Advancing Clinical Trial Design for Non-Muscle Invasive Bladder Cancer
Abstract
BACKGROUND:
Despite recent drug development for non-muscle invasive bladder cancer (NMIBC), few therapies have been approved by the US Food and Drug Administration (FDA), and there remains an unmet clinical need. Bacillus Calmette-Guerin (BCG) supply issues underscore the importance of developing safe and effective drugs for NMIBC.
OBJECTIVE:
On November 18–19, 2021, the FDA held a public virtual workshop to discuss NMIBC research needs and potential trial designs for future development of effective therapies.
METHODS:
Representatives from various disciplines including urologists, oncologists, pathologists, statisticians, basic and translational scientists, and the patient advocacy community participated. The workshop format included invited lectures, panel discussions, and opportunity for audience discussion and comment.
RESULTS:
In a pre-workshop survey, 92% of urologists surveyed considered the development of alternatives to BCG as a high drug development priority for BCG-naïve high-risk patients. Key topics discussed included definitions of disease states; trial design for BCG-naïve NMIBC, BCG-unresponsive carcinoma in situ, and BCG-unresponsive papillary carcinoma; strengths and limitations of single-arm trial designs; assessing patient-reported outcomes; and considerations for assessing avoidance of cystectomy as an efficacy measure.
CONCLUSIONS:
The workshop discussed several important opportunities for trial design refinement in NMIBC. FDA encourages sponsors to meet with the appropriate review division to discuss trial design proposals for NMIBC early in drug development.
INTRODUCTION
Bladder cancer is the sixth most common cancer diagnosed in the United States (US), causing an estimated 17,100 deaths in 2022 [1]. Approximately 75% of cases initially present with non-muscle invasive bladder cancer (NMIBC). The initial standard of care treatment for NMIBC is transurethral resection of bladder tumor (TURBT) with or without adjuvant intravesical chemotherapy or Bacillus Calmette-Guerin (BCG), according to tumor risk stratification [2, 3]. A US Food and Drug Administration (FDA) - American Urological Association (AUA) workshop held in 2013 discussed the need for more effective drugs in NMIBC and a potential drug development path forward for BCG-unresponsive NMIBC [4]. However, despite multiple subsequent trials investigating various agents using various administration and drug delivery methods, only two agents have been approved for NMIBC in the last 20 years in the US [5, 6]. Additionally, intermittent global BCG supply shortages have become a more continuous and severe issue since 2017 [7].
On November 18–19, 2021, the FDA held a public workshop with the purpose of generating further discussion on the state of treatment of NMIBC and on potential future trial designs. Representatives from the academic urology, oncology, basic and translational science, statistical, and patient advocacy communities were invited to participate in order to include a broad range of perspectives in advancing clinical trial design in NMIBC. The workshop format included invited lectures, panel discussions, and opportunity for audience discussion and comment.
This report summarizes key discussion points from the workshop and further discusses several of these issues.
BCG SUPPLY ISSUES
BCG is a cancer immunotherapy that has been used for treatment of NMIBC since 1977 [8, 9], and is FDA-approved for patients with the following categories of NMIBC: carcinoma in situ (CIS) and/or high-risk Ta/T1 defined as 1) Ta high grade (HG)>3 cm or multifocal or recurrent, or 2) T1 HG. Standard treatment with BCG involves weekly induction for 6 weeks (up to 2 consecutive inductions without complete response) and maintenance to 3 years with 3 weekly instillations at months 3, 6 and every 6 months to 3 years [10, 11].
There have been recent issues affecting the supply and availability of BCG in the US. Two substrain or seed lot-derived BCGs were approved and previously available in the US: the Connaught and TICE strains. However, production of the Connaught BCG strain (Sanofi Pasteur) paused in 2012, then stopped permanently in 2017 [12]. TICE BCG is now manufactured in a single plant in the US for global distribution to 70 countries. Although production capacity for the TICE strain has been increased by the manufacturer, demand still exceeds supply. In January 2021, the manufacturer announced plans to build a new plant that is expected to triple production of TICE BCG. The project is expected to be completed by late 2026 [13].
In response to the shortage situation, a joint statement addressing BCG utilization was issued in 2019 on behalf of multiple professional and advocacy organizations [14]. This statement provided several recommendations for patient management during BCG shortages, including prioritizing BCG use for patients with high-risk NMIBC and considering dose reductions of BCG. Additionally, intravesical chemotherapy (often doublets), are sometimes used for patients with intermediate and high-risk disease during shortage situations [15, 16].
Because of the recent BCG supply issues, many practices have had periods of limited or no access to BCG over the past several years. Multiple global BCG shortages have occurred in the past, associated with increased recurrence and progression rates [17]. In preparation for this workshop, two of the authors (SL and NH) sent a survey to urologists identified by Bladder Cancer Advocacy Network (BCAN) (n = 94), Society of Urologic Oncology (n = 1010), and Large Urology Group Practice Association (n = 2200) as routinely treating patients with NMIBC, to gain understanding of the reality of clinical practice in the setting of BCG shortage. Of 255 surveys returned, community urologists represented 60% of respondents, academic urologists represented 32%, and the remaining 10% were hybrid or other.
Survey results demonstrated that the majority of urologists were able to give a full induction course of BCG without reducing the dose of each treatment. The majority were able to give maintenance BCG to patients with high-risk NMIBC for the first 12 months, while 58% gave less than 3 years of maintenance due to the BCG shortage. One-third of academic and two-thirds of community urologists favored BCG over chemotherapy for patients with intermediate risk disease. Although the need to borrow BCG was reported infrequently, 39% reported adverse outcomes as a result of the BCG shortage.
An overwhelming majority (92%) considered the development of non-BCG–based alternative therapies as a high priority for BCG-naïve high-risk patients while 24% felt the BCG shortage was affecting their ability to enroll patients in clinical trials in the BCG-naïve, exposed or unresponsive disease states. These results highlighted the potential benefit to patients that drug development in NMIBC, even in the BCG-naïve setting, could have.
Active Clinical Trials in the BCG-Naïve NMIBC Setting
Clinical trials represent an opportunity to expand treatment options for patients. Table 1 lists selected currently enrolling trials identified via ClinicalTrials.gov for patients with BCG-Naïve NMIBC, indicating active drug development in this setting.
Table 1
Selected Active Phase 2 and 3 Clinical Trials in the BCG-Naïve NMIBC Settinga
| Trial number (Name) | Intervention Treatment | Control | Enrollmentb | Primary Endpoint | Study Start Datec |
| NCT02948543 (BCG+MM) | Mitomycin (IVES)+BCG | BCG | 500 | DFS | July 2013 |
| NCT02138734 (QUILT-2.005) | N-803 (IVES)+BCG (IVES) | BCG (IVES) | 596 | CRR and DFS | July 2014 |
| NCT03091660 (PRIME SWOG S1602) | Tokyo BCG+/- intradermal BCG priming | BCG | 1000 | TTHGR | Feb 2017 |
| NCT03528694 (POTOMAC) | Durvalumab (IV)+BCG (IVES) | BCG (IVES) | 1018 | DFS | May 2018 |
| NCT03560479 (HP002-001)d | Alpha 1 H (α-lactalumbin+oleic acid) | Placebo | 52 | AEs, cell shedding, change in papillary tumor characteristics | May 2018 |
| NCT03504163 (NMI-UTUC) | Pembrolizumab (IV)+BCG | N/A | 37 | DFS at 6 mos | June 2018 |
| NCT03664869 (FB-10) | EMDA-MMC (IVES)+BCG | BCG | 300 | Recurrence rate at 2 yrs | October 2018 |
| NCT03799835 (ALBAN) | Atezolizumab (IV)+BCG (IVES) | BCG (IVES) | 516 | RFS | January 2019 |
| NCT03636256 (NANODOCE-2017-02) | NanoDoce (IVES) | N/A | 36 | AEs | April 2019 |
| NCT04386746 (GEMDOCE) | Gemcitabine (IVES)+Docetaxel (IVES) | N/A | 26 | 3-month CRR | July 2020 |
| NCT03711032 (KEYNOTE-676 Cohort B) | Pembrolizumab (IV)+BCG (IVES) | BCG (IVES) | 1405 | CRR and EFS | December 2018 |
| NCT04165317 (CREST) | Sasanlimab (SC)+BCG | BCG | 999 | EFS | December 2019 |
| NCT05327647 (BicaBCa) | Bicalutamide (PO)+BCG | BCG | 160 | RR 3 years | June 2022 |
| NCT04493489 | Propranolol (PO)+BCG | BCG | 242 | RFS 2 years | September 2020 (not yet recruiting) |
| NCT05538663 (BRIDGE) | Gemcitabine (IVES)/Docetaxel (IVES) vs. BCG | BCG | 870 | EFS | November 2022 (not yet recruiting) |
| NCT05410730 | SHR-1501 + /- BCG | N/A | 129 | CRR and 12-month DFS | November 2022 |
| NCT05037279 (EVER) | Verity (Russian strain) BCG | TICE BCG | 540 | RFS | January 2023 (not yet recruiting) |
Abbreviations: AEs: adverse events; BCG: Bacillus Calmette-Guerin; CRR: complete response rate; DFS: disease-free survival; DLT: dose-limiting toxicity; EFS: event-free survival; EMDA-MMC: electromotive mitomycin C; intravenous: IV; IVES: intravesical; PO: per os (oral); RFS: recurrence-free survival; SC: subcutaneous; TTHGR: time to high-grade recurrence. aAs of December 1, 2022. bDenotes actual or target enrollment per ClinicalTrials.gov. cDenotes estimated or actual study start date per ClinicalTrials.gov. dWindow-of-opportunity trial, where study drug is given prior to transurethral resection. The other trials require patients to have complete resection of papillary tumors during screening.
TRIAL DESIGNS IN THE BCG-NAïVE SETTING
For patients with high-risk (HR) NMIBC, intravesical BCG given weekly for six consecutive weeks followed by maintenance BCG according to the SWOG 8507 trial schedule (see “BCG Supply Issues” section) has remained the standard of care for two decades [18]. Despite high initial complete response rates to BCG, the majority of patients with HR NMIBC experience tumor recurrences, highlighting the need for development of innovative therapeutic approaches as well as effective non-BCG options given the ongoing BCG supply shortage. Consequently, the discussion of current and future clinical trial designs within the BCG-naïve patient population focused on the HR NMIBC BCG-naïve patient population. Nuances in clinical trial design unique to intermediate-risk (IR) NMIBC populations were not discussed in detail within this workshop as several workshop participants felt that differences in the standard treatment of IR compared to HR NMIBC (e.g. shorter schedule of maintenance BCG, acceptance of either intravesical chemotherapy or BCG as standard therapy, and lower progression rates) warranted separate trials for IR NMIBC.
Determining the expected clinical efficacy with traditional BCG therapy is important for accurately powering clinical trial designs for the HR BCG-naïve NMIBC patient population. Recent randomized trials with BCG as a control arm include the EORTC 30962 trial and the 102.1 trials, both utilizing a control arm regimen of full-dose TICE BCG induction followed by maintenance therapy [19, 20]. Within these BCG therapy control arms (n = 338 [EORTC 30962], n = 98 [102.1]), one-year recurrence-free survival (RFS) rates of 75% were seen in both studies with two-year RFS rates of 62% and 65% observed respectively in the EORTC 30962 and 102.1 trials. Nuances of note include an absence of patients with CIS in the EORTC 30962 trial, only one year of maintenance BCG in the 102.1 trial, and a mixture of intermediate- and high-risk NMIBC patients in the 102.1 trial. In contrast, in the NIMBUS trial, the BCG therapy control arm (n = 175, with 8% receiving TICE, 91% receiving Medac, and 1% receiving Connaught BCG) achieved an 89% RFS rate at one year and 85% at 2 years [21]. These data may be useful for powering future study designs within the BCG-naïve HR NMIBC population, despite limitations in interpretability of time-to-event data in a single-arm in isolation.
In prior FDA publications on NMIBC trial designs, randomized clinical trials of a novel agent versus an appropriate active control (e.g., BCG) or of a novel agent added to a standard of care were recommended (Figs. 1 and 2). Accordingly, most current and proposed clinical trials in BCG-naïve HR NMIBC have aligned their studies with these designs.
Fig. 1
BCG-Naïve HR NMIBC Direct Comparison Clinical Trial Design. Abbreviations: BCG: Bacillus Calmette-Guerin; EFS: event-free survival; HR NMIBC: high-risk non-muscle invasive bladder cancer; R: randomize; SOC: standard of care; Tx: treatment.
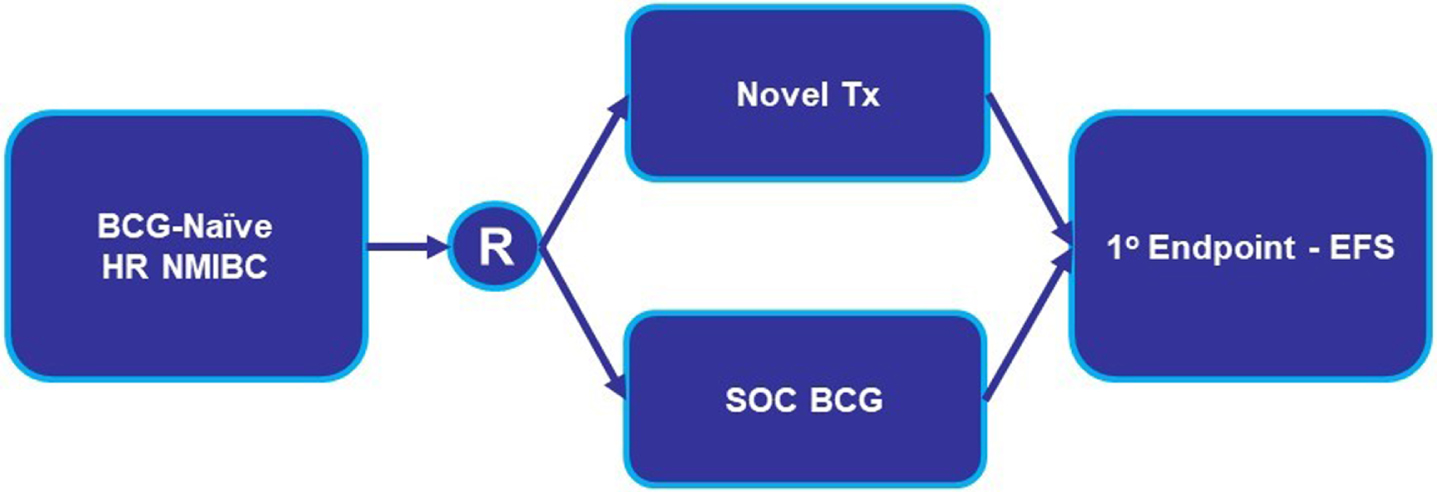
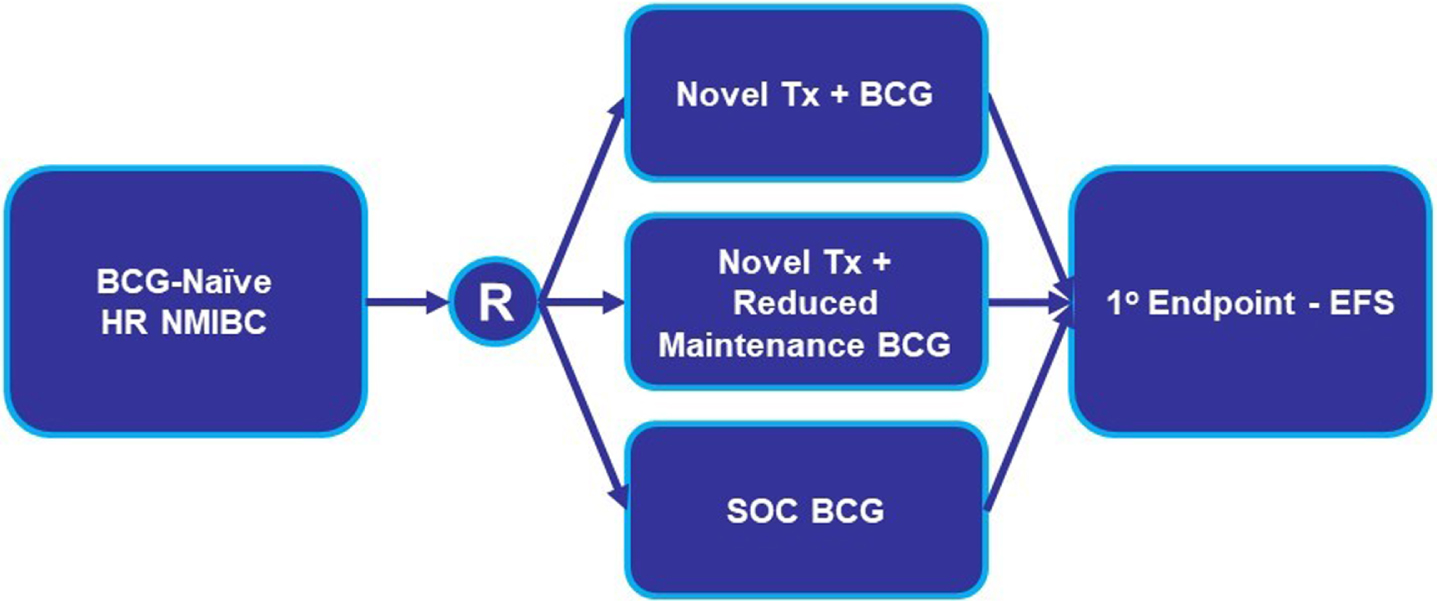
Fig. 2
BCG-Naïve HR NMIBC Add-On Clinical Trial Design. Abbreviations: BCG: Bacillus Calmette-Guerin; EFS: event-free survival; HR NMIBC: high-risk non-muscle invasive bladder cancer; R: randomize; SOC: standard of care; Tx: treatment.
In both the direct comparison and add-on trial designs, important considerations include eligibility, treatment, and efficacy assessments, as described in Table 2 [22]. Examples of several ongoing or planned direct comparison trials utilizing a non-inferiority primary endpoint and add-on trials employing superiority primary endpoint designs were highlighted during the workshop.
Table 2
Key trial design considerations in non-muscle invasive bladder cancer
| Trial Population | BCG-Naïve | BCG-Unresponsive CIS with or without Papillary | BCG-Unresponsive Papillary Only |
| Eligibility | •Uniform pre-treatment staginga | ||
| Treatment | •Schedules and durations of maintenance therapy in both the experimental and the control arms •BCG strain(s) to utilize within standard of care control armsb | •If using investigational treatment in combination with BCG, consider a) BCG strain(s) to utilize within standard of care control armsb and | •Randomized trial design needed |
| •Strategies to anticipate and to address BCG supply shortages during the conduct of a trial | |||
| Efficacy | •Definitions of events that are considered recurrences within all regions of the urinary tract (bladder, upper urinary tract, and prostatic urethra) •What degree of improvement in the primary endpoint would be considered clinically meaningful in experimental study arms •Consistent use of enhanced cystoscopy imaging modalities between each patient’s screening, any visit(s) to document initial response, and during any directed or mandatory biopsies •Importance of long-term follow-up to assess for the risk of progression and long-term disease control | ||
| Non-Inferiority Design | •Discussion with the FDA should occur to discuss key issues including the constancy assumption and appropriate primary endpoint (22) | •N/A | •N/A |
| Safety | •Detailed assessment and reporting of local vs systemic treatment-related adverse events •PROs: Pre-specified objectives | ||
Abbreviations: BCG: Bacillus Calmette-Guerin (BCG); N/A: not applicable; PROs: patient-reported outcomes. aFor example, the need to re-resect tumor base to ensure resection of all remaining papillary disease, ensure the absence of any muscle-invasive disease, and identify any concurrent CIS. bInclusion of non-FDA approved strains should be discussed with FDA in advance.
The direct comparison and add-on trial designs being proposed in the modern era for the BCG-naïve HR NMIBC population rely on a standard of care (SOC) BCG control arm therapy (Figs. 1 and 2). A control arm drug regimen should be relevant and acceptable to the region for which the data are intended [23]. Thus, sponsors or investigators proposing to employ alternative non-BCG control arms must provide sufficient data demonstrating their appropriateness as a control arm regimen for the US population if the trial is intended to support an FDA marketing application.
Two examples of potential control arm regimens that were discussed were the intravesical chemotherapy doublet regimen of gemcitabine and docetaxel (gem/doce), and the Tokyo alternative BCG strain. Clinician panelists confirmed that gem/doce is commonly used in patients with BCG-naïve NMIBC, particularly in times of greatest BCG supply shortages. To date, only retrospective clinical efficacy data with intravesical gem/doce exist in this population. Ongoing prospective trials of intravesical gem/doce in patients with BCG-naïve HR NMIBC are underway and aim to provide prospective multi-site measures of clinical efficacy. Similarly, the clinical efficacy of the Tokyo alternative BCG strain compared to standard of care TICE BCG has been studied in the fully-accrued S1602 trial with results maturing.
With most current randomized trials utilizing BCG as their standard of care control regimen, participants expressed concern that the BCG supply shortages have delayed accrual to randomized trials of BCG-naïve HR NMIBC. The question arose of whether the accelerated approval pathway could be used to address the unmet need in this setting with a single-arm trial design. Regardless of approval pathway, single-arm trials are reserved for situations in which it is unethical to randomize patients to a placebo control or a randomized trial is not feasible due to lack of equipoise or challenges in accrual in a rare disease (see section on Trial Design for BCG-Unresponsive CIS in the Modern Era). In prior FDA guidance, when a randomized trial was not feasible, a single-arm trial design assessing durable complete response rate in BCG-unresponsive HR NMIBC was considered appropriate for either regular approval or the accelerated approval pathway, in which confirmatory evidence of effectiveness is required after approval. However, the accelerated approval pathway is not intended to be limited to single-arm trials, and the requirements for accelerated approval include not only demonstration of substantial improvement over available therapy but also substantial evidence of effectiveness from adequate and well-controlled studies [24, 25]. A well-controlled study could consider a control arm therapy commonly used in clinical practice during BCG shortages, for example, reduced-dose BCG or other standard regimen. However, use of alternative (e.g., non-BCG, reduced dose BCG, or alternative BCG schedules) control arms should be supported by a rationale that includes their expected efficacy in this patient population. Furthermore, other approaches to clinical trial conduct in the setting of the BCG shortage, such as the allowance of standard of care BCG administration at a patient’s local urologist’s office rather than at a study site, have been successfully implemented in some studies.
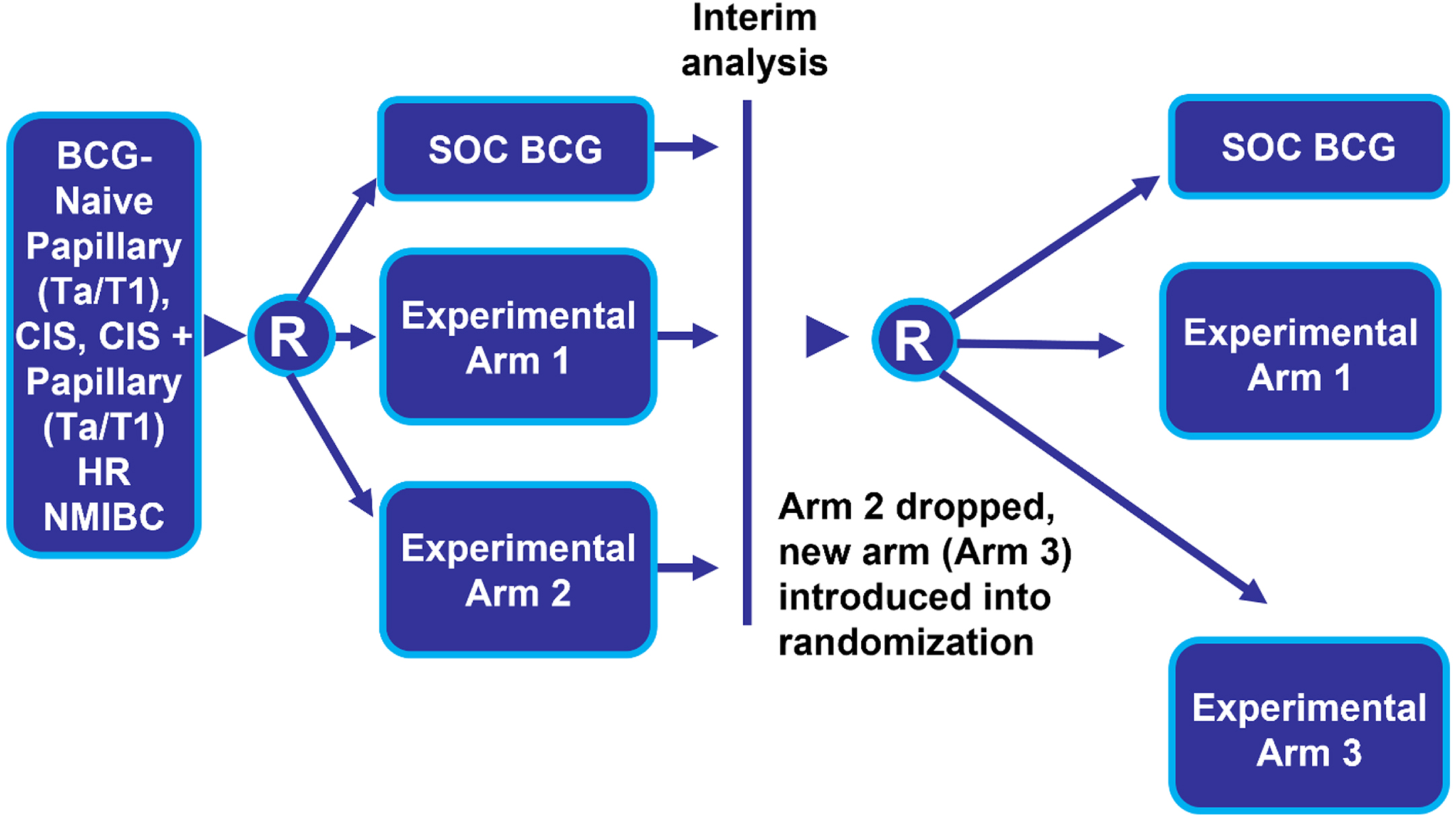
Another trial design feature that can mitigate the impact of a BCG shortage is a randomized platform design where the “common control” arm would receive the standard of care (e.g., TICE BCG). Multiple interventions or investigational drugs could then be evaluated simultaneously within a single master protocol that minimizes the number of patients randomized to the control arm (Fig. 3) [26]. This is sometimes referred to as a multi-arm, multi-stage trial. Experimental arms can be added and dropped over time based on pre-specified efficacy and futility rules. This type of trial design, especially when multiple sponsors cooperate to evaluate drugs in the setting of a common control, has been encouraged by the FDA [27].
Fig. 3
Randomized Platform Common Control Trial Design. Abbreviations: BCG: Bacillus Calmette-Guerin; CIS: carcinoma in situ; HR NMIBC: high-risk non-muscle invasive bladder cancer; R: randomize; SOC: standard of care; Tx: treatment.
DEFINITIONS OF DISEASE STATES FOLLOWING BCG
The need to more clearly and consistently define disease states following receipt of BCG was recognized as a critical step in the efforts to facilitate drug development. To this end, FDA discussed the need for a more standardized definition at the 2012 workshop. As a follow-up to that workshop, the FDA published a Guidance for Industry published in draft form in 2016 and finalized in 2018 entitled “BCG-Unresponsive Nonmuscle Invasive Bladder Cancer: Developing Drugs and Biologics for Treatment” [28]. The definition of BCG-unresponsive NMIBC in the Guidance has been used by trial protocols since that time.
A set of rigorous criteria for “adequate BCG” (“5 + 2” followed by a finding of high-risk NMIBC within a certain time period) to meet the “BCG-unresponsive” definition in the Guidance was useful because patients may expect to have disparate outcomes depending on the amount of prior BCG previously received [29]. However, the optimal definition of “adequate BCG” has not been prospectively studied, and the two categories of disease states do not comprehensively capture all patients such as those whose disease falls into the gap between BCG-naïve and BCG-unresponsive. Many patients do not receive optimal therapy per the Guidance for their initial disease for a variety of reasons including BCG shortage. To help address this issue, the International Bladder Cancer Group (IBCG) recently published a consensus document formulated by an international group of experts. In this document, the term “BCG-exposed” was introduced to describe patients who have high-grade persistent or recurrent NMIBC within 24 months of the last BCG dose but do not meet the definition of BCG-unresponsive disease [30]. Of note, the IBCG consensus stated that the comparator arm in trials with BCG-exposed NMIBC should be BCG therapy, since it will be considered standard of care to treat BCG-exposed high-risk NMIBC with additional BCG for the foreseeable future. However, eligibility and trial designs for investigation of a new drug should be discussed with FDA.
Subsection 1: Characterizing Disease States Following Partial Doses of BCG
Given that many patients are treated with reduced doses of BCG (e.g., one-third dose per instillation) during the ongoing shortage, a question that was discussed is how to view such patients in the context of trial enrollment for clinical trials in the BCG-unresponsive setting. For instance, should patients previously treated with reduced doses of BCG be enrolled in the same cohort as patients previously treated with full doses of BCG? In attempting to answer this question, some workshop participants highlighted the results of EORTC 30692, where, despite improved disease-free rates in the full-dose groups, no statistically significant difference was seen overall in intermediate- and high-risk patients receiving 1/3 vs. full-dose maintenance BCG [19]. The workshop discussion also noted the wide variability in number of colony forming units (CFUs) between each vial of BCG, which indicates challenges in ascertaining the precise dose of viable BCG dose administered on a patient level [31]. Acknowledging these data in the setting of an overall paucity of data describing outcomes with partial dose BCG, some participants still felt that the inclusion of patients with previous 1/3rd dosing in trials in both the BCG-exposed and BCG-unresponsive settings may be acceptable.
Subsection 2: Once BCG-Unresponsive, Always BCG-Unresponsive
If a patient has met criteria for BCG-unresponsive disease, sponsors may consider allowing the patient to participate in subsequent trials enrolling a BCG-unresponsive population, even if BCG was not the last drug to which the patient was exposed (i.e., not newly BCG-unresponsive). Workshop participants noted that this is not only practical for clinical trial access to patients but also based on the biological principle that patients who have demonstrated BCG-unresponsive disease are unlikely to respond to BCG again even if another agent was given after meeting BCG-unresponsive NMIBC criteria. However, consideration may also be given to duration of disease-free interval prior to recurrence, as a prolonged disease-free interval may reflect different underlying biology. Workshop participants felt that these patients should be eligible for trials and their prior treatment considered in terms of line of therapy rather than last line of therapy in an effort to maximize access to the largest number of trials for the most patients. However, sponsors are responsible for providing evidence such as pathology reports and documentation of BCG administration to demonstrate that the patient met “BCG unresponsive” criteria, even if this occurred substantially prior to enrollment. It is also critical for sponsors to assure persistent/recurrent disease at study entry.
TRIAL DESIGN FOR BCG-UNRESPONSIVE CIS IN THE MODERN ERA
Patients with BCG-unresponsive CIS can potentially be studied in either a single-arm trial or in a randomized controlled trial (RCT) [28]. Single-arm trials can be used in clinical settings when a RCT is unethical or infeasible. Historically, the standard of care for patients with BCG-unresponsive CIS has been radical cystectomy, a procedure associated with morbidity and mortality. At the time of the workshop, it was unclear whether current practice patterns were such that the FDA-approved therapies for BCG-unresponsive NMIBC could be deemed “standard of care.” Thus, randomizing patients to an active FDA-approved control was felt to be potentially challenging. Importantly, while the control arm of an RCT must be considered an accepted U.S. standard of care, it need not be an FDA-approved therapy (e.g., off-label intravesical chemotherapy). As such, the use of a randomized control trial design in this setting is considered feasible.
Single-arm trials necessitate comparison to a historical control to interpret trial results. Characteristics of patients enrolled in a specific single-arm trial may differ from those in the broader BCG-unresponsive population, which may both complicate the comparison to external control and limit the generalizability of results. Lack of a randomized comparator arm can also make differentiating drug-related adverse events from those due to the underlying disease or other causes challenging. The more limited characterization of safety seen with single arm trial designs can have important implications on the assessment of overall benefit-risk. Assessment of the contribution of effect for each therapy within a combination regimen is also impossible within a single-arm study. Assessing the contribution of each drug or drugs to the safety and efficacy of a combination therapy is essential in regulatory review, as the use of ineffective drugs in a combination may introduce toxicity without improving efficacy outcomes.
Trials in patients with BCG-unresponsive CIS are subject to variability in key aspects of trial conduct, including use of advanced cystoscopy techniques, use of mandatory vs. for-cause biopsies, operator-dependent conclusions on cystoscopy findings, frequency of focal CIS being completely resected by screening TURBT alone, and other factors. Substantial heterogeneity in disease assessments performed in a single-arm trial can hinder interpretation of trial results. In addition to balancing of known and unknown baseline factors, a randomized trial design may mitigate concerns of variability in trial conduct to better allow for interpretation of trial results.
Mandatory biopsies, also termed “random biopsies”, are not for-cause biopsies but rather prespecified at a specific time point according to a template. The role of mandatory biopsies in the evaluation of investigational drug efficacy was a key topic of discussion. Some protocols have only required for-cause biopsies, triggered by abnormal cystoscopy or cytology. Some protocols have incorporated mandatory biopsies, but at variable time points. Participants expressed a wide range of perspectives on the topic. Some felt that studies that employ mandatory biopsies, typically at the time of CR, provide the most rigorous determination of CR, particularly if the trial is single-arm. This is due to large variations in cystoscopic evaluations between urologists and limited sensitivity of urine cytology. Some data suggest that approximately 4-5% of CIS recurrences may be detected by the addition of mandatory biopsies over for-cause biopsies [32, 33]. However, concerns raised about requiring all protocols to incorporate mandatory biopsies included the perspective that the potential increase in CIS detection rate was too small to warrant the risk and burden to patients incurred by biopsy. Others doubted that mandatory biopsy minimizes bias due to variability in how these biopsies are done. Therefore, if done, a pre-specified methodology in the protocol (e.g., 5 pre-specified sites in bladder plus prostatic urethra and any visible abnormality) would be more likely to optimize inter-reader consistency.
If mandatory biopsies are an imperfect solution to inter-reader variability in sensitivity of CIS detection, the search continues for other techniques that could optimize CIS detection across investigators that could be more easily standardized across investigators and would be more tolerable to patients. The Cysview Registry Group published a study that included an analysis of a subset of patients that recurred within 12 months of completion of BCG. Among these 282 patients, 16 (13%) of recurrences were detected by blue light cystoscopy (BLC) alone and would likely have been missed with white light cystoscopy [WLC] alone) or 6% of all cystoscopies. Of those 16 recurrences, 14 were CIS. Further studies would be useful evaluating the CIS detection rate when mandatory biopsy is used in conjunction with BLC, mandatory biopsy is used in conjunction with WLC, BLC is used without mandatory biopsy, and WLC is used without mandatory biopsy. As noted in Table 2, if enhanced cystoscopy imaging modalities are used at screening, consistent use of these technologies at visit(s) documenting initial response and during any directed or mandatory biopsies is important to optimize interpretability of findings.
Another important topic was the assessment and interpretation of urine cytology. It was noted that in clinical practice, a positive urine cytology is thought to represent cancer in the urothelial tract until proven otherwise, due to high specificity of cytology. However, positive cytology in the setting of other negative pre-specified evaluations of the lower tract with positive alternative source of malignant cytology may be acceptable in the definition of CR if the investigational drug is intravesical, as this may represent abnormalities in the upper tract that are not expected to be reachable by such therapy [28].
TRIAL DESIGN FOR PATIENTS WITH PAPILLARY-ONLY HIGH-RISK BCG-UNRESPONSIVE NMIBC
The 2018 BCG-Unresponsive NMIBC Guidance states, “For patients without active disease (disease was resected at or before trial entry), FDA recommends a randomized controlled trial design using a time-to-event endpoint such as recurrence-free survival. In contrast, patients with carcinoma in situ (CIS) at trial entry can be studied in either a randomized controlled trial or a single-arm trial [28].
The basis for this recommendation is that CIS has been viewed as a diffuse disease due to field effects that can rarely be completely resected by TURBT, while papillary disease may be completely resected by TURBT. Thus, the objective of post-TURBT therapy in CIS is ablative while the objective of post-TURBT therapy in completely resected papillary disease is to prevent recurrence. Due to the logic that only a time-to-event endpoint can measure the efficacy of a drug in a patient with no visible disease at baseline and the regulatory principle that time-to-event endpoints cannot be reliably interpreted from single-arm trials, only a randomized trial would be appropriate for regulatory review purposes of a drug intended for patients with completely resected papillary disease at study baseline [34]. Furthermore, some data suggests that the molecular characteristics of papillary and CIS disease are distinct [35, 36].
Potential randomized designs for HR BCG-naïve and BCG-exposed papillary disease patients were similar to designs discussed in the Trial Designs in the BCG-Naïve Setting section: 1) Direct comparison (investigational agent vs. BCG); 2) Add-On (investigational agent+BCG vs. BCG); and 3) Modified Direct Comparison (investigational drug vs. physician choice of standard agent (a limited number of therapeutic regimens from which each investigator could choose, which could include options such as intravesical gem/doce). In any of these 3 designs, populations with CIS (with or without papillary) disease and those with papillary only could theoretically be studied in separate trials (Fig. 4) or they may be studied in one trial (Fig. 5).
Fig. 4
Two-Trial Design Simulation Investigating Papillary (Ta/T1) and CIS Patients Separately. Abbreviations: BCG: Bacillus Calmette-Guerin; CIS: carcinoma in situ; CR: complete response; EFS: event-free survival; HR NMIBC: high-risk non-muscle invasive bladder cancer; SOC: standard of care; Tx: treatment.
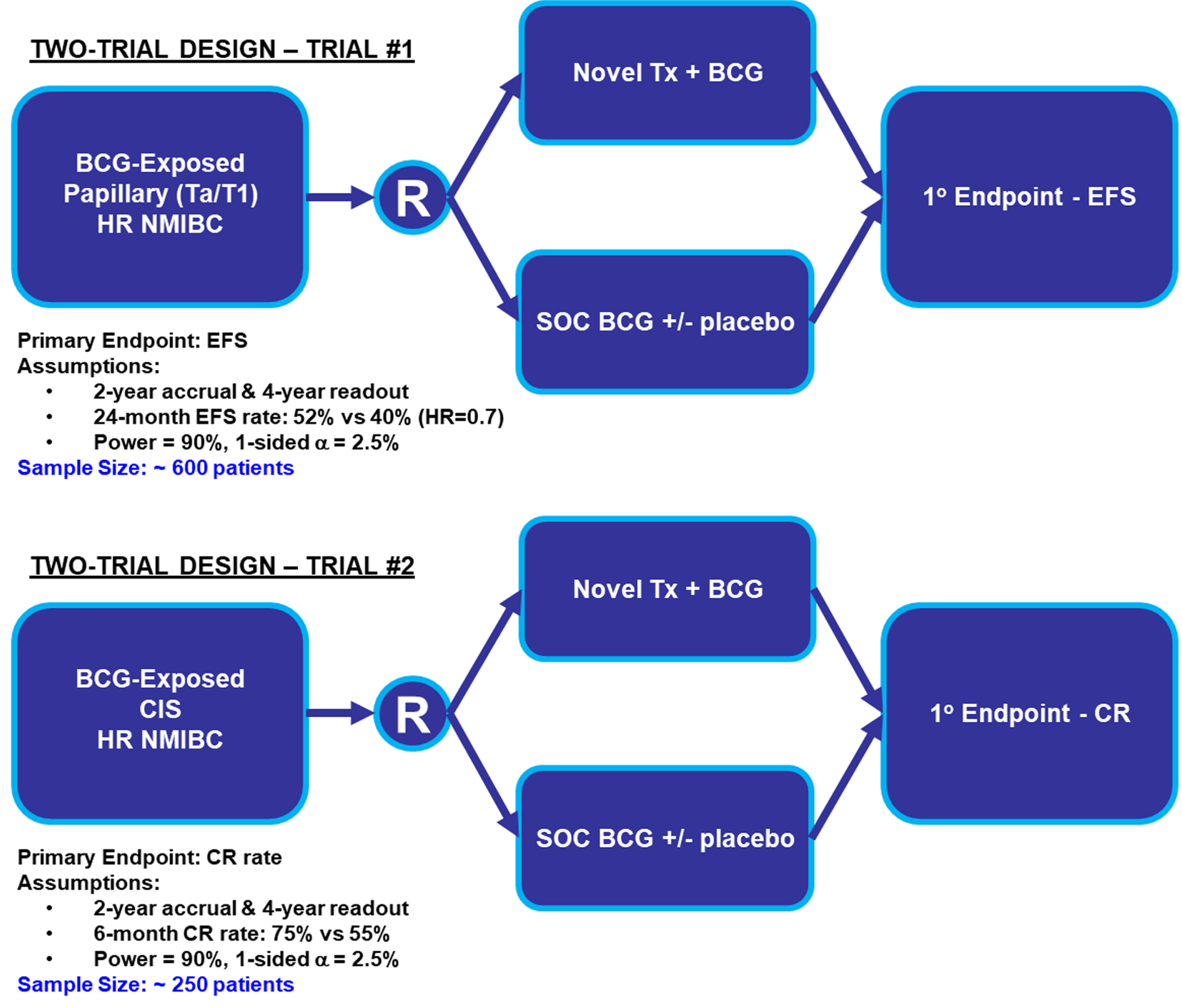
Fig. 5
One-Trial Design Simulation Investigating Papillary (Ta/T1) and CIS Patients Simultaneously. Abbreviations: BCG: Bacillus Calmette-Guerin; CIS: carcinoma in situ; CR: complete response; EFS: event-free survival; HR NMIBC: high-risk non-muscle invasive bladder cancer; SOC: standard of care; Tx: treatment.
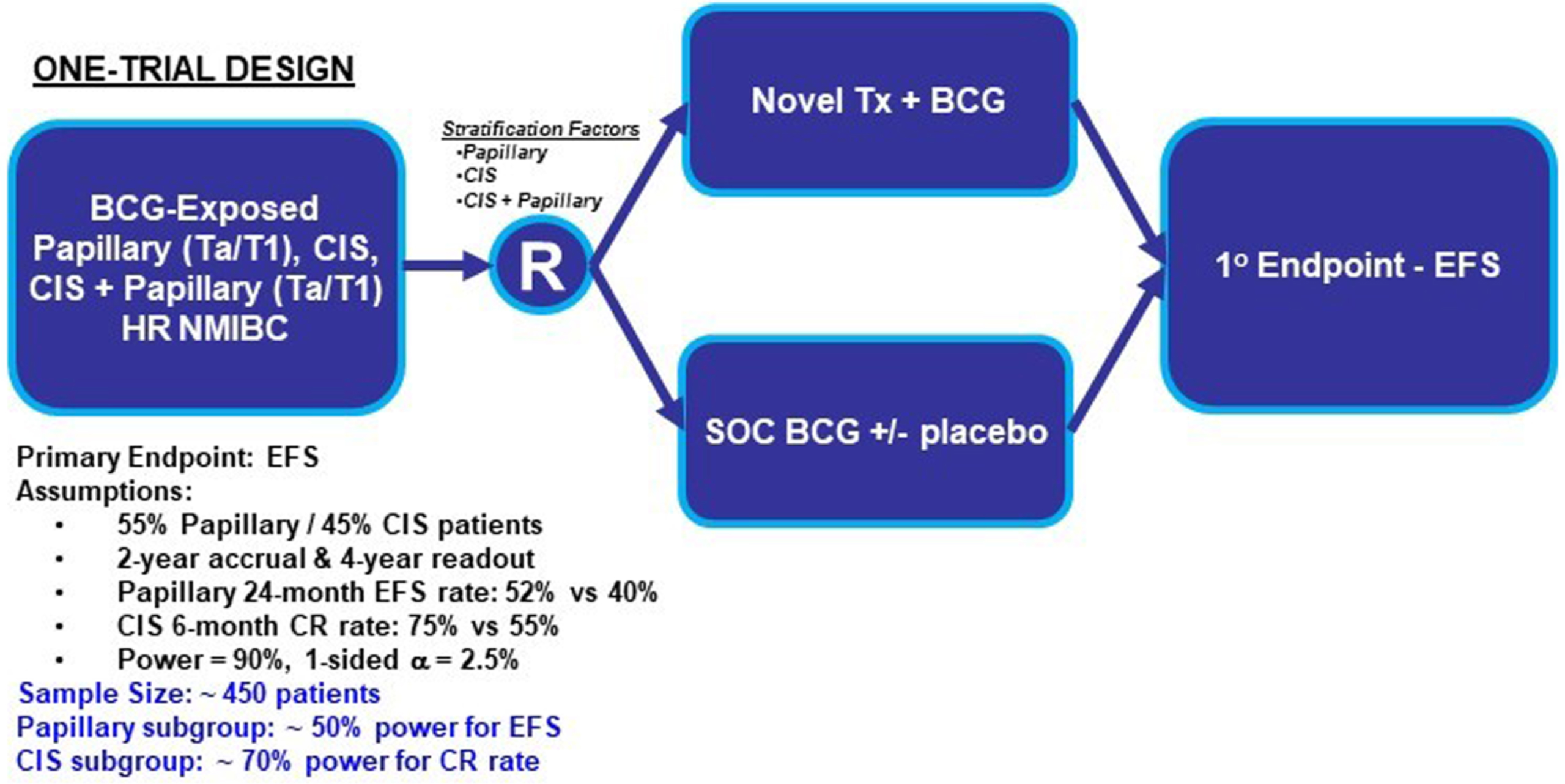
Both the two-trial and one-trial designs present advantages and disadvantages. Two-trial designs provide more homogenous populations, clearer data analyses with fewer assumptions regarding event timing, and concise conclusions within each NMIBC population. These advantages come at the cost of a larger required total sample size across the two trials. In contrast, a one-trial design requires a smaller sample size and presents operational advantages with all study sites recruiting to the same trial. Disadvantages of a one-trial design include the chance for the primary intent-to-treat analysis being driven by one patient population and the potential for subgroup analyses to be underpowered. However, a large effect size may overcome some of these limitations.
Table 2 describes important aspects of trial designs for patients with BCG-unresponsive HR papillary HR NMIBC.
REGULATORY CONSIDERATIONS FOR INCORPORATING A CYSTECTOMY-FREE SURVIVAL ENDPOINT INTO TRIAL DESIGN
Radical cystectomy [RC] is the standard of care for several categories of high-risk NMIBC including BCG-unresponsive and HR BCG-naïve disease [28, 37]. However, RC is a major procedure associated with approximately 2% 30-day and 5% 90-day mortality rates and other short- and long-term complications [38, 39]. These risks combined with the desire to preserve bladder and other associated structural function lead some patients to refuse or delay cystectomy. Given the clinical meaningfulness of RC as an outcome, there has been interest in evaluating time to cystectomy or cystectomy-free survival as a secondary endpoint [28, 40, 41].
Inherent in the meaningfulness of delay or avoidance of cystectomy is the assumption that RC is associated with short and long term symptom and functional detriments that impair quality of life. While a reasonable assumption, there are concerns around capturing and interpreting RC as an endpoint. t. Firstly, some data suggest that cystectomy results in a less substantial decrement in quality of life, following the initial post-RC recovery period, than previously assumed [42]. Additionally, when considering the alternatives to cystectomy, many workshop participants emphasized that the adverse impact on quality of life associated with conservative (i.e., bladder-sparing) therapy are often underestimated and poorly characterized in the literature.
Beyond issues related to clinical meaningfulness, when evaluating endpoint selection, regulatory considerations also emphasize maximizing accuracy of measurement and minimizing risk of bias. One example of the potential introduction of uncertainty in the measurement of this endpoint is the choice of how to determine the time point at which a patient agrees to cystectomy or a physician recommends cystectomy. This important decision naturally involves a high degree of subjectivity and patient preference. In addition, any benefits of delaying RC must be weighed against the risk of progression from NMIBC to MIBC which would result in a worsening of prognosis from 80% survival to 30% survival at 5 years [43–45]. Given these challenges, delay of cystectomy would need to be done judiciously and appropriately, with predetermined and clinically sound criteria for when a cystectomy should be offered in order to mitigate the subjectivity of the endpoint and, more importantly, reduce the likelihood of progression to more advanced MIBC or metastatic disease.
Ultimately, if cystectomy-free survival is being considered as a secondary endpoint for future regulatory review, it is critical that a) the intervention is less morbid than cystectomy, b) objective criteria for recommending cystectomy should be delineated in the protocol, c) the reasons for cystectomy and avoidance of cystectomy be comprehensively recorded, including concordance with the objective criteria noted above, and d) overall cancer-specific efficacy be carefully assessed (e.g., progression to MIBC or metastatic disease). With this framework, it is clear that demonstration of short and long-term tolerability is essential for these trials, and that the results of other clinically meaningful endpoints such as progression-free survival and overall survival would need to be consistent with cystectomy-free survival to suggest a positive benefit-risk ratio. Modern trials with extensive reporting of the reasons for cystectomy and avoidance of cystectomy may enhance understanding of patient decision-making and how to design trials with more favorable benefit-risk ratios in the future. However, workshop discussion noted that many clinicians may still feel that the interpretation of cystectomy-free survival is challenging due to risk of bias and thus the results of this endpoint may not provide overall helpful information.
PATIENT-REPORTED OUTCOMES (PROS) AND PATIENT-FOCUSED DRUG DEVELOPMENT
When choosing a PRO instrument to use in a cancer trial, it is important to select a PRO measurement tool that is proximal to the disease or treatment (i.e., symptoms or direct treatment side effects and well defined functional impacts), and that the PRO items (individual questions) are appropriate to the study. This is primarily because the more global concept of health-related quality of life can be influenced by factors beyond the trial intervention (life events, social constructs). Other considerations include the trial patient population, the type of therapy under investigation, the adverse effects of interest, and the frequency and total number of questions being asked to patients. Additionally, FDA’s draft guidance for industry provides recommendations to sponsors for collection of a core set of PROs in cancer clinical trials and related considerations for instrument selection and trial design [46]. Use of item libraries such as the PRO-CTCAE [47], the EORTC item library [48] and other measurement systems [49, 50] can allow for rigorous and tailored assessment in NIMBC studies.
During the workshop discussion, patient representatives highlighted several common and important disease-related and treatment-related symptoms: bladder function and urinary symptoms such as pain, urgency, and frequency; emotional stress, anxiety, feeling of uncertainty and fear of disease recurrence/progression; medical complications and financial burden associated with requirement for repeated surveillance procedures. These patient-reported symptom and function items could be taken into consideration in drug development programs aiming to delay time to cystectomy.
Complications associated with TURBT itself are not fully understood on a trial level and could also be better characterized in future studies. Anecdotally, TURBT is commonly associated with bladder pain/spasm after the procedure. Some of the rare but severe complications associated with TURBT are major post-operative bleeding and bladder perforation. Additionally, frequent instrumentation of the urinary tract can result in urethral stricture, decompensating the bladder function, and developing or worsening of urinary symptoms. Risk of cognitive impairment and cardiopulmonary complications associated with repetitive general anesthesia is important, particularly in patients with NMIBC where the majority of patients are older adults, and the median age at diagnosis is 73 years. The impact of repeated TURBT on patients’ symptoms and function should be better understood to facilitate rational design of clinical trials aiming to reduce the number or frequency of TURBT.
CONCLUSION
While the growth in drug development in NMIBC over the last decade has been encouraging, there remains a lack of effective available treatment options for patients and many questions about optimal trial design for NMIBC remain. Furthermore, BCG supply issues have affected clinical care and the feasibility of clinical trials for which either BCG is a required prior therapy or BCG is part of an investigational drug regimen or control arm. The NMIBC workshop underscored opportunities to refine previous work, from exploring ways to utilize randomized trial design for BCG-unresponsive NMIBC, to designing clinical trials for BCG-naïve patients that could potentially lead to drugs that could be useful to a greater number of patients (Fig. 6).
Fig. 6
Abbreviations: BCG: Bacillus Calmette-Guerin; FDA: Food and Drug Administration; NMIBC: non-muscle invasive bladder cancer; U.S.: United States.

Sponsors of clinical trials are encouraged to meet with the FDA to discuss details of their trial designs. Patient selection, endpoint definitions, and clinically relevant efficacy metrics will be critical study design considerations. Video recordings and presentations from this workshop are posted on the FDA website [51].
ACKNOWLEDGMENTS
The authors thank Marc Neilson for graphics support, Joan Todd and Caitlin Drew for project management assistance, and Julia Beaver and Amna Ibrahim for guidance.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
Conceptualized manuscript: E.C., N.H., S.P.L., and C.W.
Contributed to performance work: E.C., N.H., S.P.L., J.F., S.A., A.M.K., V.B., R.S.S., A.A.J., P.B., N.S., M.K., K.S., J.R.B., M.A.O., G.D.S., C.V., E.B., M.J.R., M.D.G., R.O., P.C.B., H.A., K.B., K.P., C.P.D., Z.F., T.D., S.P.P., A.B.S., R.B., S.P., N.A., P.G.K., and C.W.
Contributed to writing the article: E.C., N.H., S.P.L., J.F., S.A., A.M.K., V.B., E.B., Z.F., D.L.S., L.A.K., P.G.K., and C.W.
CONFLICT OF INTEREST
E.C., J.F., S.A., V.B., A.A.J., P.B., K.S., J.R.B., E.B., R.O., K.B., Z.F., T.M.D., R.B., D.L.S., L.A.K., R.P., P.G.K., and C.W. have no conflicts of interest to report.
N.M.H., S.P.L., A.M.K., R.S.S., M.A.O., M.J.R., M.D.G., P.C.B., K.P., C.P.D., R.B., and S.P.P. (Psutka) are Editorial Board members of this journal, but were not involved in the peer-review process nor had access to any information regarding its peer-review.
N.M.H. receives consulting compensation from AstraZeneca, Merck, BioGears, Seattle Genetics, Mirati, Incyte, RemGen, Janssen, Pfizer, EMD Serono, Verity Pharmaceuticals, Huron Consulting, Guidepoint, Natera, Protara Therapeutics, Astellas Pharma; research support to the institution from HTG Molecular Diagnostics, AstraZeneca, Bristol Myers-Squibb, Genentech, Seattle Genetics, Pieris, Inovio, Principia Biopharm, Incyte, and Ikena Oncology; and speaking honorarium from Medscape.
S.P.L. reports clinical trial involvement with Aura Bioscience, FKD, JBL (SWOG), Genentech (SWOG), Janssen (SWOG), Merck (Alliance), QED Therapeutics, UroGen, Vaxiion, Viventia; is on the advisory board or a consultant for Aura Bioscience, BMS, C2iGenomics, Ferring, Incyte, Pfizer/EMD Serono, Protara, Stimit, UroGen, Vaxiion, Verity; has a patent for a TCGA classifier; and receives honoraria from Grand Rounds Urology and UroToday.
A.M.K. reports clinical trial involvement with FKD, Merck, Bristol Myers Squibb, Photocure, SWOG, Adolor, Heat Biologics, Janssen, Taris, Seattle Genetics; reports laboratory research with NIH, SPORE, AIBCCR, PCORI; is on the advisory board or a consultant for TMC Innovation, Arquer Diagnostics, Asieris, Astellas, Biological Dynamics, BMS, CG Oncology, Cystotech, Eisai, Engene, Ferring, InCyte, Imvax, Imagin Medical, Janssen, Medac, Merck, Nonagen, Photocure, ProTara, Pfizer, Roche, Seattle Genetics, Sessen Bio, Theralase, US Biotest, Urogen Inc; is on the editorial board of European Urology Oncology; Journal of Urology, UroToday; is president of International Bladder Cancer Network and International Bladder Cancer Group; and has a joint patent with UT MD Anderson Cancer Center for CyPRIT (Cytokine Predictors of Response to Intravesical Therapy).
R.S.S. is a consultant for CG Oncology and Verity Pharma; and receives research support from Japanese BCG Laboratories and Merck.
N.S. is a consultant for AbbVie, Accord, Alessa Therapeutics, Amgen, Antev, Arquer, Asieris, Astellas, Astra Zeneca, AuraBiosciences, Bayer, BMS, Bioprotect, Boston Scientific, Clarity, Dendreon, Exact Imaging, FizeMedical, CG Oncology, Genentech/Roche, Ferring, Foundation Medicine, ImmunityBio, Incyte, Invitae, Janssen, Lantheus, Lilly, MDX, Merck, Minomic, Myriad, Nonagen, Novartis, Nymox, Pacific Edge, Palette Life, Photocure, Pfizer, PlatformQ, Profound Medical, Promaxo, Propella, Protara, Sanofi, Specialty Networks, Telix, Tolmar, Urogen, and Vessi.
M.K. receives grant funding from the American Cancer Society; is a consultant for Merck, Pfizer, Seagen/Astellas, Photocure, Aura, BMS, Nanology, and Janssen; and holds a patent for Nanoparticle Formulations for Enhanced Drug Delivery to the Bladder.
M.A.O. receives grant/research/clinical trial support from Abbot Molecular, Photocure, Urogen; and is a consultant or on the advisory board for Fidia Pharmaceuticals, Sesen Bio, Merck, Theralase, and Urogen.
G.D.S. is a member of the Clinical Trial Protocol Committee for Merck, BMS, Janssen, CG Oncology, Pfizer, PhotoCure, Fidia, Seagen, Protara; is or has been a scientific advisor /consultant with CG Oncology; PhotoCure; Merck; Taris Biomedical (Now Janssen); Fidia Farmaceuticals; Urogen, Ferring; Fergene, Bristol Myers Squibb; Astra Zeneca; Pfizer, Janssen; Epivax Therapeutics; EnGene Bio; Astellas; SeaGen; Verity Pharmaceuticals, Protara, xCures, Nonagen, Nanology, Imvax, Asieris; and has equity stock/options with Epivax Therapeutics, Urogen, CG Oncology, Engene Bio.
M.J.R. has intellectual property with Fina Biotech and is or has been a meeting participant or lecturer for Roche, AstraZeneca, and Bayer.
M.D.G. receives research funding from Bristol Myers Squibb, Novartis, Dendreon, Astra Zeneca, Merck, Genentech; and is on the advisory board or a consultant for Bristol Myers Squibb, Merck, Genentech, AstraZeneca, Pfizer, EMD Serono, SeaGen, Janssen, Numab, Dragonfly, GlaxoSmithKline, Basilea, UroGen, Rappta Therapeutics, Alligator, Silverback, Fujifilm, Curis, Gilead, Bicycle, Asieris, Abbvie, Analog Devices.
P.C.B is a member of the advisory board or equivalent with AbbVie, AstraZeneca, Astellas, Bayer, BMS, EMD-Serono, Ferring, Fergene, Janssen, Merck, miR Scientific, Nonagen, NanOlogy, Pfizer, Photocure, Prokarium, Protara Therapeutics, QED Bioscience, Roche, Sanofi, Sesen Bio, STIMIT, TerSera, Tolmar, Urogen, Verity; is a member of a Speaker’s bureau with Janssen, Minogue, Ferring, TerSera, Pfizer; has received a grant or honorarium from iProgen; and shares a patent with Veracyte.
H.A. provides consultation to AstraZeneca, Flare Therapeutics and Paige.AI.
K.P. receives consulting fees from Photocure.
C.P.D. reports compensation for Scientific/Advisory Committee Member for AstraZeneca Pharmaceuticals, UroGen Pharma (formerly Theracoat Ltd.); consulting fees from STIMIT Corporation; consulting fees from General Atlantic; board member position for DF/HCC Kidney Cancer SPORE Advisory Board; research funding from Cancer Prevention Research Institute of Texas (CPRIT), Department of Defense (DoD), and NIH/NCI.
S.P.P. (Porten) receives research support from Photocure, consulting compensation from Pacific Edge, grant funding from PCORI, BCAN, AHRQ, Genentech, Merck, and is on the advisory board for AstraZeneca.
A.B.S. receives grant funding from PCORI, BCAN, AHRQ, Genentech, and Merck.
S.P.P. (Psutka) reports being on Practice Guidelines Committee (Upper Tract Urothelial Carcinoma) of the American Urological Association (AUA), being AUA Core Curriculum Sr. Editor (2019-2023) and Senior Consultant (2023-); is or has been on Scientific Advisory Boards for Merck (Past), ImmunityBio (Past), Janssen (Current); has received travel funding/honoraria from Medtronic (Past), AstraZeneca (Past); receives research funding from PRIME Education, INC, Bladder Cancer Advocacy Network, National Institute on Aging, and is on the editorial boards for European Urology.
N.A. receives research funding to institution from Arnivas, Astellas, Astra Zeneca, Bavarian Nordic, Bayer, Bristol Myers Squibb, Calithera, Celldex, Clovis, Crispr, Eisai, Eli Lilly, EMD Serono, Exelixis, Genentech, Gilead, Glaxo Smith Kline, Immunomedics, Janssen, Lava, Medivation, Merck, Nektar, Neoleukin, New Link Genetics, Novartis, Oric, Pfizer, Prometheus, Rexahn, Roche, Sanofi, Seattle Genetics, Takeda, and Tracon.
REFERENCES
[1] | Siegel RL , Miller KD , Fuchs HE , Jemal A . Cancer statistics, 2022. CA Cancer J Clin. (2022) ;72: (1):7–33. |
[2] | Babjuk M , Burger M , Compérat E , et al (2022). EAU guidelines: non-muscle invasive bladder cancer. Retrieved from https://uroweb.org/guideline/nonmuscle-invasive-bladder-cancer/. Access date May 27, 2022. |
[3] | Chang SS , Boorjian SA , Chou R . et al: Diagnosis and treatment of non-muscle invasive bladder cancer: AUA/SUO guideline (Amended 2020). J Urol. (2016) ;196: :1021. |
[4] | Jarow JP , Lerner SP , Kluetz PG , Liu K , Sridhara R , Bajorin D , et al. Clinical trial design for the development of new therapies for nonmuscle-invasive bladder cancer: report of a Food and Drug Administration and American Urological Association public workshop. Urology. (2014) ;83: (2):262–4. |
[5] | Drugs@FDA. ADSTILADRIN (nadofaragene firadenovecvncg) suspension, for intravesical use. [updated 2022 Dec 16; accessed 2023 Jan 2]. Available from: https://www.fda.gov/media/29/download |
[6] | Drugs@FDA. KEYTRUDA (pembrolizumab) for injection, for intravenous use. [updated 2020 Jan 8; accessed 2022 Aug 9]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label//14s066lbl.pdf |
[7] | Lerner S . BCG Shortage: The Practice Environment. [PowerPoint presentation]. FDA Workshop: Clinical Trial Design for Non-Muscle Invasive Bladder Cancer (NMIBC). [updated 2022 Jan 18; cited 2022 Aug 9]. Available from: https://www.fda.gov/media/155367/download |
[8] | Cardillo F , Bonfim M , da Silva Vasconcelos Sousa P , Mengel J , Ribeiro Castello-Branco LR , Pinho RT . Bacillus Calmette-Guérin Immunotherapy for Cancer. Vaccines (Basel). (2021) ;9: (5). |
[9] | Morales A , Eidinger D , Bruce AW . Intracavitary Bacillus Calmette-Guerin in the treatment of superficial bladder tumors. J Urol. (1976) ;116: (2):180–3. |
[10] | National Comprehensive Cancer Network. Bladder Cancer (Version 2.2022). https://www.nccn.org/professionals/physician_gls/pdf/bladder.pdf. Accessed 2022 Aug 9. |
[11] | American Urological Association. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Joint Guideline (2020). https://www.auanet.org/guidelines/guidelines/bladder-cancer-non-muscle-invasive-guideline. Accessed 9 Aug 2022. |
[12] | Fierce Pharma. Sanofi discontinuing bladder cancer drug TheraCys after years of production issues (2016). https://www.fiercepharma.com/manufacturing/sanofi-discontinuing-bladder-cancer-drug-teracys-after-years-production-issues. Accessed 5 Jan 2023. |
[13] | Merck. Facing global shortage, Merck commits to meeting patient demand for crucial treatment option (2023). https://www.merck.com/stories/facing-a-global-shortage-merck-commits-to-meeting-patient-demand-for-a-crucial-treatment-option/#:∼:text=In%20October%20%2C%20we%20announced,for%20patients%20who%20need%20them. Accessed 10 Aug 2023. |
[14] | American Urological Association. BCG Shortage Notice (2019). https://www.auanet.org/bcg-shortage-notice. Accessed 17 Jan 2023. |
[15] | Au JL , Badalament RA , Wientjes MG , Young DC , Warner JA , Venema PL , et al. Methods to improve efficacy of intravesical mitomycin C: results of a randomized phase III trial. J Natl Cancer Inst. (2001) ;93: (8):597–604. |
[16] | Steinberg RL , Thomas LJ , Brooks N , Mott SL , Vitale A , Crump T , et al. Multi-Institution Evaluation of Sequential Gemcitabine and Docetaxel as Rescue Therapy for Nonmuscle Invasive Bladder Cancer. J Urol. (2020) ;203: (5):902–9. |
[17] | Said O , Robin O , Hakim F , Alfreda P , Lionel B , Marc C . Recurrence Rate and Cost Consequence of the Shortage of Bacillus Calmette-Guérin Connaught Strain for Bladder Cancer Patients. European Urology Focus. (2021) ;7: (1):111–116. |
[18] | Lamm DL , Blumenstein BA , Crissman JD , Montie JE , Gottesman JE , Lowe BA , et al. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J Urol. (2000) ;163: (4):1124–9. |
[19] | Oddens J , Brausi M , Sylvester R , Bono A , van de Beek C , van Andel G , et al. Final results of an EORTC-GU cancers group randomized study of maintenance bacillus Calmette-Guérin in intermediate- and high-risk Ta, T1 papillary carcinoma of the urinary bladder: one-third dose versus full dose and 1 year versus 3 years of maintenance. Eur Urol. (2013) ;63: (3):462–72. |
[20] | Arends TJ , Nativ O , Maffezzini M , de Cobelli O , Canepa G , Verweij F , et al. Results of a Randomised Controlled Trial Comparing Intravesical Chemohyperthermia with Mitomycin C Versus Bacillus Calmette-Guérin for Adjuvant Treatment of Patients with Intermediate- and High-risk Non-Muscle-invasive Bladder Cancer. Eur Urol. (2016) ;69: (6):1046–52. |
[21] | Grimm MO , van der Heijden AG , Colombel M , Muilwijk T , Martínez-Piñeiro L , Babjuk MM , et al. Treatment of High-grade Non-muscle-invasive Bladder Carcinoma by Standard Number and Dose of BCG Instillations Versus Reduced Number and Standard Dose of BCG Instillations: Results of the European Association of Urology Research Foundation Randomised Phase III Clinical Trial “NIMBUS”. Eur Urol. (2020) ;78: (5):690–698. |
[22] | U.S. Food and Drug Administration. Non-Inferiority Clinical Trials to Establish Effectiveness Guidance for Industry. [updated 2016 Nov; accessed 2022 Aug 9]. Available from: https://www.fda.gov/media/78504/download |
[23] | U.S. Food and Drug Administration and International Council for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. Guidance for Industry: E 10 Choice of Control Group and Related Issues in Clinical Trials. General principles for planning and design of multi-regional clinical trials E17. [updated 2001 May; accessed 2022 Aug 9]. Available from: https://www.fda.gov/media/71349/download |
[24] | C.F.R. tit. 21, § 314.510. [Dec. 11, 1992; accessed 2022 Aug 9]. Available from: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-D/part-314/subpart-H |
[25] | U.S. Food and Drug Administration. Guidance for Industry: Expedited Programs for Serious Conditions – Drugs and Biologics. [updated 2014 May; accessed 2022 Aug 9]. Available from: https://www.fda.gov/media/86377/download |
[26] | U.S. Food and Drug Administration. Master Protocols: Efficient Clinical Trial Design Strategies to Expedite Development of Oncology Drugs and Biologics | Guidance for Industry. [updated 2022 March; accessed 2022 Aug 9]. Available from: https://www.fda.gov/media/120721/download |
[27] | Friends of Cancer Research. Pink Sheet – FDA’s Pazdur Advocates Platform Trials For Cancer Drug Development (2019). https://friendsofcancerresearch.org/news/pink-sheet-fdas-pazdur-advocates-platform-trials-for-cancer-drug-development/. Accessed 22 Dec 2022. |
[28] | U.S. Food and Drug Administration. BCG-Unresponsive Nonmuscle Invasive Bladder Cancer: Developing Drugs and Biologics for Treatment Guidance for Industry. [updated 2018 Feb; accessed 2022 Aug 9]. Available from: fda.gov/media/101468/download. |
[29] | Li R , Tabayoyong WB , Guo CC , González GMN , Navai N , Grossman HB , et al. Prognostic Implication of the United States Food and Drug Administration-defined BCG-unresponsive Disease. Eur Urol. (2019) ;75: (1):8–10. |
[30] | Roumiguié M , Kamat AM , Bivalacqua TJ , Lerner SP , Kassouf W , Böhle A , et al. International Bladder Cancer Group Consensus Statement on Clinical Trial Design for Patients with Bacillus Calmette-Guérin-exposed High-risk Non-muscle-invasive Bladder Cancer. Eur Urol. (2022) ;82: (1):34–46. |
[31] | Kamat A . Risk Stratification, Definitions of Disease States, and BCG Unresponsive Disease. [PowerPoint presentation]. FDA Workshop: Clinical Trial Design for Non-Muscle Invasive Bladder Cancer (NMIBC). [updated 2022 Jan 18; cited 2022 Aug 9]. Available from: https://www.fda.gov/media/155373/download |
[32] | Boorjian SA , Alemozaffar M , Konety BR , Shore ND , Gomella LG , Kamat AM , et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. (2021) ;22: (1):107–17. |
[33] | Swietek N , Waldert M , Rom M , Schatzl G , Wiener HG , Susani M , et al. The value of transurethral bladder biopsy after intravesical bacillus Calmette-Guérin instillation therapy for nonmuscle invasive bladder cancer: a retrospective, single center study and cumulative analysis of the literature. J Urol. (2012) ;188: (3):748–53. |
[34] | U.S. Food and Drug Administration. Clinical Trial Endpoints for the Approval of Cancer Drugs and Biologics Guidance for Industry. [updated 2018 Dec; accessed 2022 Aug 9]. Available from: https://www.fda.gov/media/71195/download |
[35] | Garczyk S , Ortiz-Brüchle N , Schneider U , Lurje I , Guricova K , Gaisa NT , et al. Next-Generation Sequencing Reveals Potential Predictive Biomarkers and Targets of Therapy for Urothelial Carcinoma in Situ of the Urinary Bladder. Am J Pathol. (2020) ;190: (2):323–32. |
[36] | Knowles MA , Hurst CD . Molecular biology of bladder cancer: new insights into pathogenesis and clinical diversity. Nat Rev Cancer. (2015) ;15: (1):25–41. |
[37] | Chang SS , Bochner BH , Chou R , Dreicer R , Kamat AM , Lerner SP , et al. Treatment of Non-Metastatic Muscle-Invasive Bladder Cancer: AUA/ASCO/ASTRO/SUO Guideline. J Urol. (2017) ;198: (3):552–9. |
[38] | Lawrentschuk N , Colombo R , Hakenberg OW , Lerner SP , Månsson W , Sagalowsky A , et al. Prevention and management of complications following radical cystectomy for bladder cancer. Eur Urol. (2010) ;57: (6):983–1001. |
[39] | Maibom SL , Joensen UN , Poulsen AM , Kehlet H , Brasso K , Røder MA . Short-term morbidity and mortality following radical cystectomy: a systematic review. BMJ Open. (2021) ;11: (4):e043266. |
[40] | National Library of Medicine, National Institutes of Health. ClinicalTrials.gov: Vicinium Treatment for Subjects With Non-muscle Invasive Bladder Cancer Previously Treated With BCG. [updated 2021 June 11; accessed 2022 Nov 22]. Available from: https://clinicaltrials.gov/ct2/show/NCT02449239 |
[41] | Chamie K , Chang SS , Kramolowsky E , Gonzalgo ML , Agarwal PK , Bassett JC , et al. IL-15 Superagonist NAI in BCG-UnresponsiveNon–Muscle-Invasive Bladder Cancer. NEJMEvidence 0: (0):EVIDoa2200167. |
[42] | Clements MB , Atkinson TM , Dalbagni GM , Li Y , Vickers AJ , Herr HW , et al. Health-related Quality of Life for Patients Undergoing Radical Cystectomy: Results of a Large Prospective Cohort. Eur Urol. (2022) ;81: (3):294–304. |
[43] | van den Bosch S , Alfred Witjes J . Long-term cancer-specific survival in patients with high-risk, non-muscle-invasive bladder cancer and tumour progression: a systematic review. Eur Urol. (2011) ;60: (3):493–500. |
[44] | Khaled D , Taylor J , Holzbeierlein J . Salvage Therapy for Non-muscle-invasive Bladder Cancer: Novel Intravesical Agents. Urol Clin North Am. (2020) ;47: (1):119–28. |
[45] | Huguet J , Crego M , Sabaté S , Salvador J , Palou J , Villavicencio H . Cystectomy in patients with high risk superficial bladder tumors who fail intravesical BCG therapy: precystectomy prostate involvement as a prognostic factor. Eur Urol. (2005) ;48: (1):53–9; discussion 9. |
[46] | U.S. Food and Drug Administration. Core Patient-Reported Outcomes in Cancer Clinical Trials 2021 [Available from: https://www.fda.gov/media/149994/download] |
[47] | NIH Healthcare Delivery Research Program. Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE) [updated 2022 Jan 28; accessed 2022 Aug 16]. Available from: https://healthcaredelivery.cancer.gov/pro-ctcae/ |
[48] | EORTC. Item Libary - Quality of Life. [accessed 2022 Aug 16]. Available from: https://qol.eortc.org/item-library/ |
[49] | HealthMeasures. PROMIS. [accessed 2023 April 4]. Available from: https://www.healthmeasures.net/explore-measurement-systems/promis/ |
[50] | FACIT. FACIT Measures & Searchable Library. [accessed 2023 April 4]. Available from: https://www.facit.org/facit-measures-searchable-library |
[51] | U.S. Food & Drug Administration. FDA Workshop: Clinical Trial Design for Non-Muscle Invasive Bladder Cancer (NMIBC). [accessed 2023 April 5]. https://www.fda.gov/drugs/news-events-human-drugs/fda-workshop-clinical-trial-design-non-muscle-invasive-bladder-cancer-nmibc-11182021#event-materials |




