Review of the Evidence that Transcranial Electromagnetic Treatment will be a Safe and Effective Therapeutic Against Alzheimer’s Disease
Abstract
We have demonstrated in multiple studies that daily, long-term electromagnetic field (EMF) treatment in the ultra-high frequency range not only protects Alzheimer’s disease (AD) transgenic mice from cognitive impairment, but also reverses such impairment in aged AD mice. Moreover, these beneficial cognitive effects appear to be through direct actions on the AD process. Based on a large array of pre-clinical data, we have initiated a pilot clinical trial to determine the safety and efficacy of EMF treatment to mild-moderate AD subjects. Since it is important to establish the safety of this new neuromodulatory approach, the main purpose of this review is to provide a comprehensive assessment of evidence supporting the safety of EMFs, particularly through transcranial electromagnetic treatment (TEMT). In addition to our own pre-clinical studies, a rich variety of both animal and cell culture studies performed by others have underscored the anticipated safety of TEMT in clinical AD trials. Moreover, numerous clinical studies have determined that short- or long-term human exposure to EMFs similar to those to be provided clinically by TEMT do not have deleterious effects on general health, cognitive function, or a variety of physiologic measures—to the contrary, beneficial effects on brain function/activity have been reported. Importantly, such EMF exposure has not been shown to increase the risk of any type of cancer in human epidemiologic studies, as well as animal and cell culture studies. In view of all the above, clinical trials of safety/efficacy with TEMT to AD subjects are clearly warranted and now in progress.
INTRODUCTION
There are currently no effective therapeutics to delay or reverse the cognitive impairment of Alzheimer’s disease (AD). Over the past decade, the many pharmacologic interventions against AD have all failed, in part because drugs have difficulty passing the blood-brain barrier and have even less bioavailability inside neurons to affect the AD pathologic process therein [1]. This is critical because intraneuronally-produced amyloid-β (Aβ), a small toxic protein, aggregates into toxic oligomers of up to eight Aβ molecules within neurons. These Aβ oligomers appear to be the initiating pathologic agents in AD, as supported by many recent studies [2, 3]. Indeed, changes in CSF levels of Aβ oligomers are associated with progression of cognitive decline in AD patients [4]. Aβ oligomers have a high affinity for intraneuronal mitochondria, especially for mitochondrial electron transport proteins on the inner mitochondrial membrane, resulting in suppression of mitochondrial function/ATP production [5, 6]. This Aβ-induced mitochondrial dysfunction appears not only to be central to AD pathogenesis, but is also an early event therein [6–9]. Thus, we believe that any effective AD therapeutic will need to penetrate not only the blood-brain barrier, but also neuronal cell membranes and then outer mitochondrial membranes in order to address the toxic “intraneuronal” Aβ oligomerization causative to mitochondrial dysfunction of AD.
Given the many years of unsuccessful drug intervention studies against AD, investigating new and innovative “non-pharmacologic” interventions against AD are now clearly warranted. Neuromodulatory approaches have consequently emerged and are currently being clinically tested against AD. These approaches include transcranial magnetic stimulation (tMS) [10, 11], transcranial direct current stimulation (tDCS) [12], and deep brain stimulation (DBS) [13, 14] via chronically-implanted electrodes. All three of these approaches provide generalized stimulatory/inhibitory effects on neuronal activity, apparently without any direct “disease-modifying” actions against AD. To our knowledge, none of these approaches have been demonstrated to be efficacious against AD endpoints in cell culture or animal models for AD.
The newest neuromodulatory approach against AD is transcranial electromagnetic treatment (TEMT), which we have pioneered in pre-clinical electromagnetic field (EMF) treatment studies [15–19] to AD transgenic mice. Much different from tMS, TEMT (and electromagnetic treatment in general) involves interdigitated magnetic and electric waves that are perpendicular to one another and to the direction which they are propagating. These interwoven magnetic/electric waves leave an antenna source and radiate away, never to return. At the EMF frequencies we have utilized, TEMT easily penetrates the entire human forebrain to impact “intraneuronal” pathologic processes, such as intraneuronal Aβ oligomer formation. Thus, TEMT is very different technology from the magnetic fields generated by tMS, which involve magnetic energy emitted by and returned to a coil conductor source. TEMT is also superior to other neuromodulatory approaches in being able to directly impact the entire forebrain while the other three neuromodulatory approaches can only affect cortical areas directly (tDCS, tMS) or a limited sub-cortical region directly (DBS). As well, tDCS and tMS require frequent clinical visits, while theneurosurgery required for DBS is both invasive and costly. By contrast, TEMT will be administered in-home by the patient’s caregiver, treat all affected areas of the AD brain, and be available to essentially the entire AD population. Thus, TEMT has distinct advantages over other neuromodulatory approaches, which should enhance the chances for it providing true therapeutic efficacy against AD.
In 2007, our laboratory, in collaboration with multiple others, began investigating the effects of EMF treatment on cognitive function and brain Aβ pathology in AD transgenic mice. In a variety of studies, we discovered and confirmed that daily EMF treatment over periods of 1–9 months can prevent and reverse cognitive impairment, as well as reverse Aβ aggregation/deposition. These benefits apparently occurred through the complementary mechanisms of Aβ disaggregation (both small/oligomeric and fibrillar/compact forms), mitochondrial enhancement, and enhanced neuronal activity. All of these studies involved EMF treatment within the ISM radiofrequency band (902–928 MHz) reserved for Industry, Science, and Medicine and at Specific Absorption Rate (SAR) power levels below FDA/FCC limits. Although these pre-clinical studies clearly justify the TEMT clinical trial currently in progress, it is important to gauge to the extent currently possible the safety of this neuromodulatory approach for long-term use in humans. As such, the purpose of this article is to review evidence regarding the safety and efficacy of TEMT (and EMF treatment in general) as a new therapeutic intervention against neurodegenerative diseases. The review is divided into three sections, with Section I presenting pre-clinical data/studies supportive of EMF efficacy in AD animal models. Section II then presents pre-clinical data/studies that provide insight into TEMT safety. Finally, Section III describes human studies that relate to TEMT safety and potential physiologic/cognitive benefits.
SECTION I: SUPPORTIVE PRE-CLINICAL STUDIES OF EMF TREATMENT EFFICACY
Since 2010, we have published five peer-reviewed papers showing the utility of EMF treatment in AD transgenic mice (Tg; AD mice) to provide cognitive benefits, anti-aggregation effects on brain Aβ, mitochondrial enhancement, and enhanced neuronal activity. These transgenic mice overexpress the mutant Swedish form of human APP alone (APPsw) or in combination with a mutant human PS1 gene (APPsw+PS1)—both mutations are causative to the early-onset form of AD via Aβ production/aggregation. In our initial paper [15], we reported that twice daily whole body EMF treatment (pulsed at 918 MHz, 1.05 W/kg SAR) begun early in adulthood before compact Aβ plaques and cognitive impairment occur, protected AD mice from otherwise certain cognitive impairment months later; this, in a complex cognitive interference test (Fig. 1A-C). If EMF treatment was delayed until older age (when compact Aβ plaques were extensive and cognitive impairment present), daily EMF treatment over months reversed both cognitive impairment (Fig. 2A) and Aβ deposition (Fig. 2B) [16–18].
Of greater significance than the reductions in “extracellular” compact Aβ plaques was the preventive effect of TEMT on “intraneuronal” oligomeric Aβ aggregation following sonication of hippocampal homogenates from aged (14-month-old) AD mice. Over the course of 6 days, the progressive increase in the 80 kD Aβ oligomer was prevented by twice daily EMF treatment to these homogenates (Fig. 3) [15]; this result indicates that EMF treatment exerts a “direct” anti-aggregating effect on oligomeric Aβ. In addition to this in vitro prevention of Aβ oligomeric formation, aged AD transgenic mice given one month of twice-daily EMF treatment exhibited a 5–10-fold increase in “mitochondrial” soluble Aβ levels within hippocampal synaptosomes (Fig. 4) [19], which is consistent with EMF treatment-induced disaggregation of oligomeric to monomeric Aβ within these mitochondria. Thus, TEMT penetrates neurons to destabilize/disrupt oligomeric Aβ therein, possibly through destabilization of hydrogen bonds between individual Aβ monomers or through disruption of dipole-dipole coupling.
Linked to the above Aβ disaggregation were 50–150% enhancements of mitochondrial function across six established measures evaluated in the same tissue (Fig. 5) [19]. This finding suggests that EMF-induced removal of oligomeric Aβ from neuronal mitochondria results in a substantial increase in neuronal mitochondrial function—exactly the therapy needed for the mitochondrial dysfunction and hypo-metabolism present in brains of AD subjects. Importantly, EMF-induced mitochondrial enhancement was observed even in hippocampal mitochondria from normal aged mice [19], indicating that EMF treatment-induced increases in mitochondrial function (especially for Complex IV enhancement) do not require removal of oligomeric Aβ aggregates. Indeed, both young adult and aged “normal” mice exhibit enhanced cognitive function with long-term EMF treatment (Fig. 1D, 2A) [15].
As yet another mechanism of EMF action, we have reported that EMF treatment for 2 months increases “neuronal activity” by 21% within entorhinal cortex of aged AD mice and normal aged mice, while increasing cognitive performance in the same animals (Fig. 6) [16]. This EMF treatment-induced increase in neuronal activity may be at least partially responsible for the minimal 0.1–0.3°C rise in brain temperature sometimes seen during treatment sessions in aged AD mice and normal mice (see Section II).
It should be underscored that all of our pre-clinical data (which is comprehensively reviewed in [18]) was attained 2–8 h after EMF administration, indicating lasting benefits of EMF treatment beyond any daily treatment period. As detailed in Section II, all of the benefits of EMF treatment occurred through “non-thermal” mechanisms because we have shown that there are no increases in brain temperature during treatment sessions or in comparison to sham controls [17]. Importantly, the benefits of long-term EMF treatment that we began reporting in 2010 have been confirmed in publications from three independent laboratories that utilized electromagnetic treatment in AD mice [20–22].
From our collective body of pre-clinical EMF studies, we have identified three mechanisms of action associated with EMF treatment’s ability to protect against or reverse cognitive impairment in AD mice: 1) disaggregation of “intraneuronal” Aβ oligomers and extracellular Aβ plaques, 2) mitochondrial enhancement within neurons, and 3) increase in neuronal activity—all three within brain areas importantfor cognitive function such as the cerebral cortex, hippocampus, and entorhinal cortex. Critical to these beneficial effects is the ability of electromagnetic waves (at the parameters utilized) to easily penetrate deep human brain areas and all neurons therein, as we have demonstrated in human phantom FDTD computer simulation studies (Fig. 7).
It is important to note that there are currently no AD therapeutics in clinical trials that have been shown to be capable of Mechanisms 1 or 2, much less both of them. By attacking the AD-initiating processes of “intraneuronal” Aβ aggregation and suppressed energy production, and in multiple brain areas impacted by AD, TEMT is not based on a single pathogenic hypothesis like most drugs are. Regarding TEMT’s anti-Aβ aggregation actions (Mech. 1),NeuroEM has identified both direct and indirect processes that are likely involved. As for TEMT’s mitochondrial enhancement actions (Mech. 2), a direct enhancement of Complex IV activity and an indirect enhancement of overall mitochondrial function via removal/disaggregation of Aβ oligomers within mitochondria are involved. A detailed description of the multiple EMF mechanisms of action against AD will be the subject of a follow-up article.
Collectively, our pre-clinical studies of EMF treatment efficacy exceed the pre-clinical work performed for most potential AD drugs prior to their advancement to clinical trials. As such, clinical trials of safety/efficacy with TEMT administration to AD patients are now clearly warranted and are in progress.
Note, since all of our pre-clinical studies, and essentially all other animal studies, have involved “whole body” EMF treatment and not EMF treatment limited to the head/cranium, these animal studies are being referred to as “EMF treatment studies”. The term “TEMT” is reserved for human clinical studies that have had, or will have, EMF treatment limited to the head/cranium, such as with our TEMT treatment device (see http://www.neuroem.com).
SECTION II: PRE-CLINICAL ANIMAL/CELL CULTURE STUDIES OF EMF TREATMENT SAFETY
Section I presented strong evidence from our EMF treatment studies in AD (transgenic) mice that long-term EMF treatment provides both cognitive and neuropathologic benefits relevant to AD. The studies within this section will evaluate the safety of EMF treatment parameters (around 900 MHz and ≤1.6 W/kg SAR) used in our pre-clinical studies and in our currently underway clinical trial – this, from the perspectives of animal and cell culture/in vitro studies. A particular emphasis will be placed on the inability of such EMF treatment to induce, or contribute to the induction of cancer, as demonstrated by a wide breath of basic science and biophysical studies.
Animal studies from our laboratories
All of our pre-clinical studies showing cognitive benefit and neuropathologic reversal in AD mice involved an EMF treatment frequency (918 MHz) within the ISM radiofrequency band (902–928 MHz) reserved for Industry, Science, and Medicine and SAR power levels (1.05 W/kg) below FCC limits for commercial devices: EMF treatment involved two 1-hour sessions every day. Animal studies have concluded time and time again that long-term exposure to radiofrequency waves in/near this ISM band have no negative impact on health, a conclusion that is underscored by safety endpoints evaluated in our own long-term studies. Those studies indicate that far from being deleterious to cognitive function in both AD mice and normal mice, daily long-term EMF treatment for up to 8 months actually improves cognitive function while not affecting sensorimotor function or anxiety levels [15–19]. The reader is referred to these five published papers for details on the cognitive benefits and sensorimotor effects of EMF treat in both normal and AD mice.
The inability of long-term EMF treatment at 918 MHz to deleteriously affect brain homeostasis is indicated by our neurochemical analysis performed in both AD mice and normal mice following daily TEMT for over 7 months [15]. For both AD mice and normal mice, TEMT had essentially no effect on hippocampal DNA repair enzymes (OGG1, PARP), antioxidant enzyme markers (cytosolic and mitochondrial SOD, GSH/GSSH), or protein oxidative damage (protein carbonyl content). Furthermore, histologic evaluation of brains from both AD mice and normal mice in our studies revealed no histologic or cytologic abnormalities, and no cancerous growths [15–17]. As well, major peripheral organs (liver, heart, lungs, kidneys) were all normal in appearance.
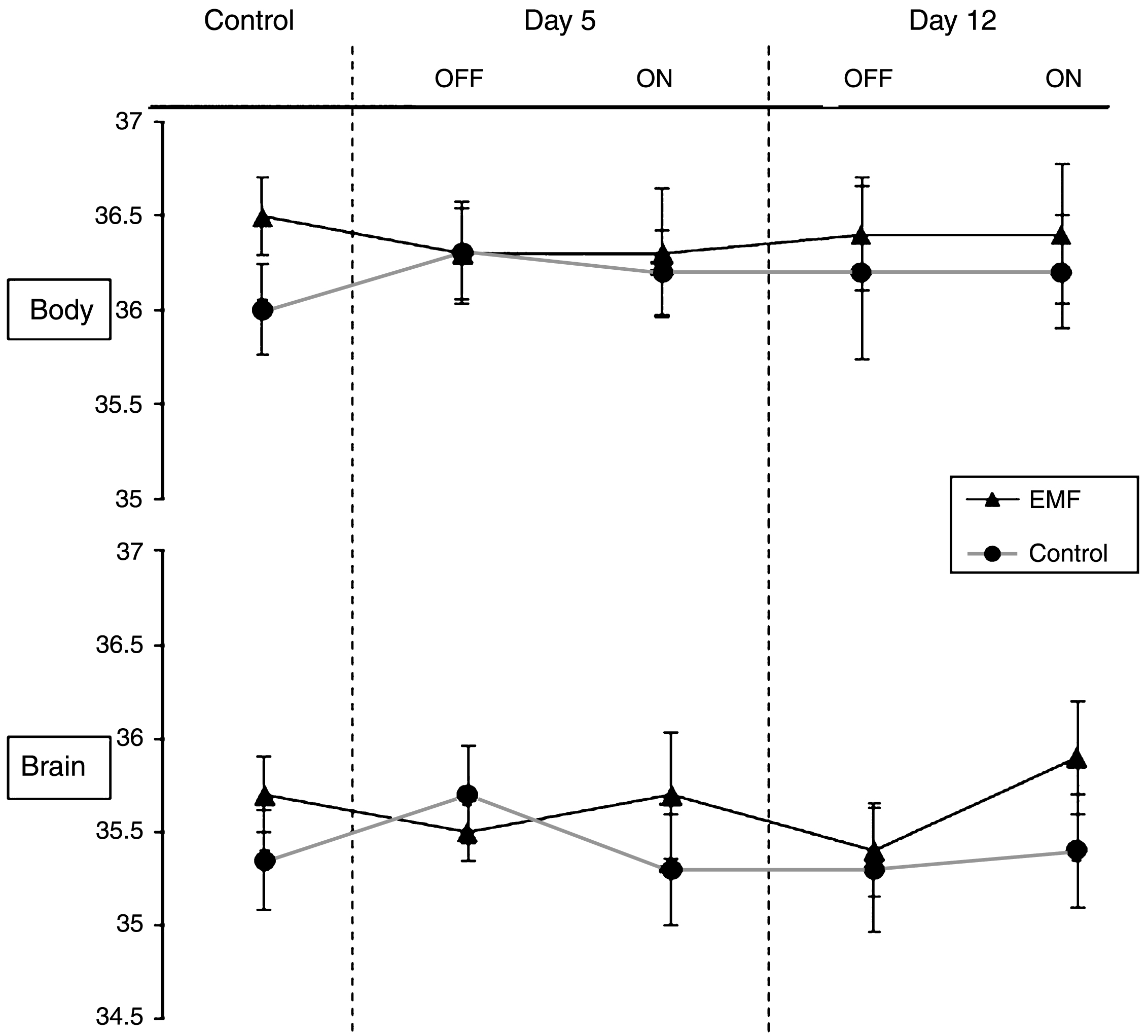
Also underscoring the safety of EMF treatment at 918 MHz and below FCC power limits for commercial devices, all of the benefits of EMF treatment that we have reported occurred without any acute or long-term increases in brain temperature; in other words, EMF treatment provided cognitive and neuropathologic benefits through “non-thermal” mechanisms. For example, acute EMF treatment (two 1-hour treatments in a single day) to several types and ages of naïve AD mice and controls revealed no change in brain temperature during or between the two treatments (Fig. 8A) [12]; this was the same brain temperature profile observed in non-treatment mice (Fig. 8B). Note the strong correlation between brain and body temperatures in this study (Fig. 8C), although brain temperature is typically around 0.3–0.4°C cooler than body temperature. Longer term TEMT treatment (daily for 12 days) to AD mice also resulted in no change in brain or body temperature, both in relation to OFF periods and compared to control mice not given EMF administration (Fig. 9) [17]. In yet another study, we attained brain temperature measurements from aged AD mice and normal mice at 1, 3, and 6 weeks into EMF treatment [17, 18]. Throughout this 6-week study period, brain temperature remained stable or was minimally elevated by 0.1–0.3°C during ON periods. Following any such brain temperature elevations, brain temperature always returned to pre-treatment levels during OFF periods. Collectively, these results suggest that clinical use of our human TEMT device will result in either no increase in brain temperature or a minimal increase of no physiologic significance. It is noteworthy that, during moderate exercise in rodents and humans, brain temperature can increase by a much more prominent 1-2°C compared to any incremental elevation induced by 900 MHz EMF exposure/administration [23].
Cancer and radiofrequency exposure: Animal and cell culture studies
Numerous studies have administered radiofrequency (RF) EMF treatment involving ≈900 MHz frequency at around 1.6 W/kg SAR to rodents in order to determine any cancer-causing effects that might arise. These full-body exposure studies have determined that such radiofrequency treatment does not initiate, nor does it promote, any type of cancer investigated. With RF treatment at these parameters extending from 5 months to life-long, four studies found no evidence for an induction of brain tumors [24–27], and another study reported no ability of such RF treatment to promote brain tumor growth initiated by a chemical carcinogen [28]. Similarly, 900 MHz RF treatment extending from several weeks to life-long did not promote chemically-induced breast cancer [29–31], nor did it promote UV radiation-induced skin cancer [32]. Indeed, no increases in any type of cancer induced by non-ionizing radiation were observed in rodents exposed to 900 MHz RF treatment for 11/2 years [33]. The National Cancer Institute’s 2015 website summarizes these studies nicely in stating, “It is generally accepted that damage to DNA is necessary for cancer to develop. However, radiofrequency energy, unlike ionizing radiation, does not cause DNA damage in cells, and it has not been found to cause cancer in animals or to enhance the cancer-causing effects of known chemical carcinogens in animals”.
The above animal studies are supported by human/rodent cell culture studies looking at DNA damage (genotoxicity) of the same 900 MHz RF treatment. Although such studies are not particularly relevant to human RF treatment because the vast majority of them are acute (less than 24 hours), they have almost universally reported no effects of 900 MHz RF exposure on indices of genotoxicity/DNA damage [34]. In this regard, RF treatment to cell cultures had no effect on DNA strand breaks [35–39] or micronucleus induction [40–42]. Relatedly, exposing brain suspensions from mice to 900 MHz RF resulted in no effects on DNA stand breaks or chromatin conformation [43]. A number of cell culture studies have measured the activity of ornithine decarboxylase (ODC), an enzymatic marker for increased cell proliferation/cancer, and found ODC activity to be similarly unaffected by RF treatment [44–46]. Krewski [47] presented multiple studies showing that RF exposure to cell cultures does not induce DNA strand breaks, chromosome aberrations, sister chromatid exchanges, or DNA repair synthesis. Verschaeve [48] reviewed the data on alleged RF-induced genetic effects and concluded that the evidence for genotoxic effects of RF exposure (which would be important for demonstrating enhanced cancer risk) is extremely weak.
Consistent with the large body of human, animal, and cell culture studies indicating no association between 900 MHz RF treatment and any type of cancer, extensive research has not established any biologic mechanisms through which such RF treatment could cause cancer, even if an association were present. There is certainly a link between some forms of electromagnetic radiation (e.g., UV radiation, x-rays, and gamma rays) and some cancers. These electromagnetic forms have extremely high frequencies that are many orders of magnitude higher than RF waves. Since the photons of these very high frequency forms of radiation carry a large amount of energy compared to RF, they can break covalent chemical bond; importantly, all carcinogenic agents act by breaking covalent bonds [49]. In sharp contrast, RF-generated photons have a much lower energy level that is insufficient to break, damage, or weaken any covalent bonds. Although RF photons can induce rotational motion of strongly dipolar residues [50] or produce resonance/vibrational effects on some molecules [51, 52], these effects are not deleterious in causing or promoting cancer. The impossibilityof radiofrequency waves, and thus our TEMT device, to induce cancer is supported by the research of none other than Albert Einstein. He won the 1905 Nobel Prize in Physics for establishing that much higher electromagnetic frequencies (UV,x-rays, gamma rays) are required to break covalent bonds in molecules and, thus, to increase cancer risk.
Cognitive function in rodents
We have performed multiple studies investigating the long-term cognitive effects of daily RF treatment to normal and AD mice [15–18]. All of these studies involved pulsed 918 MHz frequency and 1.05 W/kg SAR for two one-hour treatment periods daily, very close to the parameters built into our human TEMT device and the same daily treatment paradigm (two 1-hour periods). In none of these comprehensive studies were any cognitive impairments observed in either normal mice or AD mice in any cognitive task evaluated. Indeed, cognitive enhancement was often seen, and usually in a complex task that is measure-for-measure analogous to a human task of the same name that is used to distinguish AD and pre-AD patients from normal aged individuals—namely, the cognitive interference (CI) task. In an initial study, we found that AD mice started on daily TEMT in young adulthood were protected from otherwise inevitable cognitive impairment in the CI task at 7 months into treatment [15]. In follow-up studies involving the start of EMF treatment at older ages (when AD mice were cognitively impaired), 2–8 months of daily treatment reversed cognitive impairment in the CI task and in the Y-maze task [15–18]. Even normal mice receiving treatment in these studies showed cognitive improvement in both the CI and Y-maze tasks. In all of our studies, beneficial effects lessening brain AD neuropathology [15–18] and/or enhancing brain metabolic function [19] were observed. Although all of these mouse studies involved whole body RF treatment, mouse brains were receiving RF exposure (thus TEMT) very similar to that provided by our human TEMT device.
Other investigators have investigated cognitive endpoints in “normal” rats or mice given 900 MHz RF exposure. All of the well-designed studies involving adult animals have reported no overall effects of 10 days to 19 months RF treatment on a variety of cognitive tasks such as the 8-arm radial maze and Morris water maze [53–58]. Although one of these studies [57] reported transient cognitive impairment midway through 10 weeks of RF treatment, the authors did not find any impairment at earlier or later time points and concluded that rats can adapt to long-term RF exposure. Interestingly, one study involving RF treatment for 5 weeks to “immature” rats reported an enhancement in Morris maze memory retention [59]. Why have all other 900 MHz RF studies involving normal “adult” rodents failed to find the cognitive benefits that we have reported in normal mice? First, most of these prior studies involved shorter-term treatment (30 days or less), which our work shows is usually not sufficient for cognitive benefit in normal animals [15]. Second, in contrast to our cognitive interference task, the cognitive tasks selected have often been tasks that are relatively insensitive to various cognitive domains and not directly relevant to humans. It should be noted that some other rodent studies have actually reported cognitive impairment resulting from RF treatment [60–64]. However, most of these studies were poorly designed. For example, there was often an inexplicable delay of 2–18 months between RF treatment and cognitive testing [60, 63] or RF treatment was compromised by stressful background radio noise that was not controlled for [61]. In one of these studies, animals were given a single treatment lasting only a few seconds, then tested 12 and 18 months thereafter [63]. To summarize, well-designed RF treatment studies involving “normal” rodents have not demonstrated any long-term cognitive impairment resulting from treatment.
Other functions in rodents (immune function, oxidative markers, BBB)
Although several endpoints (immune function, oxidative markers, and blood-brain barrier [BBB] integrity) have not been analyzed to our knowledge in human RF exposure studies, studies in normal rodent studies have investigated the effects of full-body 900 MHz RF treatment on these endpoints. Regarding immune function, Johansson [65] reviewed the literature involving RF effects on the immune system (both T- and B-cell compartments) and found no effects of 900 MHz RF treatment, although effects at harmful “microwave” frequencies (e.g., 2450 MHz) were reported. With 900 MHz RF treatment for 1 month to mice, Gatta [66] reported that neither T- norB-cell compartments were affected and that a clinically relevant effect of RF treatment on the immune system was unlikely. Similarly, Nasta [67] found that the same one-month RF treatment protocol did not affect the B-cell peripheral compartment (T1 and T2 cells, mature follicular and marginal zone B-cells) or antibody (IgM and IgG) production. Most recently, Rosado [68] found no effects of 900 MHz-exposed bone marrow cells on their long-term (3-month) ability to reconstitute peripheral T and B cells, and no differences in thymocyte number, frequency, or proliferation. Collectively, these rodent studies suggest that the immunosystem will not be impacted by TEMT in humans, especially since only the head will be exposed to RF treatment.
Animal and cell culture studies have evaluated oxidative markers for evidence of oxidative stress/damage induced by 900 MHz RF treatment and have largely found little evidence for oxidative stress/damage. Seven days of 900 MHz RF exposure to rabbits resulted in no effects on all brain oxidative markers evaluated, including SOD, GSH-peroxidase, MDA, and NO [69]. Similarly, 900 MHz RF treatment to mouse cell cultures did not affect reactive oxygen species (ROS) production [70], while levels of oxidants/antioxidants (GSSH, SOD, catalase, glutathione peroxidase activity), oxidative damage/toxicity (trypan blue dye exclusion assay), and NO production were unaffected [71]. Results from these animal studies are consistent with our results showing no effects of daily RF treatment for 8 months on oxidative measures [15]. Regarding 900 MHz RF effects on the BBB, Finnie [72] reported that BBB integrity was maintained in mice after two years of daily treatment and Grafstom [73] found no evidence of BBB breakdown in rats treated once weekly for one year. By contrast, Tang [64] found damaged BBB after more acute treatment of 14–28 days. Collectively, these studies suggest that, although temporary effects of EMF on BBB integrity are possible, no long-term effects have beendemonstrated.
Thus, from the standpoints of immune function, oxidative stress, and BBB integrity, there is essentially no evidence from animal studies that 900 MHz RF treatment induces deleteriously effects.
SECTION III: HUMAN STUDIES RELATED TO TEMT SAFETY AND EFFICACY
General health studies
Particularly since 2005, many studies in normal adults have investigated the safety of cell phone use (especially GSM 900 phones) on indices of general human health such as sleeplessness, fatigue, dizziness, digestive disturbances, concentration difficulties, blood cell profiles, blood pressure, or cognitive function. The single antenna of these commercially available devices is held close to the human head during use and their electromagnetic frequency of around 900 MHz and SAR levels of <1.6 W/kg are close to those for any given antenna of the TEMT device that we have in clinical trials. In that only one antenna of the TEMT device is ON/active at any given time, the results of human studies investigating health effects of both short- and long-term GSM 900 cell phone use are especially pertinent to determining safety of our TEMT device. General health aspects of cell phone use will be considered first, followed by an analysis of the purported association between cell phones and brain cancers. It is important to note that this evaluation of human health effects of cell phones largely involves electromagnetic (RF) exposure from GSM 900 MHz cell phones, although some studies also included other cell phone technologies (e.g., GSM 1800/1900 MHz, UMTS). Obviously, GSM 900 cell phones are the closest in electromagnetic parameters to the TEMTdevice.
Valberg [74] summarized findings of the World Health Organization’s workshop on health issues potentially related to cell phone use and concluded that there is little support for adverse health effects from cell phones at or below levels established by international standards. Valberg [74] underscored that the more recent, better-designed human studies are universally negative, particularly regarding cancer development. In a very comprehensive review, Krewski [47] stated that, “All of the authoritative reviews completed within the last 2 years have concluded that there is no clear evidence of adverse health effects associated with radiofrequency fields”. In an update of their original report, Krewski and colleagues [75] again found there was no clear evidence of adverse health effects associated with radiofrequency fields/cell phones. For the period 2000–2011, Moussa [76] evaluated epidemiologic, systemic, and meta-analysis studies, and also found no consistent pattern for exposure to mobile phones being detrimental to health.
The aforementioned studies, and others, have lead prominent health organizations in the U.S. to conclude that there is no clear evidence of adverse health effects associated with radiofrequency fields. The National Institute of Environmental Health Sciences (NIEHS) states that, “The weight of the current scientific evidence has not conclusively linked cell phone use with any adverse health problems.” The FDA states that, “Studies reporting biological changes associated with radiofrequency energy have failed to be replicated and the majority of human epidemiologic studies have failed to show a relationship between RF exposure from cell phones and health problems.” The Centers for Disease Control and Prevention (CDC) states that scientific research as a whole does not support a statistically significant association between cell phone use and healtheffects.
Cognitive/physiologic studies
Regarding subjective symptoms and cognitive function, Kwon [77] conducted an extensive review of studies evaluating behavioral and neurophysiological effects of cell phone use. They found no evidence that any subjective symptoms (sleeplessness, headache, dizziness, fatigue, etc.) were induced by cell phone use; such symptoms reported in supposed hypersensitive individuals are thus psychosomatic in nature. Moreover, in over 30 published papers (most of which involved GSM 900 phones), Kwon [77] found no evidence that cell phone use resulted in any deleterious effects on cognitive function. Similarly, a meta-analysis performed by Barth et al. [78] involving 17 studies found no significant effects of GSM 900 phone exposure on cognitive abilities, a conclusion echoed by an additional meta-analysis by Valentini [79] involving 24 studies. To date, most controlled human studies reporting no deleterious cognitive effects of 900 MHz cell phone exposure have been “acute”, single exposure (3–120 min) studies [80–86], with the exception of three studies involving daily exposure for 6–27 days [87–89]. All of these studies showing no deleterious cognitive effects were exclusively in normal individuals (no AD or other neurologically-diseased subjects) and all of them involved unilateral RF exposure to only one hemisphere via a cell phone held next to the head.
No controlled human studies have investigated the cognitive effects of “long-term” and “bilateral” GSM 900 EMF treatment in normal subjects over months or years. However, two epidemiologic-based human studies have already provided indirect evidence that continued RF exposure via cell phone use could be associated with enhanced cognitive performance (executive function) in normal subjects [90] and a much reduced risk of hospitalization due to AD and vascular dementia for long-term cell phone users of 10 years or more [91]. Although involvinga very high 10,500 MHz frequency and extremely low power levels, a recent pilot study administered EMF clinically to AD patients three times a week for 5 weeks, resulting in significant improvement in a variety of cognitive measures [92]. However, the known inability of such a high EMF frequency to penetrate brain tissue, especially at the extremely low EMF utilized, suggest an unconventional mechanism may be involved in these cognitive benefits.
A number of physiologic effects have been reported with “acute” 900 MHz cell phone exposure in normal humans. First, cortical excitability is enhanced, as measured by evoked potentials [93]. Second, numerous studies have reported that acute 900 MHz cell phone exposure enhances alpha wave activity (important for basic cognitive processing) in awake cortical EEG [84, 94–96]. All of these studies suggest that neuronal activity could be beneficially enhanced by 900 MHz exposure. Since neuronal activity is coupled to glucose utilization, it is not surprising that an increase in brain glucose utilization (indexed by FDG-PET scanning) was observed in brain areas closest to the cell phone antenna [97]. In view of these diverse physiologic studies, electromagnetic waves from cell phones could actually be providing beneficial physiologic effects on brain function in normal humans.
Importantly, Wessapan [98] showed that the electromagnetic parameters we are utilizing in our clinical studies (around 900 MHz and 1.6 W/kg SAR) result in a very minimal 0.1-0.2°C increase in brain temperature in their human head FDTD simulation study. Wang [99], as well as Van Leeuwen [100], also calculated brain temperature in their FDTD simulation studies involving 900 MHz exposure and found no more than a 0.1°C rise in brain temperature. Since any potential health problems due to EMF exposure are linked to temperature increases of at least 2-3°C [19], the very minimal increase in brain temperature calculated in the FDTD studies of Wessapan [98], Wang [99], and Van Leeuwen [100] clearly indicate that the frequency (around 900 MHz) and power level (1.6 W/kg) of our clinical TEMT device is highly unlikely to have any thermally-induced health hazards associated with its use.
Thus, in terms of general health, subjective symptoms, cognitive function, and physiologic measures evaluated in humans, 900 MHz RF exposure has not been associated with any deleterious effects. In the case of cognitive function and physiologic endpoints, there is evidence that such exposure may actually be beneficial.
Brain cancer studies
The notion that GSM 900 MHz or 1800 MHz cell phones can increase the risk of brain cancer originated with a single group of Swedish researchers around 2004 and became prominent around 2008 [101,102]. Investigating the Swedish population, these researchers have repeatedly published epidemiologic studies since then concluding that GSM cell phone exposure doubles the risk of brain glioma and acoustic neuroma after 10 or more years of cell phone use [103–105]. Their most recent epidemiologic study [106] pooled two case-control studies involving Swedish patients diagnosed during 1997–2003 and 2007–2009. With cell phone exposure assessed by a self-administered questionnaire, Hardell [106] reported a 1.8x increased risk of glioma overall through 20 years. It is important to recognize that the current life-long risk of developing any form of brain cancer is about 0.5%. So even if the risk of brain cancer was doubled by long-term cell phone use (which overwhelming evidence says is not the case), the life-long risk of brain cancer would still only be a small 1% ! If NeuroEM’s TEMT device is shown to be an effective therapeutic against AD in clinical trials, the vast majority of AD patients and their families would gladly accept this claimed doubling of brain cancer risk to 1%.
Based in part on the above results reported by Swedish investigators, a working group from the World Health Organization’s International Agency for Research on Cancer (IARC) in 2011 classified radiofrequency fields emitted from mobile phones as “possibly carcinogenic to humans”. The IARC put RF fields into Category 2B, based on “limited” evidence suggesting an association between exposure from mobile phones and two types of brain cancer (glioma and acoustic neuroma) [107]. This report puts mobile phone exposure in the same potential risk Category (2B) as coffee. Any listing of carcinogenic agents by the IARC that suggests coffee is potentially carcinogenic has questionable credibility or is hopelessly out-of-date (the inclusion of coffee in Category 2B has apparently not been updated since 1991). Indeed, over the past 10 years, there has been mounting scientific evidence that coffee reduces risk of many forms of cancer, including liver cancer, rectal cancer, breast cancer, and prostate cancer [108, 109]. Following the 2011 IARC report classifying mobile phones in Category 2B, a number of investigators condemned the report as scientifically invalid and misleading. Vigayalaxmi [110] did a meta-analysis investigating the purported correlation between increased genetic damage and carcinogenesis and found that the Category 2B classification for mobile phones was not supported by genotoxicity-based evidence. Moreover, Wiedemann [111] reported that the IARC’s 2011 study was flawed because characterization of the probability of carcinogenicity was misunderstood by study participants and the respondents greatly overestimated the magnitude of the potential risk from cell phone radiofrequency exposure. In their study reporting no significant effect of intensive cell phone usage on incidence of brain cancers in Taiwan, Hsu [112] even suggested that the IARC should publish more conscientious reports to spare the public unnecessary worries.
In contrast to the above studies from a single Swedish group and the IARC’s classification, large and well-designed human epidemiologic studies performed since 2010 have concluded time and time again that long-term exposure to RF fields of around 900 MHz (typifying cell phones in the U.S.) have no negative impact on health, particularly on incidence of brain tumors. The large INTERPHONE Study [113], performed by a subsidiary of the WHO, involved 13 nations (including Sweden) with the goal of determining if RF waves from long-term cell phone use of over 10 years increased risk of brain cancers (glioma, acoustic neuroma, meningioma). This huge cased-controlled and recall-based study found no elevated risk of brain cancer with 10 or more years of cell phone use. Also, no relationship was found between lifetime number of phone calls (higher amounts of cell phone use) and brain cancer. A 2011 review of the INTERPHONE Study by the National Institute of Environmental Health and Safety (NIEHS) firmly agreed with the study’s conclusion and underscored that the INTERPHONE Study actually found an overall reduced risk of brain cancer with regular mobile phone use versus non-users [114]. Moreover, a recent extension from the INTERPHONE Study reported no relationship between location of brain tumors and regions of the brain that were exposed to the highest level of RF energy from cell phones [115].
In another huge epidemiologic study [116] with no selection bias and no recall bias, 358,000 cell phone subscribers in Denmark were followed for 17 years (1990–2007). Irrespective of whether subscribers had used cell phones for 10–13 years or more than 13 years, the incidence of brain cancers (glioma, acoustic neuroma, meningioma) was not increased. In the prospective Million Women Study (UK) involving 791,000 women, there was no increased risk of glioma, acoustic neuroma, or meningioma during 7 years of follow-up through 2011 [117, 118]. Barchana [119] actually found a decreased risk of gliomas in the Asian Pacific region after cell phones became available around 1995. Finally, Lagorio [120] recently performed a meta-analysis of 29 studies investigating cell phone use and brain cancer. In long-term cell phone users (more than 10 years), the relative risks of glioma, acoustic neuroma, and meningioma were non-significant.
Because of the aforementioned large and well-designed clinical studies, major health organizations have conclude there are no health problems (including cancer) that have been linked to radiofrequency/cell phone exposure. For example, the National Cancer Institute’s 2015 website states, “To date, there is no evidence from studies of cells, animals, or humans that radiofrequency energy can cause cancer”. Indeed, NCI’s Surveillance, Epidemiology, and End Results (SEER) Program, which tracks cancer incidence in the U.S. over time, found no increase in brain cancer incidence between 1987 and 2007, despite the dramatic increase in cell phone use in the U.S. during that time [121, 122]. Even in Sweden’s national cancer statistics, the incidence rates for glioma have not risen since 1970 [123], and glioma rates in Nordic countries from 1979 through 2008 have not increased [124], despite much increased use of cell phones in these countries. Furthermore, the U.S. FCC states that there is no scientific evidence that shows that wireless phone use can lead to cancer or to other health problems. Similar conclusions have been reached by the National Institute of Environmental Health Sciences (NIEHS), the FDA, and the Centers for Disease Control and Prevention (CDC). These organizations and the multitude of scientific studies since 2010 firmly revoke the Hardell group’s studies in Sweden, which formed the basis for the IARC’s erroneous categorization of mobile phone exposure as “possibly carcinogenic to humans”.
Thus, regarding around 900 MHz RF exposure to humans via long-term cell phone use (i.e., essentially at the same parameters as our TEMT device), many epidemiologic studies from numerous laboratories have strongly affirmed that there is no enhanced risk of brain cancers or any other cancer. Although not at the 900 MHz frequency focused on in this review, in-home RF treatment at 27 MHz to patients with various cancers was not only safe, but appeared to induce anti-tumor effects [125, 126]. Particularly for liver cancer [125], it was concluded that daily RF treatment may increase the time to radiological progression of the disease. Such studies suggest that, far from causing cancer, RF treatment may actually be therapeutic against it
CONCLUSIONS
Since pharmacologic interventions against AD have thus far been unsuccessful in slowing or reversing the AD process, non-pharmacologic therapeutics against the disease must now be seriously considered. Based on a diversity of pre-clinical studies from our laboratory in collaboration with others, the neuromodulatory approach of TEMT appears to offer unique, disease-modifying potential that could limit or reverse AD memory loss. In reviewing the evidence from animal, cell culture, and human clinical studies, this article concludes that TEMT should be a safe therapeutic against AD and other neurodegenerative diseases, even with long-term utility. Our just-initiated Phase I clinical trial involving TEMT administration to AD subjects will provide an even more definitive assessment of TEMT’s safety and potential efficacy against AD.
ACKNOWLEDGMENTS
Funds for the research and writing of this paper have been provided by NeuroEM Therapeutics, Inc. (Phoenix, AZ). We thank our primary collaborators in this work, Drs. Chuanhai Cao and Patrick Bradshaw from the University of South Florida, as well as Dr. Takashi Mori of Saitama Medical University in Japan. We also thank David Kirk (Phoenix, AZ) for his graphic design expertise in the figures.
Authors’ disclosures available online (http://j-alz.com/manuscript-disclosures/16-0165r1).
REFERENCES
[1] | Pardridge WM ((2009) ) Alzheimer’s disease drug development and the problem of the blood-brain barrier. Alzheimers Dement 5: , 427–432. |
[2] | Hayden EY , Teplow DB ((2013) ) Amyloid β-protein oligomers and Alzheimer’s disease. Alzheimers Res Ther 5: , 60. |
[3] | Bourdenx M , Koulakiotis NS , Sanoudou D , Bezard E , Dehay B , Tsarbopoulos A ((2015) ) Protein aggregation and neurodegeneration in prototypical neurodegenerative diseases: Examples of amyloidopathies, tauopathies, and synucleinopathies. Prog Neurobiol. doi: 10.1016/jpneurobio.2015.07.003 |
[4] | Jongbloed W , Bruggink KA , Kester MI , Visser PJ , Scheltens P , Blankenstein MA , Verbeeck MM , Teunissen CE , Veerhuis R ((2015) ) Amyloid-β oligomers relate to cognitive decline in Alzheimer’s disease. J Alzheimers Dis 45: , 35–43. |
[5] | Galindo MJ , Ikuta I , Zhu X , Casadesus G , Jordan J ((2010) ) Mitochondrial biology in Alzheimer’s disease pathogenesis. J Neurochem 114: , 933–945. |
[6] | Muller W , Eckert A , Curz C , Eckert G , Leuner K ((2010) ) Mitochondrial dysfunction: Common final pathway in brain aging and Alzheimer’s disease-therapeutic aspects. Mol Neurobiol 41: , 159–171. |
[7] | Chen JX , Yan SS ((2010) ) Role of mitochondrial β-amyloidin Alzheimer’s disease. J Alzheimers Dis 20: , S569–S578. |
[8] | Morais V , Strooper B ((2010) ) Mitochondrial dysfunction and neurodegenerative disorders: Cause or consequence. J Alzheimers Dis 20: , S255–S263. |
[9] | Caldwell CC , Yao J , Diaz Brinton R ((2015) ) Targeting the prodromal stage of Alzheimer’s Disease: Bioenergic and mitochondrial opportunities. Neurotherapeutics 12: , 66–80. |
[10] | Bentwich J , Dobronevsky E , Aichenbaum S , Shorer R , Peretz R , Khaigrekht M , Marton R , Rabey JM ((2011) ) Beneficial effect of repetitive transcranial magnetic stimulation combined with cognitive training for the treatment of Alzheimer’s disease: A proof of concept study. J Neural Transm 118: , 463–471. |
[11] | Rabey JM , Dobronevsky E , Airchenbaum S , Gonen O , Marton RF , Khaigrekht M ((2013) ) Repetitive transcranial magnetic stimulation combined with cognitive training is a safe and effective modality for the treatment of Alzheimer’s disease: A randomized, double-blind study. J Neural Transm 120: , 813–819. |
[12] | Boggio PS , Ferrucci R , Mameli F , Martins D , Martins O , Vergari M , Tadini L , Scarpini E , Fregni F , Priori A ((2012) ) Prolonged visual memory enhancement after direct current stimulation in Alzheimer’s disease. Brain Stimul 5: , 223–230. |
[13] | Laxton AW , Tang-Wai DF , McAndrews M , Zumsteg D , Wennberg R , Keren R , Wherrett J , Naglie G , Hamani C , Smith GS , Lozano AM ((2010) ) A Phase I trial of deep brain stimulation of memory circuits in Alzheimer’s disease. Ann Neurol 68: , 521–532. |
[14] | Kuhn J , Hardenacke K , Lenartz D , Gruendler T , Ullsperger M , Bartsch C , Mai JK , Zilles K , Bauer A , Matusch A , Schulz RJ , Noreik M , Bührle CP , Maintz D , Woopen C11 , Häussermann P , Hellmich M , Klosterkötter J , Wiltfang J , Maarouf M , Freund HJ , Sturm V ((2014) ) Deep brain stimulation of the nucleus basalis of Meynert in Alzheimer’s dementia. Mol Psychiatry 20: , 353–360. |
[15] | Arendash GW , Sanchez-Ramos J , Mori T , Mamcarz M , Lin X , Runfeldt M , Wang L , Zhang G , Sava V , Tan J , Cao C ((2010) ) Electromagnetic field treatment protects against and reverses cognitive impairment in Alzheimer’s transgenic mice. J Alzheimers Dis 19: , 191–210. |
[16] | Mori T , Arendash GW ((2011) ) Electromagnetic field treatment enhances neuronal activity: Linkage to cognitive benefit and therapeutic implications for Alzheimer’s Disease. J Alzheimers Dis Parkinsonism 1: , 2. |
[17] | Arendash GW , Mori T , Dorsey M , Gonzalez R , Tajiri N , Borlongan C ((2012) ) Electromagnetic treatment to old Alzheimer’s mice reverses β-amyloid deposition, modifies cerebral blood flow, and provides selected cognitive benefit. PLoS One 7: , e35751. |
[18] | Arendash GW ((2012) ) Transcranial electromagnetic treatment against Alzheimer’s disease: Why it has the potentialto trump Alzheimer’s disease drug development. J Alzheimers Dis 32: , 243–266. |
[19] | Dragicevic N , Bradshaw PC , Mamcartz M , Lin X , Wang L , Cao C , Arendash GC ((2011) ) Long-term electromagnetic field treatment enhances brain mitochondrial function of both Alzheimer’s transgenic mice and normal mice: A mechanism for electromagnetic field-induced cognitive benefit? Neuroscience 185: , 135–149. |
[20] | DeTaboada L , Yu J , El-Amouri S , Gattoni-Celli S , Richiere S , McCarthy T , Streeter J , Kindy MS ((2011) ) Transcranial laser therapy attenuates amyloid-β protein precursor transgenic mice. J Alzheimers Dis 23: , 521–535. |
[21] | Banaceur S , Banasr S , Sakly M , Abdelmelek H ((2013) ) Whole body exposure to 2.4 GHz WIFI signals: Effects on cognitive impairment in adult triple transgenic mouse models of Alzheimer’s disease. Behav Brain Res 240: , 197–201. |
[22] | Jeong YJ , Kang GY , Kwon JH , Choi HD , Pack JK , Kim N , Lee YS , Lee HJ ((2015) ) 1950 MHz electromagnetic fields ameliorate Aβ pathology in Alzheimer’s disease mice. Curr Alzheimer Res 12: , 481–492. |
[23] | Kiyatkin EA ((2010) ) Brain temperature homeostasis: Physiological fluctuations and pathological shifts. Front Biosci 15: , 73–92. |
[24] | Adey WR , Byus CV , Cain CD , Higgins R , Jones R , Kean C , Kuster N , MacMurray A , Stagg R , Zimmerman G , Phillips J , Haggren W ((1999) ) Spontaneous and nitrosourea-induced primary tumors of the central nervous system in Fischer 344 rats chronically exposed to 836 MHz modulated microwaves. Radiat Res 152: , 293–302 . |
[25] | Higashikubo R , Culbreth V , Spitz D , LaRegina M , Pickard W , Straube W , Moros E , Roti J ((1999) ) Radiofrequency electromagnetic fields have no effect on the in vivo proliferation of the 9L brain tumors. Radiat Res 152: , 665–671. |
[26] | Zook BC , Simmerns SJ ((2001) ) The effects of 860 MHz radiofrequency radiation on the induction or promotion of brain tumors and other neoplasms in rats. Radiat Res 155: , 572–583. |
[27] | La Regina M , Moros E , Pickard W , Straube W , Baty J , Roti J ((2013) ) The effect of chronic exposure to 835.62 MHz FDMA or 847.74 MHz CDMA radiofrequency radiation on the incidence of spontaneous tumors in rats. Radiat Res 160: , 143–151. |
[28] | Zook B , Simmens S ((2006) ) The effects of pulsed 860MHz radiofrequency radiation on the promotion of neurogenic tumors in rats. Radiat Res 165: , 608–615. |
[29] | Chagnaud J , Veyret B ((1999) ) In vivo exposure of rats to GSM-modulated microwaves: Flow cytometry analysis of lymphocyte subpopulations and of mitogen stimulation. Int J Radiation Biol 75: , 111–113. |
[30] | Bartsch H , Bartsch C , Seebald E , Deerberg F , Dietz K , Vollrath L , Mecke D ((2002) ) Chronic exposure to a GSM-like signal (mobile phone) does not stimulate the development of DMBA-induced mammary tumors in rats: Results of three consecutive studies. Radiat Res 157: , 183–190. |
[31] | Anane R , Dulou P , Taxile M , Geffard M , Crespeau F , Veyret B ((2003) ) Effects of GSM-900 microwaves on DMBA-induced mammary gland tumors in female Sprague-Dawley rats. Radiat Res 160: , 492–497 . |
[32] | Heikkinen P , Kosma V , Alhonen L , Huuskonen H , Komulainen H , Kumlin T , Laitinen J , Lang S , Puranen L , Juutilainen J ((2003) ) Effects of mobile phone radiation on UV-induced skin tumourigenesis in ornithine decarboxylase transgenic and non-transgenic mice. Int J Radiation Biol 79: , 221–233. |
[33] | Heikkinen P , Kosma V , Hongisto T , Huuskonen H , Hyysalo P , Komulainen H , Kumlin T , Lahtinen T , Lang S , Puranen L , Juutilainen J ((2001) ) Effects of mobile phone radiation on X-ray-induced tumorigenesis in mice. Radiat Res 156: , 775–785. |
[34] | Juutilainen J , Hoyto A , Kumlin T , Naarala J ((2011) ) Review of possible modulation-dependent biological effects of radiofrequency fields. Bioelectromagnetics 32: , 511–534. |
[35] | Malyapa R , Ahern E , Straube W , Moros E , Pickard W , Roti Roti J ((1997) ) Measurement of DNA damage after exposure to electromagnetic radiation in the cellular phone communication frequency band (835.62 and 847.74 MHz). Radiat Res 148: , 618–627. |
[36] | Li L , Bisht K , LaGroye I , Zhang P , Straube W , Moros E , Roti Roti J ((2001) ) Measurement of DNA damage in mammalian cells exposed in vitro to radiofrequency fields at SARs of 3-5W/kg. Radiat Res 156: , 328–332. |
[37] | Maes A , Collier M , Verschaeve L ((2001) ) Cytogenetic effects of 900 MHz 9GSM) microwaves on human lymphocytes. Bioelectromagnetics 22: , 91–96. |
[38] | Hook G , Zhang P , Lagroye I , Li L , Higashikubo R , Moros E , Strabue W , Pickard W , Baty J , Roti Roti J ((2004) ) Measurement of DNA damage and apoptosis in Molt-4 cells after in vitro exposure to radiofrequency radiation. Radiat Res 161: , 193–200. |
[39] | Luukkonen J , Juutilainen J , Naarala J ((2010) ) Combined effects of 872 MHz radiofrequency radiation and ferrous cholide on reactive oxygen species production and DNA damage in human SH-SY5Y neuroblastoma cells. Bioelectromagnetics 31: , 417–424. |
[40] | Bisht K , Moros E , Straube W , Baty J , Roti Roti J ((2002) ) The effect of 835.62 MHz FDMA or 847.74 MHz CDMA modulated radiofrequency radiation on the induction of micronuclei in C3H 10T(1/2) cells. Radiat Res 157: , 506–515. |
[41] | Zeni O , Chiavoni A , Sannino A , Antolini A , Forigo D , Bersani F , Scarfi M ((2003) ) Lack of genotoxic effects (micronucleus induction)in human lymphocytes exposed in vitro to 900 MHz electromagnetic fields. Radiat Res 160: , 152–158. |
[42] | Juutilainen J , Heikkinen P , Soikkeli H , Maki-Paakkanen J ((2007) ) Micronucleus frequency in erythrocytes of mice after long-term exposure to radiofrequency radiation. Int J Radiation Biol 83: , 213–220. |
[43] | Belyaev I , Koch C , Terenius O , Roxstrom-Lindquist K , Malmgren L , Sommer W , Salford L , Persson B ((2006) ) Exposure of rat brain to 915 MHz GSM microwaves induces changes in gene expression but not double stranded DNA breaks or effects on chromatin conformation. Bioelectromagnetics 27: , 295–306. |
[44] | Hoyto A , Sihvonen AP , Alhonen L , Juutilainen J , Naarala J ((2006) ) Modest increase in temperature affects ODC activity in L929 cells: Low-level radiofrequency radiation does not. Radiat Environ Biophys 45: , 231–235. |
[45] | Hoyto A , Sokura M , Juutilainen J , Naarala J ((2008) ) Radiofrequency radiation does not significantly affect ornithine decarboxylase activity, proliferation, orcaspase-3 activity of fibroblasts in different physiologic conditions. Int J Radiation Biol 84: , 727–733. |
[46] | Billaudel B , Taxile M , Ruffie G , Veyret B , Lagroye I ((2009) ) Effects of exposure to DAMPS and GSM signals on ornithine decarboxylase (ODC) activity: I. L-929 mouse fibroblasts. Int J Radiation Biol 85: , 510–518. |
[47] | Krewski D , Glickman B , Habash R , Habbick B , Lotz W , Mandeville R , Prato F , Salem T , Weaver D ((2007) ) Recent advances in research on radiofrequency fields and health: 2001-2003. J Toxicol Environ Health B Crit Rev 10: , 287–318. |
[48] | Verschaeve L , Juutilainen J , Lagroye I , Miyakoshi J , Saunders R , de Seze R , Tenforde T , van Rongen E , Veyret B , Xu Z ((2010) ) In vitro and in vivo genotoxicity of radiofrequency fields. Mutat Res 705: , 252–268. |
[49] | Leikind B ((2010) ) Do cell phones cause cancer? The Skeptic 15: . |
[50] | Sengwa R , Kaur K ((1999) ) Microwave absorption in oligomers of ethylene glycol. Ind J Biochem Biophys 36: , 325–329. |
[51] | Blank M , Goodman R ((2004) ) Initial interactions in electromagnetic field-induced biosynthesis. J Cell Physiol 199: , 359–363. |
[52] | Gerner C , Haudek V , Schandl U , Bayer E , Gundacker N , Hutter H , Mosgoeller W ((2010) ) Increased protein synthesis by cells exposed to a 1,800-MHz radio-frequency mobile phone electromagnetic field, detected by proteome profiling. Int Arch Occup Environ Health 83: , 691–702. |
[53] | Sienkiewicz Z , Blackwell R , Haylock R , Saunders R , Cobb B ((2000) ) Low-level exposure to pulsed 900 MHz microwave radiation does not cause deficits in the performance of a spatial learning task in mice. Bioelectromagnetics 21: , 151–158. |
[54] | Dubreil D , Jay T , Edeline J ((2002) ) Does head-only exposure to GSM-900 electroamgentic fields affect the performance of rats in spatial learning tasks? Behav Brain Res 129: , 203–210. |
[55] | Dubreil D , Jay T , Edeline J ((2003) ) Head-only exposure to GSM 900-MHz electromagnetic fields does not alter rat’s memory in spatial and non-spatial tasks. Behav Brain Res 145: , 51–61. |
[56] | Ammari M , Jacquet A , Lecomte A , Sakly M , Abdelmelek H , de Seze R ((2008) ) Effect of head-only sub-chronic and chronic exposure to 900-MHz GSM electromagnetic fields on spatial memory in rats. Brain Injury 22: , 1021–1029. |
[57] | Hao D , Yang L , Chen S , Tong J , Tian Y , Su B , Wu S , Zeng Y ((2013) ) Effects of long-term electromagnetic field exposure on spatial learning and memory in rats. Neurol Sci 34: , 157–164. |
[58] | Klose M , Grote K , Spathmann O , Streckert J , Clemens M , Hansen V , Lerchl A ((2014) ) Effects of early-onset radiofrequency electromagnetic field exposure (GSM 900 MHz) on behavior and memory in rats. Radiat Res 182: , 435–447 . |
[59] | Kumlin T , Livonen H , Miettinen P , Juvonen A , van Groen T , Puranen L , Pitkaaho R , Juutilainen J , Tanila H ((2007) ) Mobile phone radiation and the developing brain: Behavioral and morphological effects in juvenile rats. Radiat Res 168: , 471–479. |
[60] | Nittby H , Grafstrom G , Tian DP , Malmgren L , Brun A , Persson BR , Salford LG , Eberhardt J ((2008) ) Cognitive impairment in rats after long-term exposure to GSM-900 mobile phone radiation. Bioelectromagnetics 29: , 219–232. |
[61] | Fragopoulou AF , Miltiadous P , Stamatakis A , Stylianopoulou F , Koussoulakos SL , Margaritis LH ((2010) ) Whole body exposure with GSM 900MHz affects spatial memory in mice. Pathophysiology 17: , 179–187. |
[62] | Ntzouni M , Stamatakis A , Stylianopoulou F , Margaritis L ((2011) ) Short-term memory in mice is affected by mobile phone radiation. Pathophysiology 18: , 193–139. |
[63] | Jiang D , Li J , Zhang J , Xu S , Kuang F , Lang H , Wang Y , An G , Li J , Guo G ((2013) ) Electromagnetic pulse exposure induces overexpression of beta-amyloid protein in rats. Arch Med Res 44: , 178–184. |
[64] | Tang J , Zhang Y , Yang L , Chen Q , Tan L , Zuo S , Feng H , Chen Z , Zhu G ((2015) ) Exposure to 900MHz electromagnetic fields activates the mkp-1/ERK pathway and causes blood-brain barrier damage and cognitive impairment in rats. Brain Res 1601: , 92–101. |
[65] | Johansson O ((2009) ) Disturbance of the immune system by electromagnetic fields – A potentially underlying cause for cellular damage and tissue repair reduction which could lead to disease and impairment. Pathophysiology 16: , 157–177. |
[66] | Gatta L , Pinto R , Ubaldi V , Pace L , Galloni P , Lovisolo G , Marino C , Pioli C ((2003) ) Effects of in vivo exposure to GSM-modulated 900MHz radiation on mouse peripheral lymphocytes. Radiat Res 160: , 600–605. |
[67] | Nasta F , Prisco M , Pinto R , Lovisolo G , Marino C , Pioli C ((2006) ) Effects of GSM-modulated radiofrequency electromagnetic fields on B-cell peripheral differentiation and antibody production. Radiat Res 165: , 664–670. |
[68] | Rosado M , Nasta F , Prisco M , Lovisolo G , Marino C , Pioli C ((2014) ) Effects of GSM-modulated 900MHz radiofrequency electromagnetic fields on the hematopoietic potential of mouse bone marrow cells. Bioelectromagnetics 35: , 559–567. |
[69] | Irmak M , Fadillioglu E , Gulec M , Erdogan H , Yagmurca M , Akyol O ((2002) ) Effects of electromagnetic radiation from a cellular telephone on the oxidant and antioxidant levels in rabbits. Cell Biochem Funct 20: , 279–283. |
[70] | Zeni O , Di Pietro R , d’Ambrosio G , Massa R , Capri M , Naarala J , Juutilainen J , Scarfi M ((2007) ) Formation of reactive oxygen species in L929 cells after exposure to 900MHz RF radiation with and without co-exposure to 3-chloro-4-(dichloromethyl)-5-hydroxy-2(5H)-furanone. Radiat Res 167: , 306–311. |
[71] | Hook G , Spitz D , Sim J , Higashikubo R , Baty J , Moros E , Roti Roti J ((2004) ) Evaluation of parameters of oxidative stress after in vitro exposure to FMCW- and CDMA-modulated radiofrequency radiation fields. Radiat Res 162: , 497–504. |
[72] | Finnie J , Blumbergs P , Manavis J , Utteridge T , Gebski V , Davies R , Vernon-Roberts B , Kuchel T ((2002) ) Effect of long-term mobile communication microwave exposure on vascular permeability in mouse brain. Pathology 34: , 344–347. |
[73] | Grafstrom G , Nittby H , Brun A , Malmgren L , Persson B , Salford L , Eberhardt J ((2008) ) Histopathological examinations of rat brains after long-term exposure to GSM-900 mobile phone radiation. Brain Res Bull 77: , 257–263. |
[74] | Valberg P , van Deventer T , Repacholi M ((2007) ) Workgroup report: Base stations and wireless networks-radiofrequency (RF) exposures and health consequences. Environ Health Perspect 115: , 416–424. |
[75] | Habash R , Elwood J , Krewski D , Lotz W , McNamee J , Prato F ((2009) ) Recent advances in research on radiofrequency fields and health: 2004-2007. J Toxicol Environ Health B Crit Rev 12: , 250–288. |
[76] | Moussa M ((2011) ) Review on health effects related to mobile phones. Part II: Results and conclusions. J Egypt Public Health Assoc 86: , 79–89. |
[77] | Kwon M , Hamalainen H ((2011) ) Effects of mobile phone electromagnetic fields: Critical evaluation of behavioral and neurophysiological studies. Bioelectromagnetics 32: , 253–272. |
[78] | Barth A , Ponocny I , Gnambs T , Winker R ((2012) ) No effects of short-term exposure to mobile phone electromagnetic fields on human cognitive performance: A meta-analysis. Bioelectromagnetics 33: , 159–165. |
[79] | Valentini E , Ferrara M , Presaghi F , De Gennaro L , Curcio G ((2010) ) Systemic review and meta-analysis of psychomotor effects of mobile phone electromagnetic fields. Occup Environ Med 10: , 708–716. |
[80] | Preece A , Iwi G , Davies-Smith A , Wesnes K , Butler S , Lim E , Varey A ((1999) ) Effect of a 915MHz simulated mobile phone signal on cognitive function in man. Int J Radiat Biol 75: , 447–456. |
[81] | Haarala C , Ek M , Bjornberg L , Laine M , Revonsuo A , Koivisto M , Hamalainen H ((2004) ) 902MHz mobile phone does not affect short term memory in humans. Bioelectromagnetics 25: , 452–456. |
[82] | Cinel C , Boldini A , Russo R , Fox E ((2007) ) Effects of mobile phone electromagnetic fields on an auditory order threshold task. Bioelectromagnetics 28: , 493–496. |
[83] | Haarala C , Takio F , Rintee T , Laine M , Koivisto M , Revonsuo A , Hamalaminen H ((2007) ) Pulsed and continuous wave mobile phone exposure over left versus right hemisphere: Effects on human cognitive function. Bioelectromagnetics 28: , 289–295. |
[84] | Regel S , Gottselig J , Schuderer J , Tinguely G , Retey J , Kuster N , Landolt H , Achermann P ((2007) ) Pulsed radio frequency radiation affects cognitive performance and the waking electroencephalogram. Neuroreport 18: , 803–807. |
[85] | Curcio G , Valentini E , Moroni F , Ferrara M , De Gennaro L , Bertini M ((2008) ) Psychomotor performance is not influenced by brief repeated exposures to mobile phones. Bioelectromagnetics 29: , 237–241. |
[86] | Schmi M , Loughran S , Regel S , Murbach M , Bratic-Grunauer A , Rusterholz T , Bersagliere A , Kuster N , Achermann P ((2012) ) Sleep EEG alternations: Effects of different pulse-modulated radio frequency electromagnetic fields. J Sleep Res 21: , 50–58. |
[87] | Besset A , Espa F , Dauvilliers Y , Billiard M , de Seze R ((2005) ) No effect on cognitive function from daily mobile phone use. Bioelectromagnetics 26: , 102–108. |
[88] | Fritzer G , Goder R , Friege L , Wachter J , Hansen V , Hinze-Selch D , Aldenhoff J ((2007) ) Effects of short- and long-term pulsed radiofrequency electromagnetic fields on night sleep and cognitive functions in healthy subjects. Bioelectromagnetics 28: , 316–325. |
[89] | Sauter C , Dorn H , Bahr A , Hansen M , Peter A , Bajbouj M , Danker-Hopfe H ((2011) ) Effects of exposure to electromagnetic fields emitted by GSM 900 and WCDMA mobile phones on cognitive function in young male subjects. Bioelectromagnetics 32: , 179–190. |
[90] | Arns M , Van Luijtelaar G , Sumich A , Hamilton R , Gordon E ((2007) ) Electroencephalographic, personality, and executive function measures associated with frequent mobile phone use. Int J Neurosci 117: , 1341–1360. |
[91] | Schuz J , Waldemar G , Olsen J , Johansen C ((2009) ) Risks for central nervous system diseases among mobile phone subscribers: A Danish retrospective cohort study. PLoS One 4: , e4389. |
[92] | Guerriero F , Botarelli E , Mele G , Polo L , Zoncu D , Renati P , Sgarlata C , Rollone M , Ricevuti G , Maurizi N , Francis M , Rondanelli M , Perna S , Guido D , Mannu P ((2015) ) An innovative intervention for the treatment of cognitive impairment-Emisymmetric bilateral stimulation improves cognitive functions in Alzheimer’s disease and mild cognitive impairment: An open-label study. Neuropsychiatr Dis Treat 11: , 2391–2404. |
[93] | Ferreri F , Curcio G , Pasqualetti P , De Gennaro L , Fini R , Rossini P ((2006) ) Mobile phone emissions and human brain excitability. Ann Neurol 60: , 188–196. |
[94] | Croft R , Chandler J , Burgess A , Barry R , Williams J , Clark A ((2002) ) Acute mobile phone operation affects neural function in humans. Clin Neurophysiol 113: , 1623–1632. |
[95] | Croft R , Hamblin D , Spong J , Wood A , McKenzie R , Stough C ((2008) ) The effect of mobile phone electromagnetic fields on the alpha rhythm of human electroencephalogram. Bioelectromagnetics 29: , 1–10. |
[96] | Curcio G , Ferrara M , Moroni F , D’Inzeo G , Bertini M , De Gennaro L ((2005) ) Is the brain influenced by a phone call? An EEG study of resting wakefulness. Neurosci Res 53: , 265–270. |
[97] | Volkow N , Tomasi D , Wang G , Vaska P , Fowler J , Telang F , Alexoff D , Logan J , Wong C ((2011) ) Effects of cell phone radiofrequency signal exposure on brain glucose metabolism. JAMA 305: , 808–813. |
[98] | Wessapan T , Srisawatdhisukul S , Rattanadecho P ((2012) ) Specific absorption rate and temperature distributions in human head subjected to mobile phone radiation at difference frequencies. Int J Heat Mass Transf 55: , 347–359. |
[99] | Wang J , Fujiwara O ((1999) ) FDTD computation of temperature rise in the human head for portable telephones. IEEE Trans Microw Theory Tech 47: , 1528–1534. |
[100] | van Leeuwen G , Lagendijk J , Van Leersum B , Zwamborn A , Hornsleth S , Kottee A ((1999) ) Calculation of change in brain temperatures due to exposure to a mobile phone. Phys Med Biol 44: , 2367–2379. |
[101] | Hardell L , Sage C ((2008) ) Biological effects from electromagnetic field exposure and public exposure standards. Biomed Pharmacother 62: , 104–109. |
[102] | Hardell L , Carlberg M , Sodergvist F , Hansson-Mild K ((2008) ) Meta-analysis of long-term mobile phone use and the association with brain tumours. Int J Oncology 32: , 1097–1103. |
[103] | Hardell L , Carlberg M , Hansson-Mild K ((2009) ) Epidemiological evidence for an association between use of wireless phones and tumor diseases. Pathophysiology 16: , 113–122. |
[104] | Khurana V , Teo C , Kundi M , Hardell L , Carlberg M ((2009) ) Cell phones and brain tumors: A review including the long-term epidemiologic data. Surg Neurol 72: , 205–214. |
[105] | Hardell L , Carlberg M , Sodergvist F , Mild K ((2013) ) Case-control study of the association between malignant brain tumours diagnosed between 2007 and 2009 and mobile and cordless phone use. Int J Oncol 43: , 1833–1845. |
[106] | Hardell L , Carlberg M ((2015) ) Mobile phone and cordless phone use and the risk for glioma – Analysis of pooled case-control studies in Sweden 1997-2003 and 2007-2009. Pathophysiology 22: , 1–13. |
[107] | Baan R , Grosse Y , El Ghissassi F , Lauby-Secretan B , Bouvard V , Benbrahim-Tallaa L , Guha N , Freeman C , Galichet L , Straif K , WHO-IARCWorking Group ((2011) ) Carcinogenicity of radiofrequency electromagnetic fields. Lancet Oncol 12: , 624–626. |
[108] | Nawrot P , Jordan S , Eastwood J , Rotstein J , Hugenholtz A , Feeley M ((2003) ) Effects of caffeine on human health. Food Addit Contam 20: , 1–30. |
[109] | Higdon J , Frei B ((2006) ) Coffee and health: A review of recent human research. Crit Rev Food Sci Nutr 46: , 101–123. |
[110] | Vigayalaxmi Prihoda TJ ((2012) ) Genetic damage in human cells exposed to non-ionizing radiofrequency fields: A meta-analysis of the data from 88 publications (1990-2011). Mutat Res 749: , 1–16. |
[111] | Wiedemann P , Boerner F , Repacholi M ((2014) ) Do people understand IARC’s 2B categorization of RF fields from cell phones?. Bioelectromagnetics 35: , 373–378. |
[112] | Hsu MH , Syed-Abdul S , Scholl J , Jian W , Lee P , Iqbal U , Li Y ((2013) ) The incidence rate and mortality of malignant brain tumors after 10 years of intensive cell phone use in Taiwan. Eur J Cancer Prev 22: , 596–598. |
[113] | INTERPHONE Study Group (48 collaborators) ((2010) ) Brain tumour risk in relation to mobile telephone use: Results of the INTERPHONE international case-control study. Int J Epidemiol 39: , 675–694. |
[114] | Swerdlow A , Feychting M , Green A , Leeka Kheifets L , Savit D ((2011) ) Mobile phones, brain tumors, and the interphone study: Where are we now? Environ Health Perspect 119: , 1534–1538. |
[115] | Larjavaara S , Schüz J , Swerdlow A , Feychting M , Johansen C , Lagorio S , Tynes T , Klaeboe L , Tonjer SR , Blettner M , Berg-Beckhoff G , Schlehofer B , Schoemaker M , Britton J , Mäntylä R , Lönn S , Ahlbom A , Flodmark O , Lilja A , Martini S , Rastelli E , Vidiri A , Kähärä V , Raitanen J , Heinävaara S , Auvinen A ((2011) ) Location of gliomas in relation to mobile telephone use: A case-case and case-specular analysis. Am J Epidemiol 174: , 2–11. |
[116] | Frei P , Poulsen A , Johansen C , Olsen J , Steding-Jessen J , Schuz J ((2011) ) Use of mobile phones and risk of brain tumours: Update of Danish cohort study. BMJ 343: , d6387. |
[117] | Benson V , Pirie K , Schuz J , Reeves G , Berai V , Green J , Million Women Study Collaborators ((2013) ) Mobile phone use and risk of brain neoplasms and other cancers: Prospective study. Int J Epidemiol 42: , 792–802. |
[118] | Benson V , Pirie K , Schuz J , Reeves G , Beral V , Green J ((2014) ) Authors’ response to: The case of acoustic neuroma: Comment on mobile phone use and risk of brain neoplasms and other cancers. Int J Epidemiol 43: , 275. |
[119] | Barchana M , Margaliot M , Liphshitz I ((2012) ) Changes in brain glioma incidence and laterality correlates with use of mobile phones – a nationwide population based study in Israel. Asian Pac J Cancer Prev 13: , 5857–5863. |
[120] | Lagorio S , Roosli M ((2014) ) Mobile phone use and risk of intracranial tumors: A consistency analysis. Bioelectromagnetics 35: , 79–90. |
[121] | Inskip P , Hoover R , Devesa S ((2010) ) Brain cancer incidence trends in relation to cellular telephone use in the United States. Neuro Oncol 12: , 1147–1151. |
[122] | Little M , Rajaraman P , Curtis R , Devesa S , Inskip P , Check D , Linet M ((2012) ) Mobile phone use and glioma risk: Comparison of epidemiological study results with incidence trends in the United States. BMJ 344: , e1147. |
[123] | Ahlbom A , Feychting M ((2011) ) Mobile telephones and brain tumours. BMJ 343: , d6605. |
[124] | Deltour I , Auvinen A , Feychting M , Johansen C , Klaeboe L , Sankila R , Schuz J ((2012) ) Mobile phone use and incidence of glioma in the Nordic countries 1979-2008: Consistency check. Epidemiology 23: , 301–307. |
[125] | Costa FP , de Oliveira AC , Meirelles R , Machado MC , Zanesco T , Surjan R , Chammas MC , de Souza Rocha M , Morgan D , Cantor A , Zimmerman J , Brezovich I , Kuster N , Barbault A , Pasche B ((2011) ) Treatment of advanced hepatocellular carcinoma with very low levels of amplitude-modulated electromagnetic fields. Br J Cancer 105: , 640–648. |
[126] | Zimmerman JW , Jimenez H , Pennison M , Brezovich I , Morgan D , Mudry A , Costa FP , Barbault A , Pasche B ((2013) ) Targeted treatment of cancer with radiofrequency electromagnetic fields amplitude-modulated at tumor-specific frequencies. Chin J Cancer 32: , 573–581. |
Figures and Tables
Fig.1
EMF treatment, begun in young adulthood, protects AD mice (Tg) mice from cognitive impairment and improves basic memory of normal mice. Cognitive interference testing at 4-5 months (A) and 6-7 months (B) into EMF treatment revealed overall [Tg and non-Tg(NT) animals combined] cognitive benefits at the initial test point and cognitive protection of Tg mice at the later test point during the first of two test Blocks. C) Proactive interference testing during Block 2 revealed both overall benefit (at 4-5 M) and cognitive protection of Tg mice (at 6-7M). *p < 0.05 versus other group(s) at same time point; †p < 0.05 versus Tg/EMF group. D) Normal (NT) mice at 6-7 months into EMF treatment showed superior Y-maze spontaneous alternation. *p < 0.05 versus all other groups.
![EMF treatment, begun in young adulthood,
protects AD mice (Tg) mice from cognitive impairment and improves basic memory of normal mice. Cognitive
interference testing at 4-5 months (A) and 6-7 months (B) into EMF treatment revealed overall [Tg and non-Tg(NT)
animals combined] cognitive benefits at the initial test point and cognitive protection of Tg mice at the later
test point during the first of two test Blocks. C) Proactive interference testing during Block 2 revealed both
overall benefit (at 4-5 M) and cognitive protection of Tg mice (at 6-7M). *p < 0.05 versus other group(s) at same time point; †p < 0.05 versus Tg/EMF group. D) Normal (NT) mice at 6-7 months into EMF treatment showed superior Y-maze spontaneous alternation. *p < 0.05 versus all other groups.](https://ip.ios.semcs.net:443/media/jad/2016/53-3/jad-53-3-jad160165/jad-53-jad160165-g001.jpg)
Fig.2
At 8 months into EMF treatment, cognitively-impaired AD mice (Tg) mice exhibited cognitive benefits and reduced brain Aβ deposition. A) Cognitive interference testing revealed Tg/EMF mice as vastly superior to Tg controls in 3-trial recall and retroactive interference performance. Even non-transgenic (NT) mice receiving EMF exposure showed better recall performance than NT controls, particularly early in recall testing. The final 2-day block of testing is shown from four days of testing. Upper graph: *p < 0.025 versus control; Lower graph: *p < 0.05 or higher level of significance versus control. B) Long-term EMF treatment significantly reduced total Aβ deposition in entorhinal cortex and hippocampus of Tg mice. Photomicrographic examples of typical Aβ immunostained-plaques from Tg and Tg/EMF mice are provided. *p < 0.02 versus Tg control group. Scale bar = 50 μm.
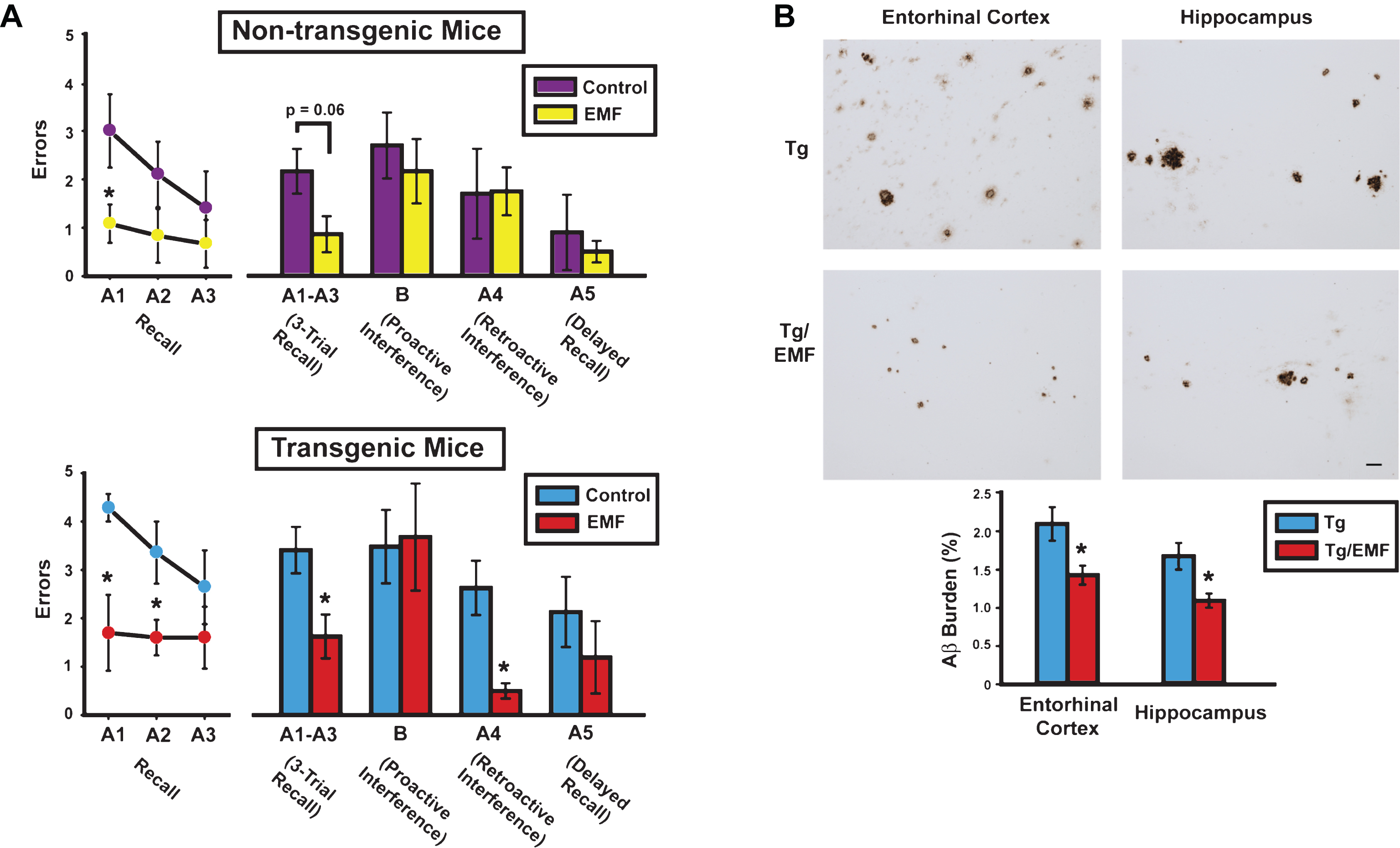
Fig.3
In Vitro EMF treatment of hippocampal homogenates from aged Tg mice results in progressively decreased Aβ oligomerization between 3 and 6 days into treatment. Western blots display the 80 kDa Aβ oligomer on top and the β-Actin protein control on bottom. Left panel shows non-treated Tg controls of progressive Aβ aggregation, while right panel shows the same homogenates treated with EMF through 6 days.
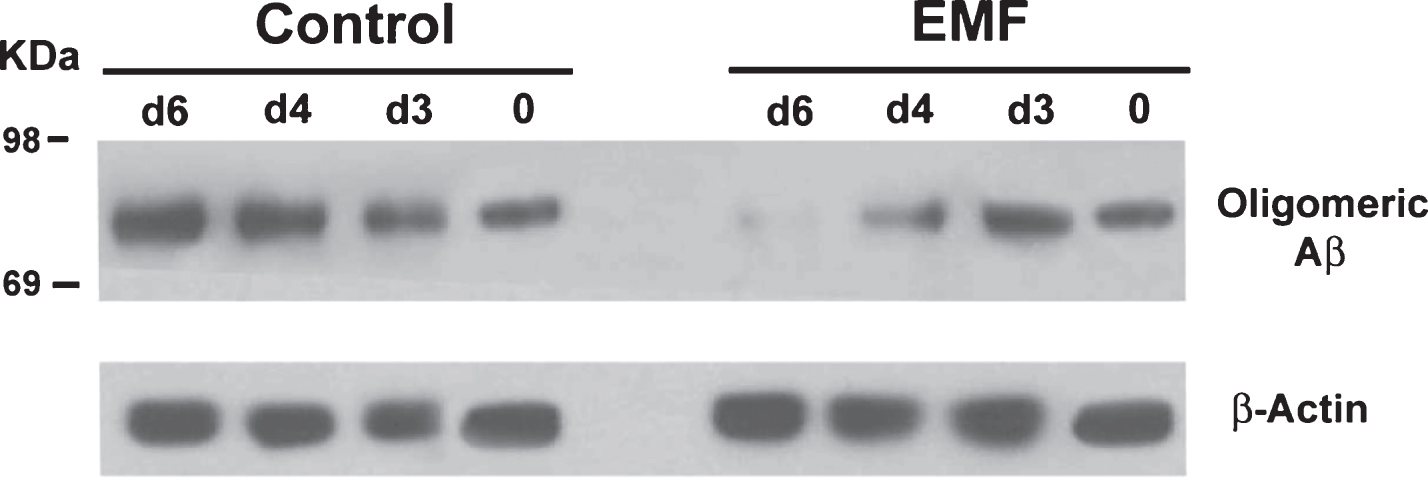
Fig.4
Long-term EMF treatment of aged AD (Tg) mice dramatically increased soluble Aβ1–40 levels in mitochondria preparations from both cortex and hippocampus. These 5–10x increases in mitochondrial Aβ are consistent with an EMF-induced disaggregation of toxic Aβ oligomers associated with intraneuronal mitochondria.
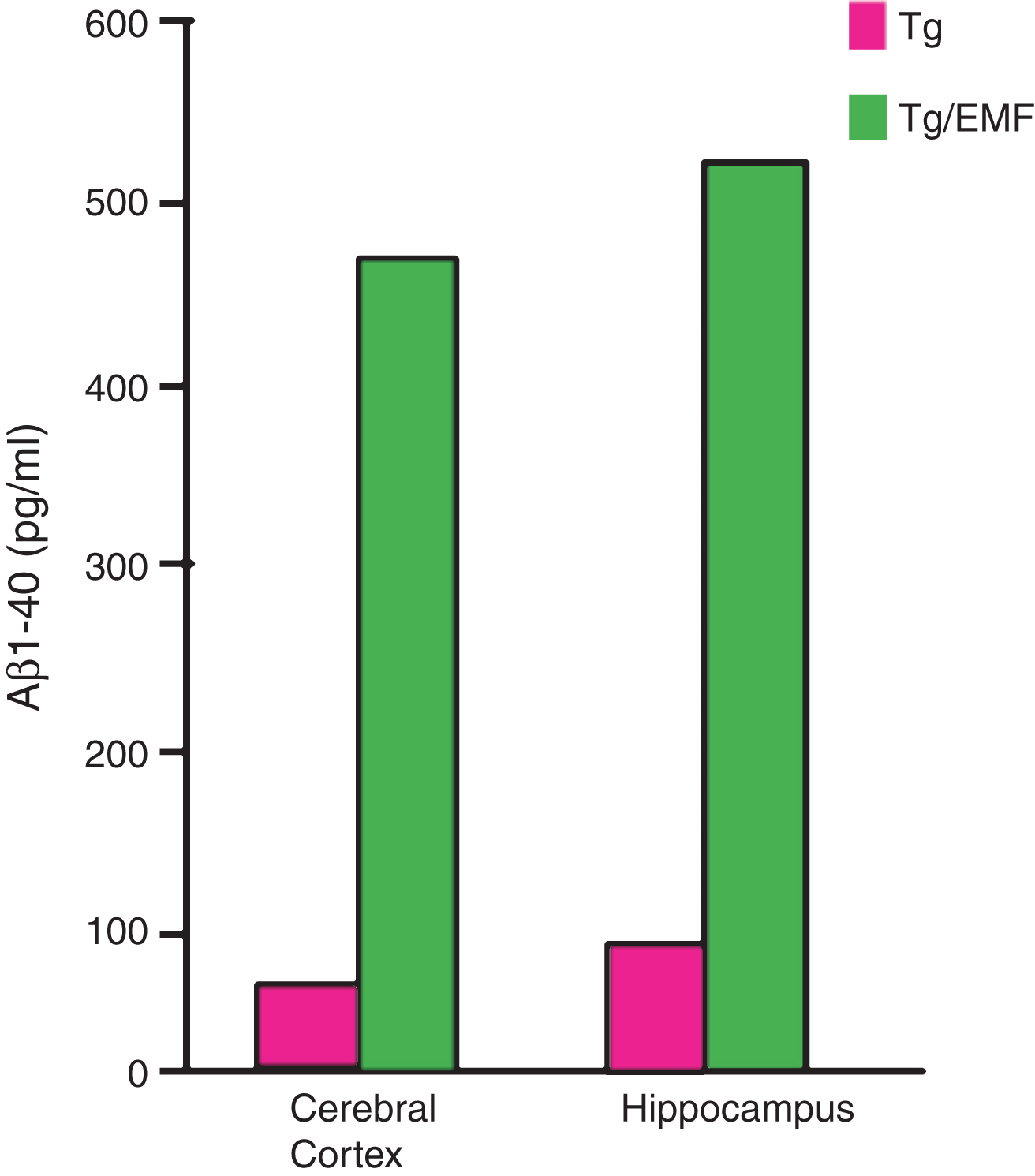
Fig.5
EMF treatment greatly enhances mitochondrial function within both cerebral cortex and hippocampus of aged AD (Tg) mice. Shown are percent changes across six measures of mitochondrial function, wherein 50–150% enhancements were induced by EMF treatment.
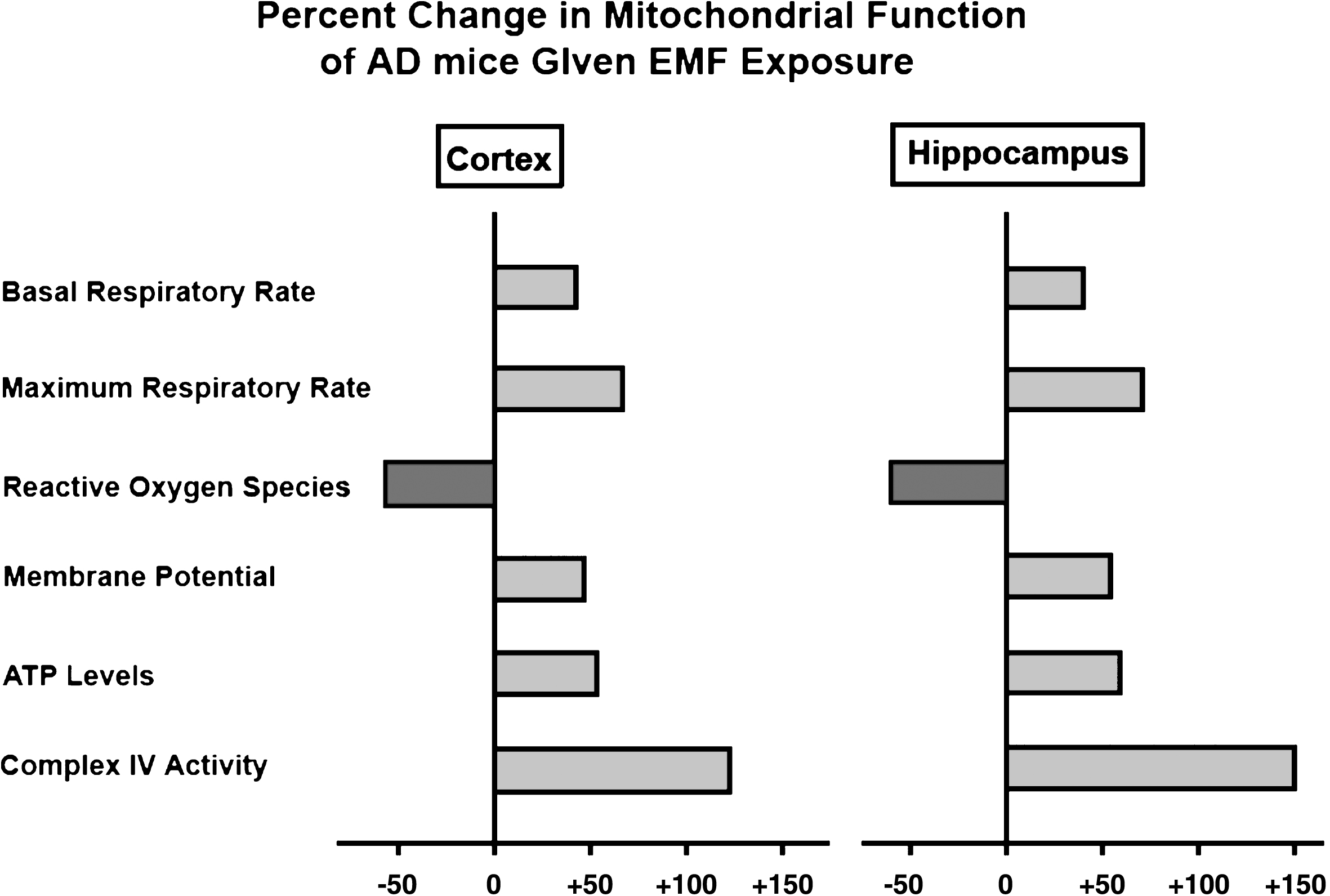
Fig.6
TEMT increases neuronal activity in entorhinal cortex of aged AD mice, as indicated by the number of cFos-stained neurons. Note increased number of active neurons in AD mice given long-term TEMT (right) compared to control AD mice not given TEMT (left). For AD and normal mice, average number of c-Fos-stained neurons in entorhinal cortex from five representative fields increased from 83 neurons per field in controls to 100 neurons per field after TEMT (↑21%; p < 0.02).
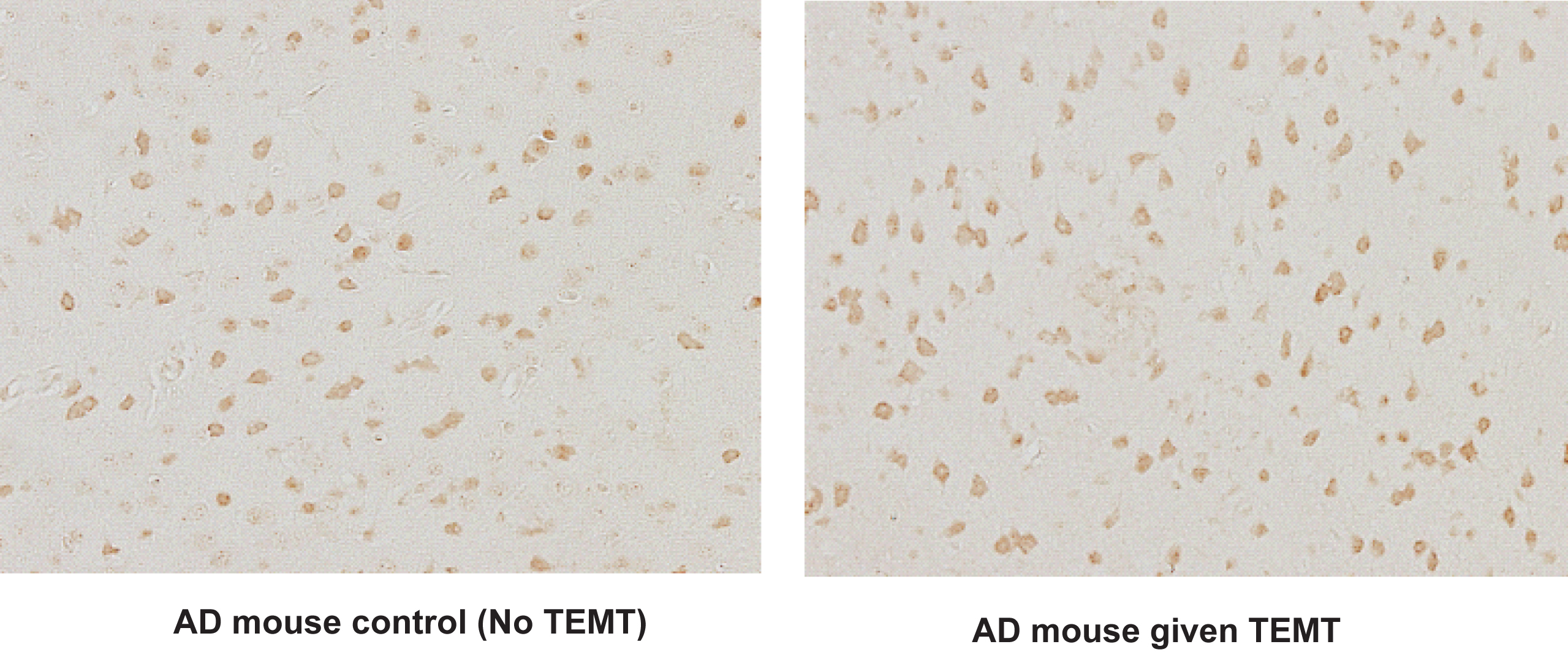
Fig.7
An FDTD computer simulation showing deep electric field penetration by an excitation element (one of eight elements) positioned on the cranium. Deep brain regions, such as the hippocampus and entorhinal cortex, are easily affected by this single element.
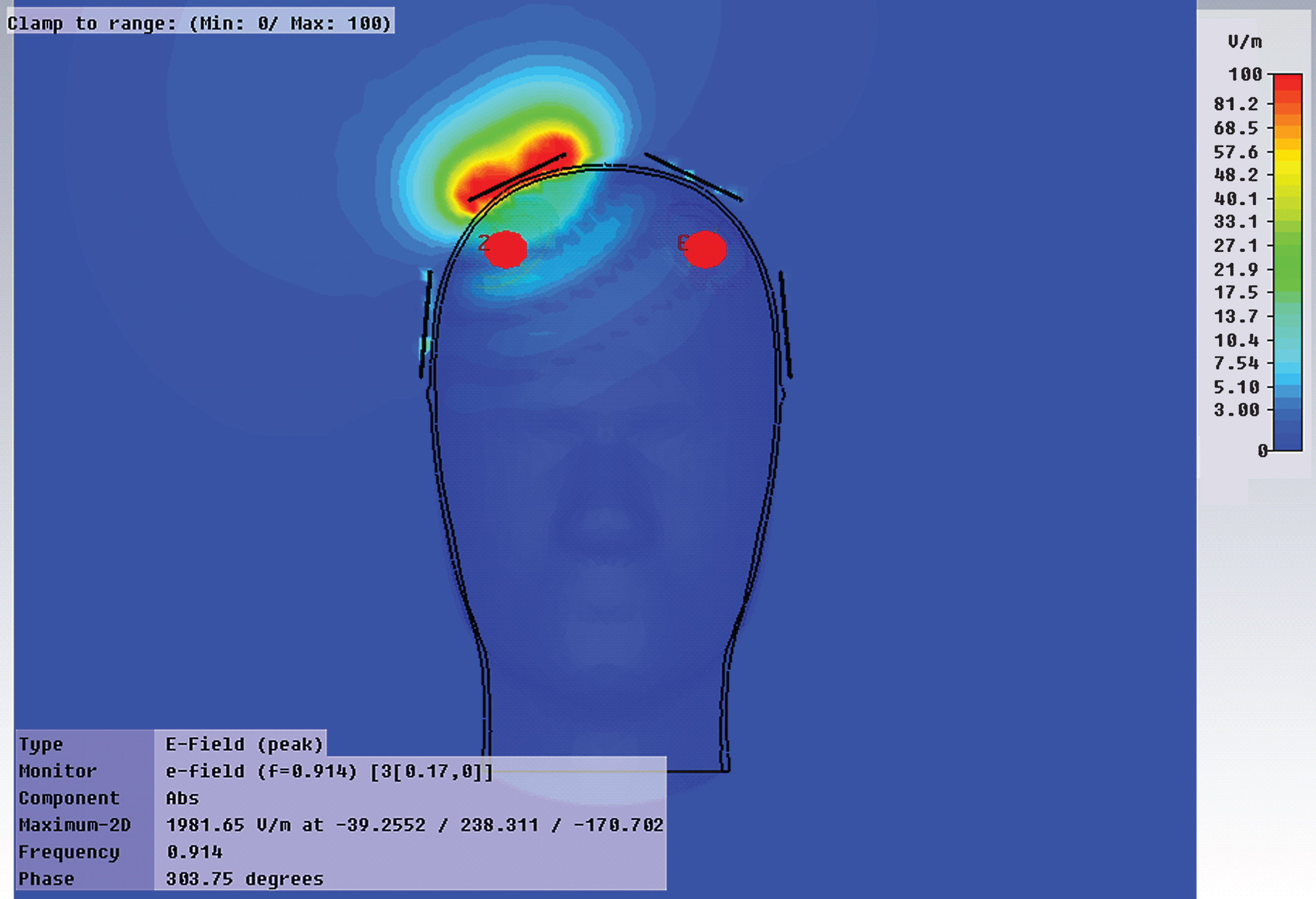
Fig.8
A, B) There are no changes in brain temperature of AD transgenic mice (both APPsw and APPsw+PS1) and normal mice (NT) during acute EMF treatment (two 1-h treatments during a single day) compared naïve Tg and NT mice of various ages. C) The strong correlation between brain and body temperature, with brain temperature typically being 0.3-0.4°C cooler.
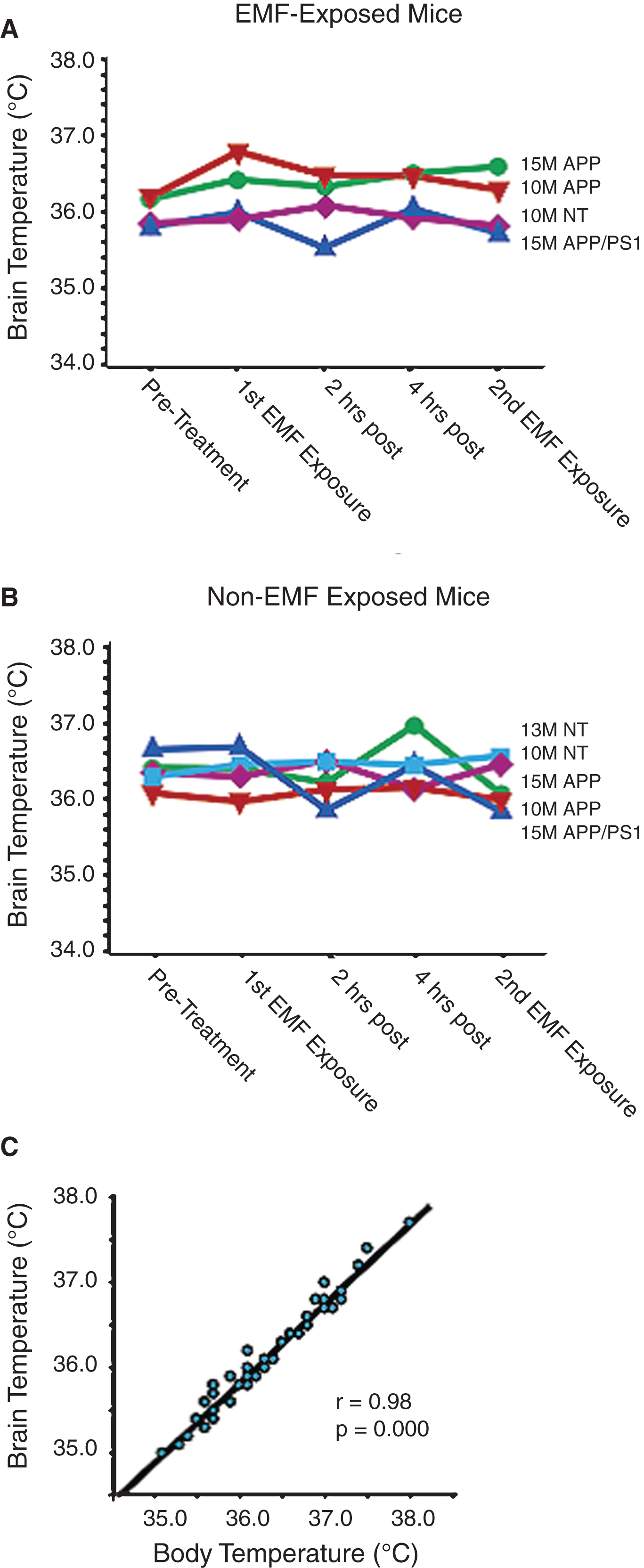
Fig.9
Body and brain temperature measurements for AD mice recorded prior to the start of EMF treatment (control), as well as at 5 days and 12 days into EMF treatment. For both control and treatment time points, there were no differences between EMF-treated and control AD mice for either body or brain temperatures. As well, no significant differences in OFF versus ON temperatures were evident in EMF-treated AD mice.