Applied current thermoacoustic imaging for biological tissues
Abstract
BACKGROUND:
The large differences of electrical characteristics can be used to reflect the physiological and pathological changes about biological tissues, and it can provide evidence for the early diagnosis and treatment of cancer in potential applications.
OBJECTIVE:
This paper describes a method called Applied Current Thermoacoustic Imaging (ACTAI) and explores the theory and demonstrates a low conductivity numerical simulation and fresh pork experimental studies.
METHODS:
In this paper, firstly, the principle of ACTAI is studied. In ACTAI, a target is applied with a microsecond width Gaussian pulse current. Then the target absorbs Joule heat and expands instantaneously, sending out thermoacoustic waves. The waves contain the conductivity information of the target. The waves received by sound transducers are processed by the time inversion method to reconstruct the sound source distribution of the target to illustrate the conductivity information of the target. Secondly, a square model with low conductivity was used as a target to conduct numerical simulation of ACTAI. Lastly, a fresh pork experiment study was conducted.
RESULTS:
The presented experimental results suggest that ACTAI can identify the conductivity changes information of the target with perfect imagery contrast and deep penetration.
CONCLUSION:
The ACTAI modality would benefit from the noncontact measurement and can be convenient for clinical application.
1.Introduction
In the past decades, more and more attention has been paid to the imaging method based on thermoacoustic effect [1, 2, 3, 4]. In the thermoacoustic effect, a biological tissue is irradiated in an electromagnetic field. The tissue absorbs some electromagnetic energy, and then because of thermo elastic expansion it subsequently leads to ultrasonic wave emission. The acoustic waves are related to the properties of the tissue, and could be collected by the ultrasound transducers setting around the tissue [5, 6]. Photoacoustic imaging [7, 8] and microwave-induced thermoacoustic imaging [9, 10] are two imaging modalities based on the thermoacoustic effect. Photoacoustic imaging, which uses light as excitation source, is only suitable for superficial applications. Compared with photoacoustic imaging, microwave-induced thermoacoustic imaging, which uses microwave radiation, obtains deeper penetration. The imaging depth depends on the tissue properties and the electromagnetic frequency used for irradiation. In principle, using lower frequencies can achieve deeper penetration [11].
To explore a potential imaging method for deeper penetration, Zheng et al. have theoretically studied the feasibility using radio frequency magnetic field below 20 MHz to electric conductance or magnetic nanoparticles targets to modulate thermoacoustic imaging, called magnetically mediated thermoacoustic imaging (MMTAI) [12, 13]. A metal sample was used to experiment with, and the theromacoustic imaging was obtained, which was similar to the sample in terms of shape, and a coherent frequency domain method was discussed in great detail. The imaging method has broad prospects for the development of thermoacoustic imaging using low frequency magnetic field, and it has been preliminarily proved by the results of numerical simulation and physical experiment. In this paper, we report a new imaging method called Applied Current Thermoacoustic Imaging (ACTAI). In ACTAI, a
2.Theory
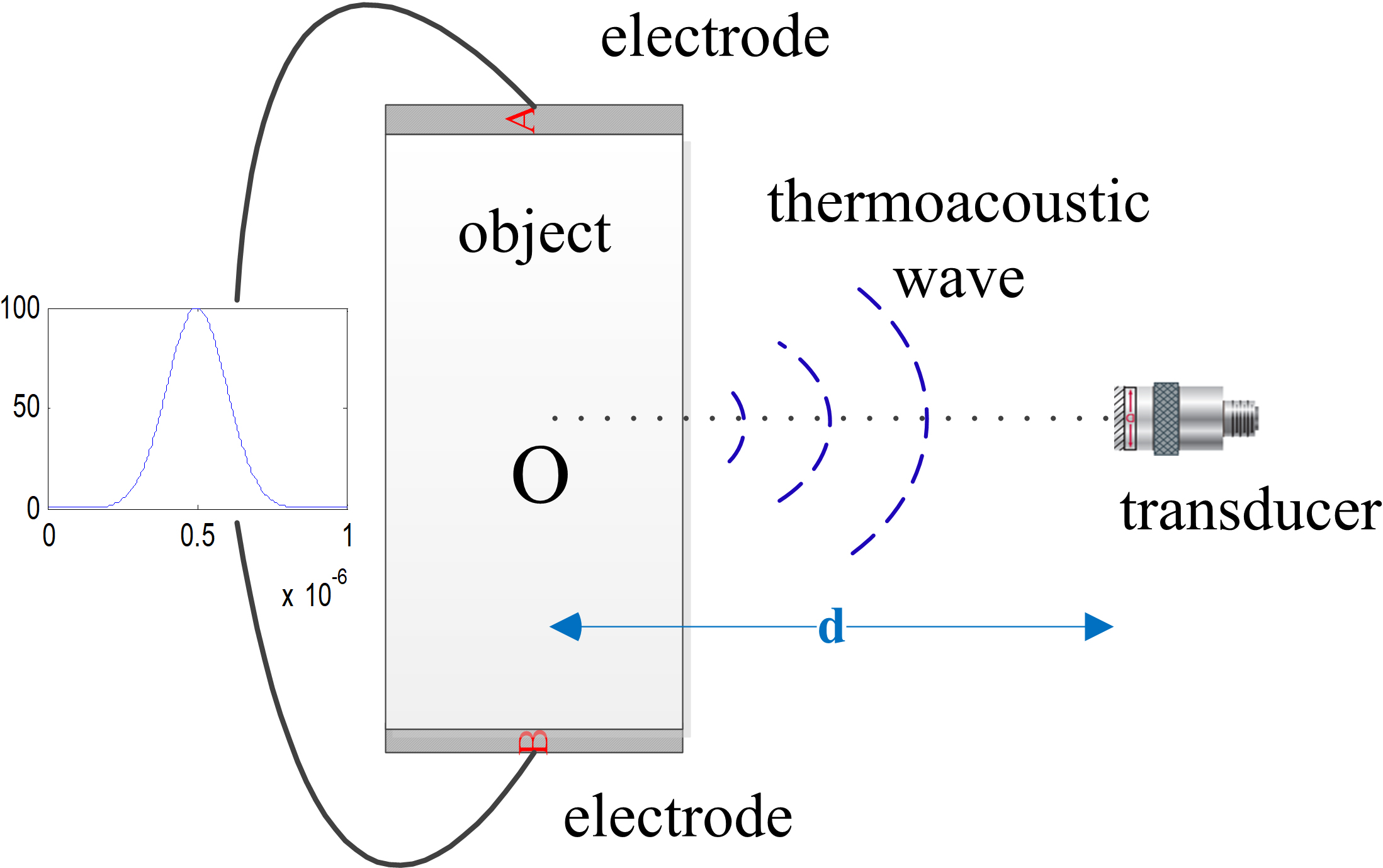
A theoretical analysis shows that the acoustic pressure has a relationship with the conductivity of the target, which is described by a detailed deduction. The conceptual framework of ACTAI is shown in Fig. 1. In ACTAI, the target is injected in a Gauss pulse current. This produces one pulse electric field
(1)
Figure 1.
The principle of ACTAI.
where
(2)
where
(3)
where
The sound pressure
(4)
where
3.Numerical simulation
In order to verify the theory of ACTAI, the coupling analysis of electromagnetic field and acoustic field for low conductivity simulation model was carried out using the finite element method. In order to simulate tumor tissue surrounded by healthy tissues, the simulation sample was made by putting a square tumor tissue with 1 cm length into a rectangle healthy tissue with a size of 30 mm
Figure 2.
The results of the simulation in the ACTAI. (a) The conductivity distribution in the target. (b) Normalized waveforms obtained by its detector.
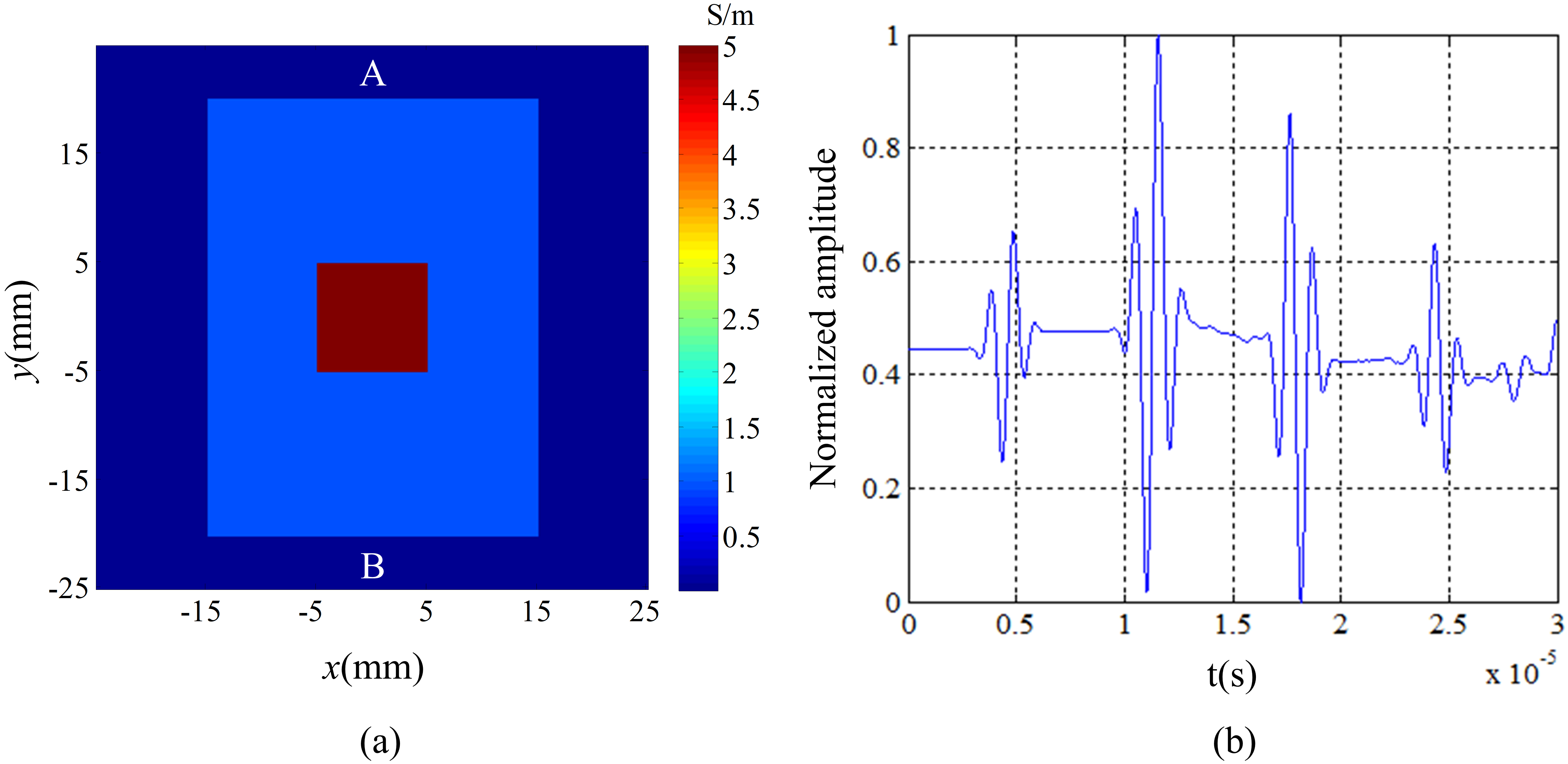
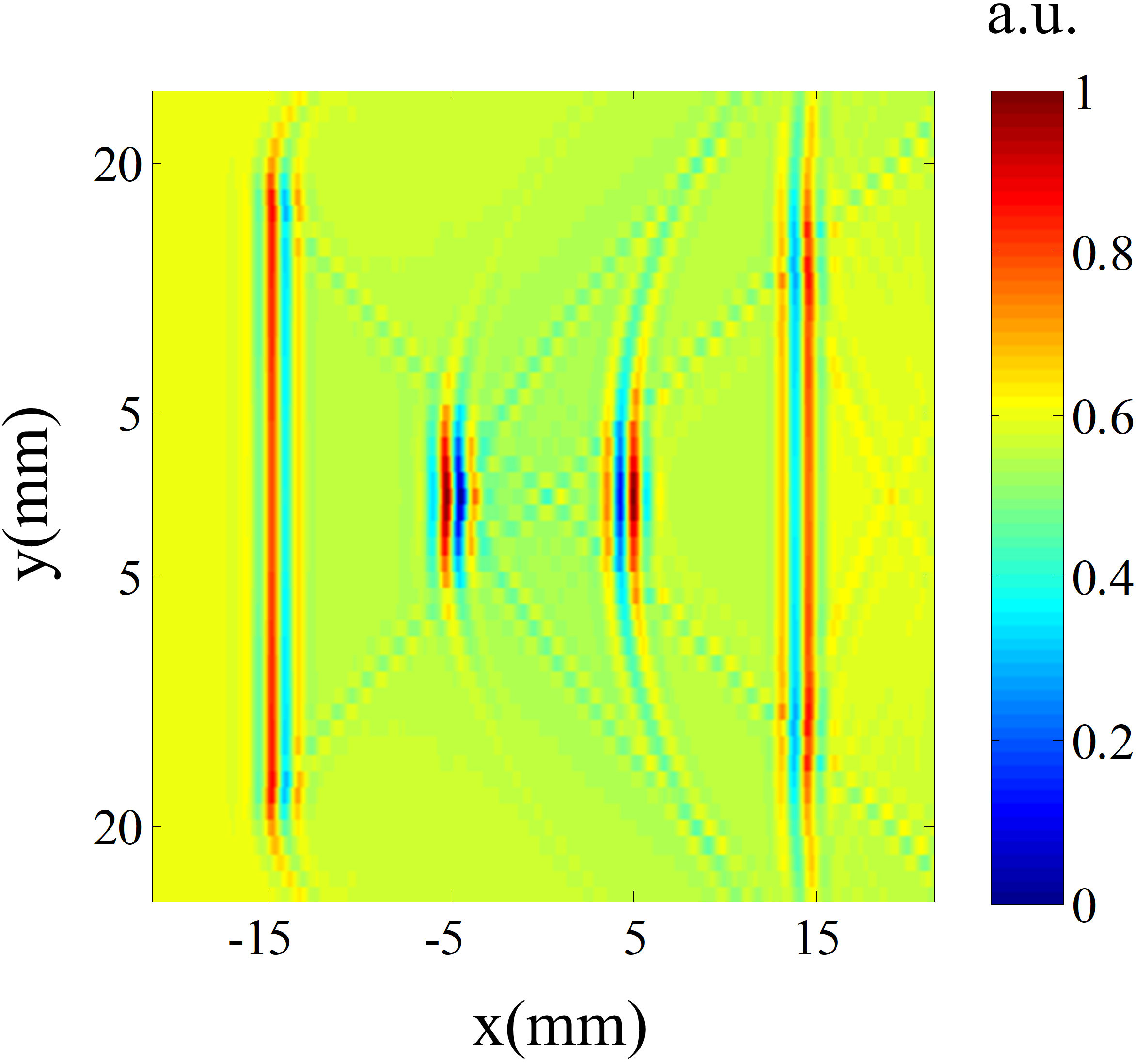
Figure 3.
B-scan image of the simulation model.
The signals received by the detectors are the convolution of the sound pressure and the impulse response of the sound transducers in practical experiments. So the simulation signals are processed with the convolution of the pressure to agree with the experiments. The normalized waveform detected by the transducer is obtained by Eq. (4) and shown in Fig. 2b. 3.5
The transducer is moved in the line
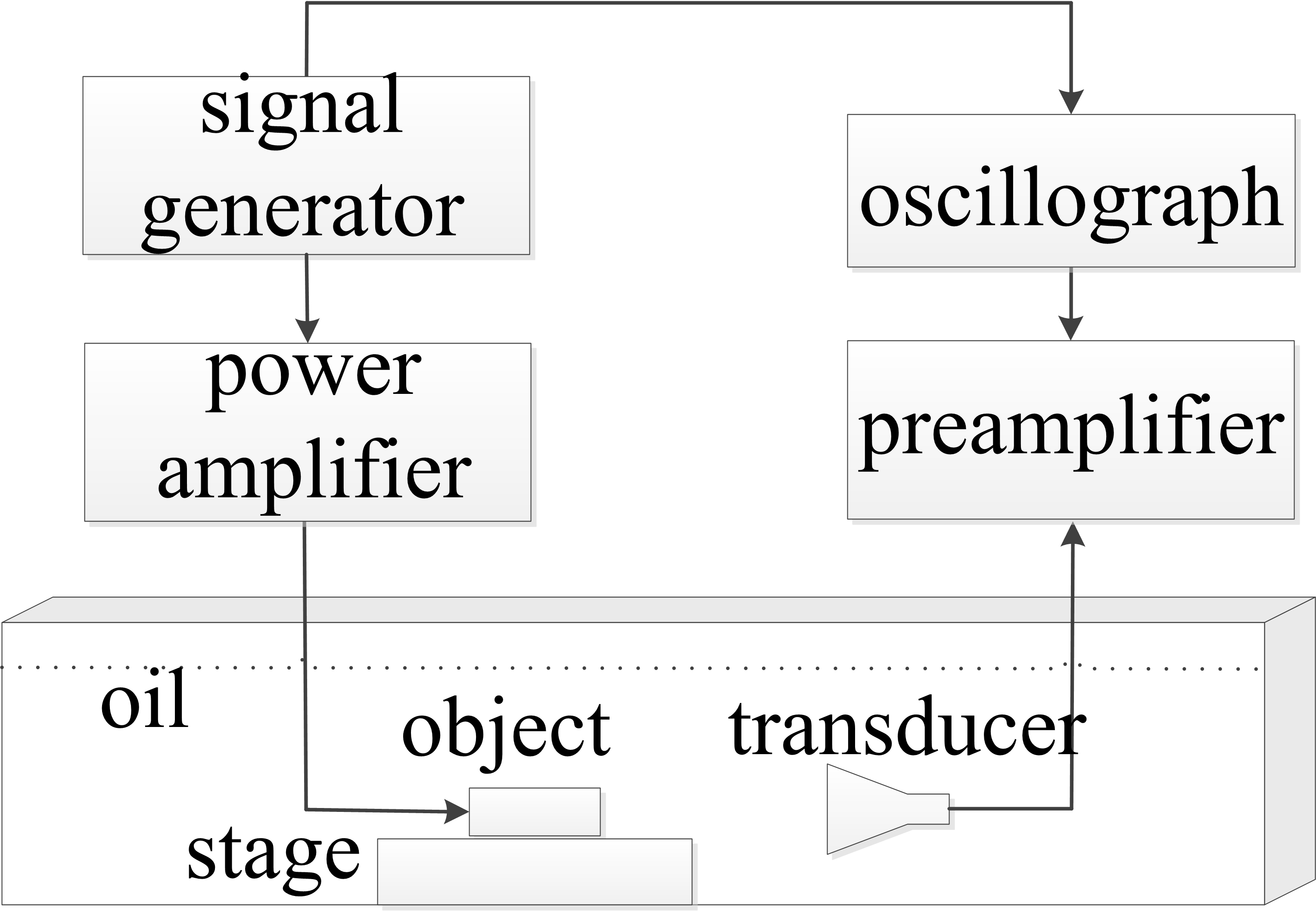
Figure 4.
Schematic view of the ACTAI system.
4.Experiments
To verify the feasibility of this method, the ACTAI experiments have been conducted using phantoms and pork. The abridged general view of the experimental system is shown in Fig. 4. In the experiment, to couple the sound waves, the transformer insulation oil was put in a plastic container to an established height for immersing the transducer and the target. The sound velocity of the transformer insulation oil is measured as 1400 m/s. The target is well fixed on a rotation mechanism and remains the same horizontal height as the center axis of the detector. The current excitation consists of a signal generator and a power amplifier, applying 1
Figure 5.
Gel phantom model. (a) Schematic diagram of the gel phantom and the transducer. (b) Acoustic waveform from the gel phantom.
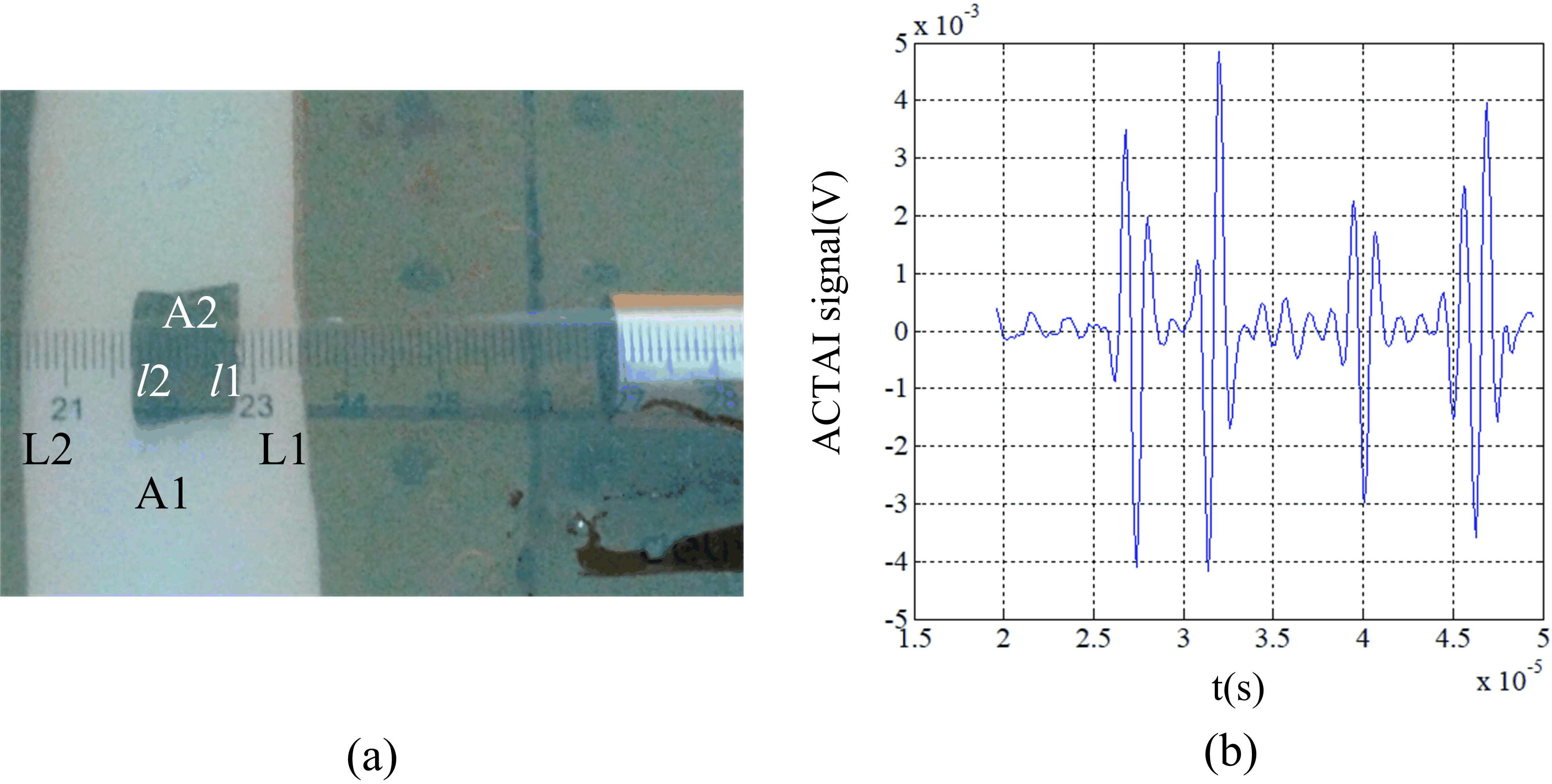
Figure 6.
Schematic diagram of the pork and the transducer.

In the experiments, the model was made of 10% cooled salinity gel as shown in Fig. 5. The phantom is buckled from the rectangular A1 model with a small rectangular A2. The length and width of A1 are 14 cm and 3 cm respectively. The length and width of A2 are 1.5 cm and 1 cm respectively. The distance between the boundary L1 of A1 and A2 is 0.7 cm. The distance between the boundary L2 of A1 and A2 is 1.3 cm. When the positional relationship between the phantom and the transducer is shown in Fig. 5a, the distance between the interface of the transducer and the boundary L1 of A1 is set to be 3.5 cm. The oscilloscope received the sound waveform shown in Fig. 5b. The horizontal axis and the vertical axis of the figure represent time (10
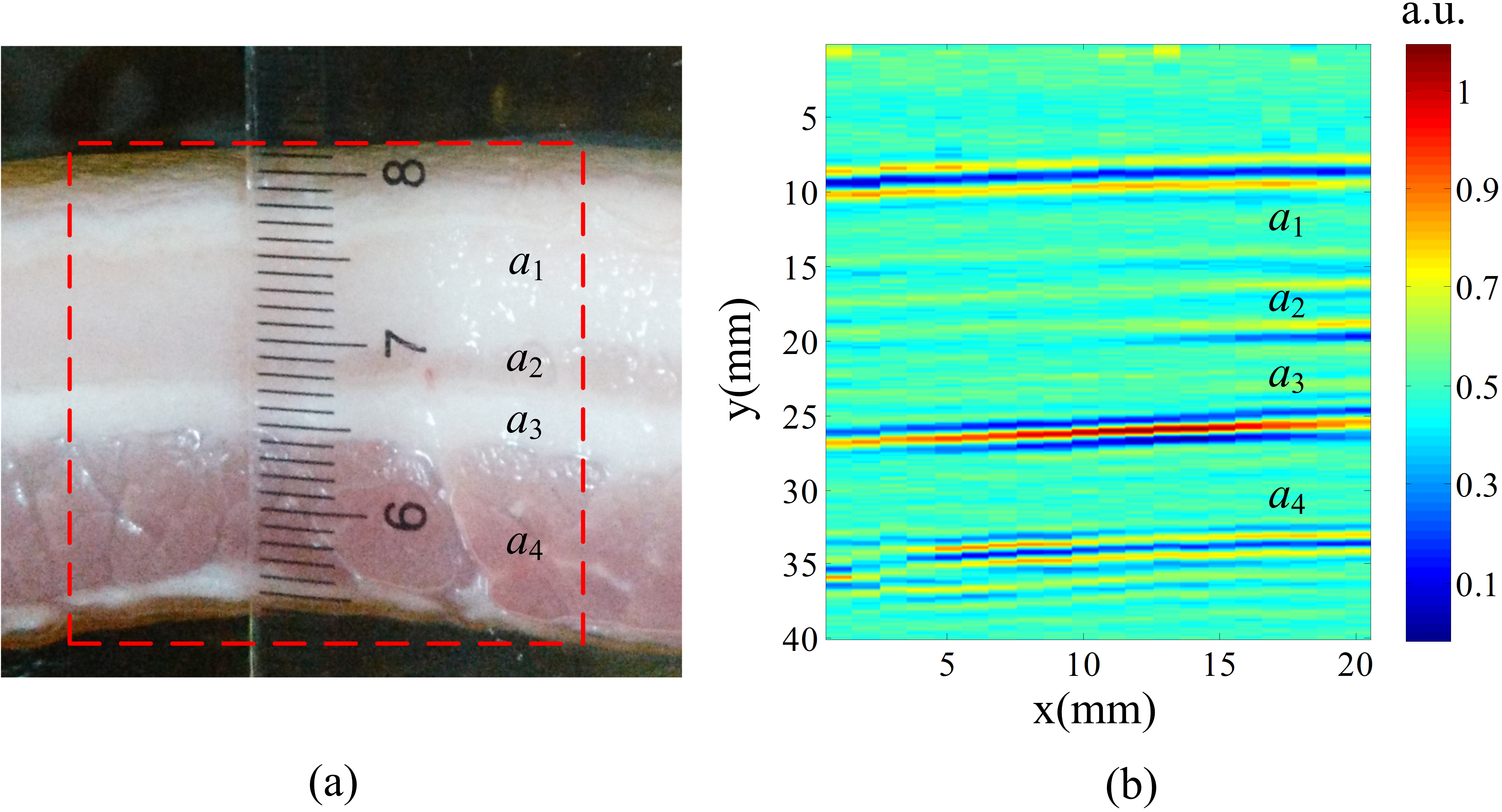
In the pork experiment, a piece of fresh pork was bought from a supermarket as the target. Figure 6 shows the positional relationship between the pork and the transducer. The length of the pork was about 2.5 cm in the y direction. The outer fat section a1 was about 1 cm length and the outer muscle section a4 was about 1 cm length in the y direction. The inner muscle section a2 and the inner fat section a3 were about 0.3 cm length in the y direction respectively. The conductivity of the fat tissue was very low around 0.02–0.03 S/m and the conductivity of the muscle was much higher about 0.55–0.62 S/m [28]. The distance between the interface of the transducer and the pork boundary close to the transducer was set to be 3.5 cm.
Figure 7.
Acoustic waveform from a piece of fresh pork.
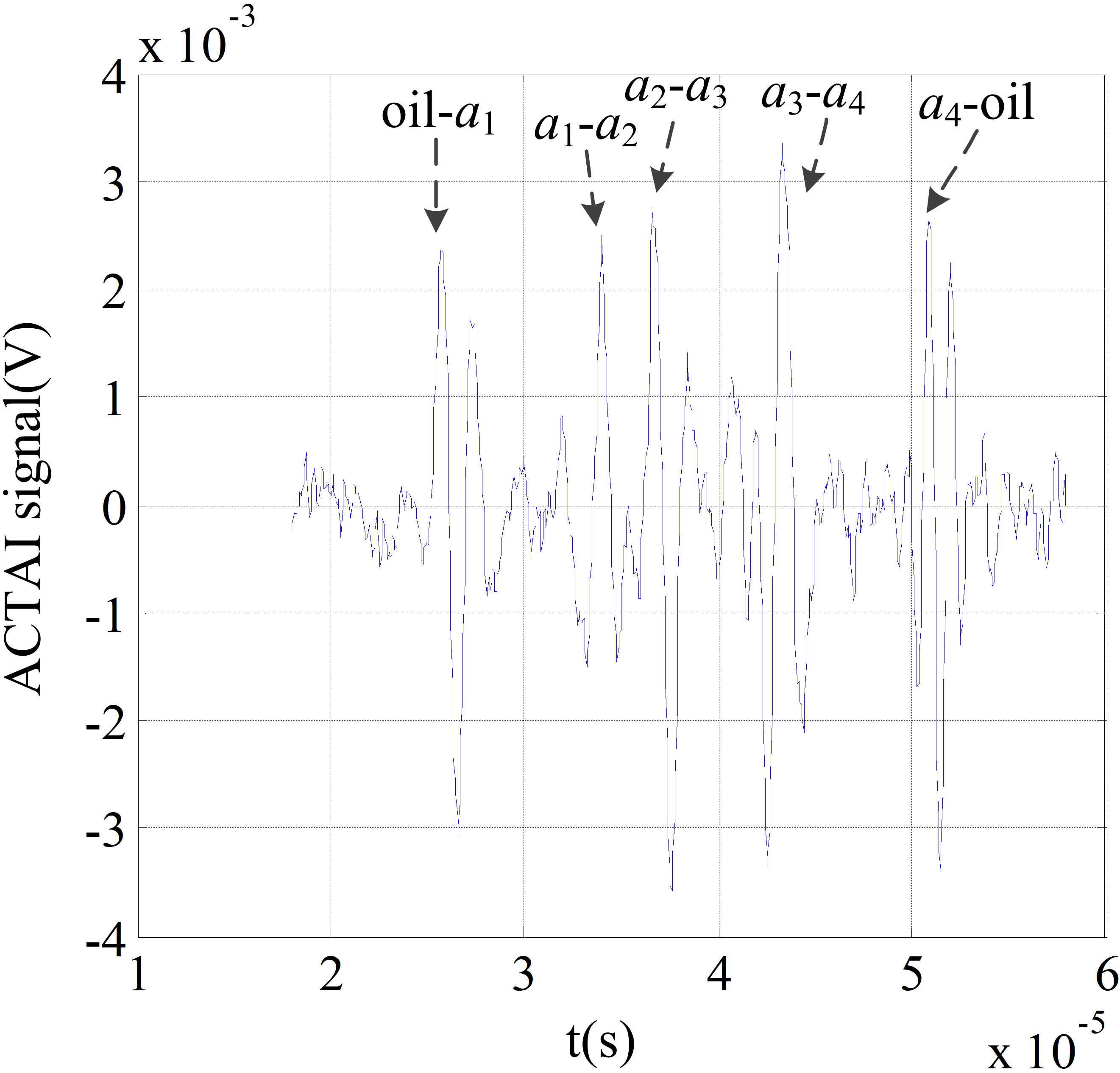
Figure 8.
Experimental results of fresh pork. (a) The imaging region. (b) B-scan ACTAI image with normalized scale of the tissue sample shown in (a).
The oscilloscope received the sound signal as shown in Fig. 7. The horizontal axis and the vertical axis of the figure represent time (10
The transducer is moved in the x direction from
5.Conclusion
The feasibility and performance of the newly proposed ACTAI method was verified by computer simulations and experiment researches in thermoacoustic imaging from noninvasive pressure measurements. The theoretical formulas of ACTAI have been derived under the assumption of acoustic homogeneity. Therefore, the detection system is needed to further improve in sensitivity, in order to facilitate the application of this method in biological imaging. However, the present simulation and experiment results show the feasibility and effectiveness of the proposed ACTAI in a practical setting. A numerical simulation on low conductivity phantom has been investigated and an experiment setup with fresh pork close to the real human tissues has been conducted. In the simulation study, the signals from a low conductivity model were conducted. In the experiment study, the acoustic signals and B-scan images of the fresh pork were presented. The fresh pork model experiment demonstrates the effectiveness of the proposed approach. These results of the simulation and experiment studies provide a new approach for conductivity reconstruction of thermoacoustic imaging, and present one novelty imaging modality for the diagnosis of early disease to deeper penetration. Further studies would be conducted to establish the performance of ACTAI in biological tissues.
Acknowledgments
This research was supported by the National Natural Science Foundation of China under Grant No. 51907013 and the Scientific and Technological Research Program of Chongqing Municipal Education Commission under Grant No. KJQN201801341.
Conflict of interest
None to report.
References
[1] | Xu M, Wang L. Photoacoustic imaging in biomedicine. Rev. Sci. Instrum. (2006) ; 77: : 041101. |
[2] | Xu M, Xu Y, Wang L. Time-domain reconstruction algorithms and numerical simulations for thermoacoustic tomography in various geometries. Trans. Biomed. Eng. (2003) ; 50: : 1086-1099. |
[3] | Guo L, Li L, Dong F, et al. Non-equilibrium plasma jet induced thermo-acoustic resistivity imaging for higher contrast and resolution. Sci Rep. (2017) ; 7. |
[4] | Yang Y, Liu G, Li Y, et al. Conductivity reconstruction for magnetically mediated thermoacoustic imaging. Journal of Medical Imaging & Health Informatics. (2018) ; 8: : 66-71. |
[5] | Jeong JJ, Choi H. An impedance measurement system for piezoelectric array element transducers. Measurement. (2017) ; 97: : 138-144. |
[6] | Choi H, Yeom JY and Ryu JM. Development of a multiwavelength visible-range-supported opto-ultrasound instrument using a light-emitting diode and ultrasound transducer. Sensors. (2018) ; 18: : 3324. |
[7] | Wang X, Pang Y, Ku G. Noninvasive laser-induced photoacoustic tomography for structural and functional imaging of the brain. Nat. Biotech. (2003) ; 21: : 803-806. |
[8] | Wang L. Tutorial on photoacoustic microscopy and computed tomography. IEEE J. Sel. Top. Quantum Electron. (2008) ; 14: : 171-179. |
[9] | Lin H, Wei Q, Jin X, et al. Thermoacoustic tomography: A novel method formerly breast tumor detection. X-Acoustics: Imaging and Sensing. (2015) ; 1. |
[10] | Mashal A, Booske J, Hagness S. Toward contrast-enhanced microwave-induced thermoacoustic imaging of breast cancer: An experimental study of the effects of microbubbles on simple thermoacoustic targets. Phys. Med. Biol. (2009) ; 54: : 641-650. |
[11] | Choi H, Woo P, Yeom JY, et al. Power MOSFET linearizer of a high-voltage power amplifier for high-frequency pulse-echo instrumentation. Sensors. (2017) ; 17: : 764. |
[12] | Feng X, Gao F, Zheng Y. Magnetically mediated thermoacoustic imaging toward deeper penetration. Appl. Phys. Lett. (2013) ; 103: : 083704-083707. |
[13] | Feng X, Gao F, Zheng Y. Modulatable magnetically mediated thermoacoustic imaging with magnetic nanoparticles. App. Phy. Lett. (2015) ; 106: : 093705. |
[14] | Gabriel C, Corthout E. The dielectric properties of biological tissues: I. Literature survey. Phys. Med. Biol. (1996) ; 41: : 2231-2279. |
[15] | Gabriel S. Compilation of the dielectric properties of body tissues at RF and microwave frequencies. Phys. Med. Biol. (1996) ; 41: : 2251-2269. |
[16] | Fear E, Hagness S, Meaney P. Enhancing breast tumor detection with near-field imaging. Microwave Magazine IEEE. (2002) ; 3: : 48-56. |
[17] | Campbell A, Land D. Dielectric properties of female human breast tissue measured in vitro at 32 GHz. Physics in Medicine & Biology. (1992) ; 37: : 193-210. |
[18] | Zhou Y, Yao J, Wang L. Tutorial on photoacoustic tomography. Journal of Biomedical Optics. (2016) ; 21: : 61007. |
[19] | Wang S, Ma R, Zhang S, et al. Translational-circular scanning for magneto-acoustic tomography with current injection. Biomedical Engineering Online. (2016) ; 15: : 10. |
[20] | Payne B, Venugopalan V, Miki B, et al. Optoacoustic tomography using time-resolved interferometric detection of surface displacement. Journal of Biomedical Optics. (2003) ; 8: : 273-280. |
[21] | Hao N, Arbabian A. Peak-power-limited frequency-domain microwave-induced thermoacoustic imaging for handheld diagnostic and screening tools. IEEE Transactions on Microwave Theory & Techniques. (2017) ; 65: : 2607-2616. |
[22] | Yang Y, Xia Z, Li Y, et al. Reconstruction of acoustic source in applied current thermoacoustic imaging. Scientia Sinica. (2018) . |
[23] | Liu G, Huang X, Xia H, et al. Magnetoacoustic tomography with current injection. Science Bulletin. (2013) ; 58: : 3600-3606. |
[24] | Liu G. Magnetic Acoustic Imaging Technology: Magnetic Acoustic Imaging Based on Ultrasonic Detection, 1st ed. Beijing, China: Science Press; (2014) . |
[25] | Wang S, Ma R, Zhang S, et al. Translational-circular scanning for magneto-acoustic tomography with current injection. Biomedical Engineering Online. (2016) ; 15: : 10. |
[26] | Xu M, Wang L. Time-domain reconstruction for thermoacoustic tomography in a spherical geometry. IEEE Transactions on Medical Imaging. (2002) ; 21: : 814. |
[27] | Liu S, Zhao Z, Zhu X, et al. Block based compressive sensing method of microwave induced thermoacoustic tomography for breast tumor detection. Journal of Applied Physics. (2017) ; 122: : 7-1810. |
[28] | Hu G, et al. Imaging biological tissues with electrical conductivity contrast below 1 Sm-1 by means of magnetoacoustic tomography with magnetic induction. Appl. Phys. Lett. (2010) ; 97: : 103705. |




