya leaves. In previous research, chlorophyll extract is known to have the ability to absorb light. By using UV-Vis and Micro-Raman Spectroscopy, we analyzed the differences in BST thin film with spinach and papaya chlorophyll extract. BST films without doping were also presented as a control sample.
1.1.Chlorophyll
Pigments or dyes, generally found in meristem tissue cells which form a chloroplast. Chloroplast has very diverse shapes and sizes [39]. Chloroplast is composed of a stroma, covered by membranes, in which small granules are scattered containing green chlorophyll pigments. Pigments in chloroplast (chlorophyll) have an essential role in the photosynthesis, it is a catalyst and absorbs the kinetic energy [33]. From the research conducted by Meyer and Anderson (1952), chlorophyll is divided into 3 types, chlorophyll-a, -b and -c. In this study, chlorophyll-a, -b and -c have the same chemical properties which are insoluble in water but can dissolve in various types of organic solvents. Chlorophyll-a is soluble in ethyl alcohol, ethyl ether, acetone, chloroform, and carbon-bisulfide, chlorophyll-b and -c also soluble with them but it is not as easy as chlorophyll-a [30]. Besides, if chlorophyll-a and chlorophyll-b are hydrolyzed, alcohol (phytol) will be obtained. The phytol group forms a third of the chlorophyll molecule and has a strong affinity for oxygen. If it is ignored, pure chlorophyll will leave a residue composed only of magnesium-oxide. Although iron (Fe) and other minerals are essential for the formation of chlorophyll in living cells, magnesium (Mg) is the only metal element as a catalyst in visible light absorption [30].
Based on the Yentsch and Menzel (1963) study, all of the chlorophyll have the fluorescence properties, when it get irradiated with a certain spectrum of light (excitation spectrum), the light is transmitted (emission spectrum) in a different spectrum. For example, chlorophyll-a dissolved in 85% acetone has a maximum excitation between wavelengths of 430–450 nm (blue-purple) and will provide maximum emission between wavelengths of 650–675 nm (dark red). The absorption spectrum of chlorophyll-a, -b and -c in ether solution is shown in Fig. 2, the maximum absorption of primers is in the blue-purple spectrum (wavelength 430–477 nm) and secondary maximum in the red spectrum (628–662 nm) [30].
1.2.UV-Vis and Raman spectroscopy
UV-Vis Spectrophotometry used to analyze the energy gap of thin film. Spectrophotometric measurements involve considerable electronic energy in the molecules analyzed, so the UV-Vis spectrophotometer is more widely used for quantitative analysis than qualitative [20]. This is appropriate with the measurement range desired by this study. Energy gap is obtained by drawing a line of the relationship between
Another instrument is Micro-Raman Spectroscopy, which used to analyze samples (solid, liquid, even biological samples). Micro-Raman Spectroscopy has an accurate level of measurement, non-destructive to the sample, so the analysis does not damage the physical sample [23,32]. In a previous study conducted by S. Y. Wang, 2006, Raman spectroscopy was used for measuring thin films doped using Cerium. The results of his research stated that Raman Spectroscopy can analyze the crystal structure, surface morphology, and stability of the Ce-doped BST film [37]. Micro-Raman Spectroscopy type in this research using DU420-BROD semicolon (indoortechnology co. Ltd., northern Ireland) because this instrument is able to measure the sample at any condition, solid as solution alike [32].
2.Materials and experimental procedures
2.1.Materials
The material needed is Barium acetate [Ba(CH3COO)2], Strontium acetate [Sr(CH3COO)2], Titanium Isopropoxide [Ti(C12O4H28)], Si (100) p-type, ethylene glycol, acetic acid, and aquadest. In this study, the ratio of solvent was 3:1 between acetic acid and ethylene glycol.
2.2.Experimental procedures
The first procedures is chlorophyll extraction, The extraction process is starting on cleaning the leaf with the running water, throwing away the leaf bones, then blending 2 part of leaf with 1 part of aquadest, after that filtering the solution and blanching at 60°C for 10 minutes, collecting the clumps, moving into the glass ware petri, and drying at 50°C.
The second is fabrication of BST and BST-Chlorophyll thin film. It was following the research conducted by Irzaman [13], as seen in Fig. 3.
Fig. 3.
Fabrication of BST and BST-Chlorophyll thin film.

3.Results and discussion
The optical properties was characterized by UV-Vis spectroscopy. The reflectance mode can be used to estimate the energy gap. Energy gap is energy needed by electron to excite from the valence band to the conduction band [9,16]. The lowest energy is mean the easiest of electron to move [11,21]. According to the research conducted by Ruzimuradov 2007, BST has 3.2 eV of energy gap [31]. In this study found that BST have 2.82 eV, it was close and less than the reference. The analysis result can be seen in Fig. 4 and Fig. 5, also summarized in Table 1.
Fig. 4.
Reflectance of Ba0.2Sr0.8TiO3film.
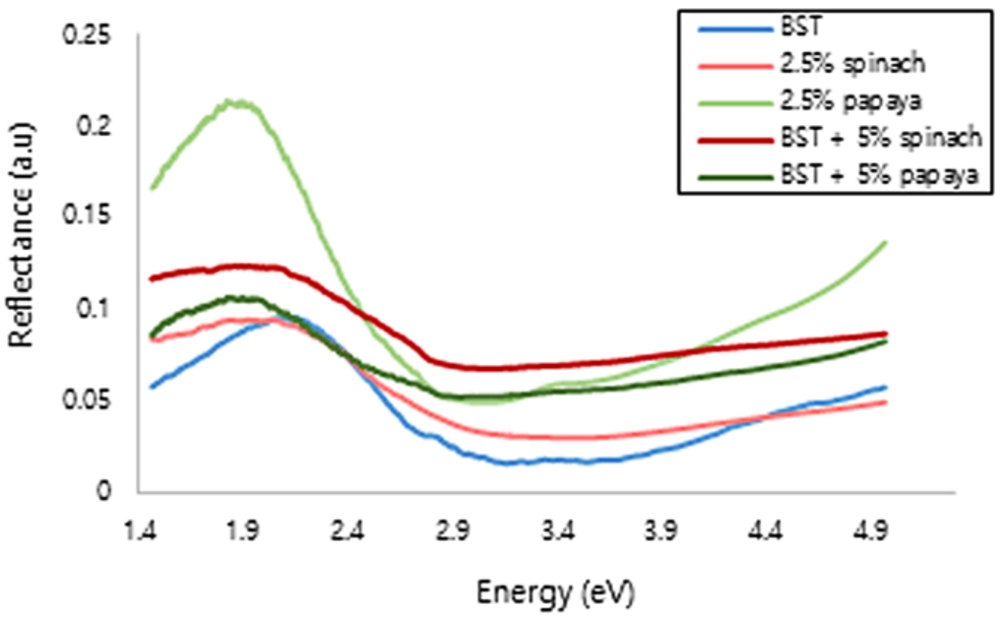
Fig. 5.
The energy gap of Ba0.2Sr0.8TiO3 film.
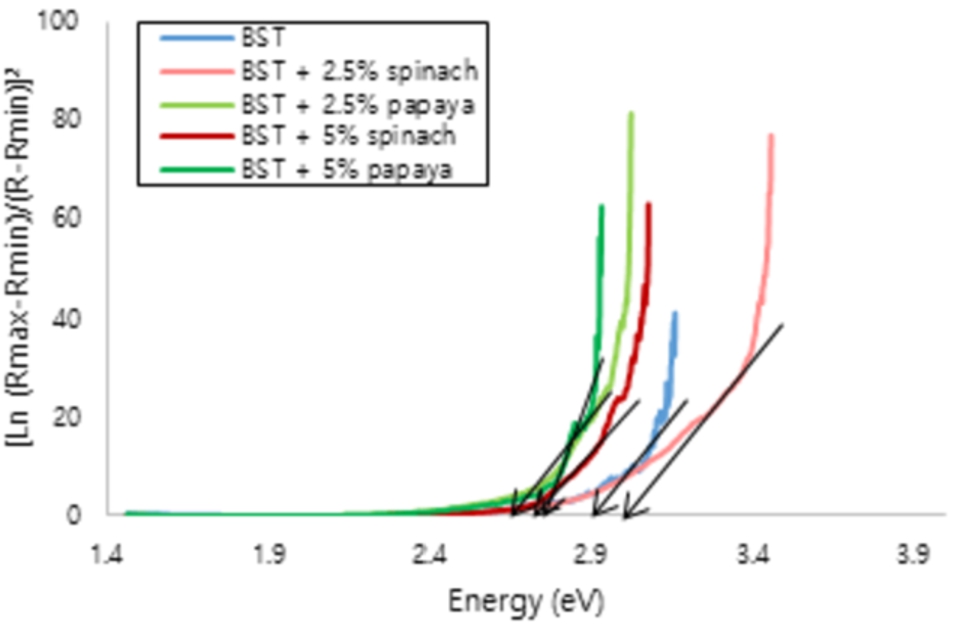
Table 1
The energy gap of BST and BST-Chlorophyll film
| Sample | Energy Gap |
| BST | 2.82 |
| BST with 2.5% spinach chlorophyll | 2.98 |
| BST with 2.5% papaya chlorophyll | 2.69 |
| BST with 5% spinach chlorophyll | 2.69 |
| BST with 5% papaya chlorophyll | 2.66 |
The reflectance as an energy is shown in Fig. 4. Based on the UV-Vis characterization, BST and BST-chlorophyll film in this research have the energy gap smaller than the reference. In Fig. 5, the energy gap can be seen from the change in potential energy or the slope of the line. If the slope of the line gets bigger, the energy gap will be even greater [4]. This research is a continuation of our previous research, BST with chlorophyll papaya, spinach, and mustard as much as 5% dissolved in acetic acid and ethylene glycol in a ratio of 3:1. The smallest energy gap found at BST with spinach 5%, it was 2.79 eV [26]. Also other studies on BST with 2.5% chlorophyll mustard, cassava, papaya and spinach dissolved with 3:1 have energy gaps more than 3 eV [25]. So, the composition of the solvent affects the energy gap. In this study, we use the 19:1 ratio of solvent and we got the smaller energy gaps. Besides, eventhough using the some ratio, BST with papaya in this study has smaller energy gap than BST with mustard chlorophyll [28] and BST with cassava chlorophyll in previous [27].
The Micro-Raman analysis in Fig. 6 shows that more than one phonon mode, and an increase of intensity due to chlorophyll presence starting at the wavenumber 400 cm−1 to 1000 cm−1. Along with differences in chlorophyll extract spinach and papaya leaves, absorption of Transverse-Optical A(TO)/ Emission of Longitudinal-Optical E(LO) Phonon [5,29] at a peak of 523 cm−1 and E(LO) at a peak of 676 cm−1, it has a different shift in peak toward greater intensity. Phonon optical frequencies and symmetry modes on tetragonal BST are available in Table 2.
Fig. 6.
Micro-Raman analysis of Ba0.2Sr0.8TiO3 film.
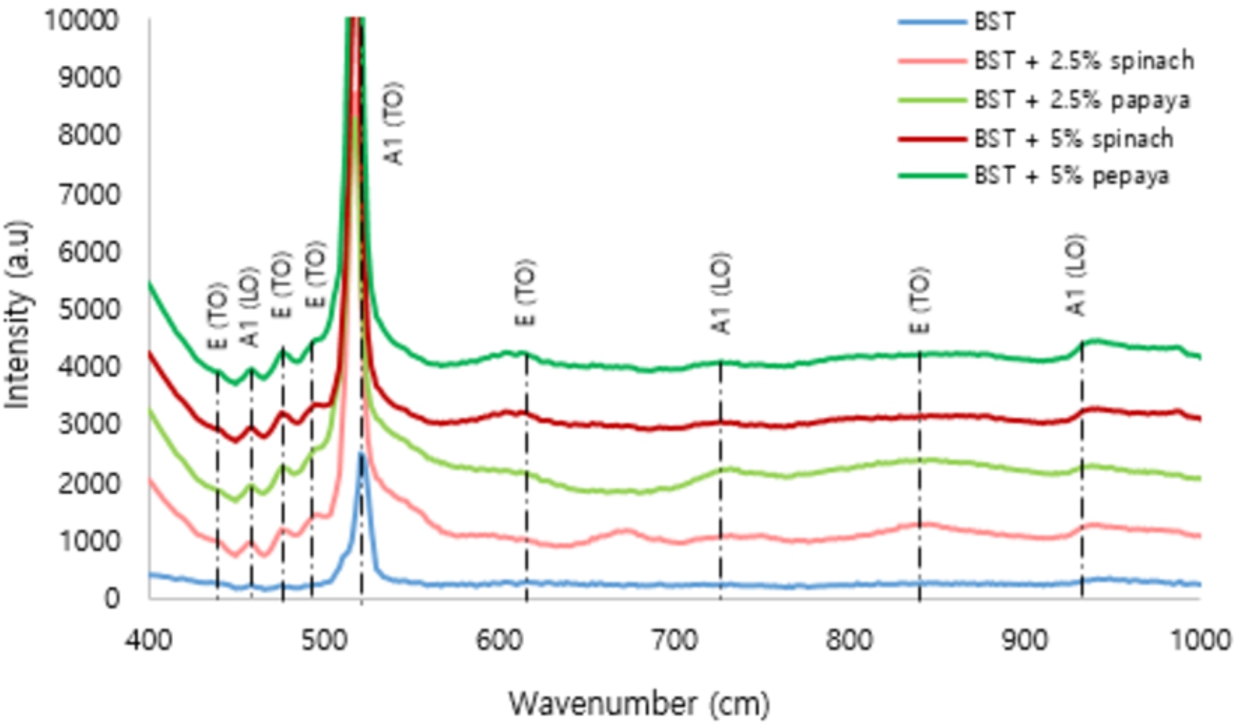
All of BST films with chlorophyll are perovskite structures from the tetragonal phase which is characterized by a peak intensity of 841 cm−1 [32]. The safe intensity of BST with chlorophyll 5% papaya higher than another concentration of chlorophyll. Therefore, polycrystalline and tetragonal structures are very large. However, the low peak was shown in chlorophyll at a frequency of 672 cm−1 is likely due to the scattering disorder activity of the TO and LO phonon branches which have a high density in the Brillouin zone boundary state [35]. The low peaks at 947 cm−1 in all BST film samples are likely due to the residual pressure and surface pressure between the film and the silicon substrate. However, the peak at 521 cm−1 to 523 cm−1 in all of samples is the optical phonon mode that undergoes degeneration. At frequency of 521 cm−1 to 523 cm−1 the peak increase or difference in peak intensity is seen. Based on research conducted by [30], one of the factors that causes the difference in intensity at frequency 521 cm−1 is caused by the release of the Mg atom compound, so that Chlorophyll can decompose both while still in plant cells (the operation of the chlorophyllase enzyme) or when the chlorophyll extraction process takes place [36].
Table 2
Comparison of the shift mode based on the optical phonon frequency and symmetry mode on tetragonal BST
| Symmetry [37] | Wavenumber (cm−1) [37] | Wavenumber (cm−1) (this study) | ||||
| BST | BST + 2.5% spinach | BST + 2.5% papaya | BST + 5% spinach | BST + 5% papaya | ||
| E(TO) | 416 | 460 | 459 | 459 | 459 | 459 |
| A1(LO) | – | 480 | 476 | 476 | 476 | 476 |
| E(TO) | – | 495 | 495 | 499 | 498 | 498 |
| E(TO) | – | 508 | 510 | 510 | 510 | 510 |
| A1(TO) | 521 | 523 | 518 | 518 | 518 | 518 |
| E(TO) | 676 | 672 | 673 | 673 | 674 | 672 |
| A1(LO) | 742 | 742 | 743 | 743 | 744 | 744 |
| E(TO) | 841 | 841 | 841 | 841 | 841 | 841 |
| A1(LO) | 947 | – | 947 | 948 | 947 | 946 |
Thin films which have good optical properties can be further investigated and applied as sensors. In the case of measuring blood glucose, thin film is used in devices as a capacitive biosensor. Capacitive sensor is an electronic sensor that works based on the concept of capacitive. This sensor works based on the changes in electrical energy charge that can be stored by the sensor due to changes in plate distance, changes in cross-sectional area and changes in the capacitive sensor dielectric volume. The concept of capacitors is the process of storing and releasing electrical energy in the form of electrical charges on a capacitor that is influenced by surface area, distance, and dielectric material. Capacitive sensor consists of two main components as an electrode, sensing electrode and reference electrode.
Capacitive biosensors measure the dielectric of the layer on the interface between the electrolyte and electrode. The glucose oxidase enzyme is immobilized onto electrodes to the oxidize glucose. In practice, the biochemical reaction of glucose with enzyme generates the free electron carriers. Therefore, the potential difference on the increased electrodes causing the voltage output that can be measured from the biosensor [7]. It can be concluded that this research is the beginning of a series of biosensor studies. Based on the analysis of optical properties and phonon modes, BST thin films have the potential to be applied as sensors, especially glucose sensors.
4.Conclusion
Fabrication of Ba0.2Sr0.8TiO3 film was deposited by spin coating technique. The analysis of UV-Vis spectroscopy and Micro-Raman Spectroscopy showed that the film has good characteristics. Addition of chlorophyll into BST solution raises the good interaction between chlorophyll atoms and BST. This result obtained that BST with 5% chlorophyll papaya have the smallest energy gap, and overall the addition of chlorophyll generates the small energy than the BST film without addition of chlorophyll. It can be conclude that BST thin films have the potential to be applied as sensors, especially glucose sensors.
Acknowledgements
We would love to thanks to Sato Laboratory, Kwansei Gakuin University, Japan for providing laboratory facilities and assisting in analyzing BST film using Micro-Raman Spectroscopy. Also, this research was funded by Hibah RISPRO LPDP, Ministry of Finance Indonesia, with the contract number PRJ-78/LPDP/ 2019, December 2, 2019.
Conflict of interest
The authors have no conflict of interest to report.
References
[1] | H.K. Adli, W.M. Khairul and H. Salleh, Linear Nonyloxy-Substituted Thiourea-Chlorophyll Thin Film as Potential Single Layer Photovoltaic Cell, Vol. 1, pp. 1–5, 2011. |
[2] | J.T. Andrews, Non-Invasive Glucose Monitoring Techniques: A review and current trends, pp. 1–47. |
[3] | I.M. Astawa, Sistem Instrumentasi Alat Ukur Kadar Gula Darah Non Invasive Berbasiskan Arduino, no. November, pp. 1–3, 2016. |
[4] | S. Ebraheem and A. El-Saied, Band Gap Determination from Diffuse Reflectance Measurements of Irradiated Lead Borate Glass System Doped with TiO 2 by Using Diffuse Reflectance Technique, vol. 2013, no. May, pp. 324–329, 2013. |
[5] | S. Garc, Sustainable luminescent solar concentrators based on organic – inorganic hybrids modi fi ed with chlorophyll †, 2018. |
[6] | M.L. Hariyanto et al., Pengaruh Komposisi Fe Terhadap Konduktivitas Ion Oksigen Pada Oksida Perovskit La 0.7 Sr 0.3 Co, vol. 3, no. 1990, pp. 978–979, 2011. |
[7] | A. Hartono, E. Sanjaya and R. Ramli, Glucose sensing using capacitive biosensor based on polyvinylidene fluoride thin film, Biosensor 8: (1) ((2018) ). doi:10.3390/bios8010012. |
[8] | Irzaman et al., Application of lithium tantalate (LiTaO 3) films as light sensor to monitor the light status in the Arduino Uno based energy-saving automatic light prototype and passive infrared sensor, Vol. 193, 2018. |
[9] | Irzaman, M. Dahrul, H. Syafutra, A. Arif, M.N. Indro and Siswadi, Synthesis and characterizations photodiode thin film barium strontium titanate (BST) doped niobium and iron as light sensor, in: The 4th Asian Physics Symposium, American Institute of Physics (AIP) Conference, Vol. 1325: , (2010) , pp. 43–46. |
[10] | Irzaman, M. Dahrul, B. Yuliarto, K.A. Hammam and H. Alatas, Ferroelectrics Letters Section 45: (4–6) ((2018) ), 49–57. doi:10.1080/07315171.2018.1537333. |
[11] | Irzaman, Y. Pebriyanto, E.R. Apipah, I. Noor and A. Alkadri, Integr. Ferroelectr. 167: (1) ((2015) ), 137–145. doi:10.1080/10584587.2015.1107358. |
[12] | Irzaman, R. Siskandar, Aminullah and H. Alatas, Characterization of BaSrTiO films as light and temperature sensors and its implementation on automatic drying system model, Integrated Ferroelectrics 168: (1) ((2016) ), 130–150. doi:10.1080/10584587.2016.1159537. |
[13] | Irzaman, R. Siskandar, N. Nabilah, Aminullah, B. Yuliarto, K.A. Hamam and H. Alatas, Ferroelectrics Journal 524: ((2018) ), 44–55. doi:10.1080/00150193.2018.1432842. |
[14] | Irzaman, H. Syafutra, A. Arif, H. Alatas, M.N. Hilaluddin, A. Kurniawan, J. Iskandar, M. Dahrul, A. Ismangil, D. Yosman et al., AIP Conference Proceedings 1586: ((2014) ), 1–13. |
[15] | Irzaman, H. Syafutra and E. Rancasa, Ferroelectrics The Effect of Ba/Sr Ratio on Electrical and Optical Properties of Ba x Sr ( 1-x ) TiO 3, no. September, pp. 37–41, 2013. |
[16] | Irzaman, I.R. Putra, Aminullah, H. Syafutra and H. Alatas, Development of Ferroelectric Solar Cells of Barium StrontuimTitanate (BaxSr1-xTiO3) for Substituting Conventional Battery in LAPAN-IPB Satellite (LISAT), Procedia, Elsevier, (2016) . |
[17] | J. Kang, J. Park and H. Lee, Pt-doped SnO2 thin film based micro gas sensors with high selectivity to toluene and HCHO, Sensors & Actuators: B. Chemical (2017). |
[18] | W.M. Khairul, M.F. Yusof, R. Rahamathullah and A.I. Daud, Single Molecule Thin Film Featuring Disubstituted Thiourea (TU) Doped with Chlorophyll as Potential Active Layer in Photovoltaic Cell, Vol. 8, pp. 8175–8190, 2013. |
[19] | S. Li et al., Prevalence of Diabetes Mellitus and Impaired Fasting Glucose, Associated with Risk Factors in Rural Kazakh Adults in Xinjiang, China, pp. 554–565, 2015. |
[20] | M. Marschewski, H. Taş, C. Otto, W. Maus-Friedrichs, A. Schmidt and O. Höfft, Adsorption study of thin films of terephthalic acid and substituted benzoic acids on HOPG studied by metastable induced electron spectroscopy and ultraviolet photoelectron spectroscopy, Journal of Electron Spectroscopy and Related Phenomena 229: ((2018) ), 26–32. doi:10.1016/j.elspec.2018.09.002. |
[21] | P. Marwoto, U.N. Semarang and D. Aryanto, Properties of ZnO: Ga Thin Films Deposited by dc Magnetron Sputtering: Influence of Ga-Doped Concentrations on Structural and Optical Properties of ZnO: Ga Thin Films Deposited by dc Magnetron Sputtering: Influence of Ga-Doped Concentration, no. December, 2016. |
[22] | I. Migdalis, D. Leslie, N. Papanas, P. Valensi and H. Vlassara, Diabetes Mellitus 2014 Diabetes Mellitus, no. May 2015, 2014. |
[23] | L.I. Murthy and W.E. Halperin, Medical screening and biological monitoring a guide to the literature for physicians, Journal of Occupational and Environmental Medicine 37: (2) ((1995) ), 170–184. doi:10.1097/00043764-199502000-00016. |
[24] | F. Niklaus and C. Vieider, MEMS-Based Uncooled Infrared Bolometer Arrays – A Review MEMS-Based Uncooled Infrared Bolometer Arrays – A Review, no. May 2014, 2008. |
[25] | E.K. Palupi, H. Alatas and Irzaman, Analysis of energy gap and the refractive index of barium strontium titanate (Ba0.2Sr0.8TiO3) films doped of chlorophyll from green leafy vegetables, IOP Conf. Series: Earth and Environmental Science 209: ((2018) ), 1–7. |
[26] | E.K. Palupi, R. Umam, B.B. Andriana, H. Sato, H. Alatas and Irzaman, Fabrication and analysis phonon mode of barium strontium titanate-chlorophyll thin film (chlorophyll extract: Green spinach, cassava, green choy sum), AIP Conference Proceedings 2202: ((2019) ), 020018. |
[27] | E.K. Palupi, R. Umam, B.B. Andriana, H. Sato, B. Yuliarto, H. Alatas and Irzaman, Micro-Raman analysis of Ba0.2Sr0.8TiO3 (barium strontium titanate) doped of chlorophyll of cassava leaf, Ferroelectrics Journal 540: ((2019) ), 227–237. doi:10.1080/00150193.2019.1611116. |
[28] | E.K. Palupi, Irzaman, H. Alatas, B. Yuliarto, R. Umam, B.B. Andriana and H. Sato, Analysis of spectroscopy: Mustard greens leaf of chlorophyll as a Ba0.2Sr0.8TiO3 (barium strontium titanate) film dopant, Integrated Ferroelectrics Journal 201: ((2019) ), 75–85. doi:10.1080/10584587.2019.1668692. |
[29] | D. Patidar, K.S. Rathore, N.S. Saxena, K. Sharma and T.P. Sharma, Energy band gap studies of CdS nanomaterials, Journal of Nano Research 3: ((2016) ), 97–102. |
[30] | S.H. Riyono, Beberapa Sifat Umum Dari Klorofil Fitoplankton, Vol. XXXII, no. 1, pp. 23–31, 2007. |
[31] | O. Ruzimuradov, M. Hojamberdiev, C. Fasel and R. Riedel, Fabrication of lanthanum and nitrogen-co-blanded SrTiO3-TiO2 heterostructured macroporous monolithic materials for photocatalytic degradation of organic dyes under visible lights, Journal of Alloys and Compounds ((2016) ). doi:10.1016/jallcom.2016. |
[32] | H. Sato, B.B. Andriana, H. Shinzawa and Y. Matsuura, probes; in vivo animal and ex vivo clinical application studies An optical biopsy system with miniaturized Raman and spectral imaging probes; in vivo animal and ex vivo clinical application studies, no. January 2009, 2014. |
[33] | J. Strickland, Measuring the production of marine phytoplankton, Bull. Fish. Res. Bd 122: (122) ((1960) ), 1–172. |
[34] | F.L. Traversa, F. Bonani, Y.V. Pershin and M. Di Ventra, Dynamic Computing Random Access Memory, 2014. |
[35] | U.D. Venkateswaran, Physical Review B 58: ((1998) ), 14256–14260. doi:10.1103/PhysRevB.58.14256. |
[36] | T.C. Vogelmann and J.R. Evans, Profiles of light absorption and chlorophyll within spinach, pp. 1313–1323, 2002. |
[37] | S.Y. Wang et al., Raman spectroscopy studies of Ce-doping effects on Ba 0. 5 Sr 0. 5 TiO 3 thin films, pp. 1–6, 2006. |
[38] | T. Wang, L. Jin, C. Li, Q. Hu and X. Wei, Relaxor Ferroelectric BaTiO3–Bi(Mg2/3Nb1/3)O3 Ceramics for Energy Storage Application, vol. 566, no. 34925, pp. 559–566, 2015. |
[39] | Y. Xiong, R. Li and Y. Min Yue, Quantitative estimation of photosynthetic pigments using new spectral indices, Journal of Forestry Research 24: (3) ((2013) ), 477–483. doi:10.1007/s11676-013-0379-0. |
[40] | Y.I. Yuzyuk, Phase Transitions in ( Ba0. 7Sr0. 3 ) TiO3 /( 001 ) MgO Thin Film Studied by Raman Scattering by Raman scattering, no. May 2014, 2003. |

![Blood glucose measuring tools [3].](https://ip.ios.semcs.net:443/media/bsi/2020/9-1-2/bsi-9-1-2-bsi200197/bsi-9-bsi200197-g001.jpg)
![The absorption spectrum of chlorophyll-a, -b, and -c uses ether solvents [30].](https://ip.ios.semcs.net:443/media/bsi/2020/9-1-2/bsi-9-1-2-bsi200197/bsi-9-bsi200197-g002.jpg)


