Liver function, quantified by the LiMAx test, as a predictor for the clinical outcome of critically ill patients treated with linezolid
Abstract
BACKGROUND:
Critically ill patients commonly suffer from infections that require antimicrobial therapy. In previous studies, liver dysfunction was shown to have an essential impact on the dose selection in these patients. This pilot study aims to assess the influence of liver dysfunction, measured by the novel LiMAx test, on clinical outcomes in critically ill patients treated with linezolid.
METHODS:
Twenty-nine critically ill patients were included and treated with linezolid. Indications for linezolid therapy were secondary or tertiary peritonitis (46.7%), bloodstream infection (6.7%) and 46.7% were other infections with gram-positive bacteria. Linezolid C
RESULTS:
Cured patients presented lower median linezolid C
CONCLUSIONS:
The LiMAx test predicts clinical failure more precisely than linezolid trough levels in critically ill surgical patients. Therefore liver failure may have a stronger impact on the outcome of critically ill surgical patients than low linezolid C
1.Introduction
Patients from the intensive care unit (ICU) frequently suffer from bacterial infections resulting in high mortality rates between 20% to 50% [1, 2, 3, 4, 5]. Thus, finding the optimal antimicrobial therapy with an appropriate dosage regime in ICU patients is of the utmost necessity to achieve a fast treatment response with high success rates.
Linezolid, an oxazolidinone antibiotic, remains an essential antimicrobial agent against most gram-positive bacteria, including resistant strains, which are particularly frequent in intensive care units [6, 7, 8, 9, 10]. While reasonable data regarding a successful therapy with linezolid were obtained in less severely ill patients, clinical observations in ICU patients have shown treatment failure by up to 30%. Further, 15% to 30% of critically ill patients receiving linezolid, have suffered from adverse effects (e.g., elevated liver enzymes, gastrointestinal disturbances, haematological toxicity) [11, 12, 13].
Linezolid is metabolised non-enzymatically and enzymatically by liver enzymes into two major inactive metabolites [14, 15]. The clearance of linezolid is approximately 65% non-renal, with up to 30% excreted unchanged in the urine [15]. Based on these data, a high variability of linezolid in patients with hepatic impairment is not expected. However, some studies showed a wide interindividual variability of linezolid plasma concentrations during treatment with the recommended flat dosing of 600 mg twice daily [11, 16]. Moreover, patients with liver dysfunction are reported to have a significantly higher risk of developing side effects, which could contribute to inadequate antimicrobial treatment [17].
The role of the liver as a modulator of drug-pharmacokinetics is well known. Some previous studies reported an alteration in serum trough concentrations of antibiotics in patients suffering from liver dysfunction [18, 19, 20]. Due to the lack of reliable liver function tests, no existing data can describe the impact of the degree of liver function on the pharmacokinetics of linezolid and the patient’s clinical outcome. In a recent pilot study, we demonstrated pharmacokinetic variability in linezolid serum concentrations depending on hepatic function using the maximal liver function capacity (LiMAx test) [21]. Patients with LiMAx values in the normal range showed significantly lower linezolid serum concentration. In this prospective observational study, we therefore aim to evaluate the clinical outcome of critically ill patients treated with linezolid depending on the hepatic function. Furthermore, potential factors predicting the outcome of these patients were investigated using a logistic regression model.
2.Methods
2.1Study design
The study was approved by the ethics review board of the Charité medical faculty in accordance with the provisions of the Declaration of Helsinki. Written informed consent was obtained from all participants or their responsible legal representatives prior to study inclusion (ethics board approval number EA4/022/13).
Patients from the surgical intensive care unit (SICU) of the Charité University Hospital were included in the study. Inclusion criteria were a medical indication (proven infection with suspected or proven involvement of gram-positive bacteria) for intravenous anti-infective therapy with linezolid and an age between 18 and 99 years. Exclusion criteria were an allergy against linezolid or methacetin, co-medication with substances metabolized by cytochrome P450 1A2 or with substances affecting the clearance of linezolid and missing informed consent.
Included patients (
2.2LiMAx
The LiMAx test was performed using the FLIP
2.3Determination of clinical outcome
Clinical outcome was assessed until seven days after the end of therapy. Clinical cure was defined as the resolution or significant improvement of clinical symptoms evaluated by an intensivist compared to the beginning of therapy such that no additional antibiotic therapy or intervention was necessary [30, 31]. Clinical failure was defined as death due to treated infection, persistence or recurrence of infection during linezolid therapy (inadequate response) and new or persistent symptoms at least 7 days after the end of therapy. The need for a change in antibiotic therapy due to adverse effects or clinical worsening and a positive culture for gram-positive bacteria at the site of primary infection reported at the end of therapy was also considered a clinical failure [32]. An uncertain outcome was recorded when there was insufficient information available to determine the outcome or the patient died from causes other than an infection.
2.4Statistical analysis
Patients were divided into two groups determined by their clinical outcome. Continuous variables are shown as the median and interquartile range. Categorical variables were investigated with Fisher’s exact test and are shown as frequencies. Depending on the distribution, tested by the Kolmogorov-Smirnov test in combination with the Shapiro-Wilk test, comparisons between clinical failure and clinical cure were performed using the Mann-Whitney U test or the independent ttest for non-connected samples. A
Variables related to the clinical outcome were analyzed by univariate analysis using Pearson’s chi-squared test. Multivariable logistic regression was performed on candidate predictor variables to identify independent risk factors for clinical cure. The receiver operating characteristic (ROC) curve was applied to calculate the best cut-off value of minimum LiMAx during linezolid therapy for the prediction of clinical outcome. Further ROC-curve analysis was performed on Bilirubin, Platelet count and INR in order to compare it with the LiMAx ROC-curve. Statistical analysis was performed with SPSS Statistics 22 (SPSS Inc., Chicago, IL, USA).
3.Results
3.1Demographic and characteristics of patient groups
A total of twenty-nine patients were observed in our clinical study. Twenty-eight patients were divided into two arms according to their evaluated clinical outcome. One remaining male patient (age 61 years) was documented as the indeterminate response due to death from reasons outside of infection. Fifteen patients were cured of the infection (11 males, 4 females, median age 57 years) and therefore included in the clinical cure group. Thirteen patients (7 males, 6 females, median age 71 years) showed a treatment failure and were included in the clinical failure group. In almost 47% of the patients, the indication of linezolid therapy was secondary or tertiary peritonitis, in nearly 7% a bloodstream infection and in the rest of the patient’ other source of infection was detected (46.7%). Gram-positive bacteria were observed in 89.3% of the patients, while in 28.6% of all cases a co-infection with multidrug-resistant gram-negative bacteria (MRGN) was observed (Table 1).
Table 1
Baseline characteristics before start of linezolid therapy
| Clinical cure | Clinical failure |
| |
|---|---|---|---|
| Patients ( | 15 | 13 | |
| Age (y) | 57 (50–62) | 71 (63.0–80.5) | 0.004 |
| Gender (m/f) | 11/4 | 7/6 | 0.433 |
| BMI (kg/m | 24.2 (20.3–27.8) | 26.8 (24.4–30.5) | 0.442 |
| Lactate (mmol/L) | 0.92 (0.67–1.29) | 2.11 (1.27–3.50) | 0.019 |
| INR | 1.37 (1.20–1.43) | 1.37 (1.24–1.99) | 0.060 |
| Total bilirubin (mmol/L) | 0.03 (0.01–0.06) | 0.17 (0.05–0.36) | 0.002 |
| Platelet counts (/nL) | 254.0 (202.0–369.0) | 103.0 (43.5–311.0) | 0.072 |
| AST (IU/L) | 36.0 (30.0–64.0) | 85.0 (56.5–215.5) | 0.006 |
| ALT (IU/L) | 25.0 (17.0–36.5) | 39.0 (28.5–139.0) | 0.043 |
| GGT (IU/L) | 152.0 (29.0–328.0) | 120.0 (54.5–267.0) | 0.964 |
| WBC (/nL) | 16.01 (9.34–26.10) | 17.67 (11.17–31.45) | 0.464 |
| CRP (mmol/L) | 7.72 (5.22–14.87) | 11.25 (3.34–12.30) | 0.418 |
| Serum creatinine (mmol/L) | 0.04 (0.03–0.07) | 0.08 (0.05–0.14) | 0.280 |
| MELD-score | 13 (9–16) | 24 (10–30) | 0.017 |
| SOFA score | 12 (8–12) | 11 (8–16) | 0.519 |
| SAPS II score | 70 (37–78) | 69 (58–85) | 0.350 |
| APACHE II score | 32 (19–33) | 28 (26–37) | 0.519 |
|
Indication for linezolid, | |||
| Secondary peritonitis | 6 (40) | 5 (38.5) | |
| Tertiary peritonitis | 1 (6.7) | 3 (23.1) | 0.625 |
| Blood stream infection | 1 (6.7) | 1 (7.7) | |
| Other | 7 (46.7) | 4 (30.7) | 0.549 |
|
Microbiological isolate, | |||
| Enterococcus faecalis | 2 (13) | 2 (15.4) | |
| Enterococcus faecium | 1 (6.7) | 1 (7.7) | |
| Gemella sp, | 1 (6.7) | 0 (0) | |
| Lactococcus lactis | 1 (6.7) | 0 (0) | |
| MRSA | 0 (0) | 1 (7.7) | |
| Staphylococcus epidermidis | 2 (13) | 0 (0) | 0.500 |
| Streptococcus mitis | 2 (13) | 0 (0) | 0.500 |
| Streptococcus salivarius | 2 (13) | 0 (0) | 0.500 |
| VRE | 2 (13) | 8 (61.5) | 0.109 |
| MDRGN | 5 (33) | 3 (23.1) | 0.727 |
|
Co-treatment, | |||
| Colistin | 0 (0) | 1 (7.7) | |
| Meropenem | 15 (100) | 13 (100) | |
| Tobramycin | 0 (0) | 1 (7.7) |
Data are presented as median and interquartile range (25th to 75th percentile) or frequencies. BMI: Body mass index; INR: International normalized ratio; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; GGT: Gamma-glutamyl transferase; WBC: White blood cell count; CRP: C-reactive protein; MELD: Model for end-stage liver disease; SOFA: Sequential organ failure assessment; SAPS: Simplified acute physiology score; APACHE II: Acute physiology and chronic health evaluation; MRSA: Methicillin-resistant staphylococcus aureus; VRE: Vancomycin-resistant enterococci; MDRGN: Multidrug-resistant gramnegative bacteria.
No significant differences were observed between the two groups at the beginning of therapy regarding disease severity (SOFA-, SAPSII-, APACHEII score), gamma-glutamyl transferase (GGT), white blood cells count (WBC), platelet counts and serum creatinine. Patients with clinical failure showed significantly higher values for age, lactate, total bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT) and MELD score. The baseline characteristics of both groups are shown in Table 1.
3.2Comparisons of liver function and linezolid serum concentration depending on the clinical outcome
Patients with clinical cure had a mean LiMAx-value of 349
Table 2
Comparisons between patients with clinical cure and clinical failure
| Clinical cure ( | Clinical failure ( |
| |
|---|---|---|---|
| LiMAx ( | 326 (204–444) | 131 (60–190) | 0.000 |
| Lactate (mmol/L) | 0.67 (0.50–0.94) | 1.67 (0.94–3.44) | 0.000 |
| INR | 1.25 (1.14–1.35) | 1.39 (1.22–1.94) | 0.043 |
| Total bilirubin (mmol/L) | 0.03 (0.02–0.04) | 0.23 (0.07–0.38) | 0.003 |
| Platelet count (/nL) | 219 (168–274) | 72 (33–175) | 0.001 |
| AST (IU/L) | 52 (27–88) | 102 (47–304) | 0.062 |
| ALT (IU/L) | 27 (19–67) | 45 (24–143) | 0.146 |
| GGT (IU/L) | 218 (48–511) | 283 (53–346) | 0.928 |
| PCHE (kU/L) | 2.5 (1.7–2.8) | 2.6 (1.6–3.9) | 0.247 |
| GLDH (IU/L) | 8.1 (3.8–26.8) | 24.4 (6.6–124.4) | 0.096 |
| MELD-score | 10 (9–17) | 28 (10–33) | 0.046 |
| Linezolid | 1.9 (0.5–3.9) | 5.1 (1.8–9.1) | 0.720 |
| Creatinine clearance (mL/min) | 86.40 (38.70–114.20) | 11.50 (1.85–42.00) | 0.006 |
| Duration of therapy (days) | 10 (7–14) | 10 (9–14) | 0.677 |
| SOFA score | 14 (12–19) | 14 (10–18) | 0.767 |
| SAPS score | 80 (68–95) | 79 (68–92) | 1.000 |
Data are presented as median and interquartile range (25th to 75th percentile). LiMAx: Maximal liver function capacity; INR: International normalized ratio; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; GGT: Gammaglutamyl transferase; PCHE: Pseudocholinesterase; GLDH: Glutamate dehydrogenase; MELD: Model for end-stage liver disease; SOFA: Sequential organ failure assessment; SAPS: Simplified acute physiology score.
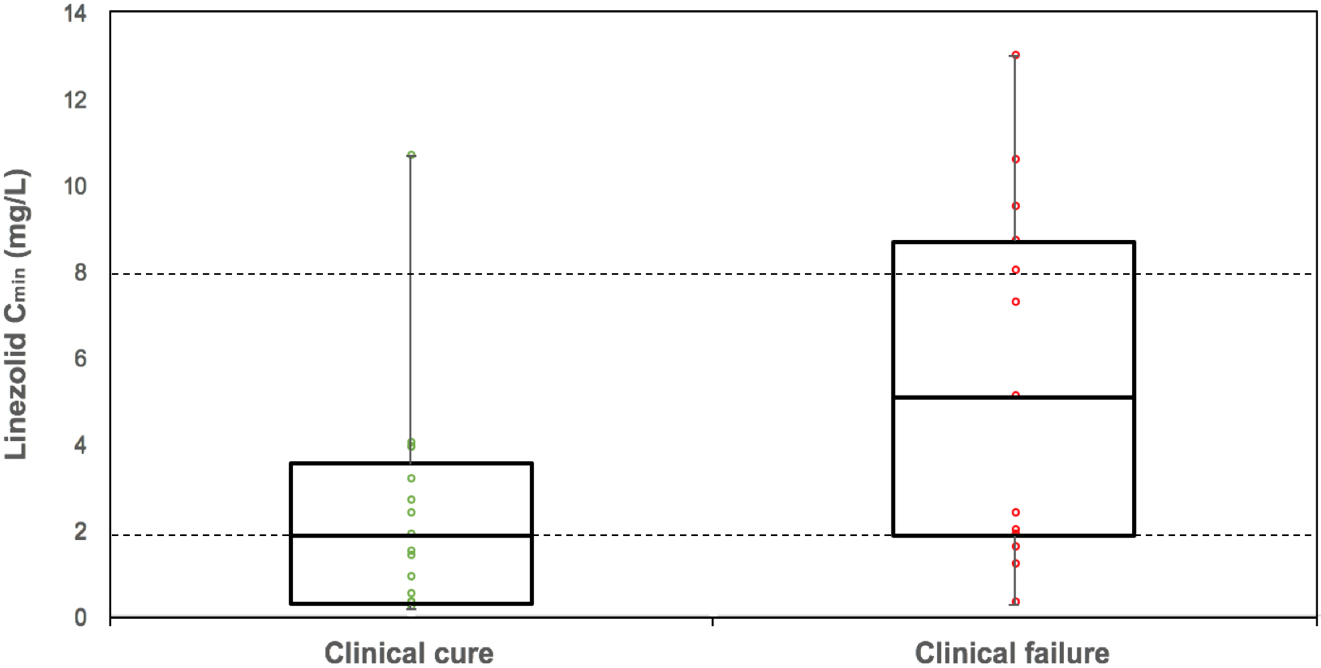
The median linezolid C
Figure 1.
Linezolid serum trough concentration depending on clinical outcome. Data are presented as box-and-whisker plots showing median with interquartile range. Linezolid overlaid with dot plot of the linezolid trough serum concentration of each patient belong to each group. Dotted lines define the lower and upper plasma target range.
3.3Independent predictors of clinical deterioration
The univariate analysis showed that LiMAx-value (
Table 3
Predictors associated with clinical outcome
| Univariate analysis | Multivariate analysis | ||||
| Sig. | OR | CI 95% | Sig. | ||
| Upper | Lower | ||||
| Linezolid C | 0.03 | ||||
| LiMAx value ( | 1.022 | 1.003 | 1.042 | 0.025 | |
| Platelet count (cells/nL) | |||||
| Lactate (mg/dL) | |||||
| Billirubin (mg/dL) | 0.03 | ||||
| INR | 0.03 | ||||
| Cl | |||||
LiMAx: Maximal liver function capacity; INR: International normalized ratio; Cl
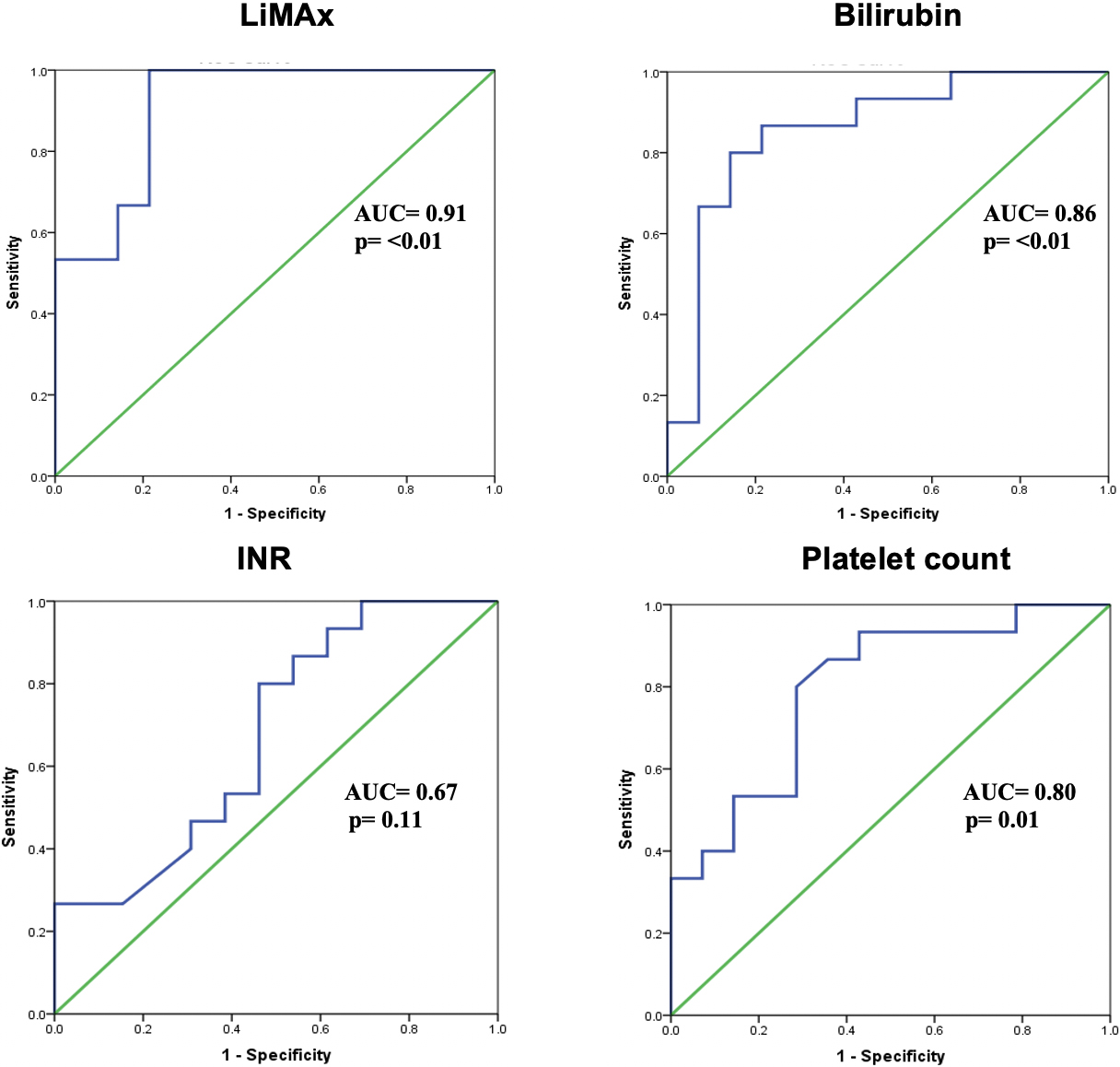
Figure 2.
ROC analysis for LiMAx, total bilirubin, INR and platelet count. Area under the receiver operating characteristic (AUROC) of LiMAx is 0.903
The final multivariable binary logistic regression model found only the LiMAx test as an independent predictor of clinical cure (Table 3). The Omnibus tests of model coefficients indicated that this model was statistically significant (Chi-square
The ROC and AUROC of LiMAx-value to predict early clinical deterioration were then constructed. The AUROC was 0.903 (96% CI 0.787–1.00) and the area under the curve was significantly different (
4.Discussion
The present study investigated the correlation between enzymatic liver function, based on the LiMAx test, linezolid C
Ide and colleagues showed that patients with preserved renal function revealed lower linezolid C
Several research groups found substantial interindividual variability in the pharmacokinetics of linezolid, particularly in critically ill patients. While most authors described a relationship between linezolid drug levels and renal function, none of the studies investigated the findings systematically due to liver failure [15, 38, 39]. However, non-renal clearance accounts for approximately 65% of the total clearance of linezolid and the influence of liver function on linezolid pharmacokinetic is highly suspected [40]. In our previous study, we could demonstrate that liver dysfunction, measured by the LiMAx test, explained 30% of the interindividual variability in linezolid clearance. These findings showed precisely the correlation between liver function and linezolid pharmacokinetic [21].
Linezolid therapy is associated with thrombocytopenia and the results of several studies indicated that linezolid-associated thrombocytopenia mainly occurred in patients with higher linezolid C
Zhang and colleagues described an elevated risk of linezolid-associated thrombocytopenia in patients with acute-on-chronic liver failure. They concluded that severe liver failure reduces the clearance of linezolid by about 50%. This leads to an increase of free linezolid in plasma and tissue with a higher risk for developing thrombocytopenia as a toxic side effect [17]. In the present study, patients of the clinical failure group revealed significantly lower platelet counts in comparison with patients of the clinical cure group (72/nL vs. 219/nL,
Several studies showed the LiMAx test as a reliable tool to quantify liver dysfunction in different clinical settings including critically ill patients [28, 52, 53, 54]. In summary, the findings revealed the LiMAx test being superior for predicting liver dysfunction compared to conventional liver function tests (e.g. dynamic indocyanine green test or static tests like bilirubin, INR, lactate etc.). Since the LiMAx test is a new diagnostic tool to measure the dynamic liver function, the costs are significantly higher than those of conventional tests (LiMAx test €C 320
There were several limitations to our study. First, the relatively small sample size (a total of 29 patients). Second, the uncontrolled nature of the study may include the risk of bias. Third, the small number of patients did not permit compelling statistical comparisons between groups. Therefore, future prospective studies that include pharmacokinetic analysis will be indispensable for determining the optimal individual linezolid strategy.
5.Conclusion
In patients with polymicrobial abdominal infections under linezolid therapy, the LiMAx test, as a marker of organ failure, can predict clinical failure more precisely than linezolid trough levels. Second, the influence of linezolid toxicity may be overestimated concerning thrombocytopenia. In critically ill patients, the impact of liver failure on the development of thrombocytopenia may be more relevant. Further prospective studies, investigating the correlation between linezolid, liver function and clinical outcome with a PK/PD design, are warranted to elucidate this issue.
Acknowledgments
The authors would like to thank the team of the Surgical Intensive Care Unit of the Chariteé for supporting the study. They would also like to thank the team of the “workgroup for the liver” for their cooperation in planning and designing the study.
Conflict of interest
MS is the inventor of the LiMAx test and has the capital interest in Humedics GmbH (Berlin, Germany), the company marketing the LiMAx test. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by research grants of the Charité University Hospital, Berlin, Germany. No funds were obtained for payment of the authors.
References
[1] | Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med [Internet]. (2001) Jul; 29: (7): 1303-10. Available from: https://insights.ovid.com/pubmed?pmid=11445675. |
[2] | Engel C, Brunkhorst FM, Bone H-G, Brunkhorst R, Gerlach H, Grond S, et al. Epidemiology of sepsis in Germany: results from a national prospective multicenter study. Intensive Care Med [Internet]. (2007) ; 33: (4): 606-18. Available from: doi: 10.1007/s00134-006-0517-7. |
[3] | Karlsson S, Varpula M, Ruokonen E, Pettilä V, Parviainen I, Ala-Kokko TI, et al. Incidence, treatment, and outcome of severe sepsis in ICU-treated adults in Finland: the Finnsepsis study. Intensive Care Med [Internet]. (2007) Feb 26 [cited 2018 Jan 8]; 33: (3): 435-43. Available from: doi: 10.1007/s00134-006-0504-z. |
[4] | Martin CM, Priestap F, Fisher H, Fowler RA, Heyland DK, Keenan SP, et al. A prospective, observational registry of patients with severe sepsis: The Canadian Sepsis Treatment and Response Registry*. Crit Care Med [Internet]. (2009) [cited 2018 Jan 8]; 37: (1): 81-8. Available from: https://insights.ovid.com/pubmed?pmid=19050636. |
[5] | Quenot J-P, Binquet C, Kara F, Martinet O, Ganster F, Navellou J-C, et al. The epidemiology of septic shock in French intensive care units: the prospective multicenter cohort EPISS study. Crit Care [Internet]. (2013) Apr 25 [cited 2018 Jan 8]; 17: (2): R65. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23561510. |
[6] | Álvarez-Lerma F, Palomar Martínez M, Olaechea Astigarraga P, Insausti Ordeñana J, López Pueyo MJ, Gracia Arnillas MP, et al. Analysis of treatments used in infections caused by gram-positive multiresistant cocci in critically ill patients admitted to the ICU. Rev Esp Quimioter [Internet]. (2012) Mar [cited 2018 Jan 8]; 25: (1): 65-73. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22488544. |
[7] | DeRyke CA, Lodise TP, Rybak MJ, McKinnon PS. Epidemiology, treatment, and outcomes of nosocomial bacteremic staphylococcus aureus pneumonia. Chest [Internet]. (2005) Sep 1 [cited 2018 Jan 8]; 128: (3): 1414-22. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0012369215521673. |
[8] | Falagas ME, Siempos II, Vardakas KZ. Linezolid versus glycopeptide or beta-lactam for treatment of Gram-positive bacterial infections: meta-analysis of randomised controlled trials. Lancet Infect Dis [Internet]. (2008) Jan 1 [cited 2018 Jan 8]; 8: (1): 53-66. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18156089. |
[9] | Jiang H, Tang R-N, Wang J. Linezolid versus vancomycin or teicoplanin for nosocomial pneumonia: meta-analysis of randomised controlled trials. Eur J Clin Microbiol Infect Dis [Internet]. (2013) Sep 10 [cited 2018 Jan 8]; 32: (9): 1121-8. Available from: doi: 10.1007/s10096-013-1867-z. |
[10] | McKenzie C. Antibiotic dosing in critical illness. J Antimicrob Chemother. (2011) ; 66: (SUPPL. 2): 25-31. |
[11] | Cattaneo D, Orlando G, Cozzi V, Cordier L, Baldelli S, Merli S, et al. Linezolid plasma concentrations and occurrence of drug-related haematological toxicity in patients with Gram-positive infections. Int J Antimicrob Agents [Internet]. (2013) Jun 1 [cited 2017 Oct 20]; 41: (6): 586-9. Available from: https://ac.els-cdn.com/S0924857913000812/1-s2.0-S0924857913000812-main.pdf?_tid=47a5b502-f47d-11e7-9d0b-00000aab0f6c&acdnat=1515420637_f17fdeaacdc0a07903362461a29bc4d9. |
[12] | Smith PF, Birmingham MC, Noskin GA, Meagher AK, Forrest A, Rayner CR, et al. Safety, efficacy and pharmacokinetics of linezolid for treatment of resistant Gram-positive infections in cancer patients with neutropenia. Ann Oncol. (2003) ; 14: (5): 795-801. |
[13] | Vinh DC, Rubinstein E. Linezolid: a review of safety and tolerability. J Infect [Internet]. (2009) Sep 1 [cited 2018 Jan 8]; 59: : S59-74. Available from: https://www.sciencedirect.com/science/article/pii/S0163445309600098?via%3Dihub. |
[14] | Dryden MS. Linezolid pharmacokinetics and pharmacodynamics in clinical treatment [Internet]. Vol. 66, Journal of Antimicrobial Chemotherapy. (2011) [cited 2018 Aug 1]. 7-15. Available from: https://academic.oup.com/jac/article-abstract/66/suppl_4/iv7/729737. |
[15] | Slatter JG, Stalker DJ, Feenstra KL, Welshman IR, Bruss JB, Sams JP, et al. Pharmacokinetics, metabolism, and excretion of linezolid following an oral dose of [(14)C]linezolid to healthy human subjects. Drug Metab Dispos. (2001) ; 29: (8): 1136-45. |
[16] | Pea F, Furlanut M, Cojutti P, Cristini F, Zamparini E, Franceschi L, et al. Therapeutic drug monitoring of linezolid: a retrospective monocentric analysis. Antimicrob Agents Chemother [Internet]. (2010) Nov [cited 2018 Jan 9]; 54: (11): 4605-10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20733043. |
[17] | Zhang YM, Yu W, Zhou N, Li JZ, Xu LC, Xie ZY, et al. High frequency of thrombocytopenia in patients with acute-on-chronic liver failure treated with linezolid. Hepatobiliary Pancreat Dis Int. (2015) ; 14: (3): 287-92. |
[18] | Dailly E, Verdier M-C, Deslandes G, Bouquié R, Tribut O, Bentué-Ferrer D. Level of evidence for therapeutic drug monitoring of ceftriaxone. Therapie [Internet]. (2012) Mar [cited 2018 Jan 9]; 67: (2): 145-9. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0040595716308265. |
[19] | Lheureux O, Trepo E, Hites M, Cotton F, Wolff F, Surin R, et al. Serum β-lactam concentrations in critically ill patients with cirrhosis: a matched case-control study. Liver Int [Internet]. (2016) Jul 1 [cited 2017 Dec 16]; 36: (7): 1002-10. Available from: doi: 10.1111/liv.13039. |
[20] | Merrell MD, Cherrington NJ. Drug metabolism alterations in nonalcoholic fatty liver disease. Drug Metab Rev [Internet]. (2011) Aug [cited 2018 Jan 9]; 43: (3): 317-34. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21612324. |
[21] | Wicha SG, Frey OR, Roehr AC, Pratschke J, Stockmann M, Alraish R, et al. Linezolid in liver failure: exploring the value of the maximal liver function capacity (LiMAx) test in a pharmacokinetic pilot study. Int J Antimicrob Agents [Internet]. (2017) Oct 1 [cited 2017 Dec 18]; 50: (4): 557-63. Available from: https://www.sciencedirect.com/science/article/pii/S0924857917302728?via%3Dihub. |
[22] | Tobin CM, Sunderland J, White LO, MacGowan AP. A simple, isocratic high-performance liquid chromatography assay for linezolid in human serum. J Antimicrob Chemother [Internet]. (2001) Nov 1 [cited 2018 Jan 10]; 48: (5): 605-8. Available from: https://academic.oup.com/jac/article-lookup/doi/10.1093/jac/48.5.605. |
[23] | WA K, EA D, DP W, JE Z. APACHE II: a severity of disease classification system. Crit Care Med [Internet]. (1985) [cited 2020 Dec 29]; 13: (10). Available from: https://pubmed.ncbi.nlm.nih.gov/3928249/. |
[24] | JL V, R M, J T, S W, A DM, H B, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the european society of intensive care medicine. Intensive Care Med [Internet]. (1996) [cited 2020 Dec 29]; 22: (7). Available from: https://pubmed.ncbi.nlm.nih.gov/8844239/. |
[25] | Le Gall J-R, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPS II) based on a european/north american multicenter study. JAMA J Am Med Assoc [Internet]. (1993) Dec 22 [cited 2020 Dec 29]; 270: (24): 2957. Available from: http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.1993.03510240069035. |
[26] | Abe S, Yoshihisa A, Takiguchi M, Shimizu T, Nakamura Y, Yamauchi H, et al. Liver dysfunction assessed by model for end-stage liver disease excluding INR (MELD-XI) scoring system predicts adverse prognosis in heart failure. PLoS One [Internet]. (2014) Jun 23; 9: (6): e100618-e100618. Available from: https://www.ncbi.nlm.nih.gov/pubmed/24955578. |
[27] | Fang JT, Tsai MH, Tian YC, Jenq CC, Lin CY, Chen YC, et al. Outcome predictors and new score of critically ill cirrhotic patients with acute renal failure. Nephrol Dial Transplant. (2008) . |
[28] | Kaffarnik MF, Lock JF, Vetter H, Ahmadi N, Lojewski C, Malinowski M, et al. Early diagnosis of sepsis-related hepatic dysfunction and its prognostic impact on survival: a prospective study with the LiMAx test. Crit Care [Internet]. (2013) Oct 31 [cited 2018 Jan 10]; 17: (5): R259. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24172237. |
[29] | Stockmann M, Lock JF, Malinowski M, Niehues SM, Seehofer D, Neuhaus P. The LiMAx test: a new liver function test for predicting postoperative outcome in liver surgery. HPB (Oxford). (2010) ; 12: (2): 139-46. |
[30] | Montravers P, Dupont H, Bedos J-P, Bret P, Tigecycline Group. Tigecycline use in critically ill patients: a multicentre prospective observational study in the intensive care setting. Intensive Care Med [Internet]. (2014) Jul 29 [cited 2018 Feb 24]; 40: (7): 988-97. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24871500. |
[31] | Moran GJ, Krishnadasan A, Mower WR, Abrahamian FM, LoVecchio F, Steele MT, et al. Effect of cephalexin plus trimethoprim-sulfamethoxazole vs cephalexin alone on clinical cure of uncomplicated cellulitis. JAMA [Internet]. (2017) May 23 [cited 2018 Feb 24]; 317: (20): 2088. Available from: http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.2017.5653. |
[32] | Solomkin J, Hershberger E, Miller B, Popejoy M, Friedland I, Steenbergen J, et al. Ceftolozane/tazobactam plus metronidazole for complicated intra-abdominal infections in an era of multidrug resistance: results from a randomized, double-blind, phase 3 trial (ASPECT-cIAI). Clin Infect Dis [Internet]. (2015) May 15 [cited 2018 Feb 24]; 60: (10): 1462-71. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25670823. |
[33] | Dong H, Xie J, Wang T, Chen L, Zeng X, Sun J, et al. Pharmacokinetic/pharmacodynamic evaluation of linezolid for the treatment of staphylococcal infections in critically ill patients. Int J Antimicrob Agents [Internet]. (2016) Sep 1 [cited 2018 Aug 1]; 48: (3): 259-64. Available from: https://www.sciencedirect.com/science/article/pii/S0924857916301479?via%3Dihub. |
[34] | Ide T, Takesue Y, Ikawa K, Morikawa N, Ueda T, Takahashi Y, et al. Population pharmacokinetics/pharmacodynamics of linezolid in sepsis patients with and without continuous renal replacement therapy. Int J Antimicrob Agents [Internet]. (2018) May 1 [cited 2018 Aug 1]; 51: (5): 745-51. Available from: https://www.sciencedirect.com/science/article/pii/S0924857918300323?via%3Dihub. |
[35] | Rayner CR, Forrest A, Meagher AK, Birmingham MC, Schentag JJ. Clinical pharmacodynamics of linezolid in seriously ill patients treated in a compassionate use programme. Clin Pharmacokinet [Internet]. (2003) [cited 2018 Aug 1]; 42: (15): 1411-23. Available from: https://link.springer.com/content/pdf/10.2165%2F00003088-200342150-00007.pdf. |
[36] | Dong HY, Xie J, Chen LH, Wang TT, Zhao YR, Dong YL. Therapeutic drug monitoring and receiver operating characteristic curve prediction may reduce the development of linezolid-associated thrombocytopenia in critically ill patients. Eur J Clin Microbiol Infect Dis [Internet]. (2014) [cited 2018 Aug 1]; 33: (6): 1029-35. Available from: https://link.springer.com/content/pdf/10.1007%2Fs10096-013-2041-3.pdf. |
[37] | Ballus J, Lopez-Delgado JC, Sabater-Riera J, Perez-Fernandez XL, Betbese AJ, Roncal JA. Surgical site infection in critically ill patients with secondary and tertiary peritonitis: epidemiology, microbiology and influence in outcomes. BMC Infect Dis [Internet]. (2015) [cited 2018 Aug 1]; 15: (1). Available from: https://bmcinfectdis.biomedcentral.com/track/pdf/10.1186/s12879-015-1050-5. |
[38] | Swoboda S, Ober MC, Lichtenstern C, Saleh S, Schwenger V, Sonntag HG, et al. Pharmacokinetics of linezolid in septic patients with and without extended dialysis. Eur J Clin Pharmacol. (2010) ; 66: (3): 291-8. |
[39] | Zoller M, Maier B, Hornuss C, Neugebauer C, Döbbeler G, Nagel D, et al. Variability of linezolid concentrations after standard dosing in critically ill patients: a prospective observational study. Crit Care [Internet]. (2014) Jul 10 [cited 2018 Jan 9]; 18: (4): R148. Available from: http://ccforum.biomedcentral.com/articles/10.1186/cc13984. |
[40] | MacGowan AP. Pharmacokinetic and pharmacodynamic profile of linezolid in healthy volunteers and patients with Gram-positive infections. J Antimicrob Chemother [Internet]. (2003) [cited 2018 Aug 1]; 51: (90002): 17ii-25. Available from: https://academic.oup.com/jac/article-abstract/51/suppl_2/ii17/2473475. |
[41] | Boak LM, Rayner CR, Grayson ML, Paterson DL, Spelman D, Khumra S, et al. Clinical population pharmacokinetics and toxicodynamics of linezolid. Antimicrob Agents Chemother [Internet]. (2014) [cited 2018 Aug 1]; 58: (4): 2334-43. Available from: http://aac.asm.org/. |
[42] | Lopez-Garcia B, Luque S, Roberts JA, Grau S. Pharmacokinetics and preliminary safety of high dose linezolid for the treatment of Gram-positive bacterial infections [Internet]. Vol. 71, Journal of Infection. W.B. Saunders; (2015) [cited 2018 Aug 1]. 604-7. Available from: https://www.sciencedirect.com/science/article/pii/S0163445315002029?via%3Dihub. |
[43] | Pea F, Viale P, Cojutti P, Del pin B, Zamparini E, Furlanut M. Therapeutic drug monitoring may improve safety outcomes of long-term treatment with linezolid in adult patients. J Antimicrob Chemother [Internet]. (2012) [cited 2018 Aug 1]; 67: (8): 2034-42. Available from: https://academic.oup.com/jac/article-abstract/67/8/2034/748220. |
[44] | Green SL, Maddox JC, Huttenbach ED. Linezolid and reversible myelosuppression [Internet]. Vol. 285, Journal of the American Medical Association. American Medical Association; (2001) [cited 2018 Aug 1]. 1291. Available from: http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.285.10.1291. |
[45] | Diamond DL, Jacobs JM, Paeper B, Proll SC, Gritsenko MA, Carithers RL, et al. Proteomic profiling of human liver biopsies: Hepatitis C virus-induced fibrosis and mitochondrial dysfunction. Hepatology [Internet]. (2007) Sep 1 [cited 2018 Aug 1]; 46: (3): 649-57. Available from: doi: 10.1002/hep.21751. |
[46] | Wynalda M, Hauer M, Wienkers L. Oxidation of the Novel Oxazolidinone Antibiotic Linezolid in Human Liver Microsomes [Internet]. (2000) [cited 2018 Aug 6]. Available from: http://dmd.aspetjournals.org/content/dmd/28/9/1014.full.pdf. |
[47] | Sasaki T, Takane H, Ogawa K, Isagawa S, Hirota T, Higuchi S, et al. Population pharmacokinetic and pharmacodynamic analysis of linezolid and a hematologic side effect, thrombocytopenia, in Japanese patients. Antimicrob Agents Chemother [Internet]. (2011) [cited 2018 Aug 6]; 55: (5): 1867-73. Available from: http://aac.asm.org/. |
[48] | Grau S, Morales-Molina JA, Mateu-de Antonio J, Marín-Casino M, Alvarez-Lerma F. Linezolid: Low pre-treatment platelet values could increase the risk of thrombocytopenia [8] [Internet]. Vol. 56, Journal of Antimicrobial Chemotherapy. 2005: [cited 2018 Aug 6]. 440-1. Available from: https://watermark.silverchair.com/dki202.pdf?token=AQECAHi208BE49Ooan9kkhW_Ercy7Dm3ZL_9Cf3qfKAc485ysgAAAZ0wggGZBgkqhkiG9w0BBwagggGKMIIBhgIBADCCAX8GCSqGSIb3DQEHATAeBglghkgBZQMEAS4wEQQMLn0QGhaV8NCPsrVlAgEQgIIBUCYzf5oMLd9aFTcMMG0Zg4bbdG0xAR5UIZDrP_YRFDpxZ2An. |
[49] | Ikuta SI, Tanimura K, Yasui C, Aihara T, Yoshie H, Iida H, et al. Chronic liver disease increases the risk of linezolid-related thrombocytopenia in methicillin-resistant Staphylococcus aureus-infected patients after digestive surgery. J Infect Chemother [Internet]. (2011) Jun 1 [cited 2018 Aug 6]; 17: (3): 388-91. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21161560. |
[50] | Munoz SJ, Stravitz RT, Gabriel DA. Coagulopathy of Acute Liver Failure [Internet]. Vol. 13, Clinics in Liver Disease. Elsevier; (2009) [cited 2018 Aug 6]. 95-107. Available from: https://www.sciencedirect.com/science/article/pii/S1089326108001128?via%3Dihub. |
[51] | Pluta A, Gutkowski K, Hartleb M. Coagulopathy in liver diseases [Internet]. Vol. 55, Advances in Medical Sciences. Elsevier; (2010) [cited 2018 Aug 6]. 16-21. Available from: https://www.sciencedirect.com/science/article/pii/S189611261460002X?via%3Dihub. |
[52] | Jara M, Bednarsch J, Malinowski M, Pratschke J, Stockmann M. Effects of oxaliplatin-based chemotherapy on liver function – an analysis of impact and functional recovery using the LiMAx test. Langenbeck’s Arch Surg [Internet]. (2016) Feb 27 [cited 2018 Aug 6]; 401: (1): 33-41. Available from: http://link.springer.com/10.1007/s00423-015-1352-5. |
[53] | Kaffarnik M, Stoeger G, Liebich J, Grieser C, Pratschke J, Stockmann M. Liver function, quantified by LiMAx test, after major abdominal surgery. comparison between open and laparoscopic approach. World J Surg [Internet]. (2018) Feb 24 [cited 2018 Jan 10]; 42: (2): 557-66. Available from: http://link.springer.com/10.1007/s00268-017-4170-9. |
[54] | Stockmann M, Lock JF. Prediction of Postoperative Outcome After Hepatectomy With a New Bedside Test for Maximal Liver Function Capacity. (2009) ; 250: (1): 119-25. |
[55] | Sakka SG. Assessing liver function [Internet]. Vol. 13, Current Opinion in Critical Care. (2007) [cited 2018 Aug 6]. 207-14. Available from: https://insights.ovid.com/crossref?an=00075198-200704000-00016. |




