SELENON-Related Myopathy Across the Life Span, a Cross-Sectional Study for Preparing Trial Readiness
Abstract
Background:
SELENON(SEPN1)-related myopathy (SELENON-RM) is a rare congenital neuromuscular disease characterized by proximal and axial muscle weakness, spinal rigidity, scoliosis and respiratory impairment. No curative treatment options exist, but promising preclinical studies are ongoing. Currently, natural history data are lacking, while selection of appropriate clinical and functional outcome measures is needed to reach trial readiness.
Objective:
We aim to identify all Dutch and Dutch-speaking Belgian SELENON-RM patients, deep clinical phenotyping, trial readiness and optimization of clinical care.
Methods:
This cross-sectional, single-center, observational study comprised neurological examination, functional measurements including Motor Function Measurement 20/32 (MFM-20/32) and accelerometry, questionnaires, muscle ultrasound, respiratory function tests, electro- and echocardiography, and dual-energy X-ray absorptiometry.
Results:
Eleven patients with genetically confirmed SELENON-RM were included (20±13 (3–42) years, 73% male). Axial and proximal muscle weakness were most pronounced. The mean MFM-20/32 score was 71.2±15.1%, with domain 1 (standing and transfers) being most severely affected. Accelerometry showed a strong correlation with MFM-20/32. Questionnaires revealed impaired quality of life, pain and problematic fatigue. Muscle ultrasound showed symmetrically increased echogenicity in all muscles. Respiratory function, and particularly diaphragm function, was impaired in all patients, irrespective of the age. Cardiac assessment showed normal left ventricular systolic function in all patients but abnormal left ventricular global longitudinal strain in 43% of patients and QRS fragmentation in 80%. Further, 80% of patients showed decreased bone mineral density on dual-energy X-ray absorptiometry scan and 55% of patients retrospectively experienced fragility long bone fractures.
Conclusions:
We recommend cardiorespiratory follow-up as a part of routine clinical care in all patients. Furthermore, we advise vitamin D supplementation and optimization of calcium intake to improve bone quality. We recommend management interventions to reduce pain and fatigue. For future clinical trials, we propose MFM-20/32, accelerometry and muscle ultrasound to capture disease severity and possibly disease progression.
ABBREVIATIONS
CIS: checklist individual strength
DEXA-scan: dual-energy X-ray absorptiometry
EK2: Egen klassifikation version 2
FEV1: forced expiratory volume in 1 second
FVC: forced vital capacity
GLS: global longitudinal strain
GTFT: graded and timed function tests
HFMS: Hammersmith functional motor scale
9 HHD: hand-held dynamometry
INQoL: Individualized Neuromuscular Quality of Life
IPA: Impact on Participation and Autonomy
LVEF: left ventricular ejection fraction
MEP: maximum expiratory pressure
14 MFM-20/32: Motor Function Measure –20/32
MiniBEST: Mini Balance Evaluation Systems Test
MIP: maximum inspiratory pressure
MRC: medical research council
PBS: Pediatric Balance Scale
PCF: peak cough flow
PedsQL: Pediatric Quality of Life Inventory
RAND36: Reasearch and Development-36
SELENON-RM: SELENON-related myopathy
SEPN1-RM: SEPN1-related myopathy
24 SNIP: sniff nasal inspiratory pressure
TDI: tissue doppler imaging
TTE: transthoracic echocardiography
TUG: Timed Up and Go
VC: vital capacity
6MWT: 6-minute walk test
10MWT: 10-meter walk test
INTRODUCTION
SELENON(SEPN1)-related congenital myopathy (SELENON-RM) is characterized by axial and proximal muscle weakness, early-onset rigidity of the spine, scoliosis, and respiratory insufficiency. Histologic disease entities in this spectrum include multi-minicore disease, congenital fiber type disproportion (CFTD), desmin-related myopathy with Mallory-body like inclusions and rigid spine muscular dystrophy (RSMD1) [1–5]. The clinical diagnosis of SELENON-RM is confirmed by two recessive (likely) pathogenic variants in the SELENON (SEPN1) gene coding for the selenoprotein N. Selenoprotein N is an endoplasmic reticulum (ER) calcium sensor that responds to diminished luminal calcium levels by refilling the ER calcium stores. SELENON-RM has striking similarities at the cellular level with classical mitochondrial diseases, with marked alterations in mitochondrial physiology and energy metabolism [6]. Currently, no curative treatment options are available, but promising preclinical trials are ongoing [7–9]. Supportive care from rehabilitation and allied health care in combination with optimal respiratory, cardiac, nutrition and orthopedic management, is essential for preventing or treating severe complications in SELENON-RM [10]. To pave the way towards clinical trials, it is important to identify and clinically and genetically characterize patients, and to select sensitive and patient friendly clinical and functional outcome measures.
There are two previous retrospective clinical studies on the natural history of patients with SELENON-RM. These studies showed that scoliosis and respiratory management, body mass abnormalities (both cachexia and obesity) and the specific type of the SELENON gene variant are the main prognostic determinants for disease progression. Further, they showed a progressive decline in functional abilities and respiratory function [1, 11]. However, both studies had major limitations, including their retrospective design, and the absence of functional measurements, muscle imaging techniques (i.e. muscle ultrasound or MRI), bone quality assessment and no standardized cardiorespiratory follow-up. Cross-sectional and prospective natural history studies including a large subset of clinical, functional, and imaging outcome measures are not available. The current cross-sectional study on SELENON-RM fills in this gap [12]. Our goal is to identify all Dutch and Dutch-speaking Belgian patients, and provide deep clinical phenotyping of our baseline cohort for prospective follow-up and recruitment for future clinical trials. We want to contribute to optimal clinical management for SELENON-RM patients across the lifespan by providing recommendations for clinical care. Further, based on the findings from our study we will propose outcome measures that can be used for measuring disease severity and possibly disease progression in SELENON-RM patients for future natural history studies and clinical trials.
MATERIALS AND METHODS
Study design
This cross-sectional study in adult and pediatric patients with SELENON-RM is the start of the 1.5-year natural history study, the LAST STRONG Study: a study on LAMA2-MD and SELENON-RM To Study Trial Readiness, Outcome measures and Natural history [12]. The methods of the cross-sectional study are similar to those in the manuscript on LAMA2-MD [12, 13]. The study has been approved by the medical ethical reviewing committee Region Arnhem –Nijmegen (NL64269.091.17; 2017-3911) and is registered at ClinicalTrials.gov (NCT04478981).
Study population
Patients were recruited non-selectively and consecutively from September 2020 to March 2021. All patients known at our neuromuscular center were personally informed of the study. Patients were further recruited through contacting all Dutch and Dutch-speaking Belgian (pediatric) neurologists, rehabilitation specialists and clinical geneticists and through promotion of our study by patient organizations. Inclusion criteria include a genetic confirmation of SELENON-RM by two recessive (likely) pathologic variants in the SELENON gene, or typical clinical and histological alterations combined with genetic confirmation in a first degree relative. Exclusion criteria were an insufficient understanding of the Dutch language or the unwillingness to provide informed consent. The subjects’ consent was obtained according to the Declaration of Helsinki. If patients did not wish or were not able to visit our center, they were offered to participate in this study through home visits.
Neurological examination and functional measurements
Patients underwent a standard neurological examination, hand-held dynamometry, goniometry, Motor Function Measurement 20/32 (MFM-20/32), Hammersmith Functional Motor Scale (HFMS), Pediatric Balance Scale (PBS), Mini Balance Evaluation Systems Test (MiniBEST), and graded and timed function tests as far as these were appropriate for their age and functional abilities [12, 4–21]. An accelerometer (GENEActiv Original, Activinsights Ltd, 87.5 Hz sampling) attached to the non-dominant hand measured in all patients the number of counts per day and the percentage of sedentary, light, moderate and vigorous activity in the home situation for seven days [12, 22–24].
Questionnaires
Patients and/or their parent(s) were asked to complete age-adapted questionnaires, including Pediatric Quality of Life Inventory (PedsQL, Generic Core Scale, Neuromuscular Module (NMM) and Multidimensional Fatigue Scale (MFS), age 2 to 18 years), Research and Development-36 (RAND36, age ≥18 years), Individualized Neuromuscular Quality of Life (INQoL, age ≥18 years), McGill pain questionnaire (age ≥12 years), Wong-Baker Faces Pain rating scale (age ≥2 years), Checklist Individual Strength (CIS, age ≥18 years), ACTIVLIM (age ≥12 years), Impact on Participation and Autonomy (IPA, age ≥18 years), and Egen Klassifikation version 2 (EK2, age ≥18 years) and were compared to normative values [12, 24–38].
Ancillary investigations
Muscle thickness and muscle echogenicity with quantitative grayscale analysis and visual grading using the Heckmatt ultrasound score of a subset of bilateral muscles were assessed by ultrasound using an Esaote MyLabTwice ultrasound scanner (Esaote SpA, Genoa, Italy) with an 3–13 MHz broadband linear transducer and a 53-mm footprint in all ages [39, 40]. Respiratory function was assessed through spirometry in the upright and supine position, and the maximum expiratory pressure (MEP), maximum inspiratory pressure (MIP) and sniff nasal inspiratory pressure (SNIP) in the upright position in all patients of five years or older [41]. Diaphragm ultrasound was performed to quantify diaphragm function through assessment of diaphragm thickness and thickening in all ages [42]. Further, cardiac assessment was done by electrocardiography and conventional transthoracic echocardiography (TTE) with speckle tracking and tissue doppler imaging (TDI) in all ages [43]. Fragmented QRS complex was defined by the presence of an additional R-wave (R′) or notching in nadir of the R-wave or the S-wave, or the presence of more than one R′ without a typical bundle branch block [44]. Left ventricular ejection fraction (LVEF) was considered as decreased if <52% in males, <54% in females and <55% in the pediatric population [45, 46]. Global longitudinal strain was considered abnormal if above –18% in adults and above –19.5% in children [47, 48]. In order to assess bone quality, a dual-energy X-ray absorptiometry (DEXA-)scan of the right femoral neck and lumbar spine, including a vertebral fracture assessment, was performed in all ages [49].
Statistical methods
Descriptive statistics were used to summarize our data in IBM SPSS Statistics 25.0.0.1 for Windows (SPSS, Inc., Chicago, IL). Values are mean±SD, unless otherwise stated. Pearson’s correlation was used to assess correlations [50]. A Pearson’s correlation was considered moderate (r = 0.40–0.59), strong (r = 0.60–0.80), or very strong (r = 0.80–1.0) [50]. A paired sample t-test was used to compare the mean echogenicity of muscles on the right and left side. An independent sample t-test was performed to compare the echogenicity between ambulant and non-ambulant patients. A non-parametric test for independent samples was used to compare the Heckmatt score between ambulant and non-ambulant participants. If reference values are available from literature, overlapping confidence intervals were checked. Differences were considered significant if p < 0.05. In case of missing data, patients were excluded from this specific sub analysis.
Data availability statement
The data that support the findings of this study are available within the article and supplementary material, and from the corresponding author, upon reasonable request.
RESULTS
Study population
We were able to contact 15 patients, of whom 11 patients were included (19.7±12.9 years(range: 3–42 years), F = 3, M = 8). Three patients did not want to participate in scientific research and one patient denied to participate for COVID-19-related reasons. Nine patients were of Dutch origin, two patients were from the Dutch-speaking part of Belgium (Flanders). One patient participated in this study through home visits only, and thus underwent solely part of the protocol (Supplementary Table 1). Key clinical characteristics are shown in Table 1.
Table 1
Clinical and genetic features of SELENON-RM patients
| ID | Sex | Age (years) | Genetic mutation | MFM-20/32 score | Ambulant (yes/no) | Respiratory support (yes/no) (FEV1%, dVC%) | Cardiac function (LVEF*, GLS**); ECG*** | Bone quality (L; F; #) | |||
| Variant DNA (protein) | Variant type | ACMG classification | Ref | ||||||||
| 1 | M | 16 | 1. c.713dup p.(Asn238fs) | 1. Frameshift | 1. Pathogenic | [2, 51] | 55.2% | No (only with walking aid) | Yes (33%, –35%) | LVEF: 58%; QRS fragmentation, iRBBB | F: –3.7 |
| 2. c.1332_1334del p.(Asn444del) | 2. In-frame deletion | 2. Pathogenic | |||||||||
| 2 | F | 25 | c.943G>A p.(Gly315Ser) (homozygous) | Missense | Pathogenic | [2, 11, 52] | 77.1% | Yes | Yes (37%, –38%) | LVEF: 66%; GLS –19.3%; | F: –1.7 |
| 3 | M | 3 | c.943G>A p.(Gly315Ser) (homozygous) | Missense | Pathogenic | [2, 11, 52] | 81.7% | Yes | No | LVEF: 66%; QRS fragmentation | L: –1.9# |
| 4 | M | 21 | 1. c.943G>A p.(Gly315Ser)2. c.1332_1334del p.(Asn444del) | 1. Missense2. in-frame deletion | 1. Pathogenic2. Pathogenic | [2, 11, 52] | 83.3% | Yes | No (41%, –30%) | LVEF: 54%; GLS: –17.3%; QRS fragmentation | L: –1.2F: –2.8# |
| 5 | F | 8 | c.1A>G (p.Met1?) (homozygous) | Loss-of-function in start codon | Pathogenic | [2] | 91.7% | Yes | Yes (72%, –5%) | LVEF: 69%; GLS: –23.2%; | L: 0.7F: –0.4 |
| 6 | M | 42 | c.943G>A p.(Gly315Ser) (homozygous) | Missense | Pathogenic | [52] | 76.0% | Yes | Yes (32%, –47%) | LVEF: 60%; GLS: –15.8%; QRS fragmentation; right heart axis | F: –3.0# |
| 7 | F | 30 | 1. c.713dup p.(Asn238fs) | 1. Frame-shift | 1. Pathogenic | [2, 51] | 43.8% | No | Yes (23%, –43%) | LVEF: 58%; GLS: –17.0%; QRS fragmentation; iRBBB | F: –2.8# |
| 2. c.1332_1334del p.(Asn444del) | 2. In-frame deletion | 2. Pathogenic | |||||||||
| 8 | M | 9 | 1. c.943G>A p.(Gly315Ser)2. c.301G>C r.(spl?) | 1. Missense mutation2. Splice site | 1. Pathogenic2. Likely pathogenic | [2, 11, 52] | 77.1% | Yes | Yes (58%, 3%) | LVEF: 57%; GLS: –24.3%; QRS fragmentation | L: 0.9 F: –2.4 |
| 9 | M | 13 | 1. c.713dup p.(Asn238fs) | 1. Frameshift | 1. Pathogenic | [2, 53] | 72.9% | Yes | Yes (42%, –10%) | LVEF: 58%; GLS: –27.6%; QRS fragmentation | L: –0.5 F: –3.5 |
| 2. c.13_22dup p.(Gln8fs) | 1. Frameshift | 2. Pathogenic | |||||||||
| 10 | M | 11 | c.943G>A p.(Gly315Ser) (homozygous) | Missense | Pathogenic | [2, 11, 52] | 75.0% | Yes | No (36%, –24%) | QRS fragmentation | L: –1.4 F: –2.3 |
| 11 | M | 39 | c.943G>A p.(Gly315Ser) (homozygous) | Missense | Pathogenic | [2, 11, 52] | 49.0% | Yes | Yes | – | # |
| Mean | M, n = 9 | 19.7±12.9 [3 to 42] | – | – | – | – | 71.2±15.1% [43.8 to 91.7] | Yes, n = 9 | Yes, n = 8/11FEV1% : 42±15% [23 to 72]dVC: –26±18% [–47.1 to 3.0] | LVEF: 61±5% [54 to 69]; normal in all.GLS: –20.6±4.4% [–27.6 to –17.0]; abnormal, n = 3 | Adults: F: –2.6±0.6 [–3.0 to –1.7] L: –1.2Children: F: –2.5±1.3 [–3.7 to –0.4] L: –0.4±1.2 [–1.9 to 0.9] |
* Decreased LVEF = <52% in males,<54% in females and <55% in the pediatric population; ** abnormal GLS = above –18% in adults and above –19.5% in children; *** QRS fragmentation = the presence of an additional R-wave (R′) or notching in nadir of the R-wave or the S-wave, or the presence of more than one R′ without a typical bundle branch block. N = number of patients; ACMG = American College of Medical Genetics; MFM-20/32 = Motor Function Measurement 20/32; FEV1% = percentage of predicted forced expiratory volume in the first second; dVC (sitting –supine) = difference in vital capacity between the sitting and the supine position; LVEF = left ventricular ejection fraction; GLS = global longitudinal strain; ECG = electrocardiogram; (i)RBBB = (incomplete) right bundle branch block; L = z-score or t-score of lumbar spine on DEXA-scan; F = z-score or t-score of femoral neck on DEXA-scan; # = (fragility) LBF(s) in medical history; ambulant is defined as the ability to walk 10 meter unaided.
4.2Genetic characteristics
All patients had genetic confirmation of SELENON-RM before enrollment in the study. The variant c.943G>A (p.(Gly315Ser)) (compound homozygous and heterozygous) was most frequently found. Other genetic information is shown in Table 1 [2, 51–53].
Neurological examination
Facial muscle weakness and symmetrical skeletal muscle weakness with a proximal to distal gradient were present in all patients (Table 1, Figs. 1A-E and 2). Contractures were seen in most of the patients, with elbow extension, knee extension and ankle dorsiflexion contractures being the most prevalent. Distal joint hypermobility was found in six patients. Rigid spine was seen in all patients (Fig. 1F). Eight participants had a high arched palate. One patient showed pseudohypertrophy of the calf muscles, while muscle atrophy was more frequently seen in others (Fig. 1G-H). Nine participants had scoliosis, of whom four had undergone spondylodesis to correct spinal deformity. Scapular dyskinesia (including scapular winging) was found in six patients (Fig. 1I).
Fig. 1
Facial muscle weakness and dysmorphic features in SELENON-RM patients. (A) neutral position, showing mild bilateral ptosis. (B) m. frontalis weakness. (C) m. orbicularis oculi weakness. (D) m. risorius weakness. (E) inadequate puckering movements, indicating m. orbicularis ori weakness. (F) inability of neck flexion and lumbar flexion, indicating rigid spine. (G) pseudohypertrophy of the calve musculature. (H) general muscle hypotrophy, most pronounced in the calves and upper legs, and spinal deformity (status after osteosynthesis). (I) scapular winging. Permission for publication of the photographs from all identifiable patients was granted.
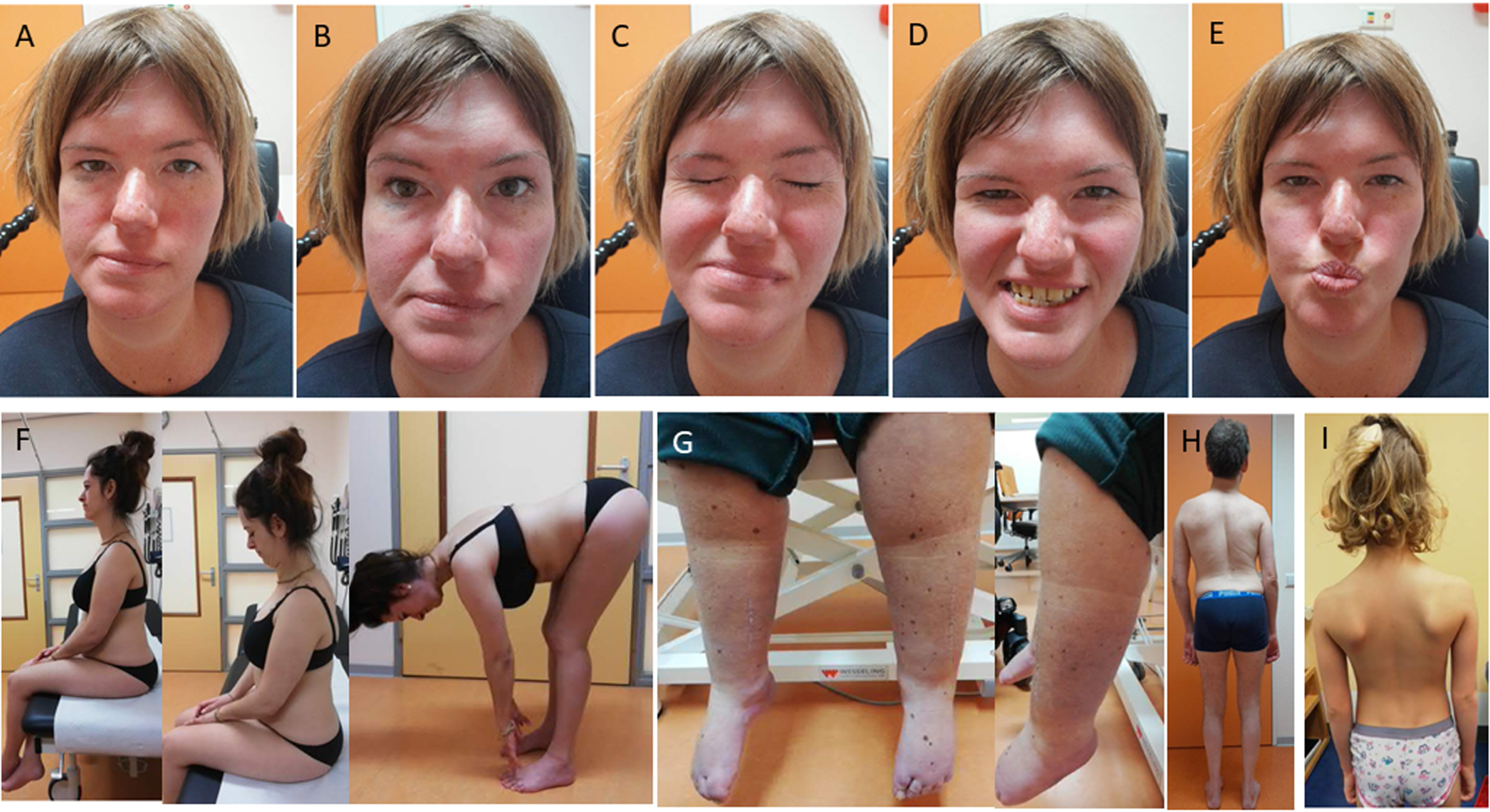
Fig.2
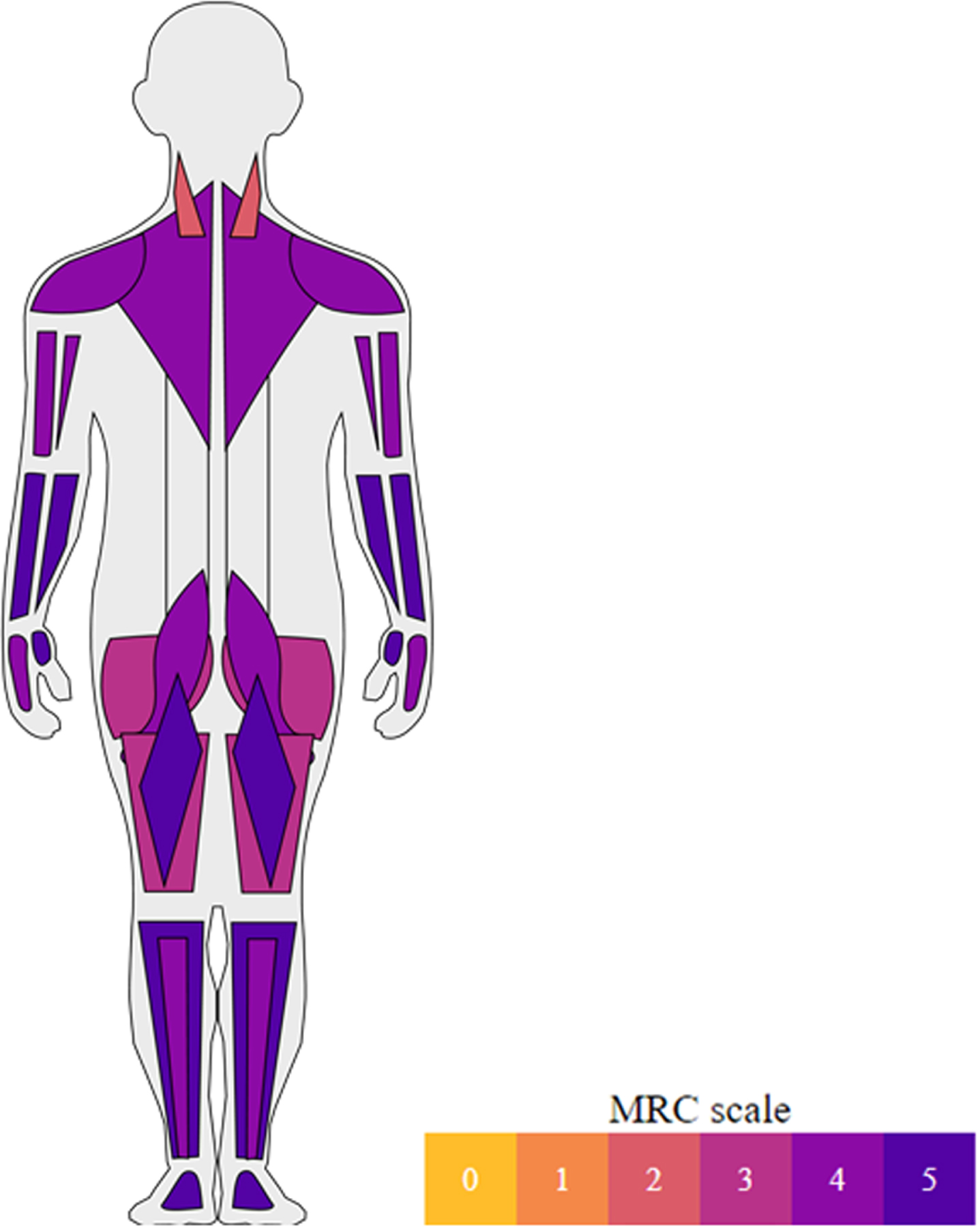
Median muscle strength according to the MRC grading scale. Median muscle strength (MRC) in neck flexor, neck extensor, deltoid, biceps brachii, triceps brachii, wrist flexor, wrist extensor, finger flexor, finger extensor, iliopsoas, gluteus, quadriceps, hamstrings, foot flexor and foot extensor muscles.
Accelerometry
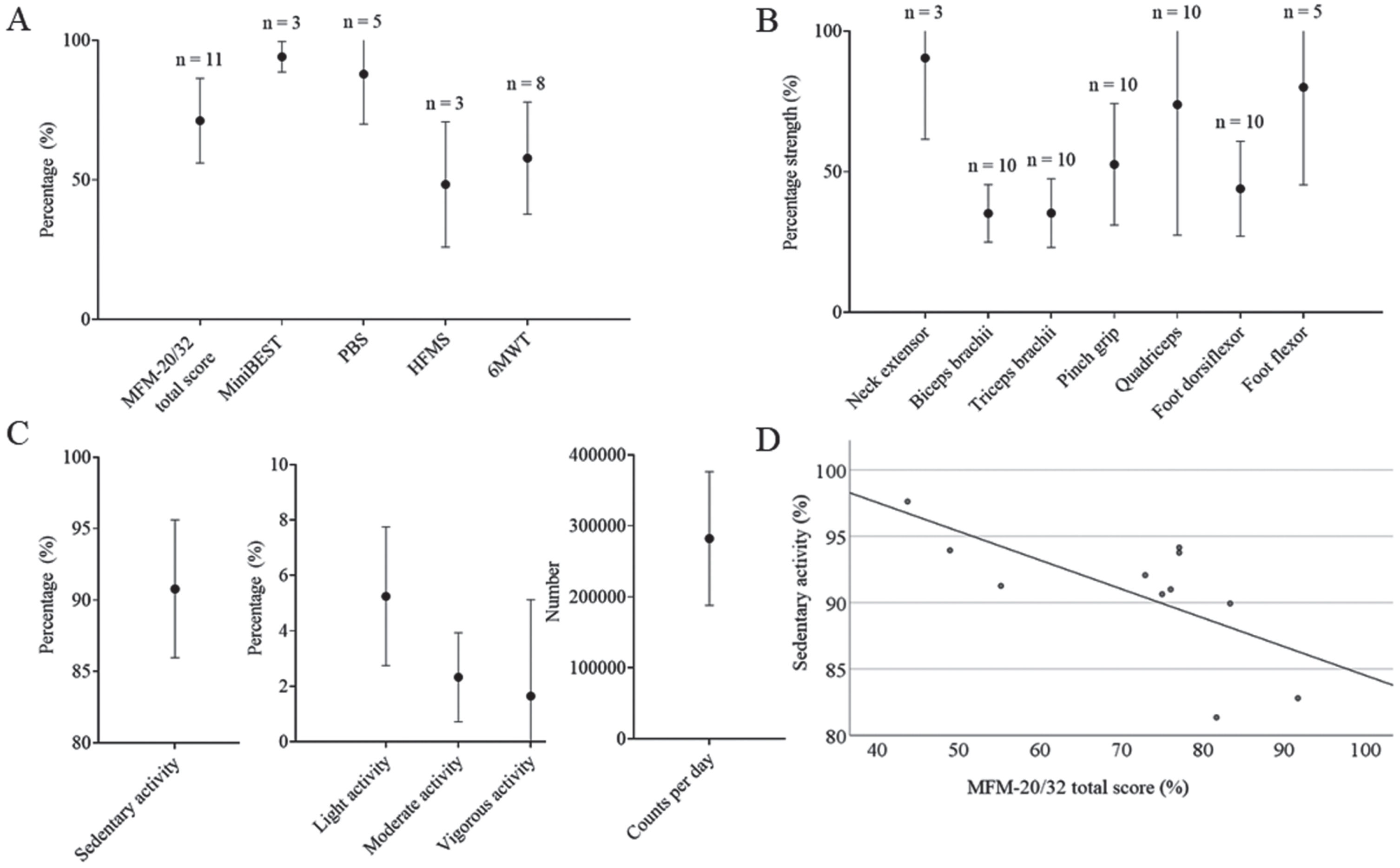
Accelerometry was performed in all patients. There was a strong correlation between the MFM-20/32 total score (primary outcome measure) and the percentage of time spent on sedentary activity (Pearson’s correlation, –0.682, p < 0.05) and light activity (Pearson’s correlation, 0.754, p < 0.01) (Fig. 3). Next to that, MFM domain 1 and domain 2 were strongly correlated with sedentary (Pearson’s, –0.654, p < 0.05 and –0.746, p < 0.01, respectively) and light (Pearson’s, 0.745 and 0.740, p < 0.01, respectively) activity.
Fig. 3
Overview on percentage of MFM-20/32 total score, MiniBEST, PBS and HFMS; the percentage of distance predicted at the 6MWT; the percentage of predicted strength (N) of several muscles; the percentage of sedentary, light, moderate and vigorous activity and the number of counts per day as measured through accelerometry (mean±SD); and the correlation between the MFM-20/32 total score and percentage of sedentary activity. (A) Percentage of MFM-20/32 total score, MiniBEST, PBS and HFMS; and the percentage of distance predicted at the 6MWT. MFM-20/32 was performed by all patients participating in this study. The MiniBEST and PBS were performed by ambulant patients, while the HFMS was only performed by patients who were not able to perform either of the two balance tests (MiniBEST or PBS) due to physical limitations (n = 3). 6MWT included all ambulant patients of 5 years and older that were seen in the hospital (n = 8). (B) Percentage of predicted strength (N) of the neck extensor, biceps brachii, triceps brachii, quadriceps, foot dorsiflexor and foot flexor muscles and the pinch grip. n = the number of patients (each patient having two muscles examined, i.e. left and right side); (C) Percentage of sedentary, light, moderate and vigorous activity and the number of counts per day as measured through accelerometry. (D) Percentage sedentary activity as measured through accelerometry was negatively correlated to the MFM-20/32 total score (Pearson’s correlation, –0.682, p < 0.05). MFM-20/32 = motor function measurement 20/32; MiniBEST = Mini Balance Evaluation Systems Test; PBS = Pediatric Balance Scale; HFMS = Hammersmith Functional Motor Scale; 6MWT = 6-minute walk test. All values are mean±SD.
Functional measurements
A detailed overview on functional measurement scores can be found in Fig. 3.
4.5.1Motor Function Measurement 20/32 (MFM-20/32)
MFM-20/32 was performed in all patients. The mean MFM-20/32 total score was 71.2±15.1% (Fig. 3). Domain 1 (standing and transfers, 50.4±26.6) was most severely affected, followed by domain 2 (proximal muscle function, 79.8±13.0%). Domain 3 (distal muscle function, 94.6±4.5%) was unaffected in the majority of the patients.
Hammersmith Functional Motor Scale (HFMS)
The HFMS was performed in the three out of 11 patients that were non-ambulant or that could not perform the balance test (Fig. 3). None of these patients could lift their head from supine position, prop on extended arms, come from supine to a sitting position, perform a four-point kneeling or crawl. Sitting on a plinth or chair and supported standing were not affected in any of them.
Mini Balance Evaluation Systems Test (MiniBEST)
Three adult patients were able and felt comfortable to perform the tasks of the MiniBEST (Fig. 3). All subscales, including anticipatory movements, reactive postural control, sensory orientation, and dynamic gait were only minimally affected. The remaining adult patients were not ambulant or had a high fall risk, whereby they could not perform balance tests.
Pediatric Balance scale (PBS)
Five children were able and felt comfortable to perform the PBS (Fig. 3). Two children achieved the maximum score. One young child (M, age 3 years) had difficulties with following the instructions. The subdomains “turning the head and trunk to look behind left and right shoulders while standing still” and “reaching forward with outstretched arm while standing” were most severely affected.
Graded and timed function tests
The Timed Up and Go (TUG) tests with (15.0±7.8 s) and without (13.2±7.09 s) a cognitive task was performed by nine out of 11 patients, with a delay of 10% or more in three patients in the cognitive task version. Five patients were able to perform the 30 seconds sit-to-stand test, with a mean number of eight stands (range: one to 16). Eight patients were able to independently raise from the ground (12.9±5.5 s), of whom three needed a nearby object for support, four only needed the support of their hands on their upper legs, and one patient only used her hands for support on the ground. The 6-minute walk test (6MWT), and climbing and descending a standardized physiotherapy stairs were not performed in one patient due to his participation through home visits. Two out of 10 patients were unable to do the 6MWT: one patient was too young and one was not ambulant. One patient (M, 16 years), defined as not ambulant since he could not walk unaided, was nevertheless able to perform the 6MWT using a walking aid (walking frame with wheels combined with an ankle-foot orthosis on the right side). The mean percentage of the predicted walked distance was 57.8±20.0% (Fig. 3). Only one participant was able to walk a distance above the lower limit of normal (523 meter, F, 8 years). Three out of 10 patients were able to climb four steps (5.8±5.8 s) and four out of 10 patients were able to descend four steps (3.8±4.5 s) on a standardized physiotherapy stairs with alternating steps and without the use of their hands. One patient could not climb or descend the standardized physiotherapy stairs at all. The remaining patients either used their hands or could not perform alternating steps.
Hand-held dynamometry
The neck flexor could not be measured with dynamometry in any of the patients due to severe neck flexor weakness (median MRC 2). The neck extensor muscles could be measured in six patients who had a MRC score ≥4. All other muscle groups could be measured in all patients of five years and older, of which the foot flexor muscles (80.0±34.7%) and quadriceps muscles (73,8±46,4%) had the highest strength percentage of predicted value (Fig. 3).
Questionnaires
A detailed overview on the questionnaires can be found in Supplementary Table 2.
RAND36
Five, adult patients filled in the RAND36 questionnaire. The subdomains scores for “physical functioning”, “role limitations due to physical health”, “vitality” and “general health” were decreased compared to healthy controls.
Individualized Neuromuscular Quality of Life (INQoL)
Five, adult patients filled in the INQoL. The subdomain “muscle weakness” was found to be most severely affected (74.7±8.6%). Patients experienced minimal problems of muscle locking (4.2±9.4%) and social relationships (14.6±14.4%). The mean Quality of Life score was 42,8±11,4%.
Checklist Individual Strength (CIS)
Four out of five patients reported to have a problematic fatigue (>76 of 140 points), i.e. a combination of subjective fatigue, reduction in motivation, reduction in activity and reduction in concentration that negatively influences the person’s performance in the occupational and home setting. The subjective fatigue subdomain was increased in all patients (≥27 of 56 points), of whom two reported severe subjective fatigue (>35 of 56 points).
Pediatric Quality of Life Inventory (PedsQL)
Six patients and/or their parents filled in the PedsQL questionnaires. One patient was too young to fill in this questionnaire (3 years), so only the proxy forms were filled in by his parents. The total score on the PedsQL Generic Core and PedsQL MFS was significantly decreased compared to healthy controls. PedsQL NMM scores were in line with SMA patients [25].
ACTIVLIM
Ten patients filled in the ACTIVLIM questionnaire. A mean of 2.6±3.5 activities were impossible to perform, 5.4±3.8 activities were difficult to perform, and 9.9±5.0 activities were easy to perform. Four patients reported that they could perform all tasks. The median total score was 28.5 out of 36.
Impact on Participation and Autonomy (IPA)
Five patients filled in the IPA questionnaire. The subdomain “autonomy outdoors” was found to be most severely affected. Generally, two out of five patients experienced major problems and three out of five patients experienced minor problems in participation and autonomy.
Egen klassification scale 2 (EK2)
Seven patients filled in the EK2 questionnaire. The mean EK2, a scale to measure physical function over time, was 7.7±7.7 out of 51 points. A score of zero indicates the highest physical function and a score of 51 the lowest.
McGill pain questionnaire
Six out of seven patients indicated that they might experience some sort of recurrent pain on a normal day with normal activities, with pain in the back and/or neck or shoulder region being the most outstanding. Two patients indicated that they could not sleep due to the pain, that they woke up in the morning with pain, and that it hinders them with normal movements and daily activities. Only one patient indicated that she had to rest because of the pain, that it hindered her leisure activities, that she felt listless and that she had to use analgesics.
Wong-Baker faces Pain rating scale
Five out of 11 patients indicated to have not had any pain or only minimal pain at the moment of examination or during the previous week. Four patients experienced pain every day in the week prior to the examinations. Three patients indicated to have had extreme pain (numerical rating scale ≥8/10) in the back and/or shoulder region in the week prior to the examinations.
Muscle ultrasound
Echogenicity and Heckmatt score
Muscle ultrasound was performed in all patients. An overview on the echogenicity (z-score) and Heckmatt score per muscle can be found in Fig. 4 and Table 2. We found no significant difference in echogenicity and Heckmatt score between left and right sided muscles. The echogenicity was most severely increased in the sternocleidomastoid and the biceps brachii. The soleus and temporalis were found to be least affected. We found a correlation between the echogenicity and the Heckmatt score of a subset of muscles and the MFM-20/32 total score, MRC score and age. Further, non-ambulant patients had an increased echogenicity and Heckmatt score in the flexor carpi radialis compared to ambulant patients.
Fig. 4
Overview on echogenicity (z-score and Heckmatt score) per muscle. The z-score of the echogenicity (A) and the Heckmatt score (B) in the temporalis, sternocleidomastoid, biceps brachii, flexor carpi radialis, rectus abdominis, rectus femoris, vastus lateralis, tibialis anterior, biceps femoris, gastrocnemius medial head and soleus muscles are shown.
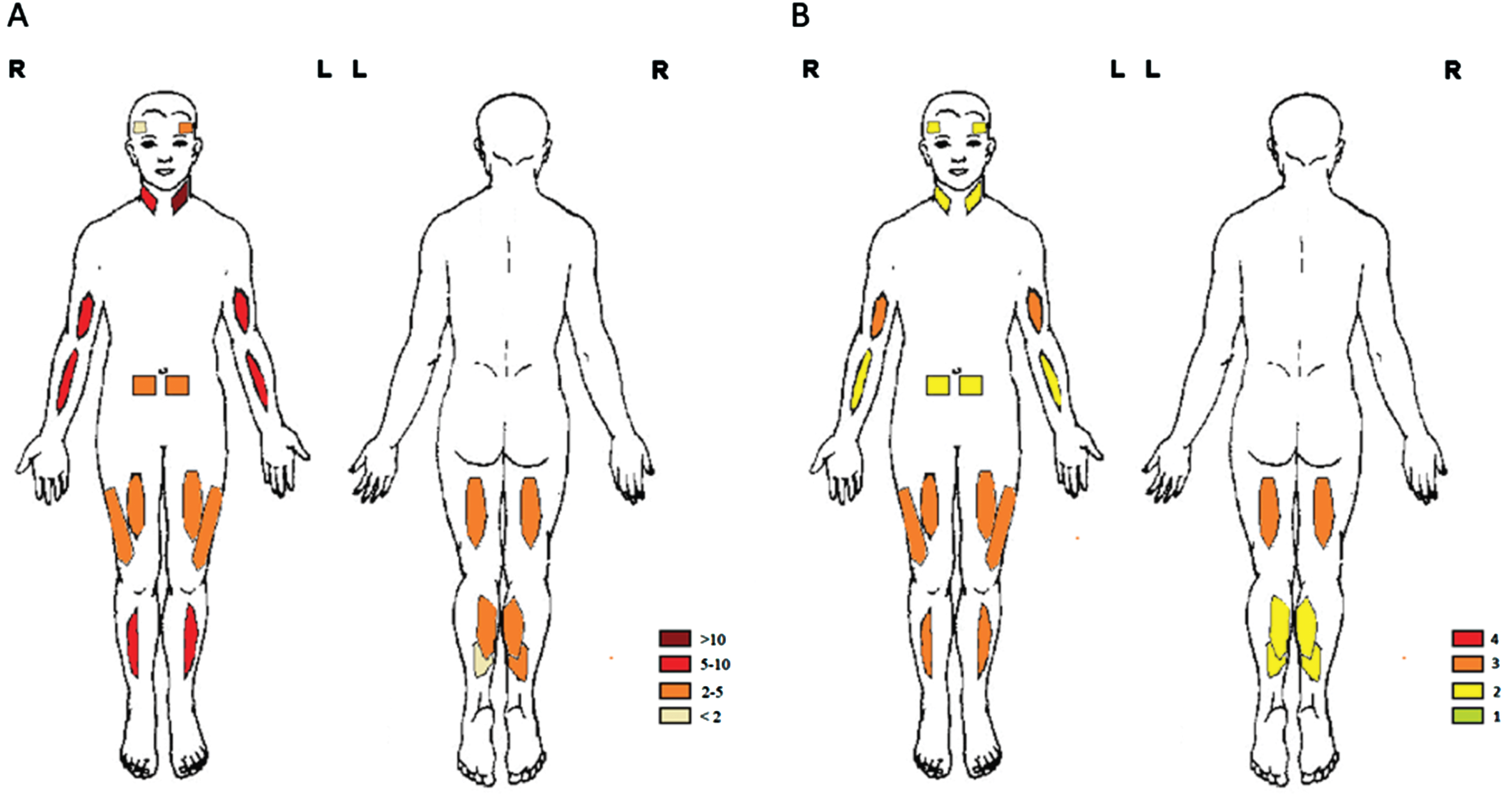
Table 2
Muscle ultrasound in SELENON-RM patients
| Muscle | Echogenicity z-score | Heckmatt score | Muscle thickness |
| (mean) | (median [IQR]) | z-score (mean) | |
| Biceps brachii | 7.06 | 3 [2.25–4.0] | –0.08 |
| Biceps femoris | 2.38 | 3 [3–3.75] | 0.33 |
| Flexor carpi radialis | 5.70 | 2 [2 – 3] | –1.25 |
| Gastrocnemius | 2.59 | 2 [2 – 2] | 0.17 |
| Rectus abdominis | 3.69 | 2 [2 – 3] | –0.81 |
| Rectus femoris | 3.96 | 3 [2 – 3] | –1.09 |
| Soleus | 1.93 | 2 [2 – 2] | –0.51 |
| Sternocleidomastoid | 10.07 | 2 [2–2.75] | –1.58 |
| Temporalis | 2.16 | 2 [1 – 2] | 0.15 |
| Tibialis anterior | 5.24 | 3 [3 – 3] | –0.81 |
| Vastus lateralis | 4.82 | 3 [3 – 3] | –0.75 |
| Correlation (Pearson’s correlation) | |||
| MFM-20/32 total score | Echogenicity | Heckmatt score | |
| Flexor carpi radialis, –0.750, p < 0.01 | Biceps brachii, –0.576, p < 0.01 | ||
| Flexor carpi radialis, –0.874, p < 0.01 | |||
| MRC | Echogenicity | Hackmatt score | |
| Biceps brachii left, –0.897, p < 0.01 | Biceps brachii left, –0.802, p < 0.01 | ||
| Biceps brachii right, –0.668, p < 0.05 | |||
| Soleus right, 0.688, p < 0.05 | |||
| Age | Echogenicity | Heckmatt score | |
| Rectus abdominis, –0.574, p < 0.01 | Biceps brachii, 0.567, p < 0.01 | ||
| Soleus, –0.594, p < 0.01 | Biceps femoris, 0.720, p < 0.01 | ||
| Sternocleidomastoid muscle, 0.581, p < 0.01 | |||
| Muscle thickness | Echogenicity | Heckmatt score | |
| Gastrocnemius, –0.480, p < 0.05 | Gastrocnemius, –0.445, p < 0.05 | ||
| Sternocleidomastoid, –0.464, p < 0.05 | Sternocleidomastoid, –0.665, p < 0.01. | ||
| Temporalis, –0.601, p < 0.01 | |||
| Muscle | Echogenicity - | Echogenicity - Non- | Echogenicity - |
| Ambulant (mean) | ambulant (mean) | Significance (2-tailed) | |
| Flexor carpi radialis | 4.79 | 9.30 | p < 0.05 |
IQR = interquartile range; MFM-20/32 = Motor Function Measurement 20/32; MRC = Medical Research Council.
Fig. 5
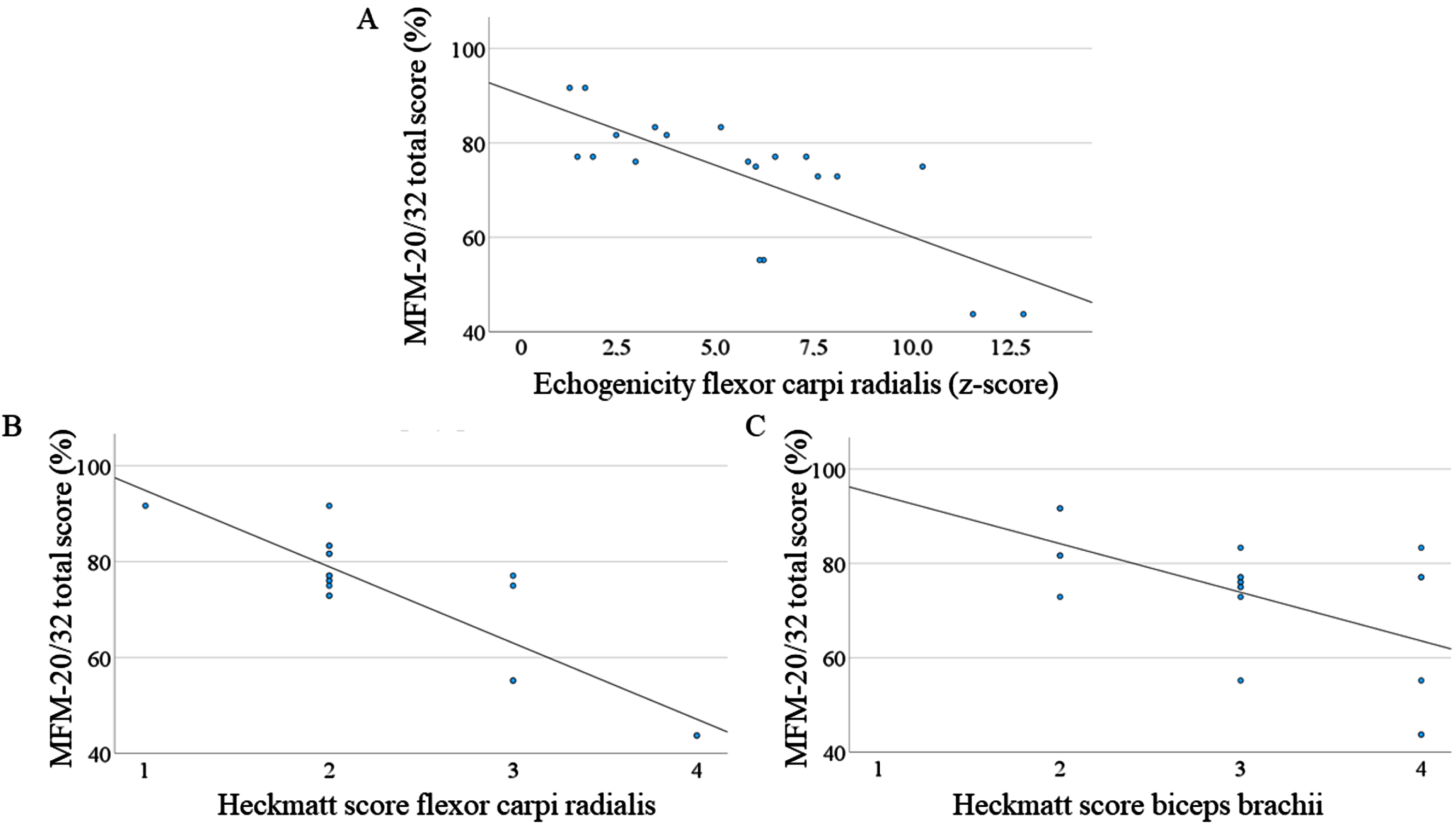
Correlation between MFM-20/32 total score and muscle ultrasound (echogenicity and Heckmatt score). (A) Correlation between the z-score of echogenicity of flexor carpi radialis muscles and the total MFM-20/32 score (Pearson’s correlation, –0.750, p < 0.01). (B) Correlation between Heckmatt score of flexor carpi radialis muscles and the total MFM-20/32 score (Pearson’s correlation, –0.874, p < 0.01). (C) Correlation between Heckmatt score of biceps brachii muscles and the total MFM-20/32 score (Pearson’s correlation, –0.576, p < 0.01). MFM-20/32 = Motor Function Measurement 20/32.
Muscle thickness
We found a significant difference in muscle thickness between the left and right side for the temporalis (p < 0.05), while there was no difference in muscle thickness for all other studied muscles.
Respiratory function tests
Spirometry was performed in nine patients (Table 3). All patients had a decreased FEV1% (41.6±14.8%) in the upright position. The mean decrease in vital capacity (VC) between the upright and supine position was 25.5±17.7%. The MIP and MEP was abnormal in all patients (44.8±19.7% and 48.4±6.3% of predicted, respectively) (Table 3). Eight out of 11 patients used (nocturnal) non-invasive ventilation, which was started in all at a pediatric age. Only one adult patient (M, age 21) was not in need of (nocturnal) non-invasive ventilation.
Table 3
Respiratory function tests in SELENON-RM
| Respiratory function tests (mean±SD (range)) | |
| FEV1 (percentage predicted) | 41.6±14.8% (23 to 72) |
| FVC (percentage predicted) | 38.2±13.7% (21 to 67) |
| PEF | 192±53 L/min (143 to 291) |
| PCF | 190±56 L/min (124 to 296) |
| dVC sitting –supine | 25.5±17.7% (–47.1 to 3.0) |
| MIP (percentage predicted) | 44.8±19.7% (16.2 to 71.2) |
| MEP (percentage predicted) | 48.4±6.3% (35.8 to 56.8) |
| SNIP | 30.3±12.1 (13 to 43) (M); 37.7±17.2 (18 to 50) (F) mmH2O |
FEV1% = percentage predicted of the forced expiratory volume in the first second; FVC% = percentage predicted of the forced vital capacity; PEF = peak expiratory flow; PCF = peack cough flow; dVC (sitting –supine) = difference in vital capacity between the sitting and supine position; MIP = maximal inspiratory pressure; MEP = maximal expiratory pressure; SNIP = sniff nasal inspiratory pressure.
Diaphragm ultrasound
Ultrasound of the diaphragm was performed in 10 patients. In all these patients, the diaphragm was nondetectable at the end of expiration, representing severe diaphragm atrophy. Furthermore, there was no visible increase in diaphragm thickness during inspiration. The latter was accompanied by a paradoxical breathing pattern.
Cardiac assessment
ECG and echocardiography were performed in 10 patients. QRS fragmentation was seen in eight patients.An incomplete right bundle branch block was seen in two patients (age 16 and 30 years), in the absence of arrhythmia, and did not require any treatment. Five out of 10 patients showed sinus arrythmia, while no other arrhythmias were observed in our cohort. Echocardiography showed abnormal global longitudinal strain in three out of seven patients (adult: –17.3%, –15.8% and –17.0%). Left ventricular ejection fraction (LVEF) was normal in all patients, and none of the patients used cardiac medication. No abnormalities of the valves, right ventricle volume/function or atria were seen in any of the patients.
Bone quality
A DEXA-scan was performed in 10 participants. The mineral bone density of the femoral neck was reduced in three out of four pediatric patients (z-score -2.5±1.3 [-3.7 to -0.4]) and in all adults (t-score 2.6±0.6 [-3.0 to -1.7]). Measurement of the mineral bone density of the lumbar spine could be performed in only five pediatric patients (z-score -0.4±1.2 [-1.9 to 0.9]) and one adult patient (t-score –1.2) due to the presence of spondylodesis material. Six participants had experienced one or more fragility fractures of the long bones. The number of retrospective fractures was strongly correlated to the lumbar bone mineral density (–0.926, p < 0.01), but not to the femoral bone density or ambulation status. No other causes for decreased bone quality (i.e. hormonal abnormalities, malnutrition, medication related) could be identified. One patient had received treatment with bisphosphonates prior to participation in this study.
DISCUSSION
We here present a cross-sectional study of characteristics of SELENON-RM patients, showing 1) axial and limb muscle weakness with a proximal to distal gradient, 2) joint contractures and distal joint hypermobility, 3) increased echogenicity on muscle ultrasound reflecting fatty replacement and/or fibrosis, 4) respiratory impairment with severe diaphragm atrophy and dysfunction, requiring non-invasive ventilation in the majority of the patients, 5) decreased bone quality, 6) problematic fatigue; and 7) pain, mostly localized in the back, neck or shoulder region. LVEF was normal in all patients, yet QRS fragmentation and abnormal global longitudinal strain, which both are well-known predictors of worse clinical outcome in other neuromuscular diseases, were frequently present [44, 54–56]. Based on these cross-sectional cohort data, we have several recommendations for clinical practice and future research, which will be discussed below.
Optimizing clinical care for SELENON-RM patients
Based on the respiratory impairment with prominent diaphragm atrophy and dysfunction in all patients, we recommend minimally annual respiratory function tests in (a)symptomatic patients. This should be started in pre-schoolers to early detect impairment of respiratory function and to enable timely respiratory training, lung volume recruitment techniques and/or non-invasive ventilatory support. Due to the severe diaphragm dysfunction, we strongly advise to perform spirometry in both the upright and the supine position. In case of symptoms suggestive of nocturnal hypoventilation or abnormal spirometry we advise polysomnography. Our results and recommendations are in line with previous studies showing that most of the SELENON-RM patients, even while being ambulant, need nocturnal non-invasive ventilation due to early diaphragm dysfunction. These studies recommend yearly respiratory evaluations [1, 57]. The underlying pathophysiology of the prominent diaphragm dysfunction in SELENON-RM is unknown. It might be related to the impaired mitochondrial function and increased oxidative stress [58, 59]. Furthermore, peak cough flow was severely impaired in our cohort. Reduced cough capacity is a major risk factor for respiratory morbidity and mortality in neuromuscular diseases [60]. We therefore propose timely initiation of lung volume recruitment techniques such as air stacking [61].
QRS fragmentation and abnormal GLS are risk factors for impaired cardiovascular outcomes including decreased LVEF and ventricular arrythmias in neuromuscular disorders [44, 54–56]. Considering the prevalence of QRS fragmentation and abnormal GLS in our cohort, we therefore recommend cardiac surveillance by screening of (a)symptomatic patients every two years with ECG, Holter and echocardiography or in the presence of new cardiac symptoms. LVEF was normal in all SELENON-RM patients, which is in line with the results of our recent scoping review [62]. Villar-Quiles et al. recommended annual cardiac assessment due to the possibility of pulmonary hypertension or secondary right ventricle failure [1]. In our cohort, pulmonary hypertension, a complication of untreated respiratory impairment, was not observed. This can be explained by the optimal respiratory follow-up in our SELENON-RM cohort. We therefore suggest cardiac assessment every two years, provided that respiratory evaluations are performed yearly to early detect and treat impaired respiratory functions, and that clinical signs for pulmonary hypertension are absent.
In our study, we frequently observed decreased bone quality. Although our recent scoping review showed that decreased bone quality is prevalent in congenital myopathies, this is a new finding in SELENON-RM, and so far only general recommendations for congenital myopathies were previously reported [10, 63]. Decreased bone quality can be caused by a combination of physical inactivity, atrophied muscles, and nutritional issues and decreased outdoor time leading to deficiency of calcium and vitamin D [64–69]. Furthermore, there may also be a direct adverse effect of mitochondrial disfunction at the level of skeletal maturation and bone turnover [70]. Next, excessive intracellular reactive oxygen species levels, which might be caused by insufficient or absent selenoproteins, are thought to contribute to the development of osteoporosis by inhibiting osteoblastic differentiation of bone marrow stromal cells (BMSCs) [71]. Based on general guidelines for low bone quality, we advise appropriate vitamin D and calcium supplementation in all patients [10, 72]. Moreover, a DEXA-scan is recommended at diagnosis and subsequently every one to three years, depending on the bone mineral density, and in the presence of a new LBF or bone pain. In case of a fragility long bone fracture or vertebral fracture, or a z-score < –2 for the paediatric population or t-score < –1 for adults on the DEXA-scan, we advise to refer patients to paediatrics or internal medicine for personalized follow-up of bone quality. To this end, DEXA-scans, blood (calcium, phosphate, alkaline phosphatase, vitamin D) and urine (calcium and creatinine) tests, and consideration of additional therapies are indicated.
We observed a high prevalence of problematic fatigue in both adult and paediatric patients. Fatigue is a common feature in patients with neuromuscular disorders, and it has been previously reported to negatively influence physical endurance and motor performance in SELENON-RM [1, 6]. Various management interventions are likely to reduce experienced fatigue. Recommendations include physical therapy, optimization of cardiorespiratory function, treatment of pain and correction of risk factors (including obesity, poor nutrition and inactivity) [73, 74]. Further, self-management programs combining aerobic training, energy conservation management, and relapse prevention have been shown to improve social participation and functional endurance in patients with neuromuscular diseases and chronic fatigue [75, 76]. Moreover, cognitive behavioural therapy (CBT) can ameliorate experienced chronic fatigue in patients with neuromuscular diseases [77, 78].
Finally, patients frequently reported pain in the neck, shoulder and/or back region. Some patients even reported that normal movements and daily activities were impaired due to pain, and that they had experienced extreme pain (NRS ≥8/10) in the week prior to the investigations. Spinal deformities (scoliosis, hyperlordosis), spinal rigidity, and weak neck flexor and paraspinal muscles were considered as the major causes for the reported pain. In the two previous natural history studies, the presence of pain was unaddressed [1, 11]. Based on our results, we advise to pay special attention to pain management in SELENON-RM patients, to explore and treat its underlying causes, to provide optimal rehabilitative care including physiotherapist or occupational therapist, and to refer to a team specialized in pain management if needed.
Recommendations for research
Our cross-sectional study further provides insights into important clinical and functional outcome measures for assessing disease severity and progression of disease in future clinical trials. We show that MFM-20/32 is suitable (covering the clinical feature of interest) and feasible (being able to be performed by patients) for measuring disease severity in SELENON-RM across the life span. Measuring disease progression by long-term follow-up with MFM-20/32 has not yet been performed in SELENON-RM patients. However, it is responsive for measuring disease progression in other neuromuscular diseases [79–81]. We also found that the 6MWT could be performed by most of the patients, had large range in percentage predicted values, and had no flooring or ceiling effect. We thus expect that the 6MWT is of value to measure disease severity in ambulant SELENON-RM patients without significant cardiorespiratory comorbidities [82–84]. Further, the timed function test ‘rise from the floor’ had a wide range of values in the absence of a flooring or ceiling effect. Patients also indicated that they retrospectively experience increasing difficulties with rising from the ground. An annual decrease in the timed function test ‘rise from the floor’ was previously reported in SELENON-RM patients [11]. In Duchenne muscular dystrophy, the time to rise from the floor was relevant for predicting disease progression in the group above seven years old [85]. This timed function test might thus be of added value to measure disease severity and disease progression in patients with SELENON-RM. The usability of MFM-20/32, the 6MWT and the timed function test ‘rise from the floor’ for measuring disease progression in our cohort of SELENON-RM patients will be evaluated in the 1.5-year follow-up natural history study [12].
We conclude that the HFMS, the MiniBEST and the PBS are not suitable for assessing disease severity in SELENON-RM. The HFMS was performed in only a small subset of patients, with a limited score range, limiting us to asses disease severity. Further, all patients that were able and felt comfortable to perform the balance tests had a near normal score, leading to a ceiling effect.
Accelerometry had strong correlations with functional abilities measured through the MFM-20/32, which is in line with our results in the LAMA2-MD cohort [13]. We propose accelerometry to monitor physical activity, serve as a reliable and non-invasive method to estimate physical abilities, and guide the effect of rehabilitative care and future therapies in SELENON-RM [86–88].
Results of the muscle ultrasound provide new insights into the distribution of muscle involvement. The sternocleidomastoid and the biceps brachii muscles were most severely affected. Previous studies using MRI have shown severe inconsistencies in muscle involvement in SELENON-RM patients. For example, Hankiewicz et al. reported in a cohort of 9 patients (age range: 8 to 21 years) that the sternocleidomastoid muscle was not affected, while Tordjman et al. showed in a cohort of 8 patients (age range: 8 to 21) severe fatty infiltration [89, 90]. This stresses the need for deep clinical phenotyping using imaging modalities. The severe involvement of the biceps brachii muscle on muscle ultrasound is not in line with previous studies using MRI [89, 90]. This discrepancy between muscle ultrasound and MRI in the biceps brachii muscle has also been observed in other neuromuscular diseases [13, 91]. Muscles with an increased echogenicity and normal appearance on MRI are consistent with intramuscular fibrosis, especially in the absence of fatty replacement [91]. Our next step will include the assessment of muscle fattening on whole body MRI and correlate it to the findings on muscle ultrasound.
The results provide suggestions for patient-reported outcome measures: RAND36 to describe the quality of life, CIS to monitor fatigue and McGill pain questionnaire to assess pain in adult SELENON-RM patients. We advise PedsQL Generic Core scale, NMM and MFS (self-report and proxy-report) in the paediatric population.
RAND36 showed reduced physical functioning, role limitations due to physical health, vitality and general health compared to healthy controls, which is in line with observations in other neuromuscular disorders [92]. In line with the RAND36, the INQoL reported muscle weakness as the most prominent symptom and the CIS showed increased problematic fatigue. Since the CIS is more sensitive to measure fatigue and compromises several subdomains, we advise to use the CIS as a measure for fatigue in addition to the general RAND36 quality of life measurement [31]. Next to fatigue-related questionnaires, muscle fatigability can be measured through clinical and electrophysiological endurance tests, using isokinetic dynamometry combined with surface EMG [93]. Muscle fatigue can be an underlying cause for general fatigue and is another dimension to be assessed and treated in future clinical trials.
The majority of the patients indicated that they experience pain in the back, neck or shoulder region. The McGill pain questionnaire can be a good method to quantify and follow-up pain. Further, pain can negatively influence quality of life and fatigue in neuromuscular diseases, which are both decreased in SELENON-RM [94, 95]. We therefore suggest to examine the relation between pain, quality of life and fatigue in SELENON-RM and to investigate whether adequate pain treatment results in improvement of quality of life and fatigue.
Strength and limitations
11 SELENON-RM patients were included in our study, which is far less than the 132 patients from the cohort reported in Villar-Quiles et al. [1]. However, major strengths of our cross-sectional cohort study include the unselected cohort, the prospective design and the large set of examinations for selecting clinical, functional and imaging outcome measures. The study has frequent missing data that are inherent to the study population: some patients were too young to perform some of the functional tests. One patient participated by home visits only, therefore missing data from ancillary investigations.
Generalizability of the data
To our best knowledge, the cohort reported by Villar-Quiles et al. is the largest cohort of SELENON-RM patients reported so far and shows high similarities to our cohort both in age and clinical phenotype. In both cohorts, the clinical phenotype was marked by severe axial muscle weakness, spinal rigidity and scoliosis, with relatively preserved limb strength. Additionally, respiratory failure, requiring respiratory support while being ambulant, was frequently seen. Moreover, joint contractures and hyperlaxity were prevalent, while only a small minority of the cases were non-ambulant and none of the patients had intellectual disabilities or central nervous system involvement. Contrarily, we did not see any patients with cardiomyopathy or right ventricle failure. We did observe more subtle cardiac abnormalities including QRS fragmentation and abnormal GLS.
Conclusion
Based on the results of this cross-sectional cohort study, we recommend routine cardiorespiratory screening and bone quality assessment in (a)symptomatic patients for prevention, early detection and treatment of complications. We further recommend appropriate management interventions to reduce experienced fatigue and pain. For longitudinal follow-up and clinical trials, we suggest the following measures to assess disease severity and possibly disease progression: the MFM-20/32, accelerometery and muscle ultrasound. The 6MWT and time to raise from the ground might be of added value in ambulant patients. For a more definite selection of longitudinal clinical and functional outcome measures, we need longitudinal data from our ongoing 1.5-year natural history study LAST STRONG, of which data collection will be completed in 2023. International consensus on definite selection of outcome measures have to be reached in a consortium meeting involving key leader physicians and researchers.
ACKNOWLEDGMENTS
We thank all patients and their relatives for participation in our study. We thank Marit Boxum and Daniëlle Franken for their help in contacting patients and requesting medical data from other hospitals. Several authors of this publication are members of the Radboudumc Neuromuscular Center (Radboud-NMD), Netherlands Neuromuscular Center (NL-NMD) and European Reference Network for rare neuromuscular diseases (EURO-NMD).
FUNDING
The study is financially supported by a competitively awarded, peer-reviewed grant from Stichting Spieren voor Spieren and Stichting Stofwisselkracht, The Netherlands.
COMPETING INTERESTS
The authors declare that they have no competing interests.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was registered at clinicaltrials.gov (NCT04478981). This study was approved by the medical ethical reviewing committee of Region Arnhem-Nijmegen (NL-number NL64269.091.17, dossier number 2017-3911; date of approval of last amendment: 8 July 2020). From all patients, or in case of children their parent or legal guardian, informed consent has been obtained for participating in our study.
CONSENT FOR PUBLICATION
From all patients written consent for publication of their pictures has been obtained.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available within the article and supplementary material, and from the corresponding author, upon reasonable request.
AUTHORS’ CONTRIBUTION
KB: manuscript writing and revision, study concept design, inclusion of patients, (co-)performing all medical examinations, collection of all data and performing all (statistical) analyses. JG, BE, CE, NV critical revision of manuscript, study concept design and supervision of KB. JD, NA study protocol on muscle ultrasound and respiratory function tests. FU, FH, RN study protocol on cardiac examination. AD, JD, MJ interpretation of low bone quality. All authors read and approved the manuscript.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JND-221673.
REFERENCES
[1] | Villar-Quiles RN , von der Hagen M , Métay C , Gonzalez V , Donkervoort S , Bertini E , et al. The clinical, histologic, and genotypic spectrum of SEPN1-related myopathy: A case series. Neurology. (2020) ;95: (11):e1512–e27. |
[2] | Ferreiro A , Quijano-Roy S , Pichereau C , Moghadaszadeh B , Goemans N , Bönnemann C , et al. Mutations of the selenoprotein N gene, which is implicated in rigid spine muscular dystrophy, cause the classical phenotype of multiminicore disease: Reassessing the nosology of early-onset myopathies. Am J Hum Genet. (2002) ;71: (4):739–49. |
[3] | Clarke NF , Kidson W , Quijano-Roy S , Estournet B , Ferreiro A , Guicheney P , et al. SEPN Associated with congenital fiber-type disproportion and insulin resistance. Ann Neurol. (2006) ;59: (3):546–52. |
[4] | Ferreiro A , Ceuterick-de Groote C , Marks JJ , Goemans N , Schreiber G , Hanefeld F , et al. Desmin-related myopathy with Mallory body-like inclusions is caused by mutations of the selenoprotein N gene. Ann Neurol. (2004) ;55: (5):676–86. |
[5] | Ziyaee F , Shorafa E , Dastsooz H , Habibzadeh P , Nemati H , Saeed A , et al. A novel mutation in SEPN1 causing rigid spine muscular dystrophy A Case report. BMC Medical Genetics. (2019) ;20: (1):13. |
[6] | Filipe A , Chernorudskiy A , Arbogast S , Varone E , Villar-Quiles R-N , Pozzer D , et al. Defective endoplasmic reticulum-mitochondria contacts and bioenergetics in SEPN1-related myopathy. Cell Death & Differentiation. (2021) ;28: (1):123–38. |
[7] | Moulin M , Ferreiro A . Muscle redox disturbances and oxidative stress as pathomechanisms and therapeutic targets in early-onset myopathies. Semin Cell Dev Biol. (2017) ;64: :213–23. |
[8] | Arbogast S , Beuvin M , Fraysse B , Zhou H , Muntoni F , Ferreiro A . Oxidative stress in SEPN1-related myopathy: From pathophysiology to treatment. Ann Neurol. (2009) ;65: (6):677–86. |
[9] | Smeitink J , van Maanen R , de Boer L , Ruiterkamp G , Renkema H . A randomised placebo-controlled, double-blind phase II study to explore the safety, efficacy, and pharmacokinetics of sonlicromanol in children with genetically confirmed mitochondrial disease and motor symptoms (“KHENERGYC”). BMC Neurology. (2022) ;22: (1):158. |
[10] | Wang CH , Dowling JJ , North K , Schroth MK , Sejersen T , Shapiro F , et al. Consensus statement on standard of care for congenital myopathies. J Child Neurol. (2012) ;27: (3):363–82. |
[11] | Silwal A , Sarkozy A , Scoto M , Ridout D , Schmidt A , Laverty A , et al. Selenoprotein N-related myopathy: A retrospective natural history study to guide clinical trials. Ann Clin Transl Neurol. (2020) ;7: (11):2288–96. |
[12] | Bouman K , Groothuis JT , Doorduin J , van Alfen N , Udink Ten Cate FEA , van den Heuvel FMA , et al. Natural history, outcome measures and trial readiness in LAMA2-related muscular dystrophy and SELENON-related myopathy in children and adults: Protocol of the LAST STRONG study. BMC Neurol. (2021) ;21: (1):313. |
[13] | Bouman K, Groothuis JT, Doorduin J, van Alfen N, Udink Ten Cate FEA, van den Heuvel FMA, et al. LAMA2-Related Muscular Dystrophy Across the Life Span: A Cross-sectional Study. Neurol Genet. (2023) ;9: :e200089. DOI:10.1212/nxg.0000000000200089. |
[14] | Beenakker EA , van der Hoeven JH , Fock JM , Maurits NM . Reference values of maximum isometric muscle force obtained in 270 children aged 4-16 years by hand-held dynamometry. Neuromuscul Disord. (2001) ;11: (5):441–6. |
[15] | van der Ploeg RJ , Fidler V , Oosterhuis HJ . Hand-held myometry: Reference values. J Neurol Neurosurg Psychiatry. (1991) ;54: (3):244–7. |
[16] | Soucie JM , Wang C , Forsyth A , Funk S , Denny M , Roach KE , et al. Range of motion measurements: Reference values and a database for comparison studies. Haemophilia. (2011) ;17: (3):500–7. |
[17] | Bérard C , Payan C , Hodgkinson I , Fermanian J . A motor function measure for neuromuscular diseases. Construction and validation study. Neuromuscul Disord. (2005) ;15: (7):463–70. |
[18] | de Lattre C , Payan C , Vuillerot C , Rippert P , de Castro D , Bérard C , et al. Motor function measure: Validation of a short form for young children with neuromuscular diseases. Arch Phys Med Rehabil.. (2013) ;94: (11):2218–26. |
[19] | Main M , Kairon H , Mercuri E , Muntoni F . The Hammersmith functional motor scale for children with spinal muscular atrophy: A scale to test ability and monitor progress in children with limited ambulation. Eur J Paediatr Neurol. (2003) ;7: (4):155–9. |
[20] | Franjoine MR , Gunther JS , Taylor MJ . Pediatric balance scale: A modified version of the berg balance scale for the school-age child with mild to moderate motor impairment. Pediatr Phys Ther. (2003) ;15: (2):114–28. |
[21] | Franchignoni F , Horak F , Godi M , Nardone A , Giordano A . Using psychometric techniques to improve the Balance Evaluation Systems Test: The mini-BESTest. J Rehabil Med. (2010) ;42: (4):323–31. |
[22] | Roscoe CMP , James RS , Duncan MJ . Calibration of GENEActiv accelerometer wrist cut-points for the assessment of physical activity intensity of preschool aged children. Eur J Pediatr. (2017) ;176: (8):1093–8. |
[23] | Phillips LRS , Parfitt G , Rowlands AV . Calibration of the GENEA accelerometer for assessment of physical activity intensity in children. Journal of Science and Medicine in Sport. (2013) ;16: (2):124–8. |
[24] | Esliger DW , Rowlands AV , Hurst TL , Catt M , Murray P , Eston RG . Validation of the GENEA accelerometer. Med Sci Sports Exerc. (2011) ;43: (6):1085–93. |
[25] | Engelen V , Haentjens MM , Detmar SB , Koopman HM , Grootenhuis MA . Health related quality of life of Dutch children: Psychometric properties of the PedsQL in the Netherlands. BMC Pediatr. (2009) ;9: :68. |
[26] | Iannaccone ST , Hynan LS , Morton A , Buchanan R , Limbers CA , Varni JW . The PedsQL in pediatric patients with Spinal Muscular Atrophy: Feasibility, reliability, and validity of the Pediatric Quality of Life Inventory Generic Core Scales and Neuromuscular Module. Neuromuscul Disord. (2009) ;19: (12):805–12. |
[27] | Gordijn M , Cremers EM , Kaspers GJ , Gemke RJ . Fatigue in children: Reliability and validity of the Dutch PedsQL™ Multidimensional Fatigue Scale. Qual Life Res. (2011) ;20: (7):1103–8. |
[28] | Aaronson NK , Muller M , Cohen PD , Essink-Bot ML , Fekkes M , Sanderman R , et al. Translation, validation, and norming of the Dutch language version of the SF-36 Health Survey in community and chronic disease populations. J Clin Epidemiol. (1998) ;51: (11):1055–68. |
[29] | Seesing FM , van Vught LE , Rose MR , Drost G , van Engelen BG , van der Wilt GJ . The individualized neuromuscular quality of life questionnaire: Cultural translation and psychometric validation for the Dutch population. Muscle Nerve. (2015) ;51: (4):496–500. |
[30] | Vanderiet K , Adriaensen H , Carton H , Vertommen H . The McGill Pain Questionnaire constructed for the Dutch language (MPQ-DV). Preliminary data concerning reliability and validity. Pain. (1987) ;30: (3):395–408. |
[31] | Wong DL , Baker CM . Pain in children: Comparison of assessment scales. Pediatr Nurs. (1988) ;14: (1):9–17. |
[32] | Worm-Smeitink M , Gielissen M , Bloot L , van Laarhoven HWM , van Engelen BGM , van Riel P , et al. The assessment of fatigue: Psychometric qualities and norms for the Checklist individual strength. Journal of Psychosomatic Research. (2017) ;98: :40–6. |
[33] | Vercoulen JH , Swanink CM , Fennis JF , Galama JM , van der Meer JW , Bleijenberg G . Dimensional assessment of chronic fatigue syndrome. J Psychosom Res. (1994) ;38: (5):383–92. |
[34] | Vandervelde L , Van den Bergh PY , Goemans N , Thonnard JL . ACTIVLIM: A Rasch-built measure of activity limitations in children and adults with neuromuscular disorders. Neuromuscul Disord. (2007) ;17: (6):459–69. |
[35] | Cardol M , de Haan RJ , van den Bos GA , de Jong BA , de Groot IJ . The development of a handicap assessment questionnaire: The Impact on Participation and Autonomy (IPA). Clin Rehabil. (1999) ;13: (5):411–9. |
[36] | Steffensen B , Hyde S , Lyager S , Mattsson E . Validity of the EK scale: A functional assessment of non-ambulatory individuals with Duchenne muscular dystrophy or spinal muscular atrophy. Physiother Res Int. (2001) ;6: (3):119–34. |
[37] | van der Zee K , Sanderman R . Het meten van de algemene gezondheidstoestand met de RAND-36. Noordelijk Centrum voor Gezondheidsvraagstukken, Reeks Meetinstrumenten. (1993) ;3: :1–28. |
[38] | Varni JW , Burwinkle TM , Seid M , Skarr D . The PedsQL 4. 0 as a pediatric population health measure: Feasibility, reliability, and validity. Ambul Pediatr. (2003) ;3: (6):329–41. |
[39] | Varni JW , Burwinkle TM , Limbers CA , Szer IS . The PedsQL as a patient-reported outcome in children and adolescents with fibromyalgia: An analysis of OMERACT domains. Health Qual Life Outcomes. (2007) ;5: :9. |
[40] | Wijntjes J , van Alfen N . Muscle ultrasound: Present state and future opportunities. Muscle Nerve. (2021) ;63: (4):455–66. |
[41] | Wijntjes J , van der Hoeven J , Saris CGJ , Doorduin J , van Alfen N . Visual versus quantitative analysis of muscle ultrasound in neuromuscular disease. Muscle Nerve. (2022) ;66: (3):253–61. |
[42] | Laveneziana P , Albuquerque A , Aliverti A , Babb T , Barreiro E , Dres M , et al. ERS statement on respiratory muscle testing at rest and during exercise. Eur Respir. (2019) ;53: (6). |
[43] | van Doorn JLM , Wijntjes J , Saris CGJ , Ottenheijm CAC , van Alfen N , Doorduin J . Association of diaphragm thickness and echogenicity with age, sex, and body mass index in healthy subjects. Muscle Nerve. 2022. |
[44] | Lang RM , Badano LP , Mor-Avi V , Afilalo J , Armstrong A , Ernande L , et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. (2015) ;16: (3):233–70. |
[45] | Cho MJ , Lee JW , Lee J , Shin YB , Lee HD . Relationship between fragmented QRS complexes and cardiac status in duchenne muscular dystrophy: Multimodal validation using echocardiography, magnetic resonance imaging, and holter monitoring. Pediatr Cardiol. (2017) ;38: (5):1042–8. |
[46] | Kosaraju A , Goyal A , Grigorova Y , Makaryus AN . Left Ventricular Ejection Fraction StatPearls. Treasure Island (FL): StatPearls Publishing Copyright © 2022,StatPearls Publishing LLC.;(2022) . |
[47] | Tissot C , Singh Y , Sekarski N . Echocardiographic evaluation of ventricular function-for the neonatologist and pediatric intensivist. Front Pediatr. (2018) ;6: :79. |
[48] | Truong VT , Phan HT , Pham KNP , Duong HNH , Ngo TNM , Palmer C , et al. Normal ranges of left ventricular strain by three-dimensional speckle-tracking echocardiography in adults: A systematic review and meta-analysis. J Am Soc Echocardiogr. (2019) ;32: (12):1586–97.e5. |
[49] | Levy PT , Machefsky A , Sanchez AA , Patel MD , Rogal S , Fowler S , et al. Reference ranges of left ventricular strain measures by two-dimensional speckle-tracking echocardiography in children: A systematic review and meta-analysis. J Am Soc Echocardiogr. (2016) ;29: (3):209–25.e6. |
[50] | Whittaker LG , McNamara EA , Vath S , Shaw E , Malabanan AO , Parker RA , et al. Direct comparison of the precision of the new hologic horizon model with the old discovery model. Journal of Clinical Densitometry.. (2018) ;21: (4):524–8. |
[51] | Evans JD . Straightforward statistics for the behavioral sciences. Belmont, CA, US: Thomson Brooks/Cole Publishing Co; (1996) xxii, 600-xxii, p. |
[52] | Westra D , Schouten MI , Stunnenberg BC , Kusters B , Saris CGJ , Erasmus CE , et al. Panel-based exome sequencing for neuromuscular disorders as a diagnostic service. J Neuromuscul Dis. (2019) ;6: (2):241–58. |
[53] | Nicolau S , Liewluck T , Tracy JA , Laughlin RS , Milone M . Congenital myopathies in the adult neuromuscular clinic: Diagnostic challenges and pitfalls. Neurol Genet. (2019) ;5: (4):e341. |
[54] | Scoto M , Cirak S , Mein R , Feng L , Manzur AY , Robb S , et al. SEPN1-related myopathies: Clinical course in a large cohort of patients. Neurology. (2011) ;76: (24):2073. |
[55] | Pietrasik G , Zareba W . QRS fragmentation: Diagnostic and prognostic significance. Cardiol J. (2012) ;19: (2):114–21. |
[56] | Oreto L , Vita GL , Mandraffino G , Carerj S , Calabrò MP , Manganaro R , et al. Impaired myocardial strain in early stage of Duchenne muscular dystrophy: Its relation with age and motor performance. Acta Myol. (2020) ;39: (4):191–9. |
[57] | Verdonschot JAJ , Henkens M , Wang P , Schummers G , Raafs AG , Krapels IPC , et al. A global longitudinal strain cut-off value to predict adverse outcomes in individuals with a normal ejection fraction. ESC Heart Fail. (2021) ;8: (5):4343–5. |
[58] | Caggiano S , Khirani S , Dabaj I , Cavassa E , Amaddeo A , Arroyo JO , et al. Diaphragmatic dysfunction in SEPN1-related myopathy. Neuromuscular Disorders. (2017) ;27: (8):747–55. |
[59] | Mangner N , Garbade J , Heyne E , van den Berg M , Winzer EB , Hommel J , et al. Molecular mechanisms of diaphragm myopathy in humans with severe heart failure. Circ Res. (2021) ;128: (6):706–19. |
[60] | van den Berg M , Hooijman PE , Beishuizen A , de Waard MC , Paul MA , Hartemink KJ , et al. Diaphragm atrophy and weakness in the absence of mitochondrial dysfunction in the critically Ill. Am J Respir Crit Care Med. (2017) ;196: (12):1544–58. |
[61] | Bianchi C , Baiardi P , Khirani S , Cantarella G . Cough peak flow as a predictor of pulmonary morbidity in patients with dysphagia. Am J Phys Med Rehabil. (2012) ;91: (9):783–8. |
[62] | Belli S , Prince I , Savio G , Paracchini E , Cattaneo D , Bianchi M , et al. Airway clearance techniques: The right choice for the right patient. Front Med (Lausanne). (2021) ;8: :544826. |
[63] | Bouman K , Gubbels M , van den Heuvel FMA , Groothuis JT , Erasmus CE , Nijveldt R , et al. Cardiac involvement in two rare neuromuscular diseases: LAMA2-related muscular dystrophy and SELENON-related myopathy. Neuromuscul Disord 2022. |
[64] | Bouman K , Dittrich ATM , Groothuis JT , van Engelen BGM , Janssen MCH , Voermans NC , et al. Bone quality in patients with a congenital myopathy: A scoping review. J Neuromuscul Dis. (2023) ;10: (1):1–13. |
[65] | Iolascon G , Paoletta M , Liguori S , Curci C , Moretti A . Neuromuscular diseases and bone. Front Endocrinol (Lausanne). (2019) ;10: :794. |
[66] | Takata S , Yasui N . Disuse osteoporosis. J Med Invest. (2001) ;48: (3-4):147–56. |
[67] | Qin Y , Peng Y , Zhao W , Pan J , Ksiezak-Reding H , Cardozo C , et al. Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA- A novel mechanism in muscle-bone communication. J Biol Chem. (2017) ;292: (26):11021–33. |
[68] | Chou E , Lindeback R , D’Silva AM , Sampaio H , Neville K , Farrar MA . Growth and nutrition in pediatric neuromuscular disorders. Clin Nutr. (2021) ;40: (6):4341–8. |
[69] | Kooi-van Es M , Erasmus CE , de Swart BJM , Voet NBM , van der Wees PJ , de Groot IJM , et al. Dysphagia and dysarthria in children with neuromuscular diseases, a prevalence study. J Neuromuscul Dis. (2020) ;7: (3):287–95. |
[70] | Bian Q , McAdam L , Grynpas M , Mitchell J , Harrington J . Increased rates of vitamin D insufficiency in boys with duchenne muscular dystrophy despite higher vitamin D(3) supplementation. Glob Pediatr Health. (2019) ;6: :2333794x19835661. |
[71] | Gandhi SS , Muraresku C , McCormick EM , Falk MJ , McCormack SE . Risk factors for poor bone health in primary mitochondrial disease. J Inherit Metab Dis. (2017) ;40: (5):673–83. |
[72] | Zeng H , Cao JJ , Combs GF Jr. . Selenium in bone health: Roles in antioxidant protection and cell proliferation. Nutrients (2013) ;5: (1):97–110. |
[73] | Ward LM , Konji VN , Ma J . The management of osteoporosis in children. Osteoporos Int. (2016) ;27: (7):2147–79. |
[74] | Lou J-S , Weiss MD , Carter GT . Assessment and management of fatigue in neuromuscular disease. American Journal of Hospice and Palliative Medicine®.. (2010) ;27: (2):145–57. |
[75] | Kalkman JS , Schillings ML , Zwarts MJ , van Engelen BG , Bleijenberg G . The development of a model of fatigue in neuromuscular disorders: A longitudinal study. J Psychosom Res. (2007) ;62: (5):571–9. |
[76] | Veenhuizen Y , Cup EHC , Jonker MA , Voet NBM , van Keulen BJ , Maas DM , et al. Self-management program improves participation in patients with neuromuscular disease: A randomized controlled trial. Neurology. (2019) ;93: (18):e1720–e31. |
[77] | Kim S , Xu Y , Dore K , Gewurtz R , Larivière N , Letts L . Fatigue self-management led by occupational therapists and/or physiotherapists for chronic conditions: A systematic review and meta-analysis. Chronic Illn. (2022) ;18: (3):441–57. |
[78] | Voet N , Bleijenberg G , Hendriks J , de Groot I , Padberg G , van Engelen B , et al. Both aerobic exercise and cognitive-behavioral therapy reduce chronic fatigue in FSHD: An RCT. Neurology. (2014) ;83: (21):1914–22. |
[79] | Okkersen K , Jimenez-Moreno C , Wenninger S , Daidj F , Glennon J , Cumming S , et al. Cognitive behavioural therapy with optional graded exercise therapy in patients with severe fatigue with myotonic dystrophy type A multicentre, single-blind, randomised trial. Lancet Neurol. (2018) ;17: (8):671–80. |
[80] | Hafner P , Schmidt S , Schädelin S , Rippert P , Hamroun D , Fabien S , et al. Implementation of Motor Function Measure score percentile curves - Predicting motor function loss in Duchenne muscular dystrophy. European Journal of Paediatric Neurology. (2022) ;36: :78–83. |
[81] | Jain MS , Meilleur K , Kim E , Norato G , Waite M , Nelson L , et al. Longitudinal changes in clinical outcome measures in COL6-related dystrophies and LAMA2-related dystrophies. Neurology. (2019) ;93: (21):e1932–e43. |
[82] | Vuillerot C , Payan C , Girardot F , Fermanian J , Iwaz J , Bérard C , et al. Responsiveness of the motor function measure in neuromuscular diseases. Arch Phys Med Rehabil.-6.e. (2012) ;93: (12):1–6.e1. |
[83] | Alfano L LL BK , Flanigan K , Cripe L , Mendell J . Role of motivation on performance of the 6-minute walk test in boys with Duchenne muscular dystrophy. Dev Med Child Neurol. (2015) ;57: :57–8. |
[84] | Heresi GA , Dweik RA . Strengths and limitations of the six-minute-walk test: A model biomarker study in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. (2011) ;183: (9):1122–4. |
[85] | McDonald CM , Henricson EK , Han JJ , Abresch RT , Nicorici A , Elfring GL , et al. The 6-minute walk test as a new outcome measure in Duchenne muscular dystrophy. Muscle Nerve. (2010) ;41: (4):500–10. |
[86] | Mazzone ES , Coratti G , Sormani MP , Messina S , Pane M , D’Amico A , et al. Timed rise from floor as a predictor of disease progression in duchenne muscular dystrophy: An observational study. PLoS One. (2016) ;11: (3):–e0151445–e. |
[87] | de Vries PR , Janssen M , Spaans E , de Groot I , Janssen A , Smeitink J , et al. Natural variability of daily physical activity measured by accelerometry in children with a mitochondrial disease. Mitochondrion. (2019) ;47: :30–7. |
[88] | Bortolani S , Brusa C , Rolle E , Monforte M , De Arcangelis V , Ricci E , et al. Technology outcome measures in neuromuscular disorders: A systematic review. Eur J Neurol. (2022) ;29: (4):1266–78. |
[89] | Goode AP , Hall KS , Batch BC , Huffman KM , Hastings SN , Allen KD , et al. The impact of interventions that integrate accelerometers on physical activity and weight loss: A systematic review. Ann Behav Med. (2017) ;51: (1):79–93. |
[90] | Hankiewicz K , Carlier RY , Lazaro L , Linzoain J , Barnerias C , Gómez-Andrés D , et al. Whole-body muscle magnetic resonance imaging in SEPN1-related myopathy shows a homogeneous and recognizable pattern. Muscle Nerve. (2015) ;52: (5):728–35. |
[91] | Tordjman M , Dabaj I , Laforet P , Felter A , Ferreiro A , Biyoukar M , et al. Muscular MRI-based algorithm to differentiate inherited myopathies presenting with spinal rigidity. Eur Radiol. (2018) ;28: (12):5293–303. |
[92] | Mul K , Horlings CGC , Vincenten SCC , Voermans NC , van Engelen BGM , van Alfen N . Quantitative muscle MRI and ultrasound for facioscapulohumeral muscular dystrophy: Complementary imaging biomarkers. J Neurol. (2018) ;265: (11):2646–55. |
[93] | Winter Y , Schepelmann K , Spottke AE , Claus D , Grothe C , Schröder R , et al. Health-related quality of life in ALS, myasthenia gravis and facioscapulohumeral muscular dystrophy. J Neurol. (2010) ;257: (9):1473–81. |
[94] | González-Izal M , Malanda A , Gorostiaga E , Izquierdo M . Electromyographic models to assess muscle fatigue. J Electromyogr Kinesiol. (2012) ;22: (4):501–12. |
[95] | Abresch RT , Carter GT , Jensen MP , Kilmer DD . Assessment of pain and health-related quality of life in slowly progressive neuromuscular disease. American Journal of Hospice and Palliative Medicine®. (2002) ;19: (1):39–48. |
[96] | Morís G , Wood L , FernáNdez-Torrón R , González Coraspe JA , Turner C , Hilton-Jones D , et al. Chronic pain has a strong impact on quality of life in facioscapulohumeral muscular dystrophy. Muscle Nerve. (2018) ;57: (3):380–7. |




