Treating neuroendocrine neoplasms in the setting of HIV infection
Abstract
BACKGROUND:
There is a paucity of knowledge regarding neuroendocrine neoplasms (NEN) in patients with HIV infection.
OBJECTIVE:
To explore the incidence, characteristics and treatment outcomes of NEN in HIV-positive individuals.
METHODS:
This is a single-center, descriptive cohort study. Patients with HIV and biopsy-confirmed NEN were identified from our data registry. Data were collected retrospectively from medical records. Progression-free and overall survival (OS) were estimated using the Kaplan-Meier method.
RESULTS:
Nineteen HIV-positive patients with neuroendocrine tumors (NET) (n = 14), neuroendocrine carcinomas (NEC) (n = 2) or Merkel cell carcinoma (MCC) (n = 3) were included (median age at NEN diagnosis, 53 years). In 15 (79%) patients, HIV diagnosis preceded NEN diagnosis by a median of 11 years and 14 were receiving antiretroviral therapy (ART). Of those with data available, 75% had a viral load < 50 copies/ml and mean CD4 771 cells/mm3. The median OS in the NEC/MCC cohort was 8 months (range 5–29). The median OS in the NET cohort was not reached but based on the 25th centile, 75% of patients are expected to survive for at least 57 months. Treatment outcomes will be described.
CONCLUSIONS:
Within the limits of a small descriptive cohort study, we found no evidence in the era of ART that patients with HIV and NEN are diagnosed at a younger age, nor have a poorer prognosis compared with the wider NEN population. Thus, they should receive maximal NEN therapies to support best outcomes.
1Introduction
Neuroendocrine neoplasms (NEN) are rare tumors with an incidence of 2.5–5 cases per 100,000 persons per year [1, 2]. However, the incidence of NEN is increasing, and given their long survival durations, the prevalence of NEN has now eclipsed gastric, pancreatic, esophageal and hepatobiliary adenocarcinomas [1, 3]. Given the wide distribution of neuroendocrine cells, tumors may originate in almost any organ but are commonest in the gastroenteropancreatic (GEP) system (57%) and lung (27%) [3].
The histological classification of NEN is dependent on their site of origin. For the commonest subtype, GEP NEN, the WHO 2010 classification [4] divides them into 3 grades as determined by a proliferation-based grading index of Ki67 and/or mitotic count/10 high-power fields. Grade 1 (G1) tumors are well-differentiated, with < 2 mitosis per 10 high power fields and Ki67 < 3%. Grade 2 (G2) are well-differentiated with 2–20 mitosis per 10 high power fields and/or a Ki67 of 3–20%. Grade 3 (G3) contains both well-differentiated neuroendocrine tumors (NETs) and poorly differentiated neuroendocrine carcinomas (NECs) with > 20 mitoses per 10 high power fields and/or Ki67 > 20%. The 2019 WHO classification [5] has recently been adopted for G3 gastroenteropancreatic NEN which differentiates between G3 well-differentiated NET and G3 poorly differentiated NEC based on distinct differences in prognosis.
Given the biological heterogeneity of NEN, management is complex and a multidisciplinary approach in a center with NEN expertise is critical. For well-differentiated NET, the mainstay of treatment where possible is surgery with curative intent, including metastasectomy for oligometastatic disease. Hepatic-directed therapies, such as transarterial embolization, are also frequently used depending on site, size and number of metastases. Systemic treatment options most commonly include somatostatin analogues in the first-line setting for both anti-tumor effect and control of hormonal syndromes [6, 7]. Other systemic therapies for diffuse disease include molecular targeted therapy such as sunitinib [8] and everolimus [9, 10], peptide receptor radionuclide therapy (PRRT) [11] as well as cytotoxic chemotherapy for more aggressive tumors [12]. Treatment choice is influenced by a number of factors including primary site of origin, the functional status of the tumor, distribution and volume of disease, tumor morphology and histological grade, and patient factors such as performance status and renal function.
1.1HIV infection and cancer
A strong association has been described between HIV infection and a number of virally-mediated malignancies designated AIDS-defining cancers, namely, Kaposi’s sarcoma, non-Hodgkin’s lymphoma and cervical cancer. The widespread adoption of antiretroviral therapy (ART) has dramatically altered the incidence and prognosis of AIDS-defining cancers although, due to increased life expectancy, a significant increase in non-AIDS defining cancers, such as lung cancer and head and neck cancer, has been reported which now cause greater morbidity and mortality [13–15].
1.2HIV infection and neuroendocrine neoplasms
There is a well-defined association between HIV infection and Merkel cell carcinoma (MCC) [16]. MCC is a rare skin cancer which arises in cutaneous neural crest cells and occurs more frequently after organ transplantation or B-cell malignancy [17]. It is frequently associated with Merkel cell polyomavirus infection, a common viral infection without long-term sequelae in the general population, but associated with increased pathogenic potential in the immunocompromised [18, 19]. In a cohort of 309,365 individuals with acquired immunodeficiency syndrome (AIDS) linked to cancer registries, six cases were identified, corresponding to a relative risk of 13.4 (95% CI 4.9–29.1) compared to the general population [20].
With respect to immunodeficiency and NETs, an association between X-linked hyper-IgM syndrome (XHIGM) and NET has been described but is not seen in other types of primary immune deficiency, despite an association observed with other gastrointestinal neoplasms. Nicolaides et al. [21] described 10 patients with XHIGM and NETs, suggesting an occurrence rate of 0.02–0.06% i.e. a very rare phenomenon compared to other genetic conditions such as multiple endocrine neoplasia type 1, where the risk of developing GEP NEN is 30–70% [22].
There is little in the literature describing NEN in patients with HIV infection. Nicolaides et al. [21] described 12 HIV infected individuals who developed NET (n = 5), NEC (n = 6) and MCC (n = 1). Compared to the general NEN population, they were younger at the time of NEN diagnosis, between the 2nd and 4th decade of life; however, only half of patients were receiving ART. Three cases of rectal small cell carcinoma, a carcinoid tumor of the appendix and a NEC of the tail of the pancreas, were described. A further case series included four patients with GEP NEN ranging from well-differentiated NET to two aggressive metastatic NECs, and one patient had an intra-parotid lymph node MCC [23].
We describe the largest and most comprehensive cohort thus far reported in the literature of 19 HIV positive patients treated at a single institution and certified European NET Centre of Excellence; the clinical manifestations, treatment challenges and outcomes will be described.
2Methods
This descriptive cohort study included all HIV positive patients with biopsy-confirmed NET, NEC or MCC identified from the Royal Free Hospital NET registry and hospital records spanning the years January 1999–June 2020. Data were collected retrospectively from medical records and comprised patient demographics, date of diagnosis of HIV and date of diagnosis of NEN, HIV status (CD4 count and HIV viral load at time of NEN diagnosis, CD4 nadir), grade and stage of NEN, co-morbidities, radiological findings, cancer treatment received, modification of ART in preparation for cancer treatment and patient outcomes. Medical records were evaluated until date of death.
Given the anticipated small sample size and consequently limited statistical power, the intent of our analysis was descriptive and hypothesis generating. Continuous variables were expressed as median (range) or mean (standard deviation) and categorical variables as frequencies (percentages). Progression-free survival (PFS) and overall survival (OS) were estimated using the Kaplan-Meier method and comparison of curves was performed using the log-rank test. PFS was calculated from date of commencement of NEN therapy to date of first radiological progression or NEN-related death. Subjects without disease progression or death were censored at the date of last imaging assessment. OS was calculated from date of NEN histological diagnosis to date of NEN-related death. Subjects who remained alive were censored at last date of contact. Patients were followed for disease progression and survival every 3–6 months. Tumour assessment was performed with contrast-enhanced CT or MRI at baseline and then every 3–6 months. One patient was diagnosed with a G1 NET, one month before succumbing to metastatic esophageal cancer and was censored at date of death. One patient moved abroad after commencing third line systemic therapy; however the date of death was recorded. Statistical analyses were performed using SAS software, version 9.4 (SAS Institute, Cary, NC, USA) and Microsoft Excel, version 16.43. For all analyses, a two-sided p value < 0.05 was considered to be statistically significant.
3Results
3.1Patient population
Nineteen HIV positive patients with biopsy-confirmed NET (n = 14), NEC (n = 2) or MCC (n = 3) were included (Table 1). Sixteen (84%) patients were male with a median age at NEN diagnosis of 53 years (range 24–68).
Table 1
Patient characteristics at time of NEN diagnosis (n = 19)
| Patient | Gender | Age (yrs) | Site of primary and type | Morphology | Mitotic count/2mm2 | Ki67 | Stage of NEN | CD4 count at NEN diagnosis (cells/mm3) | HIV viral load at NEN diagnosis (copies/ml) | CD4 nadir (cells/mm3) | On ART | Second primary malignancy | Opportunistic infection history | Comorbidities | Current status | Months since NEN diagnosis |
| 1 | M | 24 | Bronchial (typical) | WD | <1 | 2% | Locoregional disease | HIV diagnosed after NET | 158 | No | – | – | – | Alive | 217 | |
| 2 | F | 68 | Bronchial (typical) | WD | 0 | <2% | Localised disease, background DIPNECH | NA | NA | NA | Yes | – | – | Latent HBV, current smoker | Alive | 12 |
| 3 | M | 50 | Bronchial (typical) | WD | 1 | 2% | Localised disease | 970 | <50 | 60 | Yes | – | – | – | Alive | 73 |
| 4 | M | 67 | Bronchial (typical) | WD | 0 | 2% | Localised disease | 10 | 691,831 | 10 | No | – | Late latent syphilis, esophageal candida, CMV viremia, PCP | T2DM, IHD, depression, latent HBV | Alive | 6 |
| 5 | M | 44 | Bronchial (typical) | WD | 1 | NR | Localised disease | HIV diagnosed after NET | 284 | Yes | – | – | T1DM, dyslipidemia, HTN | Alive | 88 | |
| 6 | M | 56 | Midgut | WD | <1/0 | 2% /18% * | Metastatic | 871 | <40 | 114 | Yes | – | – | IHD, HTN, renal colic | Deceased | 51 |
| 7 | M | 34 | Midgut | WD | <1 | 5% | Metastatic | 540 | <50 | 289 | Yes | – | – | dyslipidemia, primary hyperparathyroidism and renal calculus, current smoker | Alive | 87 |
| 8 | F | 46 | Midgut | WD | <2 | 10% | Metastatic | NA | NA | NA | Yes | – | Tuberculosis | Alive | 47 | |
| 9 | M | 65 | Pancreas | WD | 2 | 8% | Localised disease | 482 | <40 | 358 | Yes | – | – | IHD, aortic aneurysm, peripheral neuropathy, current smoker | Alive | 30 |
| 10 | M | 46 | Pancreas | WD | NR | 3% | Metastatic | 794 | <40 | 582 | Yes | – | – | – | Deceased | 57 |
| 11 | M | 53 | Pancreas | WD | NR | 2% | Localised disease | 30 | 91,453 | 30 | No | Non-small cell lung cancer, prostate cancer | Cerebral tuberculosis | Current smoker, autoimmune haemolytic anemia | Alive | 158 |
| 12 | M | 36 | Rectal (polyp) | WD | NR | <2% | Localised disease | NA | NA | NA | Yes | – | – | – | Alive | 21 |
| 13 | F | 45 | Paraganglioma (SDHA mutation), L carotid body tumour, non-functiona | WD | <4 | <5% | Localised disease | 592 | <40 | 266 | Yes | – | – | Cervix CIN1 and HPV | Alive | 10 |
| 14 | M | 62 | Unknown primary (extensive lymphadenopathy as only site of disease) | WD | <1 | <2% | Metastatic | NA | NA | NA | Yes | Esophageal cancer, Kaposi’s sarcoma | – | – | Deceased | 1 |
| 15 | M | 54 | Bronchial (LCNEC) | PD | 60 | 60% | Metastatic | 1738 | <40 | NA | Yes | – | – | NASH, HTN, COPD, osteopenia | Deceased | 23 |
| 16 | M | 41 | Unknown primary | PD | NR | 30% | Metastatic | 153 | 261,804 | NA | Poor compliance | – | – | HCV, ITP | Deceased | 5 |
| 17 | M | 58 | Merkel Cell | PD | 42 | 60% | Metastatic | 720 | <50 | 90 | Yes | Head and neck cancer | – | Obesity, familial hyperhomocysteinemia and PE, dyslipidemia, OSA, current | Deceased | 29 |
| 18 | M | 60 | Merkel Cell | PD | NR | NR | Metastatic | 230 | <50 | 20 | Yes | Anal SCC, Kaposi’s sarcoma, SCC of skin with neck node involvement | Cryptococcus, PCP | – | Deceased | 8 |
| 19 | M | 57 | Merkel Cell | PD | 15 | 80% | Metastatic | NA | NA | NA | Yes | – | – | – | Deceased | 6 |
NR not reported, NA not available, WD well-differentiated, PD poorly differentiated, DIPNECH diffuse idiopathic pulmonary neuroendocrine cell hyperplasia, CMV cytomegalovirus, PCP pneumocystis jirovecii pneumonia, T2DM type 2 diabetes mellitus, IHD ischemic heart disease, HTN hypertension, HBV hepatitis B, HCV hepatitis C, CIN1 cervical intraepithelial neoplasia grade 1, HPV human papilloma virus, SCC squamous cell carcinoma, NASH non-alcoholic steatohepatitis, COPD chronic obstructive pulmonary disease, ITP idiopathic thrombocytopenic purpura, PE pulmonary embolus, OSA obstructive sleep apnea. *Biopsy on peritoneal disease at diagnosis was consistent with G1 disease; biopsy of liver metastasis 2 years later showed high G2 disease.
Fourteen patients (74%) had a well-differentiated NET; the commonest primary tumor site was gastroenteropancreatic (3 midgut, 3 pancreas and 1 rectal), followed by five bronchial, one head and neck paraganglioma associated with a germline SDHA mutation and one unknown primary. Most well-differentiated NET (9/14, 64%) had low proliferative indices (Ki67 < 3%) and presented with localized or locoregional disease. The remaining five patients were all grade 2 (Ki67 3–20%); four of these patients presented with distant metastatic disease, one patient had a localized pancreatic primary which was successfully resected. One patient had a peritoneal biopsy at diagnosis consistent with a G1 NET of midgut origin, but a liver biopsy performed two years later in view of more aggressive disease behavior confirmed a G2 NET (Ki67 18%). Two patients with high volume liver metastases (primary midgut and primary lung, respectively) suffered from severe carcinoid syndrome.
Five patients (26%) had more aggressive G3 histology comprising two patients with poorly differentiated NEC (lung primary with Ki67 60%, and unknown primary with Ki67 30%) and three with MCC, all presenting with metastatic disease (Table 1).
3.2HIV characteristics and sequelae
Fifteen (79%) patients were diagnosed with HIV prior to their NEN diagnosis by a median of 11 years (range 8–28): two patients had a diagnosis of NEN and HIV simultaneously and two had a diagnosis of NET prior to the HIV diagnosis, but may have been living with undiagnosed HIV at the time.
At time of NEN diagnosis the majority of patients with known HIV were receiving ART (14/15; 93%); one patient was non-compliant with therapy. Viral load and CD4 count were available for 12 patients within six months of their NEN diagnosis; nine of these patients had an undetectable viral load and mean CD4 count of 771 cells/mm3 (Table 1). Three patients had advanced HIV at the time of their NEN diagnosis: one patient with a poorly differentiated metastatic NEC of unknown primary had an HIV viral load of 261,804 copies/ml and CD4 count 150 cells/mm3 and was non-compliant with ART; one patient had an incidental finding of an 8 mm low-grade bronchial NET (typical carcinoid) at the time of presenting with advanced HIV (viral load 691,831 copies/ml, CD4 count 10 cells/mm3) and multiple sequelae (late latent syphilis, esophageal candida, CMV viremia, Pneumocystis jirovecii pneumonia (PCP)) and another patient had an incidental finding of a G1 tail of pancreas NET after presenting with advanced HIV (viral load 91,453 copies/ml, CD4 count 30 cells/mm3) and cerebral tuberculosis. Three other patients had a history of an AIDS-defining illness prior to NEN diagnosis: two with Kaposi’s sarcoma and one with tuberculosis.
Non-AIDS defining cancers were seen in four patients comprising head and neck cancer, non-small cell lung cancer, prostate cancer, locally advanced anal squamous cell carcinoma, locally advanced squamous cell carcinoma of the skin and metastatic squamous cell carcinoma of the esophagus.
All patients with known HIV diagnoses were receiving ART at the time of commencement of systemic therapy for their NEN.
3.3NEN treatment outcomes and prognosis
3.3.1Bronchial NET
In the well-differentiated NET cohort (Table 2), patients with bronchial NETs (n = 5) presented with localized or locoregional disease and low-grade histopathology (typical carcinoid), and four proceeded to pulmonary lobectomy; all were alive after a median of 73 months (range 6–217) follow-up. Two patients developed distant recurrence. One patient with a solitary positive lymph node at the time of lobectomy (TNM stage [24] T2a N1 M0 R0) developed multiple sub-centimeter liver lesions eight years later, suspicious for NET metastases (arterially-enhancing on MRI and avid on 68Gallium DOTATATE PET). This patient was commenced on systemic therapy with long-acting somatostatin analogues and the liver lesions remain stable ten years to date. A second patient who had a pulmonary lobectomy but without lymph node clearance (TNM not available) developed high volume liver and bone metastases 5.3 years later complicated by carcinoid syndrome. He was treated with long-acting somatostatin analogues and peptide receptor radionuclide therapy (PRRT) with 177Lutetium DOTA-Octreotate and achieved a sustained partial objective response and amelioration of his syndrome. Both patients had an undetectable HIV viral load at time of NET recurrence.
Table 2
Therapy received and outcomes in the well-differentiated NET cohort (n = 14)
| Patient | Primary | Ki67 | Stage | Surgical resection of primary and stage | Outcome | 1st line therapy | Outcome | 2nd line therapy | Outcome | 3rd line therapy | Outcome | 4th line therapy | ART | Status | Months since NEN diagnosis |
| 1 | Bronchial | 2% | Locoregional disease | Yes (T2a N1 M0 R0) | Distant recurrence of disease 8 yrs following surgery | SSTA | Stable disease after 10 years surveillance | – | – | – | – | – | Emtricitabine, rilpivirine, tenofovir | Alive | 217 |
| 2 | Bronchial | < 2% | Localised disease | Yes (T1a Nx M0 R0) | No recurrence after 12 months follow up. DIPNECH lung nodules stable. | – | – | – | – | – | – | – | Nevirapine, emtricitabine, tenofovir | Alive | 12 |
| 3 | Bronchial | 2% | Localised disease | Yes (T1a N0 M0 R0) | Disease free at 5 yrs follow up | – | – | – | – | – | – | – | Abacavir, dolutegravir, ritonavir, darunavir | Alive | 73 |
| 4 | Bronchial | 2% | Localised disease | No –active surveillance | – | – | – | – | – | – | – | – | Emtricitabine, tenofovir, raltegravir | Alive | 6 |
| 5 | Bronchial | 1 mitosis/2 mm2* | Localised disease | Yes (TNM stage not available) | Distant recurrence of disease 5.3 yrs following surgery | SSTA and PRRT (177Lu) | Persistent partial response after 20 months after cycle 1 | – | – | – | – | – | Rilpivirine, emtricitabine, tenofovir | Alive | 88 |
| 6 | Midgut | 2% /18% | Metastatic | No | – | SSTA | PFS 8 mo | PRRT | PFS 12 mo | FCarboStrep | PFS 5 months | FOLFIRI; discontinued after cycle 1 due to severe diarrhea and AKI | Genvoya, switched to dolutegravir/Descovy due to fall in CD4 on chemotherapy; then switched to dolutegravir/ lamivudine due to AKI | Deceased | 51 |
| 7 | Midgut | 5% | Metastatic | Yes | – | SSTA for small volume residual disease | Stable disease after 7 years | – | – | – | – | – | Emtricitabine, tenofovir, darunavir, ritonavir | Alive | 87 |
| 8 | Midgut | 10% | Metastatic | Yes (pT3 N1 V1 R0 M1) | – | RFA to residual solitary liver metastasis | No recurrence 7 months post RFA | – | – | – | – | – | Efavirenz, emtricitabine, tenofovir | Alive | 47 |
| 9 | Pancreas | 8% | Localised | Yes (Whipple’s procedure, T3 N0 M0 R0) | No recurrence after 24 months follow-up | – | – | – | – | – | – | – | Darunavir, cobicistat, tenofovir, abacavir switched to dolutegarvir, emtricitabine, tenofovir, post-operatively | Alive | 30 |
| 10 | Pancreas | 3% | Metastatic | No | – | SSTA and sunitinib | PFS 14 mo | FCiSt (6 cycles) | PFS 14 mo | FCiSt re-challenge | Moved abroad | – | Efavirenz, emtricitabine, tenofovir | Deceased | 57 |
| 11 | Pancreas | 2% | Localised | No | Surveillance. Stable disease after 158 months follow-up. | – | – | – | – | – | – | – | Emtricitabine, tenofovir, darunavir, cobicistat | Alive | 158 |
| 12 | Rectal polyp | 2% | Localised | Cold snare polypectomy | No recur-rence on surveillance | – | – | – | – | – | – | – | Emtricitabine, tenofovir, efavirenz | Alive | 21 |
| 13 | Head and neck paraganglioma | < 5% | Localised | Yes | No recur-rence on surveillance | – | – | – | – | – | – | – | Elvitegravir, cobicistat, emtricitabine, tenofovir | Alive | 10 |
| 14 | Unknown primary | < 2% | Metastatic | No (synchronous esophageal cancer) | – | – | – | – | – | – | – | – | Emtricitabine, tenofovir, ritonovir, darunavir | Deceased secondary to metastatic esophageal cancer | 1 |
*Ki67 not reported. SSTA somatostatin analogue. PFS progression free survival. RFA radiofrequency ablation. FCarboStrep 5-fluorouracil/carboplatin/streptozocin. FCiSt 5-fluorouracil/cisplatin/streptozocin. DIPNECH diffuse idiopathic pulmonary neuroendocrine cell hyperplasia. PRRT peptide receptor radionuclide therapy. mo months. AKI acute kidney injury.
3.3.2Midgut NET
Three patients had midgut primaries. Two of these patients had relatively small volume metastatic disease and underwent surgical resection of the primary tumor followed by systemic therapy with somatostatin analogues in one case (stable disease after seven years on treatment) and radiofrequency ablation to treat a residual solitary liver metastasis in the other (no evidence of recurrence after seven months follow-up). The third case progressed despite first-line somatostatin analogue therapy and behaved more aggressively (Ki67 2% on first biopsy and 18% on second biopsy). This patient had somatostatin receptor-expressing disease and also suffered severe carcinoid syndrome; he was treated with PRRT with 177Lutetium DOTA-Octreotate alongside somatostatin analogues. Disease progression occurred promptly after the fourth cycle and thus chemotherapy with 5-fluorouracil (5FU), carboplatin and streptozocin was commenced which was well tolerated until the fifth and sixth cycle when a drop in CD4 count occurred. He was commenced on PCP prophylaxis and his antiretroviral therapy elvitegravir/cobicistat/emtricitabine/tenofovir was switched to dolutegravir/emtricitabine/tenofovir. He proceeded to fourth-line therapy with 5FU/irinotecan but experienced life-threatening diarrhea complicated by acute kidney injury (creatinine 426 umol/L; eGFR <15 ml/min) six days following the first cycle, which he survived. Emtricitabine/tenofovir was switched to lamivudine in view of the kidney injury, and chemotherapy was discontinued. The patient died due to progressive metastatic disease 7 months later.
3.3.3Pancreatic NET
Of the three patients with well-differentiated pancreatic NET, one patient had localized disease and proceeded to a Whipple’s resection (T3 N0 M0, Ki67 8%) with no evidence of disease recurrence after 24 months follow-up. The second patient had a localized 10×11 cm tail of pancreas NET (Ki67 2%) discovered incidentally but was lost to follow-up for 13 years; a follow-up CT chest, abdomen and pelvis surprisingly showed stable appearances. The third patient had metastatic disease (Ki67 3%) and completed three lines of systemic therapy including somatostatin analogues, sunitinib and 5FU/cisplatin/streptozocin chemotherapy before eventually succumbing to progressive metastatic disease 57 months after diagnosis.
3.3.4Poorly differentiated NEC and MCC
Four out of five patients with poorly differentiated NEC or MCC received first-line therapy with platinum-based chemotherapy regimens with a median PFS of 5.5 months (range 2–11) (Table 3).
Table 3
Therapy received and outcomes in the poorly differentiated NEC and MCC cohort (n = 5)
| Patient | Primary | Ki67 | Stage | 1st line therapy | PFS | 2nd line therapy | PFS | 3rd line therapy | PFS | 4th line therapy | PFS | ART | Status | Months since NEN diagnosis |
| 15 | Lung LCNEC | 60% | Metastatic | Carboplatin/etoposide | 6 mo | FOLFIRI | 2 mo | CAV | 2 mo | Pembro-lizumab | 5 mo | Efavirenz, emtricitabine, tenofovir switched to raltegravir, emtricitabine, tenofovir prior to chemotherapy | Deceased | 23 |
| 16 | Unknown primary NEC | 30% | Metastatic | FCiSt | 2 mo | – | – | – | – | – | – | Boosted darunavir, raltegravir, rilpiverine switched to Triumeq due to non-compliance | Deceased | 5 |
| 17 | Merkel | 60% | Metastatic | Carboplatin/etoposide | 11 mo | Avelumab | 3 mo | – | – | – | – | Emtricitabine, tenofovir, nevirapine | Deceased | 29 |
| 18 | Merkel | NR | Metastatic | Avelumab | 26 days | – | – | – | – | – | – | Dolutegravir, rilpivirine, tenofovir | Deceased | 8 |
| 19 | Merkel | 80% | Metastatic | Carboplatin/etoposide | 5 mo | – | – | – | – | – | – | Receiving ART (regimen unrecorded) | Deceased | 6 |
LCNEC large cell neuroendocrine carcinoma. FCiSt 5-fluorouracil/cisplatin/streptozocin. FOLFIRI 5-fluorouracil/folinic acid/irinotecan. CAV cyclophosphamide/doxorubicin/vincristine. NR not reported.
Recently, immunotherapy (avelumab, pembrolizumab, nivolumab) has become the new standard of care for patients with metastatic MCC based on several clinical trials [25]. Two patients in our cohort with metastatic MCC and well-controlled HIV received avelumab, however, both did poorly: one patient with high-volume disease treated in the first-line setting died 26 days after cycle 1 due to progressive disease and the other patient treated in the second-line setting after chemotherapy had a PFS of only 3 months (Table 3). One patient with metastatic large cell NEC of the lung received pembrolizumab in the third-line setting but died 5 months after its commencement. There were no immune-related toxic effects. Changes in viral load or CD4 count status were unfortunately not documented.
3.3.5Overall survival
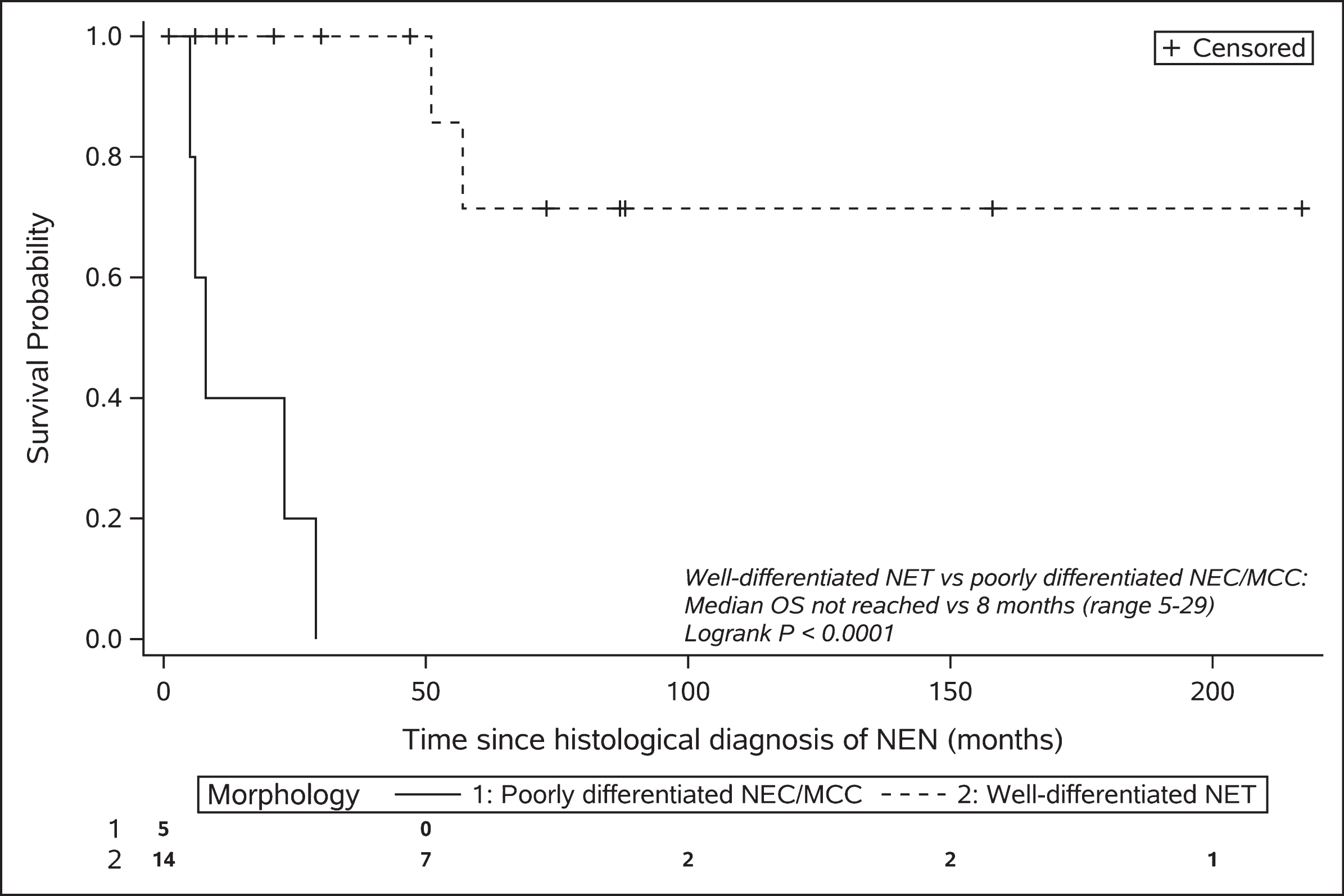
The median follow-up for patients still alive was 47 months (range 6–217) after NEN diagnosis. Of the entire cohort (n = 19), eight patients had died. Five of these patients had poorly differentiated NEC or MCC, one patient had a metastatic G2 midgut NET and died 51 months after diagnosis, one patient had a metastatic G2 pancreatic NET and died 57 months after diagnosis, and one patient with a G1 NET of unknown primary died from a synchronous esophageal squamous cell carcinoma 1 month after NET diagnosis. The median overall survival in the NEC/MCC cohort was 8 months (range 5–29). The median overall survival in the well-differentiated NET cohort has not been reached but, based on the 25th centile, 75% of patients are expected to survive for at least 57 months (Fig. 1).
Fig. 1
Kaplan-Meier plot of overall survival stratified by morphology.
4Discussion
HIV and concomitant NEN is an uncommon occurrence. The Royal Free Neuroendocrine Tumour Unit is a large international referral center and has treated approximately 3,500 patients with NEN over the last 10 years. Within that time, we identified 19 patients with concomitant HIV, giving an HIV prevalence of close to 0.5% in the NEN population. This might be an underestimate given that our unit, similar to many others, does not routinely screen patients for HIV infection. However, this is somewhat higher than the HIV prevalence in adults in the United Kingdom (0.17%) [26].
This cohort was predominantly male (84%) and HIV comorbidities and second primary cancer diagnoses were common. The characteristics of NEN including diagnosis in the sixth decade of life, and the predominance of well-differentiated (14/19; 74%) as opposed to poorly differentiated tumors, mirror those of the general NEN population [1, 27–30], and this likely reflects the fact that the majority of our cohort with known HIV were receiving treatment with ART (14/15; 93%).
In terms of overall survival, patients with NETs of the lung (n = 5 typical carcinoid) were all alive after a median follow-up of 73 months (range 6–217), and had the best prognosis owing to low grade histopathology and low rates of metastases at diagnosis. This is supported by the general NET literature, which documents a 5-year survival rate for typical pulmonary carcinoid tumors of 87–90% [31, 32], although late recurrences have been reported [3, 33] as demonstrated in two of our patients, and thus long-term follow-up is important.
The patients with well-differentiated midgut and pancreatic NETs also had a relatively good prognosis, with 4/6 (67%) alive after a median follow-up of 54 months (range 30–158; 3-year survival rate 100%; median OS not reached). This is not inferior to survival rates in well-differentiated (G1/G2) midgut and pancreatic NET in the wider NET population, with a 3-year survival rate reported as 80% and 62% respectively [1]. Three patients, with localized disease or small volume metastatic disease, did well after resection of the primary followed by liver-directed therapy or somatostatin analogues. Two patients with extensive metastatic disease had a more difficult course requiring multiple sequential therapies.
The five patients with poorly differentiated NEC or MCC, as anticipated, demonstrated a much poorer prognosis compared with the well-differentiated NET population (Fig. 1). The median PFS following platinum-based chemotherapy (5.5 months, range 2–11) and median overall survival (8 months, range 5–29) was not dissimilar to the wider NEC and MCC populations [34–37].
These findings must be interpreted in light of the limited statistical power available in analyses of rare diseases. Our study is limited by the small number of cases available and the retrospective, single-cohort design. This study also reflects a single-institution experience and while this allows for more homogeneous clinical practice the results may not be generalizable to other cancer centers. The Royal Free Neuroendocrine Tumour Unit is a large international referral center, and while this has permitted focus on a very rare subgroup of patients there may also be a potential bias towards rapid and comprehensive evaluation of neuroendocrine neoplasms. A multicenter, prospective study is required to validate our findings with inclusion of a formal non-HIV comparator group.
4.1Special considerations in the management of patients with NEN and HIV
4.1.1Drug-drug interactions
Many studies have shown that the combined treatment of cancer and HIV is safe and improves outcomes [38, 39]. However, drug-drug interactions and the need to maximize ART/cancer tre-atment efficacy must be taken into consideration [40]. Evidence-based, drug-drug interaction resources, such as the University of Liverpool HIV Drug Interactions Database (https://www.hiv-druginteractions.org/checker), are useful tools for the clinic.
Many drugs used in ART (e.g. protease inhibitors [PIs], non-nucleoside reverse transcriptase inhibitors [NNRTIs] and chemokine receptor antagonists) have the potential to cause drug interactions due to their ability to induce or inhibit the cytochrome P450 (CYP450) enzyme system. Many chemotherapy agents and molecular targeted therapies are also metabolized through the same pathway, hence there is a high potential for drug interactions with CYP450-metabolised ART. Focusing on NEN therapies, the antimetabolite agents (e.g. 5-fluorouracil), antitumor antibiotics (e.g. streptozocin) and platinums are not metabolized by the CYP450 system and therefore are unlikely to be altered by ART. In contrast, camptothecins (e.g. irinotecan) are eliminated by non-enzymatic routes and are substrates of CYP450 and UGT isozymes, and are consequently more likely to be altered by CYP450-metabolised ART [41]. Bidirectional drug interactions with CYP450-metabolised ART can also be anticipated with alkylating agents (e.g. temozolomide), epipodophyllotoxins (e.g. etoposide), tyrosine-kinase inhibitors (e.g. sunitinib), mTOR inhibitors (e.g. everolimus) and dexamethasone. While there are no randomized trials to guide the optimal ART regimen to be co-administered with these agents, ART that is metabolized independently of the CYP450 system (e.g. NRTIs, raltegravir, enfuvirtide) or that are substrates but do not induce or inhibit CYP450 enzymes (e.g. rilpivirine) are preferred [40, 42]. An evidence-based and regularly updated summary detailing the above described interactions between cancer therapies and all generations of ART can be found on the Liverpool drug interactions website (https://www.hiv-druginteractions.org/prescribing-resources -> treatment selector (by therapeutic indication) -> cancer therapies).
In our cohort, several ART regimens were used concurrently with various anti-cancer therapies, including somatostatin analogues, molecular targeted therapy with sunitinib, chemotherapy with carboplatin/etoposide, 5FU/cisplatin/streptozocin or 5FU/irinotecan and the anti-programmed cell death-1 (PD-1)/-programmed death ligand-1 (PDL-1) inhibitors, avelumab and pembrolizumab (Table 2 and Table 3). No significant drug-drug interactions were identified in our cohort; however, to minimize this risk every anti-cancer therapy and ART regimen was reviewed by a specialist pharmacist. One patient receiving dolutegravir/emtricitabine/tenofovir developed life-threatening diarrhea 6 days following cycle 1 5FU/irinotecan. There is no known interaction between dolutegravir/emtricitabine/tenofovir and irinotecan and an interaction between these drug regimens was considered unlikely. Although not formally tested, dihydropyrimidine dehydrogenase deficiency was also considered unlikely as the patient had previously received 5FU/carboplatin/streptozocin, alongside the same ART regimen, without harm. It was therefore concluded that the diarrhea was most likely secondary to the irinotecan.
4.1.2Immune checkpoint inhibitors and HIV
Three patients in our NEC/MCC cohort received immunotherapy with PD-1/PDL-1 inhibitors; however, outcomes were relatively poor in this small cohort. In the past, HIV positive patients have been excluded from checkpoint inhibitor clinical trials given concern regarding immune-related toxicity. Nevertheless, observational studies including a phase II single arm prospective study in patients with HIV and solid organ malignancies [43], suggest similar response rates and toxicity profiles compared to the non-HIV population [44–46]. Furthermore, animal models have shown that inhibiting PD-1 signaling can reduce HIV viral loads [47] and may play a role in immune reconstitution by restoring HIV-specific CD8 + T-cell function [48, 49]. A systematic review reporting the tolerance profile of immune checkpoint inhibitors, their effects on HIV viral loads, CD4 + cell count, HIV reservoirs and/or HIV-specific CD8 + T cells, has been recently reported [50]. Thirty-one articles were included, with a total of 176 participants. The side effect profile was similar to the wider population, with 12% experiencing serious adverse events and 49% non-severe adverse events. Plasma viral loads remained stable in 91.9% of patients, increased in 5.8% and decreased in 2.3%. CD4 + counts remained stable in 60.7%, increased in 24.6% and decreased in 14.7%. Most of the trials did not characterize HIV-specific CD8 + T cells as well as viral load, but in a small number of patients, transient plasma viral load increases were noted followed by a boost in HIV-specific CD8 + T cells then associated with a decrease in HIV-DNA.
4.1.3Overlapping toxicity
Anticancer drugs without potential for drug-drug interactions can share side effects with ART that are worsened when administered together, although no overlapping toxicities were seen in our cohort. For example, didanosine, stavudine, maraviroc and zidovudine can cause hepatotoxicity and should be avoided in combination with NEN chemotherapy drugs that rely on hepatic metabolism (e.g. etoposide, irinotecan) [41]. In contrast, the NRTIs, abacavir, emtricitabine, lamivudine and tenofovir, and the NNRTI, efavirenz are less likely to be hepatotoxic [41].
The molecular targeted therapies cause less myelosuppression compared with chemotherapy but may, however, have other potentially overlapping toxicities with ART [13]. Sunitinib, approved for the use in metastatic pancreatic NET, can cause QT prolongation, hypertension and increased cardiovascular events [51]. The boosted PIs, as well as rilpivirine and efavirenz, can also cause QT prolongation and should be avoided in combination with QT-prolonging molecular targeted agents given the risk of arrhythmia and sudden death [15, 41]. The mTOR inhibitor, everolimus, used in patients with gastrointestinal, pancreatic and bronchial NET, is associated with an increased risk of hyperglycemia and hypertriglyceridemia [9, 10]. PI drugs are also associated with hypertriglyceridemia, hypercholesterolemia, insulin resistance and hyperglycemia, and such risk factors may synergize.
4.1.4Prophylaxis against opportunistic infection
Chemotherapy drugs, especially lymphotoxins such as temozolomide which is often used in patients with progressive pancreatic NET, can cause CD4 count depletion and increase the risk of opportunistic infections including cytomegalovirus and other herpes reactivations, mycobacteria infections and invasive fungal infections [15]. Opportunistic infection prophylaxis may therefore need to be commenced during chemotherapy, and regular monitoring of CD4 counts in HIV patients receiving chemotherapy is vital. This may also be important in patients treated with PRRT. PRRT with 177Lutetium, used to treat patients with well-differentiated, somatostatin receptor-expressing NETs, can also cause significant lymphopenia [11] and patients with high volume bone metastases and prior treatment with chemotherapy may be more prone to PRRT-induced myelosuppression. Two patients in our cohort were treated with PRRT. Neither developed myelosuppression throughout four cycles of PRRT; however, one patient had a drop in CD4 count to 114 cells/mm3 during his subsequent line of therapy with 5FU/carboplatin/streptozocin. He was commenced on PCP prophylaxis with pentamidine which was later stopped after the completion of chemotherapy and CD4 recovery.
4.1.5Hepatitis B reactivation
Two patients in our cohort had antibodies to hepatitis B core antigen. In the setting of chemotherapy, these patients should be commenced on anti-viral prophylaxis to prevent HBV reactivation [52]; however, it is essential that ART is commenced prior to lamivudine or tenofovir monotherapy which can lead to the development of ART resistance [40].
5Conclusions
Within the limits of a small descriptive cohort study, we found no evidence in the era of ART that patients with HIV and NEN are diagnosed at a younger age, nor do they have a poorer prognosis compared with the wider NEN population. Thus, patients with HIV and NEN should be managed similarly, with pursuit of surgical resection in the setting of localized or small volume metastatic disease which offers the only chance of cure. In the unresectable disease or metastatic setting, the full breadth of liver-directed therapies and systemic therapies should also be offered to these patients to support best NEN outcomes, alongside optimization of their ART regimen to minimize the risk of drug interactions and treatment-related complications. Nevertheless, this patient cohort has unique characteristics with a high incidence of comorbidities and significant potential for therapy drug-drug interactions and overlapping toxicities. Their management by a multidisciplinary team including oncologists, HIV physicians, endocrinologists and specialist pharmacists, is paramount.
Author contributions
AH and ML collected the data. AH performed the data analysis. AH and KS drafted the manuscript. All authors critically revised and approved the final manuscript.
Conflict of interest
The authors declare no Conflict of interest.
Funding
This project was funded by the Royal Free Charitable Trust (Quiet Cancer) Grant 311 and the Parasol Foundation Trust.
M.L. is funded by the China post-doctoral Science Foundation
References
[1] | Dasari A. , Shen C. , Halperin D. , Zhao B. , Zhou S. , Xu Y. , et al., Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States, JAMA Oncol 3: ((2017) ), 1335–42. |
[2] | Genus T.S.E. , Bouvier C. , Wong K.F. , Srirajaskanthan R. , Rous B.A. , Talbot D.C. , et al., Impact of neuroendocrine morphology on cancer outcomes and stage at diagnosis: a UK nationwide cohort study 2013–2015, Br J Cancer 121: ((2019) ), 966–972. |
[3] | Yao J.C. , Hassan M. , Phan A. , Dagohoy C. , Leary C. , Mares J.E. , et al., One hundred years after “carcinoid”: Epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States, J Clin Oncol 26: ((2008) ), 3063–3072. |
[4] | Bosman F. , Carneiro F. , Hruban R. and Theise N. , (Eds.). WHO Classification of Tumours of the Digestive System (4th edn.). World Health Organization; 2010. |
[5] | Klimstra D.S. , Kloppell G. and La Rosa S.R.G. , Classification of neuroendocrine neoplasms of the digestive system. WHO Classif Tumours Dig Syst Tumours, 5th ed, WHO Classif Tumours Editor Board (Ed), Int Agency Res Cancer, Lyon 2019. |
[6] | Rinke A. , Müller H.H. , Schade-Brittinger C. , Klose K.J. , Barth P. , Wied M. , et al., Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: A report from the PROMID study group, J Clin Oncol 27: ((2009) ), 4656–4663. |
[7] | Caplin M.E. , Pavel M. , Ćwikła J.B. , Phan A.T. , Raderer M. , Sedláčková E. , et al., Lanreotide in metastatic enteropancreatic neuroendocrine tumors, N Engl J Med 371: ((2014) ), 224–33. |
[8] | Raymond E. , Dahan L. , Raoul J.L. , Bang Y.J. , Borbath I. , Lombard-Bohas C. , et al., Sunitinib malate for the treatment of pancreatic neuroendocrine tumors, N Engl J Med 364: ((2011) ), 501–513. |
[9] | Yao J.C. , Shah M.H. , Ito T. , Bohas C.L. , Wolin E.M. , Van Cutsem E. , et al., Everolimus for advanced pancreatic neuroendocrine tumors, N Engl J Med 364: ((2011) ), 514–523. |
[10] | Yao J.C. , Fazio N. , Singh S. , Buzzoni R. , Carnaghi C. , Wolin E. , et al., Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): A randomised, placebo-controlled, phase 3 study, Lancet 387: ((2016) ), 968–977. |
[11] | Strosberg J. , El-Haddad G. , Wolin E. , Hendifar A. , Yao J. , Chasen B. , et al., Phase 3 trial of 177Lu-dotatate for midgut neuroendocrine tumors, N Engl J Med 376: ((2017) ), 125–135. |
[12] | Kunz P.L. , Catalano P.J. , Nimeiri H. , Fisher G.A. , Longacre T.A. , Suarez C.J. , et al., A randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors: A trial of the ECOG-ACRIN Cancer Research Group (E2211), J Clin Oncol American Society of Clinical Oncology (ASCO) 36: ((2018) ), 4004–4004. |
[13] | Deeken J.F. , Tjen-A-Looi A J.F. , M.A. Rudek J.F. , Okuliar C. , Young M. , Little R.F. , et al., The Rising Challenge of Non-AIDS-Defining Cancers in HIV-Infected Patients, Clin Infect Dis. Oxford University Press (OUP) 55: ((2012) ), 1228–1235. |
[14] | Mitsuyasu R.T. , Non–AIDS-defining malignancies in HIV, Top HIV Med 16: ((2008) ), 117–121. |
[15] | Torres H.A. and Mulanovich V. , Management of HIV infection in patients with cancer receiving chemotherapy,, Clin Infect Dis 59: ((2014) ), 106–114. |
[16] | Izikson L. , Nornhold E. , Iyer J.G. , Nghiem P. and Zeitouni N.C. , Merkel cell carcinoma associated with HIV: review of 14 patients,, AIDS. Ovid Technologies (Wolters Kluwer Health) 25: ((2011) ), 119–121. |
[17] | Harms P.W. , Harms K.L. , Moore P.S. , DeCaprio J.A. , Nghiem P. , Wong M.K.K. , et al., The biology and treatment of Merkel cell carcinoma: current understanding and research priorities,, Nat Rev Clin Oncol, Springer Science and Business Media LLC 15: ((2018) ), 763–776. |
[18] | Wieland U. and Kreuter A. , Merkel cell polyomavirus infection and Merkel cell carcinoma in HIV-positive individuals, Curr Opin Oncol, Ovid Technologies (Wolters Kluwer Health) 23: ((2011) ), 488–493. |
[19] | Liu W. , MacDonald M. and You J. , Merkel cell polyomavirus infection and Merkel cell carcinoma, Curr Opin Virol, Elsevier BV 20: ((2016) ), 20–7. |
[20] | Engels E.A. , Frisch M. , Goedert J.J. , Biggar R.J. and Miller R.W. , Merkel cell carcinoma and HIV infection, Lancet. Elsevier BV 359: ((2002) ), 497–498. |
[21] | Nicholaides R.E. and de la Morena M.T. , Inherited and acquired clinical phenotypes associated with neuroendocrine tumors. Curr Opin Allergy Clin Immunol, Ovid Technologies (Wolters Kluwer Health) 17: ((2017) ), 431–442. |
[22] | Thakker R.V. , Newey P.J. , Walls G.V. , Bilezikian J. , Dralle H. , Ebeling P.R. , et al., Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1), J Clin Endocrinol Metab 97: ((2012) ), 2990–3011. |
[23] | Lito P. , Marotti J. , Dezube B.J. , Pantanowitz L. , Aboulafia D.M. , Campbell V. , et al., Gastroenteropancreatic Neuroendocrine Tumors in Patients With HIV Infection: A Trans-Atlantic Series, Am J Med Sci. Elsevier BV 337: ((2009) ), 1–4. |
[24] | Rami-Porta R. , Asamura H. and Travis W.D.R.V. , AJCC Cancer Staging Manual, AJCC cancer staging manual (2017), pp. 431. |
[25] | Garcia-Carbonero R. , Marquez-Rodas I. , de la Cruz-Merino L. , Martinez-Trufero J. , Cabrera M.A. , Piulats J.M. , et al., Recent Therapeutic Advances and Change in Treatment Paradigm of Patients with Merkel Cell Carcinoma, Oncologist Alphamed Press 24: ((2019) ), 1375–1383. |
[26] | Public Health England. Prevalence of HIV infection in the UK in 2018, Health Protection Report 13: (39) ((2018) ), 2019. |
[27] | Caplin M.E. , Baudin E. , Ferolla P. , Filosso P. , Garcia-Yuste M. , Lim E. , et al., Pulmonary neuroendocrine (carcinoid) tumors: European Neuroendocrine Tumor Society expert consensus and recommendations for best practice for typical and atypical pulmonary carcinoids, Ann Oncol 26: ((2015) ), 1604–1620. |
[28] | Öberg K. , Knigge U. , Kwekkeboom D. and Perren A. , Neuroendocrine gastro-entero-pancreatic tumors: ESMO clinical practice guidelines for diagnosis, treatment and follow-up, Ann Oncol Narnia 23: ((2012) ), vii124–30. |
[29] | Niederle B. , Pape U.F. , Costa F. , Gross D. , Kelestimur F. , Knigge U. , et al., ENETS consensus guidelines update for neuroendocrine neoplasms of the jejunum and ileum , Neuroendocrinology. S. Karger AG 103: ((2016) ), 125–38. |
[30] | Fraenkel M. , Kim M.K. , Faggiano A. and Valk G.D. , Epidemiology of gastroenteropancreatic neuroendocrine tumours, Best Pract Res Clin Gastroenterol. Bailliere Tindall Ltd 26: ((2012) ), 691–703. |
[31] | Travis W.D. , Rush W. , Flieder D.B. , Falk R. , Fleming M.V. , Gal A.A. , et al., Survival analysis of 200 pulmonary neuroendocrine tumors with clarification of criteria for atypical carcinoid and its separation from typical carcinoid, Am J Surg Pathol 22: ((1998) ), 934–944. |
[32] | Fink G. , Krelbaum T. , Yellin A. , Bendayan D. , Saute M. , Glazer M. , et al., Pulmonary carcinoid: Presentation, diagnosis, and outcome in 142 cases in Israel and review of 640 cases from the literature, Chest 119: ((2001) ), 1647–1651. |
[33] | Lou F. , Sarkaria I. , Pietanza C. , Travis W. , Roh M.S. , Sica G. , et al., Recurrence of pulmonary carcinoid tumors after resection: Implications for postoperative surveillance, Ann Thorac Surg 96: ((2013) ), 1156–1162. |
[34] | Walter T. , Tougeron D. , Baudin E. , Le Malicot K. , Lecomte T. , Malka D. , et al., Poorly differentiated gastro-entero-pancreatic neuroendocrine carcinomas: Are they really heterogeneous? Insights from the FFCD-GTE national cohort, Eur J Cancer 79: ((2017) ), 158–165. |
[35] | Sorbye H. , Welin S. , Langer S.W. , Vestermark L.W. , Holt N. , Osterlund P. , et al., Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): The NORDIC NEC study, Ann Oncol 24: ((2013) ), 152–160. |
[36] | Iyer J.G. , Blom A. , Doumani R. , Lewis C. , Tarabadkar E.S. , Anderson A. , et al., Response rates and durability of chemotherapy among 62 patients with metastatic Merkel cell carcinoma, Cancer Med 5: ((2016) ), 2294–301. |
[37] | Allen P.J. , Bowne W.B. , Jaques D.P. , Brennan M.F. , Busam K. and Coit D.G. , Merkel cell carcinoma: Prognosis and treatment of patients from a single institution, J Clin Oncol 23: ((2005) ), 2300–2309. |
[38] | Barta S.K. , Xue X. , Wang D. , Tamari R. , Lee J.Y. , Mounier N. , et al., Treatment factors affecting outcomes in HIV-associated non-Hodgkin lymphomas: A pooled analysis of 1546 patients, Blood American Society of Hematology 122: ((2013) ), 3251–3262. |
[39] | Lavolé A. , Chouaïd C. , Baudrin L. , Wislez M. , Raguin G. , Pialoux G. , et al., Effect of highly active antiretroviral therapy on survival of HIV infected patients with non-small-cell lung cancer, Lung Cancer. Elsevier 65: ((2009) ), 345–50. |
[40] | Welz T. , Wyen C. and Hensel M. , Drug Interactions in the Treatment of Malignancy in HIV-Infected Patients, Oncol Res Treat. S. Karger AG 40: ((2017) ), 120–127. |
[41] | Rudek M.A. , Flexner C. and Ambinder R.F. , Use of antineoplastic agents in patients with cancer who have HIV/AIDS,, Lancet Oncol 12: ((2011) ), 905–912. |
[42] | Mounier N. , Katlama C. , Costagliola D. , Chichmanian R.M. and Spano J.P. , Drug interactions between antineoplastic and antiretroviral therapies: Implications and management for clinical practice, Crit Rev Oncol Hematol 72: ((2009) ), 10–20. |
[43] | González-Cao M. , Moran T. , Dalmau J. , Garcia-Corbacho J. , Bernabé R. , Juan O. , et al., Phase II study of durvalumab (MEDIin cancer patients HIV-1-infected, J Clin Oncol. American Society of Clinical Oncology (ASCO) 37: ((2019) ), 2501–2501. |
[44] | Shah N.J. , Al-Shbool G. , Blackburn M. , Cook M. , Belouali A. , Liu S.V. , et al., Safety and efficacy of immune checkpoint inhibitors (ICIs) in cancer patients with HIV, hepatitis B, or hepatitis C viral infection,, J Immunother Cancer 7: ((2019) ), 353. |
[45] | Kim C. and Cook M.R. , Safety and Efficacy of Immune Checkpoint Inhibitor Therapy in Patients with HIV Infection and Advanced-Stage Cancer: A Systematic Review, JAMA Oncol 5: ((2019) ), 1049–1053. |
[46] | Uldrick T.S. , Gonçalves P.H. , Abdul-Hay M. , Claeys A.J. , Emu B. , Ernstoff M.S. , et al., Assessment of the Safety of Pembrolizumab in Patients with HIV and Advanced Cancer - A Phase 1 Study. JAMA Oncol, American Medical Association 5: ((2019) ), 1332–1339. |
[47] | Seung E. , Dudek T.E. , Allen T.M. , Freeman G.J. , Luster A.D. and Tager A.M. , PD-1 Blockade in Chronically HIV-1-Infected Humanized Mice Suppresses Viral Loads. Apetrei C, editor,, PLoS One 8: ((2013) ), e77780. |
[48] | Grabmeier-Pfistershammer K. , Steinberger P. , Rieger A. , Leitner J. and Kohrgruber N. , Identification of PD-1 as a unique marker for failing immune reconstitution in HIV-1-infected patients on treatment, J Acquir Immune Defic Syndr 56: ((2011) ), 118–124. |
[49] | Velu V. , Shetty D.D. , Larsson M. and Shankar E.M. , Role of PD-1 co-inhibitory pathway in HIV infection and potential therapeutic options, Retrovirology 12: ((2015) ), 14. |
[50] | Abbar B. , Baron M. , Katlama C. , Marcelin A-G. , Veyri M. , Autran B. , et al., Immune checkpoint inhibitors in people living with HIV. What about anti-HIV effects? AIDS. Ovid Technologies (Wolters Kluwer Health) 34: ((2020) ), 167–175. |
[51] | Bello C.L. , Mulay M. , Huang X. , Patyna S. , Dinolfo M. , Levine S. , et al., Electrocardiographic characterization of the QTc interval in patients with advanced solid tumors: Pharmacokinetic-pharmacodynamic evaluation of sunitinib, Clin Cancer Res 15: ((2009) ), 7045–7052. |
[52] | Bower M. , Palfreeman A. , Alfa-Wali M. , Bunker C. , Burns F. , Churchill D. , et al., British HIV Association guidelines for HIV-associated malignancies, HIV Med 15: ((2014) ), 1–92. |




