Prevalence, aetiology, and impact of paediatric feeding disorders in preterm infants admitted to a neonatal intensive care unit in Cyprus
Abstract
BACKGROUND:
Advancements in neonatal care have resulted in increased survival for preterm infants, with associated risk for paediatric feeding disorders (PFDs), the prevalence of which is relatively unexplored. Risk factors for developing PFDs in this population must be identified.
OBJECTIVE:
The aim of this study was to determine the epidemiology and risk factors for PFDs in preterm infants with Extremely Low Birth Weight (ELBW); Very Low Birth Weight (VLBW) and Low Birth weight (LBW) in the only neonatal intensive care unit (NICU) in Cyprus.
METHODS:
This study comprised 2 phases: Phase 1, a retrospective 2-year file audit, informing methodology for Phase 2, a prospective epidemiological study. Profiles of 1027 preterm infants were obtained in Phase 1. In Phase 2, clinical assessment data on 458 preterm infants (N = 224) were analyzed.
RESULTS:
The prevalence of PFDs was 36.5%. All preterm infants with ELBW and 69%with VLBW exhibited PFDs. Risk factors were birth weight (BW), gestational age (GA), bronchopulmonary dysplasia (BPD), neurological disorders, structural anomalies, and congenital heart disease (CHD).
CONCLUSIONS:
This unique epidemiological data for one country will inform NICU service provision and direct international research on PFDs in neonates.
1Introduction
Preterm birth is defined as infants born alive before 37 weeks of pregnancy. There are three sub-categories based on gestational age (GA): extremely preterm (< 28 weeks), very preterm (28 to < 32 weeks), and moderate to late preterm (32 to < 37 weeks) (Howson, Kinney, & Lawn, 2012). It is estimated that a significant number of the 500,000 infants born prematurely in Europe and North America each year (Zeitlin, Szamotulska, Drewniak, Mohangoo, Chalmers, Sakkeus, et al., 2013) spend the first few weeks to months of their lives in a neonatal intensive care unit (NICU). Significant risks for long-term cognitive, motor, sensory, nutritional, growth, and behavioural impairments are well documented in this population (Harding, Levin, Crossley, Murphy, & Van den Engel-Hoek, 2019; Kallionen, Eadon, Murphy, & Baird, 2017). Many of these infants will also have a Paediatric Feeding Disorders (PFD) at the outset because of their prematurity (Pados, Hill, Yamasaki, Litt, & Lee, 2021; Jadcherla, Wang, Vijayapal, & Leuthner, 2010). Paediatric Feeding Disorders (PFDs) are defined as “impaired oral intake that is not age-appropriate, and is associated with medical, nutritional, feeding skills, and/or psychosocial dysfunction” (Goday, Huh, Silverman, Lukens, Dodrill, Cohen, et al, 2019, p.g.124). PFDs can result in pneumonia, as milk can be misdirected into the lungs (aspiration) during swallowing. Acquiring skills for safe oral feeding is complex and very preterm infants typically have lengthy hospital stays, until they can demonstrate the ability to feed and maintain nutritional intake to support growth and development (Hoogewerf, Ter Horst, Groen, Nieuwenhuis, Bos, & van Dijk, 2017; Törölä, Lehtihalmes, Yliherva, & Olsén, 2012). Family-centred neurodevelopmental care in the NICU is evidenced as critical to overall developmental outcomes of preterm infants in the NICU (Altimer & Phillips, 2016), however, not all NICUs focus on environmentally protective, individualized, preventative care models that encompass early intervention and early promotion of oral feeding, despite the evidence that some therapeutic interventions result in shorter length of hospital stay (LOS) (Harding, Cockerill, Cane, & Law, 2018; Harding & Cokerill, 2015). Many of these infants will continue to have longer term PFDs (Schädler, Süss-Burghart, Toschke, von Voss, & von Kries, 2007) and remain at high-risk for persistent feeeding problems beyond the NICU (Rommel, De Meyer, Feenstra, & Veereman-Wauters, 2003). Additionally, infants born before 30 weeks GA present with a higher incidence of oral sensorimotor feeding problems at 1 months’ corrected age than their term-born peers (Sanchez, Spittle, Slattery, & Morgan, 2016). In addition to prematurity, there are many factors that may impact determination of oral feeding readiness, such as, an infants’ ability to maintain physiologic stability and behavioural state, though identification is often challenging amongst NICU staff (Harding, Mynard, & Hills, 2018). Understanding the prevalence of PFDs in preterm infants is important for service planning and provision of early neonatal feeding interventions. Identifying the more at-risk profiles of these infants will further facilitate the development of early intervention neonatal services in the NICU.
The aim of this study was to examine the prevalence, aetiology, and impact of PFDs in premature infants, with ELBW, VLBW, and LBW, in one national NICU over a specific time period. The context of this study is Cyprus, which according to European Perinatal Health Report (Euro-Peristat Project, 2013) and the Cypriot Ministry of Health database (Cyprus Health Monitoring Unit, 2016), is among countries with the highest preterm birth rate in Europe (Howson, Kinney, McDougall, Lawn, & Born Too Soon Preterm Birth Action Group, 2013).
2Methods
This is a two-phase investigation combining a retrospective and a prospective study at a level III NICU. Ethical approval was obtained from the Health Sciences Ethics Committee, Trinity College Dublin, Ireland, the data collection site, and the Cyprus Bioethics committee. Phase 1, a retrospective file audit of preterm infants admitted to the NICU over a two-year period, provided a profile of all infants, including the number of preterm infants born in Cyprus and admitted to the NICU in this time period. This information was critical for the planning and implementation of Phase 2. In Phase 2, an assessment of Oral Feeding Readiness (OFR) (Appendix A) and a Clinical Feeding and Swallowing Evaluation (CFSE) (Appendix B) were utilized to determine the prevalence, aetiology, and impact PFDs in a cohort of preterm infants, and were completed over a one-year period. A prospective audit, seeking the same data as in Phase 1, continued in Phase 2 to allow evaluation of the representativeness of Phase 2 data.
2.1Phase 1
A specifically devised proforma (Appendix C) was used to capture anonymized data of all preterm infants admitted to the NICU from January 1, 2009 to December 31, 2010. This provided retrospective data on GA; birth weight (BW); gender; multiple births; date of admission; date of discharge; LOS; and mortality. The first author logged data for each year separately. All anonymized information was coded and then inputted into a Microsoft Excel database, analysed and checked for accuracy by both authors. All data were password protected and stored on cite.
2.2Phase 2
This prospective cohort study was conducted in the NICU over an 18-month period. Inclusion criteria were all preterm infants with extremely low birth weight (ELBW), very low birth weight (VLBW), and low birth weight (LBW) admitted to the NICU over a one-year period. Preterm infants who were considered palliative (i.e., considered critically ill by the attending neonatologist and not expected to survive) were excluded from the OFR and CFSE assessments, but their data was captured in the file audit for the year. Participants were recruited via a gatekeeper and legal guardians provided consent. The OFR assessment was completed by first author and bedside nurses, who were trained by the first author. Training was provided by first author and included review and bedside practice of study tools for all nursing staff, one month prior to initiation of data collection, in addition to written materials. All CFSEs were completed by the first author, as there were no other SLPs in Cyprus that were trained in assessment of feeding and swallowing in paediatric population, at the time of the study. It is of note that SLP services, that is, bedside clinical and instrumental assessments of swallowing, were not provided at this NICU prior to this study. A Fiberoptic Endoscopic Evaluation of Swallowing (FEES) was planned as an instrumental assessment in collaboration with the attending Paediatric Pulmonologist, however, it was considered by the ethics review committee as being invasive and therefore was not utilized. Participants were all preterm infants with ELBW and VLBW admitted to the NICU were evaluated. One in 3 LBW infants were randomly selected, according to a random number scale, by a clinical nurse manager. This was determined because of study feasibility with a large number of LBW preterm infants admitted annually to the NICU.
In the absence of standardized OFR and CFSE tools in preterm infants at the time of the data collection, two instruments were adapted from key components of various scales and assessment tools, as one standalone was not considered to provide comprehensive information for this population. These were: a) the Oral Feeding Skills of Preterm Infants (OFS) (Lau & Smith, 2011), b) the Early Feeding Skills (EFS) Assessment (Thoyre, Shaker, & Pridham, 2005), c) the Supporting Oral Feeding in Fragile Infants (SOFFI) Method (Philbin & Ross, 2011) and d) Cue-Based Oral Feeding Clinical Pathway (Kirk, Alder, & King, 2007) and e) The Modified Brazelton Behavioural State scale (1984), that was already being utilized by nursing and therefore was adopted as part of the study. Infants were recruited from January 1st to December 31st 2013. Data collection continued until April 30, 2014, as infants recruited in December 2013, with less than 30 weeks GA were only eligible for CFSE in 2014.
The OFR tool consisted of three subsections: a) Behavioural state observed at rest and during daily cares, b) Physiologic stability at rest and during daily cares, and c) Non-Nutritive Sucking (NNS) assessment. When the aforementioned criteria in the OFR tool were established, the CFSE was administered. In the event that an infant was not able to establish OFR for a total of 3 consecutive assessments over a period of 2–7 days, the infant was described as having a PFD.
The CFSE comprised three subsections: a) Sucking pattern, b) Physiologic stability and behavioural state, before, during, and after oral feeding, and c) Signs and symptoms of swallowing impairments during and after oral feeding. In addition to the documentation of all the above observations and measurements, any significant change in vital signs (i.e., heart rate (HR), respiratory rate (RR) and oxygen saturation (SpO2) levels), as per the specific NICU’s policies and procedures were recorded. A significant variation was characterised as a 5–10%variation from baseline measures, as per the NICU’s protocol.
Table 1
Profile of preterm infants with ELBW, VLBW or LBW in Cyprus NICU in 2009, 2010, and 2013
| 2009 | 2010 | 2013 | p* | |
| Male: Female∞ | 0.904 | |||
| 287:233 | 288:219 | 237:221 | ||
| (55.2%: 44.8%) | (56.8%: 43.2%) | (51.7%: 48.3%) | ||
| Birth Weight# | 0.505 | |||
| Range | 530 g–2500 g | 530 g–2500 g | 500–2500 g | |
| Mean | 1750 g | 1750 g | 1799.54 g | |
| SD | ±458.01 g | ±458.01 g | ±460.01 g | |
| Gestational age# | 0.156 | |||
| Range | 24 wks–36 wks +5 days | 23 wks–36 wks +5 days | 23 wks–36 wks +6days | |
| Mean | 33 wks +2 days | 33 wks +4 days | 33 wks +0 days | |
| SD | ±2 wks +5 days | ±2 wks +6 days | ±2 wks +5 days | |
| Length of Stay# | 0.151 | |||
| Range | 0 –208 days | 0 –130 days | 0–363 days | |
| Mean | 25 days | 20 days | 28.65 days | |
| SD | ±26.2 days | ±22.2 days | ±35.22 days | |
| Mortality∞ | 0.001 | |||
| 19/520 (4.4%) | 23/507 (4.5%) | 4/458 (0.9%) | ||
| Multiple Births∞ | 0.117 | |||
| 199 (45.6%) | 162 (40.1%) | 214 (46.7%) |
*p value < 0.05 considered statistically significant. ∞Pearson Chi squared and Fisher’s exact test. #Kruskal Wallis non parametric test. ELBW extremely low birth weight, VLBW very low birth weight, LBW low birth weight.
2.3Data analysis
2.3.1Phase 1
Descriptive statistics of frequencies and percentages were used for Phase 1 data. Data were analysed for each year separately and examined as a whole, using the mean, standard deviation (SD), and range for variables.
2.3.2Phase 2
Analysis was conducted using Microsoft EXCEL (2010) and then transferred to SPSS version 22.0 (IBM SPSS Statistics for Windows,) for statistical analysis. Data from January 1st, 2013 to April 30th, 2014 were analysed from using the range, mean, SD for variables and tabulation of frequencies. T-tests were used to assess normally distributed continuous variables to test for the differences between 2009, 2010, and 2013. A p value of ≤0.05 was set for statistical significance.
3Results
3.1Profile of preterm infants admitted to the NICU 2009, 2010, and 2013
The profiles of all preterm infants admitted to the NICU in 2009 and 2010 (Phase 1) were compared to those of 2013 (Phase 2). Data from 1,485 preterm infants were collated. Five hundred and twenty preterm infants were admitted to the NICU in 2009, 507 preterm infants in 2010, and 458 preterm infants in 2013 (Table 1).
The male to female ratio was consistent across all 3 years (55.2%: 44.8%in 2009; 56.8%: 43.2%for 2010; 51.2%: 48.3%in 2013). GA ranges were also consistent across all three years. BW ranged from 500 g to 2,500 g, (mean: 1,799.54 g, SD: 460.01 g) across all 3 years. Multiple births accounted for 45.6%of all pre-term births in 2009; 40.1%in 2010 and 46.7%in the 2013 cohort. Overall, the profile of preterm infants for 2009, 2010, and 2013 were very similar, specifically on BW, GA, gender distribution, and multiple births. There was a significant decrease in mortality in 2013, however, there was a marked increase in the range of LOS in the NICU for that year.
Table 3
Primary and comorbid diagnoses upon discharge from the NICU in 2013 - Structural Equation Modelling (SEM) analysis
| Respiratory | Neurologic | IUGR | BPD | CHD | GI | Genetic | Structural Anomaly | Sepsis | Jaundice | AOP | |
| Respiratory | 34 | ||||||||||
| Neurological | 7 | 2 | |||||||||
| IUGR | 21 | 1 | 9 | ||||||||
| BPD | 23 | 3 | 3 | 0 | |||||||
| CHD | 24 | 2 | 3 | 2 | 0 | ||||||
| GI | 5 | 0 | 0 | 0 | 2 | 3 | |||||
| Genetic | 2 | 0 | 3 | 0 | 1 | 0 | 0 | ||||
| Structural Anomaly | 7 | 0 | 1 | 0 | 3 | 0 | 0 | 2 | |||
| Sepsis | 23 | 2 | 5 | 8 | 1 | 2 | 0 | 1 | 0 | ||
| Jaundice | 81 | 1 | 12 | 17 | 12 | 4 | 0 | 2 | 13 | 13 | |
| AOP | 17 | 1 | 2 | 7 | 2 | 0 | 1 | 0 | 5 | 11 | 0 |
NICU neonatal intensive care unit, IUGR intrauterine growth restriction, BPD bronchopulmonary dysplasia, CHD congenital heart disease, GI gastrointestinal, AOP anaemia of prematurity
3.2Profile of preterm infants assessed for paediatric feeding disorders.
Of the 458 preterm infants admitted to the NICU in 2013, 224 were assessed for PFDs using the CFSE, following establishment of OFR, between January 1st and April 30th, 2014. Cross-tabulation of preterm infants with PFDs in comparison to those who were within normal limits (WNL) were extrapolated using the 1:3 ratio of the preterm infants with LBW and categorized according to their BW classification (Table 2).
Table 2
Results of CFSE according to birth weight classification
| WNL | PFD | |||
| N | Percent % | N | Percent % | |
| ELBW | 0 | 0 | 28 | 100 |
| VLBW | 23 | 31.1 | 51 | 68.9 |
| LBW | 268 | 75.4 | 88 | 24.6 |
| TOTAL | 291 | 63.5 | 167 | 36.5 |
CFSE clinical feeding and swallowing evaluation, ELBW extremely low birth weight, VLBW very low birth weight, LBW low birth weight, WNL within normal limits, PFD paediatric feeding disorder.
According to this analysis, 36.5%(n = 167) of preterm infants exhibited PFDs. This is taken as the prevalence of PFDs in this population for 2013. All preterm infants with ELBW and over two-thirds of preterm infants with VLBW had PFDs.
As seen in Table 3, preterm infants with PFDs, primarily exhibited respiratory difficulties and jaundice, with a significant number with co-occurring Bronchopulmonary Dysplasia (BPD), Intrauterine Growth Restriction (IUGR), Congenital Heart Disease (CHD), sepsis, and Anaemia of Prematurity (AOP).
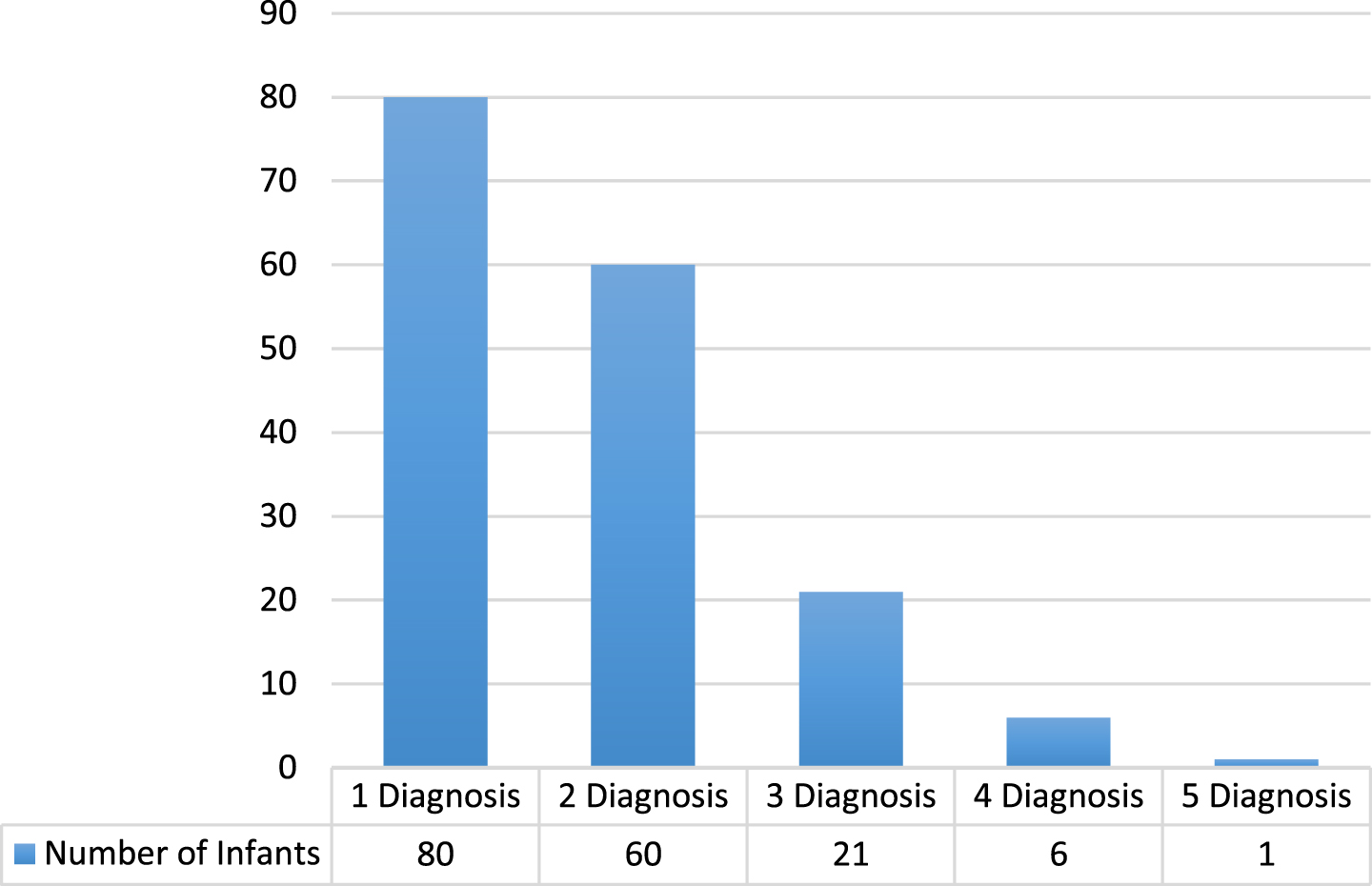
One criterion for discharge from the NICU is establishment of full oral feeding. Therefore, it was important to compare the LOS of preterm infants whose feeding and swallowing was WNL with those who had a PFD. There was a statistically significant difference between the LOS of those preterm infants with a PFD (mean: 58.35 days, SD±13.61) compared with 18.58 days (SD±13.61) for those infants were considered to be WNL. Additionally, medical diagnoses and co-morbidities were examined for all preterm infants discharged from the NICU in 2013. Almost half (47.4%) of those who exhibited PFDs presented with one medical diagnosis, whereas over one third (36%) had 2 co-occurring medical diagnoses, 12.7%with 3 diagnoses, approximately 4%with 4 or more diagnoses (Fig. 1).
Fig. 1
Number of Comorbid Diagnoses per Preterm Infant with a Paediatric Feeding Disorder in 2013.
4Discussion
Epidemiological research in paediatric dysphagia is scarce. This research is unique in that it profiles a cohort of preterm infants over a three-year period, with a primary aim of establishing the prevalence, aetiology, and impact of PFDs in preterm infants with ELBW, VLBW and LBW. The stability of the population profile, as confirmed by the audit data over the three-year period, lends credibility to the prospective study in that this participant profile is typical of preterm infants admitted previously to the NICU in Cyprus. The prevalence of PFDs in this cohort of preterm infants, was 36.5%. These findings are comparable to those by Uhm, Chang, Cheon, & Kwon (2013) who found that 40%of preterm infants whose GA was between 25–37 weeks exhibited evidence of aspiration. Similarly, Lee, Chang, Yoo, Ahn, Seo, Choi, . . . & Park (2011) found that 26.8%(n = 11) of their cohort of preterm infants with VLBW were found to have impaired airway protection on Videofluoroscopic Swallow Study. The aetiology and extent of PFDs in preterm infants poses a challenge (Reilly & Ward, 2005). It is well recognized that preterm infants admitted to the NICU present with a wide range of medical complexities, which often co-occur with other conditions (Irace, Dombrowski, Kawai, Watters, Choi, Perez, et al., 2019; Jadcherla, 2016; Gianni, Sannino, Bezze, Plevani, di Cungo, Roggero, et al., 2015; Da Costa, van der Schans, Zweens, Boelema, van der Meij, Boerman, et al, 2010; Rommel et al,2003; Hawdon, Beauregard, Slattery, & Kennedy, 2000). This study found that preterm infants with PFDs primarily exhibited respiratory difficulties and neonatal jaundice, with a significant number presenting with co-occurring BPD, IUGR, CHD, sepsis, and AOP. Respiratory disorders were diagnosed in 40%of this cohort and BPD in an additional 4%. This finding is not surprising given that respiratory disorders, BPD, and CHD are known to directly affect the integrity of feeding and swallowing (Jadcherla, et al., 2009). Wang et al., (2004), in a study of late preterm infants (i.e., 34 to 36 weeks and 6 days GA), found that respiratory disorders had a significant impact on feeding and swallowing (Wang, Dorer, Fleming, & Catlin, 2004). The demands of a precisely coordinated suck-swallow-breathe sequence (Mizuno, Nishida, Taki, Hibino, Murase, Sakurai, & Itabashi, 2007; Gewolb & Vice, 2006) placed on the preterm infant, poses significant challenges during oral feeding, especially when a respiratory disorder is present.
Given that almost half of those who exhibited PFDs in this study presented with one medical diagnosis, the medical complexities of this population are clear. Current research suggests that co-occurring diagnoses and co-morbidities may have an effect on feeding and swallowing ability, however, research to date has neither quantified, nor established a correlation between the number of co-occurring diagnoses and the extent of the PFD in preterm infants. In a recent study by Edney, Jones, & Boaden (2019), they found that an infants’ medical history can be significant in identifying those at risk of feeding difficulties. This is important from an epidemiological perspective, particularly when examining risk factors that predispose preterm infants to developing PFDs.
LOS is often associated with the extent of a preterm infants’ medical stability and feeding and swallowing ability (McNeil, 2008). Given that attainment of full oral feeding is one of the primary criteria for discharge from the NICU, this research found a significant correlation between LOS; BW; and GA of preterm infants with PFDs. LOS of preterm infants with PFDs was significantly longer (58.35 days, SD±13.61days) than preterm infants whose feeding was WNL (18.58 days, SD±13.61 days). A large, multicentre retrospective study in Finland (Korvenranta, Linna, Häkkinen, Peltola, Andersson, Gissler, et al., 2007) on preterm infants born less than 32 weeks gestation and weighing less than 1501 g, found that mean LOS was 53 days and confirmed a correlation between increasing LOS with subsequent decreasing GA and BW, indicating that this may lead to more significant morbidity. It is noteworthy that the mortality rate for 2013 in this study was significantly lower than in 2009 and 2010, whereas there was a significant increase in the LOS in 2013, particularly in the range of LOS (i.e., 0–363 days). A decrease in mortality may potentially have caused an increase in the LOS of critically ill preterm infants.
GA and BW have long been cited in the literature as risk factors for developing PFDs (Irace, et al., 2019; Wood, Costeloe, Gibson, Hennessy, Marlow, & Wilkinson, 2003; Rommel, et al., 2003). Wood, et al., (2003), who prospectively assessed 283 preterm infants born < 26 weeks gestation at 30 months corrected age, concluded that 25–30%of all preterm infants are at risk for poor feeding and swallowing outcomes. The results of this research also determined a significant correlation of GA and BW, indicating that these variables are considered risk factors for developing PFDs. In addition, this research is consistent with similar findings, which report that the lower the BW and GA, the higher the probability (25–40%) of preterm infants presenting with PFD (Törölä, et al., 2012; St. Pierre, Khattra, Johnson, Cender, Manzano, & Holsti, 2010; Jadcherla, et al., 2009). According to the results of this research, neurological disorders (p < 0.004); CHD (p < 0.005); and structural anomalies (p < 0.028) were considered significant risk factors for preterm infants developing a PFD. Additionally, it was found that over two-thirds (65.5%) of preterm infants with a medical diagnosis of CHD, exhibited PFDs.
A key objective of this study was to use the findings to facilitate planning and provision of early intervention services in the NICU. In a study on preterm birth rates in the U.S.A., it was reported that the societal cost of NICU care is estimated at $26 billion annually (Duryea, McIntire, & Leveno, 2015). In addition, Underwood et al., (2007) estimated that 15%of preterm infants required at least one rehospitalisation per year, within the first year of life (Underwood, Danielsen, & Gilbert, 2007). Though the exact cost of NICU stay was not available for this cohort and will vary from country to country, the implications of these figures emphasizes how crucial early intervention is to preterm infants in the NICU, which not only aims to improve the long-term developmental outcomes of preterm infants, but also to reduce the length of hospital stay in the NICU and hence the cost.
There were some limitations to the study. Firstly, although instrumental evaluation of swallowing was planned, determination of PFDs did not include instrumental assessments of swallowing in this cohort. This is a limitation of epidemiological studies in this population, as it is not always ethical to subject preterm infants to additional instrumental investigations if sufficient information on feeding and swallowing is available clinically. Secondly, one researcher completed the clinical assessments of feeding and swallowing lending the possibility of bias. However, this did ensure consistency in the observations and the scoring during the assessments. A final limitation is that the researchers were unable to follow up these infants longitudinally to determine their outcomes in the longer term and the resolution of PFDs. It is recommended that future studies consider this.
This epidemiological study profiles preterm infants born in Cyprus over a three period and is unique in terms of providing prevalence data for an entire population. The WHO (2014) statistical reports show that there are many similarities regarding preterm birth rates and infant mortality rates among countries, therefore the research data can be generalizable to other countries (Liu, Oza, Hogan, Chu, Perin, & Zhu, 2016; Howson, et al., 2012). In addition, it adds to the literature on PFDs in preterm infants and, ultimately, should be useful in determining risk profiles for these infants, as well as contributing to future service planning.
Acknowledgments
We would like to thank all the families and NICU staff of the Archbishop Makarios III Hospital in Nicosia, Cyprus, for their unending support during this research. Additionally, we would like to extend our gratitude to Dr. Andreas Hadjidemetriou, Dr. Christina Karaoli, Dr. Panayiotis Yiallouros, Mr. Phivos Ioannou, and Dr. Leda Hadjihanna for their guidance and assistance throughout. There is no funding source.
Authorship
Both authors, Panayiota Senekki-Florent (PSF) and Margaret Walshe (MW) contributed to the study conception and design. Material preparation, data collection and analysis were performed by PSF. The first draft of the manuscript was written by PSF and MW. Both authors, PSF and MW, commented on previous versions of the manuscript. Both authors read and approved the final manuscript.
Conflict of interest
The authors have no conflict of interest to report.
Given her role as an Associate Editor, Margaret Walshe had no involvement nor access to information regarding the peer review of this article.
Ethical considerations
(a) Retrospective Chart Review Study: This retro-spective chart review involving human participants was in accordance with the ethical standards of the institution and national research committee and with the 1963 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of the University of Dublin, Trinity College, approved this study. (b) Prospective Study: All procedures performed involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the University of Dublin, Trinity College and the Bioethics Committee of Cyprus (EEBK/EΠ/2014/04). In-formed consent was obtained from all parents/care-breakgivers of preterm infants included in the study.
Supplementary material
[1] Appendices are available in the electronic version of this article: https://dx.doi.org/10.3233/ACS-210025.
References
1 | Altimier, L , & Phillips, R. ((2016) ). The neonatal integrative developmental care model: advanced clinical applications of the seven core measures for neuroprotective family-centered developmental care. Newborn and Infant Nursing Reviews, 16: (4), 230–244. https://doi.org/10.1053/j.nainr.2016.09.030. |
2 | Brazelton, T.B. ((1984) ). Neonatal Behavioral Assessment Scale. (2nd ed.). Blackwell Scientific, London. |
3 | Cyprus Health Monitoring Unit. (2016). Perinatal Health Indicators for the Year 2014. https://www.moh.gov.cy/Moh/MOH.nsf/All/8DC461429CBC4DE7C22579CE002EF07D/$file/Perinatal%20Health%20Report%202016_Cyprus%20Maternity%20Units%202014.pdf. |
4 | Da Costa, S , van der Schans, C.P , Zweens, M.J , Boelema, S.R , van der Meij, E , Boerman, M.A , & Bos, A.F. ((2010) ). The maturation of sucking patterns in preterm, small-for-gestational age infants. Journal of Pediatrics, 157: , 603–609. https://doi.org/10.1159/000281106. |
5 | Duryea, E.L , McIntire, D.D , & Leveno, K.J. ((2015) ). The rate of preterm birth in the United States is affected by the method of gestational age assignment. American Journal of Obstetrics and Gynecology, 213: , 1.e1–1.e5. https://doi.org/10.1016/j.ajog.2015.04.038. |
6 | Edney, S.K , Jones, S , & Boaden, E. ((2019) ). Screening for feeding difficulties in the neonatal unit: Sensitivity and specificity of gestational age vs. medical history. . Journal of Neonatal Nursing, 25: (3), 116–120. https://doi.org/10.1016/j.jnn.2018.10.004. |
7 | Euro-Peristat project with SCPE and Eurocat. (2010). European Perinatal Health Report: The health and care of pregnant women and babies in Europe in 2010. https://www.europeristat.com/images/doc/EPHR2010_w_disclaimer.pdf. |
8 | Gewolb, I.H , & Vice, F.L. ((2006) ). Maturational changes in the rhythms, patterning, and coordination of respiration and swallow during feeding in preterm and term infants. Dev Med Child Neurol, 48: (7), 589–94. https://doi.org/10.1017/S001216220600123X. |
9 | Giannì, M , Sannino, P , Bezze, E , Plevani, L , di Cugno, N , Roggero, P , et al, ((2015) ). Effect of co-morbidities on the development of oral feeding ability in pre-term infants: a retrospective study. Scientific Reports, 5: , 1–8. https://doi.org/10.1038/srep16603. |
10 | Goday, P.S , Huh, S.Y , Silverman, A , Lukens, C.T , Dodrill, P , Cohen, S.S , et al, ((2019) ). Pediatric Feeding Disorder. Consensus definition and conceptual framework. Journal of Pediatric Gastroenterology and Nutrition, 68: (1), 124–129. https://doi.org/10.1097/MPG.0000000000002188. |
11 | Harding, C , & Cockerill, H. ((2015) ). Managing eating and drinking difficulties (dysphagia) with children who have learning disabilities: What is effective? Clinical Child Psychology and Psychiatry, 20: (3), 395–405. https://doi.org/10.1177/1359104513516650. |
12 | Harding, C , Cockerill, H , Cane, C , & Law, J. ((2018) ). Using non-nutritive sucking to support feeding development for premature infants: A commentary on approaches and current practice. Journal of Pediatric Rehabilitation Medicine, 11: (3), 147–152. https://doi.org/10.3233/PRM-170442. |
13 | Harding, C , Levin, A , Crossley, S.L , Murphy, R , & Van den Engel-Hoek, L. ((2019) ). Effects of early communication intervention on speech and communication skills of preterm infants in the neonatal intensive care unit (NICU): a systematic review. Journal of Neonatal Nursing, 25: (4), 177–188. https://doi.org/10.1016/j.jnn.2019.04.004. |
14 | Harding, C , Mynard, A , & Hills, E. ((2018) ). Identification of premature infant states in relation to introducing oral feeding. Journal of Neonatal Nursing, 24: (2), 104–110. https://doi.org/10.1016/j.jnn.2017.11.018. |
15 | Hawdon, J.J , Beauregard, N , Slattery, J , & Kennedy, G. ((2000) ). Identification of neonates at risk of developing feeding problems in infancy. Developmental Medicine & Child Neurology, 42: , 235–239. https://doi.org/10.1017/s0012162200000402. |
16 | Hoogewerf, M , Ter Horst, H.J , Groen, H , Nieuwenhuis, T , Bos, A.F , & van Dijk, M.W.G. ((2017) ). The prevalence of feeding problems in children formerly treated in a neonatal intensive care unit. Journal of Perinatology, 37: (5), 578–584. https://doi.org/10.1038/jp.2016.256. |
17 | Howson, C.P , Kinney, M.V , McDougall, L , & Lawn, J.E , Born Too Soon Preterm Birth Action Group ((2013) ). Born too soon: preterm birth matters. Reproductive Health, 10 Suppl1: (Suppl 1), S1. https://doi.org/10.1186/1742-4755-10-S1-S1. |
18 | Irace, A.L , Dombrowski, N.D , Kawai, K , Watters, K , Choi, S , Perez, J , et al, ((2019) ). Evaluation of aspiration in infants with Laryngomalacia and recurrent respiratory and feeding difficulties. JAMA Otolaryngology, Head & Neck Surgery, 145: (2), 146–151. https://doi.org/10.1001/jamaoto.2018.3642. |
19 | Jadcherla S. ((2016) ). Dysphagia in the high-risk infant: potential factors and mechanisms. American Journal Clinical Nutrition, 103: (2), 622S–8S. https://doi.org/10.3945/ajcn.115.110106. |
20 | Jadcherla, S.R , Wang, M , Vijayapal, A.S , & Leuthner, S.R. ((2009) ). Impact of prematurity and co-morbidities on feeding milestones in neonates: a retrospective study. Journal of Perinatology, 30: (3), 201–208. https://doi.org/10.1038/jp.2009.149. |
21 | Kallioinen, M , Eadon, H , Murphy, M.S , & Baird, G. ((2017) ). Developmental follow-up of children and young people born preterm: summary of NICE guidance. BMJ 358: . https://doi.org/10.1136/bmj.j3514. |
22 | Kirk, A.T , Alder, S.C , & King, J.D. ((2007) ). Cue-based oral feeding clinical pathway results in earlier attainment of full oral feeding in premature infants. Journal of Perinatology, 27: (9), 572–8. https://doi.org/10.1038/sj.jp.7211791. |
23 | Korvenranta, E , Linna, M , Häkkinen, U , Peltola, M , Andersson, S , & Gissler, M , et al, ((2007) ). Differences in the length of initial hospital stay in very preterm infants. Acta Pædiatrica, 96: , 1416–1420. https://doi.org/10.1111/j.1651-2227.2007.00471.x. |
24 | Lau C, , & Smith, EO ((2011) ). A novel approach to assess oral feeding skills of preterm infants. Neonatology, 100: (1), 64–70. https://doi.org/10.1159/000321987. |
25 | Lee, J.H , Chang, Y.S , Yoo, H.S , Ahn, S.Y , Seo, H.J , Choi, S.H , Jeon, G.W , Koo, S.H , Hwang, J.H , & Park, W.S. ((2011) ). Swallowing dysfunction in very low birth weight infants with oral feeding desaturation. World Journal of Pediatrics, 7: (4), 337–343. https://doi.org/10.1007/s12519-011-0281-9. |
26 | Liu, L , Oza, S , Hogan, D , Chu, Y , Perin, J , Zhu, J , et al, ((2016) ). Global, regional, and national causes of under- mortality in: an updated systematic analysis with implications for the Sustainable Development Goals. Lancet, 388: (10063), 3027–3035. https://doi.org/10.1016/S0140-6736(16)31593-8. |
27 | McNeil, D.A. ((2008) ). The Incidence and Correlates of Feeding Problems in Premature Infants Post Neonatal Intensive Care. Unpublished doctoral dissertation, University of Calgary, Calgary, Canada. Retrieved from ProQuest Dissertations and Theses database (NR44537). |
28 | Mizuno, K , Nishida, Y , Taki, M , Hibino, S , Murase, M , Sakurai, M , & Itabashi, K. ((2007) ). Infants with bronchopulmonary dysplasia suckle with weak pressures to maintain breathing during feeding. Pediatrics, 120: (4), e1035–42. https://doi.org/10.1542/peds.2006-3567. |
29 | Pados, B.F , Hill, R.R , Yamasaki, J.T , Litt, J.S , & Lee, C.S. ((2021) ). Prevalence of problematic feeding in young children born prematurely: a meta-analysis. BMC Pediatrics, 21: (1), 110. https://doi.org/10.1186/s12887-021-02574-7. |
30 | Philbin, K.M , & Ross, E.S. ((2011) ). The SOFFI Reference Guides: Text, algorithms, and appendices: A manualized method for quality bottle feedings. Journal of Perinatology and Neonatal Nursing, 25: (4), 360–380. https://doi.org/10.1097/JPN.0b013e31823529da. |
31 | Reilly, S , & Ward, E. ((2005) ). The epidemiology of dysphagia: Describing the problem-are we too late?. Advances in Speech-Language Pathology, 7: (1), 14–23. https://doi.org/10.1080/14417040500055110. |
32 | Rommel, N , De Meyer, A.M , Feenstra, L , & Veerman-Wauters, G. ((2003) ). The complexity of feeding problems in 700 infants and young children presenting to a tertiary care institution. Journal of Pediatric Gastroenterology and Nutrition, 37: , 75–84. https://doi.org/10.1097/00005176-200307000-00014. |
33 | Sanchez, K , Spittle, A.J , Slattery, J.M , & Morgan, A.T. ((2016) ). Oromotor feeding in children born before weeks’ gestation and term-born peers at months’ corrected age. Journal of Pediatrics, 178: , 113–118.e1. https://doi.org/10.1016/j.jpeds.2016.07.044. |
34 | Schädler, G , Süss-Burghart, H , Toschke, A.M , von Voss, H , & von Kries, R. ((2007) ). Feeding disorders in ex-prematures: causes - response to therapy -long term outcome. European Journal of Pediatrics, 166: (8), 803–808. https://doi.org/10.1007/s00431-006-0322-x. |
35 | St. Pierre, A , Khattra, P , Johnson, M , Cender, L , Manzano, S , & Holsti, L. ((2010) ). Content validation of the infant malnutrition and feeding checklist for congenital heart disease: A tool to identify risk of malnutrition and feeding difficulties in infants with congenital heart disease. Journal of Pediatric Nursing, 25: (5), 367–374. https://doi.org/10.1016/j.pedn.2009.04.009. |
36 | Thoyre, S.M , Shaker, C , & Pridham, K.F. ((2005) ). The early feeding skills assessment for preterm infants. Neonatal Network, 24: (3), 7–16. https://doi.org/10.1891/0730-0832.24.3.7. |
37 | Törölä, H , Lehtihalmes, M , Yliherva, A , & Olsén, P. ((2012) ). Feeding skill milestones of preterm infants born with extremely low birth weight (ELBW).&. . Development, 35: , 187–194. https://doi.org/10.1016/j.infbeh.2012.01.005. |
38 | Uhm, K.E , Yi, S.H , Chang, H.J , Cheon, H.J , & Kwin, J. Y. ((2013) ). Videofluoroscopic swallowing study findings in full-term and preterm infants with dysphagia. Annals of Rehabilitation Medicine, 37: (2), 175. https://doi.org/org/10.5535/arm.2013.37.2.175. |
39 | Underwood, M.A , Danielsen, B , & Gilbert, W.M. ((2007) ). Cost, causes and rates of rehospitalization of preterm infants. Journal of Perinatology, 27: , 614–619. https://doi.org/10.1038/sj.jp.7211801. |
40 | Wang, M.L , Dorer, D.J , Fleming, M.P , & Catlin, E.A. ((2004) ). Clinical outcomes of near-term infants. Pediatrics, 114: , 372–376. https://doi.org/10.1542/peds.114.2.372. |
41 | Wood, N.S , Costeloe, K , Gibson, A.T , Hennessy, E.M , Marlow, N , & Wilkinson, A.R. ((2003) ). The EPICure study: growth and associated problems in children born at weeks of gestational age or less. Archives of Disability & Child Fetal Neonatology Education, 88: , F492–F500. https://doi.org/10.1136/fn.88.6.F492. |
42 | Zeitlin, J , Szamotulska, K , Drewniak, N , Mohangoo, A , Chalmers, J , Sakkeus, L , Irgens, L , Gatt, M , Gissler, M , & Blondel, B. ((2013) ). Preterm birth time trends in Europe: a study of countries. BJOG, 120: , 1356–1365. https://doi.org/10.1111/1471-0528.12281. |




