Eighteenth International Kidney Cancer Symposium, November 15-16, 2019, Trump National Doral Miami Hotel, Miami, Florida
CONTENTS
01 A phase 2 open label study of cabozantinib in patients with advanced or unresectable renal cell carcinoma pretreated with one immune-checkpoint inhibitor: the BREAKPOINT trial S1
02 A Phase 2 Study of Neoadjuvant Cabozantinib in Patients with Locally Advanced Non-metastatic Clear Cell Renal Cell Carcinoma S1
03 A phase 3 study (COSMIC-313) of cabozantinib in combination with nivolumab and ipilimumab in patients with previously untreated advanced or metastatic renal cell carcinoma (RCC) of intermediate or poor risk S2
04 Adrenal Metastases as Sanctuary Sites in Advanced RCC S3
05 Age-related trends in initial systemic therapy for metastatic clear cell renal cell carcinoma (mRCC) S3
06 Agnostic Transcriptional Profiling of The Cancer Genome Atlas Data Identifies Distinct and Cooperative Role of TP63 Isoforms in Renal Cancer Subtypes That Drive Progression and Predict Clinical Outcomes S4
07 An Exploratory Study of 89Zr-DFO-Atezolizumab ImmunoPET/CT in Patients with Locally Advanced or Metastatic Renal Cell Carcinoma S5
08 Association Between Depth of Response and Overall Survival: Exploratory Analysis in Patients With Previously Untreated Advanced Renal Cell Carcinoma (aRCC) in CheckMate 214 S5
09 Association of academic rank and productivity with metrics of Twitter utilization amongst kidney cancer experts S7
10 Association of clinical benefit (CB) from first-line (1L) treatment and CB in further lines or overall survival (OS) in metastatic renal cell carcinoma (mRCC) S7
11 Associations between inflammation, depression, and cancer stage in patients with resected renal cell carcinoma S8
12 Bilateral Multifocal Clear cell papillary renal cell carcinoma with discordant histopathology S9
13 Body composition and outcomes to Immune-Oncology agents in 165 patients with metastatic clear cell renal cell carcinoma S9
14 BONSAI trial: a prospective trial evaluating a first line treatment with cabozantinib in metastatic collecting duct carcinoma. S10
15 Brain Metastasis from Renal Cell Carcinoma: an Institutional Study S11
16 Choosing First-Line Treatment for Metastatic Renal Cell Carcinoma (mRCC) in the Immuno-oncology (IO) Era: Systematic Review and Network Meta-Analysis S11
17 Chromosome 8 Deficient Clear Cell RCC: An Analysis of Clinicopathologic Characteristics S12
18 Circulating Tumor DNA (ctDNA) results in 110 Patients with Advanced Renal Cell Carcinoma S13
19 Clinical correlates of oncogenic events in translocation renal cell carcinoma S14
20 Combination cabozantinib and nivolumab treatment in patients with refractory metastatic renal cell carcinoma (mRCC) S16
21 Combining immunotherapy and VEGFR inhibition improves the outcomes of elderly and favorable risk patients with metastatic renal cell carcinoma S16
22 Connections between BAP1 and the Type I Interferon Pathway in ccRCC S17
23 Cytoreductive Nephrectomy for Symptomatic Primary Tumors: Important Therapeutic Endpoint in the Management of mRCC S17
24 Development and validation of a risk score based on patient characteristics to predict major complications after partial nephrectomy S18
25 DNA hypomethylating agents activate ERV expression in kidney cancer to potentially enhance response to immunotherapies S19
26 Effects of immune checkpoint inhibitors on the morphology of renal cell carcinoma S20
27 Efficacy of Nivolumab (NIVO) plus Ipilimumab (IPI) in metastatic Renal Cell Carcinoma (RCC) and brain metastases. S21
28 Efficacy of Systemic Treatment After Cabozantinib Failure in Metastatic Renal Cell Carcinoma S22
29 Exploratory Analysis of Adherence to Anticancer Therapies in Metastatic Renal Cell Carcinoma in the United States (US) S23
30 Fine Needle Aspiration Based Immune Organoids Recapitulate TME in ccRCC Cancer S23
31 Genomic sequencing of high-grade unclassified renal cell carcinoma allows molecular stratification and improves prognostication S24
32 Identification of Oncocytic Neoplasms with Cyclin D1 Immunohistochemistry S25
33 Immunologic correlates of previously-treated metastatic renal cell carcinoma (mRCC) patients (pts) treated on a phase II trial of intermittent nivolumab therapy S26
34 Improved response rate of Cabozantinib after immune checkpoint therapy in patients with metastatic Renal Cell Carcinoma S26
35 Incidence of Occult Brain Metastases Detected at Study Screening in Patients with Advanced Renal Cell Carcinoma S27
36 Interim results of a phase 2 study of sapanisertib in patients with previously treated advanced renal cell carcinoma S28
37 Is anesthesia associated with oncological outcomes after nephrectomy S29
38 Local and Regional Recurrences of Clinically Localized Renal Cell Carcinoma: A 15 Year Institutional Experience with Prognostic Features and Oncologic Outcomes S30
39 Long-Term Follow-Up of Nivolumab Versus Everolimus in Patients With Advanced Renal Cell Carcinoma (aRCC): The Phase 3 CheckMate 025 Trial S31
40 Loss of SETD2 drives mitochondrial dysfunction in clear cell renal cell carcinoma S32
41 Loss of tumor suppressor SETD2 alters identity of centromeric chromatin and promotes generation of neocentromeres S32
42 Mitotic defects and delays are characteristic features of SETD2 loss S33
43 Molecular chaperones Hsp70 and Hsp90 stabilize and maintain HIF2a activity in ccRCC S34
44 Multicenter Randomized Phase III Trial of Deferred Cytoreductive Nephrectomy in Synchronous Metastatic Renal Cell Carcinoma Receiving Checkpoint Inhibitors: a DaRenCa and NoRenCa Trial Evaluating
the Impact of Surgery or No Surgery. The NORDIC-SUN-Trial S34
45 Need for Sex Disaggregated Data in Outcomes Reporting for the Treatment of Renal Cell Carcinoma with Immunotherapy S35
46 Neutrophil-to-lymphocyte ratio in unclassified renal cell carcinoma is associated with outcome and differs between molecular subgroups S36
47 Nuances in surgical selection for cytoreductive nephrectomy among IMDC intermediate-poor risk patients S37
48 Patient-reported experience of diagnosis, management, and burden of renal cell carcinomas: North American Results from a global patient survey in 43 countries S38
49 PDIGREE: An adaptive phase 3 trial of PD-inhibitor nivolumab and ipilimumab (IPI-NIVO) with VEGF TKI cabozantinib (CABO) in metastatic untreated renal cell cancer (Alliance A031704) S39
50 Pembrolizumab Plus Axitinib vs Sunitinib as First-Line Therapy for Metastatic Renal Cell Carcinoma: Outcomes by IMDC Risk and Sarcomatoid Subgroups of the Phase 3 KEYNOTE-426 Study S40
51 Perspective Matters: Patient-reported ECOG performance status (PS) is lower than oncologist-reported PS and is associated with worse psychosocial outcomes S41
52 PET-CT as predictor of response in the clear-cell metastatic renal cell carcinoma (mRCC) treatment S41
53 Phase 2 study of lenvatinib plus pembrolizumab for disease progression after PD-1/PD-L1 immune checkpoint inhibitor in metastatic clear cell renal cell carcinoma (mccRCC): results of an interim analysis S42
54 Phase II Trial of Intermittent Therapy in Patients (pts) with Metastatic Renal Cell Carcinoma (mRCC) Treated with Front-line Ipilimumab and Nivolumab (Ipi/Nivo) S43
55 Physician Rationale for Prescribing Tyrosine Kinase Inhibitors and Immuno-Oncology Treatments in the First-Line Treatment of Advanced Renal Cell Carcinoma in the United States: Interim Results from a Cross-Sectional Real-World Survey S44
56 Pilot study to evaluate the biologic effect of the probiotic CBM588 in combination with nivolumab/ipilimumab for patients with mRCC S45
57 Prognostic significance and immune correlates of CD73 expression in renal cell carcinoma (RCC) S45
58 Prognostic Values of Pretreatment Lymphocite-to-Monocite Ratio in Patients with in Non-Metastatic Renal Cell Carcinoma: A Systematic Review and Meta-Analysis S46
59 PROSPER: A phase III randomized study comparing perioperative nivolumab vs. observation in patients with renal cell carcinoma (RCC) undergoing nephrectomy (ECOG-ACRIN 8143) S47
60 Protocol for a feasibility study of a cohort embedded randomised controlled trial comparing NEphron Sparing Treatment (NEST) for small renal masses S48
61 Quality of Life in Previously Untreated Patients With Advanced Renal Cell Carcinoma (aRCC) in Checkmate 214: Updated Results S49
62 Radiomic signatures of CD8+ T cell infiltration and PD-L1 expression in clear-cell renal cell carcinoma (ccRCC) S50
63 Real-World Clinical Outcomes Of Patients With Metastatic Renal Cell Carcinoma (mRCC) Receiving First-Line (1L) Therapy In The United States (US) Veteran Population S51
64 Real-world results from one year of therapy with tivozanib S51
65 Real-World Treatment Patterns Among Patients With Metastatic Renal Cell Carcinoma Receiving First-Line Therapy in the United States Veteran Population S52
66 Renal cell carcinoma induced by end stage renal disease: A population-based study in Sweden S52
67 Resectable non-clear cell renal cell carcinoma: 25 years of single-center experience S53
68 Safety and efficacy of nivolumab plus ipilimumab (NIVO+IPI) in patients with advanced renal cell carcinoma (aRCC) with brain metastases: interim analysis of CheckMate 920 S54
69 Safety and Feasibility of Surgery Following Preoperative Immunotherapy in Patients with Metastatic Renal Cell Carcinoma S54
70 Treatment-Free Survival, With and Without Toxicity, as a Novel Outcome Applied to Immuno-Oncology Agents in Advanced Renal Cell Carcinoma S55
Abstracts
01A phase 2 open label study of cabozantinib in patients with advanced or unresectable renal cell carcinoma pretreated with one immune-checkpoint inhibitor: the BREAKPOINT trial
First Author: Mennitto, Alessia
Co-Authors: Procopio, Giuseppe; Sepe, Pierangela; Claps, Melanie; De Braud, Filippo; Verzoni, Elena
Author Company: Fondazione IRCCS Istituto Nazionale Tumori
Background: Antiangiogenic therapy has been a milestone in the treatment of metastatic renal cell carcinoma (mRCC) for many years. Recently, the positive results with immunotherapy are changing the frontline standard of care of these patients but prospective data are lacking to determine the efficacy of vascular endothelial growth factor (VEGF)-targeted therapy in patients progressed to immune checkpoint inhibitors (ICI). Among VEGF pathway blockers, the multi-kinase inhibitor cabozantinib has shown prolonged survival in pre-treated mRCC patients.
Methods: This is an open label, single arm, multicenter, phase II study of cabozantinib (60 mg orally once daily) for patients with mRCC pre-treated with a PD1/PDL1 inhibitor. Forty-nine patients will be enrolled at 5 italian centers and stratified according to Heng prognostic group, duration of first-line and type of previous therapy received (ICI monotherapy or combined with a tyrosin-kinase inhibitor (TKI) or another ICI).Main inclusion criteria: histological diagnosis of predominant clear cells RCC and one previous treatment with a PD1/PDL1 inhibitor in both first line or adjuvant setting (recurrence during the adjuvant treatment or within 6 months post-treatment). Primary endpoint: to assess the progression free survival (PFS) of cabozantinib. Secondary endpoints: evaluation of overall survival (OS), objective response rate (ORR) and safety profile of the drug. Exploratory objectives: to investigate PD-L1 expression by immunohistochemistry in tumor samples; to analyze the immunological signature/profile of tumor cells and the state of circulating immune cells, as well as the modulating activity of cabozantinib on local and systemic tumor immunity; to explore bone formation and reabsorption markers in patients with or without bone metastases. Statistical analysis: a Brookmeyer-Crowley like test will be performed to detect an increment of the median PFS time from 3.8 months (H0: median PFS < 3.8 months) to 7.4 months (H1: median PFS > 7.4 months) with a power of 90% and one-sided alpha of 5%. The large sample critical value detecting the increment of the PFS median survival time will be 5.54 months. A non-parametric approach will be used to estimate survival functions for time-to-event endpoints (i.e. PFs and OS). Regression models will be used to detect and estimate statistical association between baseline biomarkers and outcomes.The study is open with 5 patients enrolled at time of submission. Clinical trial information: NCT03463681
02A Phase 2 Study of Neoadjuvant Cabozantinib in Patients with Locally Advanced Non-metastatic Clear Cell Renal Cell Carcinoma
First Author: Master, Viraj
Co-Authors: Bilen, Mehmet; Jiang, James; Brown, Jacqueline; Harik, Lara; Sekhar, Aarti; Kissick, Haydn; Jansen, Caroline; Maithel, Shishir; Kucuk, Omer; Carthon, Bradley
Author Company: Emory University School of Medicine
Background: Cabozantinib (XL-184) is a small molecule inhibitor of the tyrosine kinases c-Met, AXL and VEGFR2 that has been shown to reduce tumor growth, metastasis, and angiogenesis. 1 After the promising results from the METEOR and CABOSUN trials, cabozantinib was approved for use in the first- and second-line setting in patients with advanced RCC. 2,3 Previously, targeted therapies with sunitinib and sorafenib have been used in the neoadjuvant setting for tumor size reduction and facilitating nephrectomies. 4 In a prospective phase 2 clinical trial, Karam et al. reported that axitinib, when given prior to surgery, resulted in significant shrinking of kidney cancers, facilitating surgical resections. 5 The increased response rates with cabozantinib in mRCC, along with the other neoadjuvant TKI data, strongly support an expanded role for cabozantinib in the neoadjuvant setting.Trial Design: This is a single center, phase II study of neoadjuvant cabozantinib in patients with locally advanced non-metastatic clear cell renal cell carcinoma. Eligible patients are required to have a renal mass consistent with a clinical stage = T3Nx or TanyN+, or deemed unresectable by surgeon and with clear cell component on pre-treatment biopsy of the primary tumor. Eligible patients are also required to have an ECOG performance status of 0-1. Patients will receive treatment with cabozantinib 60mg PO daily for 12 weeks, and undergo surgery after 3 weeks of washout. The primary endpoint is overall response rate (ORR) by RECIST 1.1. Secondary endpoints include safety, tolerability, clinical outcome (DFS, OS), and surgery related outcomes. Pre and post treatment tissue and blood will be collected for biomarker analysis. 17 patients will be enrolled in this study.ClinicalTrials.gov Identification: NCT04022343
References
[1] Yakes FM, Chen J, Tan J, et al. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther. 2011;10(12):2298-2308.
[2] Choueiri TK, Escudier B, Powles T, et al. Cabozantinib versus everolimus in advanced renal cell carcinoma (METEOR): final results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2016;17(7):917-927.
[3] Choueiri TK, Halabi S, Sanford BL, et al. Cabozantinib Versus Sunitinib As Initial Targeted Therapy for Patients With Metastatic Renal Cell Carcinoma of Poor or Intermediate Risk: The Alliance A031203 CABOSUN Trial. J Clin Oncol. 2017;35(6):591-597.
[4] Bindayi A, Hamilton ZA, McDonald ML, et al. Neoadjuvant therapy for localized and locally advanced renal cell carcinoma. Urol Oncol. 2018;36(1):31-37.
[5] Karam JA, Devine CE, Urbauer DL, et al. Phase 2 trial of neoadjuvant axitinib in patients with locally advanced nonmetastatic clear cell renal cell carcinoma. Eur Urol. 2014;66(5):874-880.
03A phase 3 study (COSMIC-313) of cabozantinib in combination with nivolumab and ipilimumab in patients with previously untreated advanced or metastatic renal cell carcinoma (RCC) of intermediate or poor risk
First Author: Choueiri, Toni K.
Co-Authors: Scheffold, Christian; Wang, Fong; Powles, Thomas; Motzer, Robert J.
Author Company: Dana-Farber Cancer Institute/Brigham and Women’s Hospital and Harvard University School of Medicine, Exelixis, Inc., Barts Cancer Institute, Queen Mary University of London, Memorial Sloan Kettering Cancer Center
Background: Cabozantinib (C) inhibits tyrosine kinases involved in tumor growth, angiogenesis, and immune regulation, including MET, VEGFR, and TAM kinases (TYRO3, AXL, MER), and may promote an immune-permissive tumor environment, thus enhancing response to immune checkpoint inhibitors. C has shown preliminary clinical activity and tolerability as a doublet combination with the PD-1 inhibitor nivolumab (N) and as part of a triplet combination with N and the CTLA-4 inhibitor ipilimumab (I) in patients with advanced RCC.1,2 C is approved for patients with advanced RCC, and N+I is approved as a combination therapy in patients with previously untreated advanced RCC of intermediate or poor risk. Here we present the study design of a phase 3 trial of the triplet combination C+N+I vs N+I in previously untreated patients with metastatic or advanced RCC of International Metastatic RCC Database Consortium (IMDC) intermediate or poor risk (NCT03937219).
Methods: This randomized, controlled, double-blind, phase 3 study evaluates the efficacy and safety of C+N+I vs N+I in previously untreated patients with metastatic or advanced IMDC intermediate- or poor-risk RCC. Patients will be randomized 1:1 to receive C+N+I or N+I in combination with placebo. Patients will receive C (40 mg orally QD) + N (3 mg/kg IV Q3W) x 4 doses + I (1 mg/kg IV Q3W) x 4 doses, followed by C (40 mg orally QD) + N infusion (480 mg IV flat dose Q4W). Patients in the control arm will receive C-matched placebo and the same treatment regimen for N+I as in the experimental arm. In both trial arms, N will be administered for a maximum of 2 years. Key eligibility criteria include histologically confirmed advanced or metastatic RCC with a clear cell component, intermediate- or poor-risk RCC per IMDC criteria, measurable disease per RECIST 1.1, Karnofsky Performance Status =70%, adequate organ and marrow function, and age =18 years. Exclusion criteria include prior systemic therapy for unresectable locally advanced or metastatic RCC and uncontrolled significant illnesses. Randomization is stratified by IMDC prognostic score and geographic region. The primary efficacy endpoint is duration of progression-free survival (PFS) (using RECIST 1.1) per blinded independent central review; the secondary efficacy endpoint is duration of overall survival (OS). Additional endpoints include objective response rate (ORR), safety, correlation of biomarker analyses with outcomes, and pharmacokinetics of C in combination with N+I. The first patient was enrolled in June 2019, and enrollment is ongoing.
04Adrenal Metastases as Sanctuary Sites in Advanced RCC
First Author: Asad, Mohammad Fahad B
Co-Authors: Shah, Harsh; Dickow, Brenda; Suisham, Stacey; Varney, Cheryl; Fontana, Joseph; Vaishampayan, Ulka
Author Company: Karmanos Cancer Inst/Wayne State Univ, Detroit MI
Introduction: Immune therapy has induced durable remissions in advanced renal cancer. Various patterns of response are noted, with complete response or residual sites of metastases, or relapse and progression in multiple areas. We reviewed a case series of 7 patients with advanced renal cancer who presented with adrenal masses as the predominant areas of relapse/metastases.
Methods: Regulatory approval was obtained and informed consent was waived. Medical records were reviewed to collect patient information regarding demographics, sites of metastases, IMDC prognostic risk characteristics, therapy, response rates and progression free and overall survival.
Results: 6 out of 7 patients had relapse in adrenal glands post immune therapy and 1 patient presented with adrenal metastasis post nephrectomy. Median age is 53 years, range 41 to 59 years and all the patients in the study were males. All patients had IMDC intermediate risk characteristics. The other sites of metastases were: 3 pts with lung, 3 bone, 2 lymph node metastases. The immunotherapy used was high dose interleukin-2 (IL-2) in 3 patients, immune checkpoint inhibition (ICI) in 4 and anti-vascular axitinib with pembrolizumab combination in 4 patients. The adrenal metastases were treated with surgery and cryotherapy in one patient each. and antivascular therapy; pazopanib (1) Cabozantinib (2) and bevacizumab (1) in four patients. Genomic profiling was available on 5 pts. PDL-1 positivity and BAP-1 mutations were not noted in the cases while PBRM-1 in 2 pts, SETD2 in 2 and VHL in 3 patients .TMB assessment showed 6 Mut/MB in 2 pts and 7 Mut/MB in 1 pt. Al patients demonstrate an ongoing response to systemic therapy. Two patients require continuous steroid replacement after local therapy for adrenal metastases. The mean time from RCC diagnosis to adrenal metastasis is 83.1 months and the mean time from initial metastatic disease to one or contralateral adrenal metastasis is 34.8 months. All the patients are alive and continue in follow up.
Conclusions: Adrenal metastases were noted to be a distinct pattern of relapse or sanctuary site in advanced RCC treated with immune therapy. The relapse/ residual disease in adrenals was typically noted despite remission in other sites with immune therapy. Local therapy management was effective and systemic management with anti VEGF therapies also demonstrated response. Association with PBRM-1 mutations is suggested. Tumor environment milieu of glucocorticoids maybe potentially responsible for adrenal sites of relapse. Further investigation should focus on this unique pattern of relapse and optimal management.
05Age-related trends in initial systemic therapy for metastatic clear cell renal cell carcinoma (mRCC)
First Author: Osterman, Chelsea
Co-Authors: Deal, Allison; Milowsky, Matthew; Rose, Tracy
Author Company: University of North Carolina at Chapel Hill
Background: Historically, only about half of patients with mRCC received systemic treatment for their disease, largely due to the potential side effects associated with available therapies. The immune checkpoint inhibitor (ICI) nivolumab was approved as second line therapy for mRCC in 2015, and is generally well tolerated. We hypothesize that the introduction of ICIs as a treatment option has allowed more total patients with mRCC to receive systemic treatment, even prior to first-line approval. Furthermore, we hypothesize that early adoption of first-line immunotherapy for mRCC disproportionately increased in elderly patients, suggesting that it is perceived as more tolerable for this population.
Methods: We utilized patient registry data from the National Cancer Database (NCDB). This database contains information collected from over 1,500 Commission on Cancer accredited facilities across the United States. Patients with stage 4 clear cell RCC were included. Patients aged 70 years or older were considered “elderly.” Patients coded as receiving chemotherapy were assumed to have received targeted therapy. Systemic treatments were compared for patients in 2016 (the year after nivolumab approval but prior to front-line combination therapy approval) with patients in 2011 (5 years prior as a control). Comparisons of initial treatment by year of diagnosis were performed using chi2 and logistic regression analysis.
Results: A total of 8,342 patients were identified for inclusion in the study. Median age at diagnosis was 65 years and 67.9% of patients were male. In 2016, 10.09% of patients received initial immunotherapy and 51.52% received targeted therapy, while the remainder of patients did not receive any type of systemic treatment. This is compared to 2.71% and 49.93%, respectively, in 2011, and represents a significant rise in the use of immunotherapy (p < 0.001) but unchanged use of targeted therapy (p = 0.15). The increase in use of initial immunotherapy over this time period was significantly greater among elderly patients compared to non-elderly (pinteraction < 0.001), and this remained significant after controlling for sex, race, and comorbidity index.Conclusions: The introduction of immune checkpoint inhibitors for management of advanced ccRCC increased the total percentage of patients receiving any systemic therapy, even prior to approval of ICIs in the first line setting. In particular, there has been a higher rate of adoption of immunotherapy for elderly patients compared to younger ones. This is likely partly due to the relatively increased tolerability of immunotherapy compared to targeted therapy allowing patients to receive systemic treatment that would otherwise not have been candidates. Further studies are required to explore the shifting treatment landscape of advanced ccRCC as additional agents continue to be approved at a rapid pace.
06Agnostic Transcriptional Profiling of The Cancer Genome Atlas Data Identifies Distinct and Cooperative Role of TP63 Isoforms in Renal Cancer Subtypes That Drive Progression and Predict Clinical Outcomes
First Author: Abbas, Hussein
Co-Authors: Ngoc Hoang Bao Bui, Kimal Rajapakshe, Justin Wong, Preethi Gunaratne, Kenneth Y. Tsai, Cristian Coarfa, Elsa R. Flores
Author Company: MD Anderson Cancer Center
Abstract: Common transcriptional signatures that predict outcomes across different cancers are not well characterized. TP63, a family member of TP53, is required to maintain stem cell pluripotency and suppresses the metastatic potential of cancer cells through multiple mechanisms. In order to identify shared signatures across tumors in light of TP63 signatures, we conducted cross-species mouse-human analysis and utilized all 33 cancers from The Cancer Genome Atlas to generate 17 cancer initiation and 27 cancer progression tissue-specific signatures. Using this agnostic approach, we identified a pleiotropic role of TP63 isoforms (TAp63 and ΔNp63) that were mostly notable in genitourinary cancers, specifically renal clear cell carcinoma, kidney chromophobe, renal papillary and bladder cancers. We found a distinctive role of ΔNp63 as a suppressor of tumor progression by cooperating with TAp63 to modulate key biological pathways, principally cell cycle regulation, extra cellular matrix remodeling, epithelial to mesenchymal transition, and the enrichment of pluripotent stem cells. Importantly, these TAp63 and ΔNp63 signatures can prognosticate progression and survival, even within specific stages, in bladder and renal carcinomas. Our data describe a novel approach for understanding transcriptional activities of TP63 isoforms across a large number of cancer types, potentially enabling the identification of patient subsets most likely to benefit from therapies predicated on manipulating specific TP63 isoforms as well as predicting outcomes.
07An Exploratory Study of 89Zr-DFO-Atezolizumab ImmunoPET/CT in Patients with Locally Advanced or Metastatic Renal Cell Carcinoma
First Author: Bowman, Isaac A.
Co-Authors: Dakanali, Marianna; Mulgaonkar, Aditi; Sun, Xiankai; Oz, Orhan K.; Brugarolas, James
Author Company: UT Southwestern Medical Center
Background: Immune checkpoint inhibitors (ICIs) against CTLA-4, PD-1, and PD-L1, have fundamentally changed the treatment landscape for metastatic renal cell carcinoma (RCC) over the last several years. Despite this, responses to single-agent anti-PD-1 occur in only 25% of patients, and the risk of toxicity, particularly with combined anti-PD-1/CTLA-4 agents, is formidable. Biomarkers such as PD-L1 expression assessed by immunohistochemistry and tumor infiltrating lymphocyte density may predict response, but require invasive biopsies that fail to interrogate fully the complex tumor microenvironment throughout the patient. A number of efforts are underway to better identify patients who will benefit from immune checkpoint inhibition. Among these are the use of radiolabeled antibodies against PD-L1. While under investigation in several tumor types, to our knowledge this is the first such effort in RCC, despite the clear evidence of clinical efficacy of ICIs in RCC. We propose to combine the specificity of anti-PD-L1 antibodies with the sensitivity, resolution, and quantification offered by positron emission tomography to develop immunoPET (iPET). iPET would allow real-time monitoring of the interaction between the tumor and its microenvironment in-vivo, and may correlate with clinical response and toxicity with ICIs.
Methods: The therapeutic anti-PD-L1 antibody atezolizumab is conjugated to Zirconium-89 via desferrioxamine (DFO). This novel radiotracer (89Zr-DFO-Atezolizumab) will be administered to two cohorts of approximately 20 patients. The first cohort will be patients with localized RCC before they undergo surgery, and the second cohort will include patients with metastatic RCC prior to treatment with an ICI. Subjects must have pathologically confirmed RCC (cohort 2) or imaging consistent with localized RCC who are planning to undergo surgical resection. The main exclusion criteria are existing use of an ICI or conditions that would preclude treatment with an ICI including autoimmune disease or chronic steroid use > 10 mg/day prednisone equivalents. All subjects will receive an intravenous injection of 89Zr-DFO-Atezolizumab followed 7 days (+/- 1 day) later by a PET/CT scan. Subjects will be followed per standard clinical practice for up to 5 years. Subjects in cohort 2 will receive treatment with ICI therapy per clinician discretion. Subjects will receive a repeat 89Zr-DFO-Atezolizumab PET/CT scan if disease recurrence is documented (cohort 1) or should progression or toxicity occur after treatment with an ICI (cohort 2). Biopsy of sites of disease recurrence (cohort 1) will be standard, and encouraged for sites of disease progression (cohort 2). The co-primary endpoints will be an exploratory analysis of 89Zr-DFO-Atezolizumab uptake on PET/CT with PD-L1 expression assessed by IHC in patients undergoing nephrectomy (cohort 1) and to evaluate 89Zr-DFO-Atezolizumab uptake across metastatic sites and explore the relationship with response or toxicity while receiving ICI therapy (cohort 2). An investigational new drug approval has been granted for 89Zr-DFO-Atezolizumab (IND 143266) and accrual will commence in August 2019. NCT04006522
08Association Between Depth of Response and Overall Survival: Exploratory Analysis in Patients With Previously Untreated Advanced Renal Cell Carcinoma (aRCC) in CheckMate 214
First Author: Gruenwald, Viktor
Co-Authors: Choueiri, Toni; Rini, Brian; Powles, Thomas; George, Saby; Grimm, Marc-Oliver; McHenry, Brent; Maurer, Matthew; Motzer, Robert; Hammers, Hans; Tannir, Nizar; Albiges, Lawrence
Author Company: University Hospital Essen, Brigham and Women’s Hospital, Cleveland Clinic, Barts Cancer Institute, Roswell Park Cancer Institute, University Hospital of Jena, Bristol-Myers Squibb Company, Memorial Sloan Kettering, UT Southwestern Medical Center, MD Anderson Cancer Center, Institut Gustave Roussy
Background: Depth of response (DepOR; maximum percent reduction from baseline in sum of target lesion diameters) has shown prognostic value for long-term survival in multiple malignancies. Among aRCC patients in CheckMate 214, objective response and complete response rates were higher and more durable, and overall survival (OS) was greater (intention-to-treat: hazard ratio [HR] 0.71, P=0.0003; intermediate/poor-risk patients: HR 0.66, P<0.0001) for nivolumab + ipilimumab (NIVO+IPI) versus sunitinib (SUN) at 30-month minimum follow-up. This exploratory analysis evaluated the relationship between DepOR and OS in CheckMate 214 to determine a potential DepOR threshold predictive of long-term OS with NIVO+IPI.
Methods: Patients with previously untreated aRCC were randomized 1:1 to NIVO+IPI (3 mg/kg + 1 mg/kg intravenously) every 3 weeks for 4 doses, followed by NIVO (3 mg/kg intravenously) every 2 weeks, or SUN 50 mg/day orally for 4 weeks (6-week cycles). An exploratory analysis of OS by DepOR quartiles was conducted (quartile 0, no reduction; quartile 1, >0 to =25%; quartile 2, >25 to =50%; quartile 3, >50 to =75%; quartile 4, >75 to =100%).
Results: Of 550 and 546 patients randomized to NIVO+IPI or SUN, 479 and 459, respectively, had postbaseline target lesion measurements. Overall, greater DepOR was associated with improved OS in both arms (Table). Patients on NIVO+IPI with >50 to =75% (quartile 3) tumor reduction had similar OS as those with >75% (quartile 4) reduction, whereas only quartile 4 patients achieved comparable OS benefits with SUN (203/550 [37%] NIVO+IPI vs 46/546 [8%] SUN patients). Receiver operating characteristic analysis supported a >50% DepOR threshold for greatest OS benefit with NIVO+IPI. Additional analyses of the relationship between DepOR and outcomes across arms will be presented.
Conclusions: The relationships between DepOR and OS are distinct for NIVO+IPI versus SUN, with a greater percentage of NIVO+IPI patients having prolonged OS. Similar notable OS benefits in NIVO+IPI DepOR quartile 3 and quartile 4 suggest that a DepOR threshold >50% may be a useful indicator of potential for long-term survival with NIVO+IPI in aRCC patients. Prospective analyses to determine clinical applications are needed. Originally presented at the European Society for Medical Oncology (ESMO) Congress; September 27–October 1, 2019; Barcelona, Spain.
Clinical Trials registration: NCT02231749
Funding source: Bristol-Myers Squibb
| DepOR | NIVO+IPI N=479 | SUN N=459 | ||||
| n (%) | Median OS, months (95% CI) | OS probability vs quartile 0, HR (95% CI) | n (%) | Median OS, months (95% CI) | OS probability vs quartile 0, HR (95% CI) | |
| Quartile 0a | 105 (22) | 26.9 (18.6.NE) | – | 94 (20) | 12.4 (9.5.15.0) | – |
| Quartile 1 | 107 (22) | NR (29.9.NE) | 0.63 (0.43.0.93)b | 137 (30) | 31.9 (22.1.37.8) | 0.48 (0.35.0.67)b |
| Quartile 2 | 64 (13) | NR (26.1.NE) | 0.65 (0.42.1.01)b | 109 (24) | NR (NE) | 0.21 (0.14.0.32)b |
| Quartile 3 | 96 (20) | NR (NE) | 0.22 (0.13.0.38)b | 73 (16) | NR (NE) | 0.18 (0.11.0.29)b |
| Quartile 4 | 107 (22) | NR (NE) | 0.18 (0.11.0.32)ab | 46 (10) | NR (NE) | 0.08 (0.03.0.17)b |
aAmong quartile 0 patients, median OS was longer and OS probabilities were notably higher with NIVO+IPI vs SUN.
bP<0.0001 vs quartile 0 in the same arm.
CI, confidence interval; NE, not estimable; NR, not reached.
09Association of academic rank and productivity with metrics of Twitter utilization amongst kidney cancer experts
First Author: Salgia, Nicholas
Co-Authors: Feng, Matthew; Prajapati, Dhruv; Harwood, Richard; Nissanoff, Moshe; Dara, Yash; Ruel, Nora; Salgia, Meghan; Salgia, Sabrina; Pal, Sumanta
Author Company: City of Hope Comprehensive Cancer Center
Association of academic rank and productivity with metrics of Twitter utilization amongst kidney cancer experts Nicholas J. Salgia, Matthew Feng, Dhruv Prajapati, Richard Harwood, Moshe Nissanoff, Yash Dara, Nora Ruel, Meghan Salgia, Sabrina Salgia, Sumanta K. Pal Department of Medical Oncology and Experimental Therapeutics, City of Hope Comprehensive Cancer Center, 1500 East Duarte Road, Duarte, CA, 91010, USA.
Background: Twitter has become an increasingly popular platform for physicians to share knowledge and connect with peers and patients. We have previously characterized Twitter content pertaining to kidney cancer (Sedrak et al JCO CCI 2019). In the current study, we sought to determine associations between academic rank, academic productivity and indices of Twitter activity amongst physicians classified as kidney cancer experts.MethodsWe defined kidney cancer experts as physicians who (1) maintained an appointment at an academic center, (2) listed expertise online (including in their Twitter biography) in kidney cancer, and/or (3) had = 2 MEDLINE citations emerging with the joint search term “kidney cancer.” All experts considered in this study had active Twitter accounts. Demographic data were collected, including academic rank (tenure status, years on faculty, specialty, and US News & World Report ranking of affiliated cancer center) and academic productivity (H-index and number of publications). Twitter metrics (number of tweets, number following, number of followers, cumulative likes, and time on platform) were collected as well. Pearson correlation coefficients were calculated to assess association between numeric variables and Kruskwall-Wallis tests were performed for categorical variables. Lastly, a general linear model was created for the prediction of the ln(followers) based on variable parameters.
Results: Amongst 59 kidney cancer experts identified, 14 (23.7%) were assistant professors, 26 (44.1%) were associate professors, and 19 (32.2%) were full professors. Associate professors experienced a greater median number of followers (2289) versus assistant professors (1253) and full professors (1108) (p=0.04) as well as number following (659 vs. 290 vs. 266, p=0.08). Urologists had a greater median number of followers (2021) than medical oncologists (941) (p=0.008). With respect to academic productivity, ln(followers) was correlated with ln(H-index) (r=0.25, p=0.06). The development of a general linear model to predict ln(followers) utilizing previously listed demographic data, academic data, and Twitter metrics as predictors resulted in R-square=0.77. Ln(followers) is more positively associated with associate professors compared to assistant or full professors (t=2.04, p=0.05) and the interaction of ln(H-index) * ln(publications) is positively associated with ln(followers) (t=2.20, p=0.03).ConclusionsA combination of Twitter metrics, academic rank, and academic productivity resulted in a well-fit linear model to predict the number of Twitter followers. Associate professors, the majority of whom have been in practice for 11-20 years, demonstrated
10Association of clinical benefit (CB) from first-line (1L) treatment and CB in further lines or overall survival (OS) in metastatic renal cell carcinoma (mRCC)
First Author: Dizman, Nazli
Co-Authors: Gustavo Bergerot, Paulo; Decat Bergerot, Cristiane; Philip, Errol; Salgia, Nicholas; Hsu, JoAnn; Pal, Sumanta K.
Author Company: City of Hope Comprehensive Cancer Center
Background: Clinical benefit (CB) rate is a commonly reported parameter in clinical trials and a potential indicator of anti-tumor activity. However, there appears to be a controversy regarding whether CB is associated with an OS benefit. Herein, we sought to identify a relationship between CB in first-line (1L) therapy and CB in subsequent line therapy and overall survival (OS) in patients (pts) with metastatic renal cell carcinoma (mRCC).
Methods: Clinical data of consecutive mRCC pts treated at the City of Hope Comprehensive Cancer Center between 2010-2019 were retrospectively collected. Pts who received =2 lines of treatment were included in the analysis. CB was comprised of complete response (CR), partial response (PR) and stable disease (SD). Pts with no clinical benefit (NCB) were those who experienced progressive disease as best response. Comparison of response across treatment lines was assessed by Chi-square test. Survival analysis was performed by Kaplan-Meier function. Multivariable Cox regression analysis was performed to control for potential confounders.
Results: In total, 198 (M:F 144:54) pts were included. Median age was 60 years and 156 pts (78.8%) had clear cell disease. Per IMDC criteria, 61 pts (30.8%) had favorable risk while 137 (69.9%) pts had intermediate/poor risk disease. 1L treatment was targeted therapy (TT) in 92.4% of patients while 2% received immunotherapy (IO) and 4% received combination IO and TT. CB rate in 1L was 66.2% with 44.4% SD, 19.7% PR and 2% CR. Median progression free survival in 1L was 6.0 (95% CI, 4.6 - 7.4) months. Median OS was 37.3 (95% CI, 29.1 – 45.5) months. CB in 1L was related with CB in 2L (p=0.003). However, no relationship was observed between 1L CB and 3-4L CB rates. Median OS in pts with CB in 1L was 48.6 (95% CI, 32.8 – 64.4) months versus 24.1 (95% CI, 18.4 – 29.9) months in pts with NCB in 1L (p=0.000). Univariate cox regression survival analysis showed that CB in 1L and IMDC risk category were the two factors associated with OS. The effect of 1L CB on OS remained significant after adjustment of IMDC risk categories with hazard ratio of 2.07 (95% CI, 1.38 – 3.12) and p<0.001.
Conclusions: In our experience, pts who obtained CB from 1L therapy had significantly longer overall survival, independent of IMDC risk score. CB in 1L predicted CB in 2L, but predictive capability in subsequent therapy could not be established.
11Associations between inflammation, depression, and cancer stage in patients with resected renal cell carcinoma
First Author: Le, Thien-Linh
Co-Authors: Patil, Dattatraya; Sandberg, Alex; Lin, Fangyi; Lee, Grace; Ogan, Kenneth; Master, Viraj
Author Company: Emory University Department of Urology
Abstract: Associations between inflammation, depression, and cancer stage in patients with resected renal cell carcinoma
Introduction and Objectives: Depression has been shown to be prevalent in renal cell carcinoma (RCC) more than many other solid organ cancers. C-reactive protein (CRP), a marker of systemic inflammation, has been associated with poor outcomes in a variety of disease states including depression and nearly all malignancies. Physiologically, CRP is elevated through both IL-6 mediated hepatic production and intra-tumoral, and persistent CRP elevation after presumed curative nephrectomy is prognostic of recurrence. We sought to examine the pre-operative associations between depression, inflammation, and cancer severity in patients with RCC and, subsequently, test if nephrectomy results in improvement of depression.
Methods: We identified patients presenting for preliminary evaluation of a renal mass and prospectively administered Personal Health Questionnaire-8 (PHQ-8) surveys. These surveys were also administered to those who presented for post-nephrectomy follow-up. Exposures tested included the inflammatory marker CRP, tumor stage (AJCC), ECOG functional status (Eastern Cooperative Oncology Group), and demographic factors. The primary outcomes were clinical depression (PHQ-8 score = 10) and post-operative improvement in depression (at-least 2-point reduction in PHQ-8 score).
Results: 81 total patients completed a pre-operative PHQ-8 survey and underwent nephrectomy for localized or metastatic RCC. Of these, 44 patients also completed a post-operative PHQ-8 at follow-up. Elevated CRP before or after surgery was significantly associated with higher stage RCC (AJCC stage T3 and T4) (p < 0.001, p = 0.007 respectively). Additionally, RCC patients with elevated CRP before surgery were more likely to have depression (OR = 4.01, p = 0.011), even when controlling for tumor stage (OR = 47.25, p = 0.006). Patients who had depression before surgery had a mean improvement in PHQ-8 score of 3.5 points after surgery, whereas patients without depression experienced a worsening of PHQ-8 scores by a mean of 0.4 points after surgery (p = 0.007). Only ECOG functional status was significantly associated with post-operative improvement in PHQ-8 scores after multivariate analysis (OR = 8.76, p = 0.047).
Conclusions: We found that inflammation, as measured by CRP, is associated with higher tumor stage and higher rates of depression in patients with RCC. Patients with comorbid depression and renal cell carcinoma experienced improvement in PHQ-8 scores after nephrectomy, while patients without depression before surgery experienced a marginal worsening of PHQ-8 scores after nephrectomy. More research is needed to further elucidate the associations, causative or otherwise, between inflammation, depression, and renal cell carcinoma.
12Bilateral Multifocal Clear cell papillary renal cell carcinoma with discordant histopathology
First Author: Jakobsson, Rasmus
Co-Authors: Lundstam, Sven; Svensson, Hjalmar; Johansson, M.D., Martin
Authors Company: University of Gothenburg
Background: Renal cell carcinomas (RCC) are an evolving group of tumors comprising at least 14 distinct histological entities according to the latest WHO classification (2016). Clear cell papillary renal cell carcinoma (CCPRCC) is a recently defined RCC form with supposedly indolent behavior and unproved metastatic potential.
Methods: Case description: The patient is a 51-year-old male with a history of hypertension. Due to diffuse abdominal pain of transitory nature, a CT-scan was performed revealing bilateral small renal masses, four in the right kidney and six in the left, with maximum diameter of 35 mm. Physical examination and routine lab were normal along with equal split function. MRI and CT thorax showed nothing additional. A biopsy from the left kidney indicated CCRPCC. Open left partial nephrectomy was performed with resection of 6 tumors. Histology showed clear cells growing in papillary and tubular/acinar structures. Immunohistochemistry revealed diffuse CK7 positivity and basal “cup-shaped” carbonic anhydrase IX positivity compatible with CCPRCC. However, diffuse CD10 positivity was also noted along with non-linear, non-reversed polarity nuclei. These findings speak against a CCPRCC diagnosis. Due to ipsilateral renal infarction radical nephrectomy was performed after 1 month. The patient underwent routine genetic evaluation without finding of any genetic disorders, including von Hippel-Lindaus disease. RNA and DNA was isolated from bio banked tumor tissue. Whole genome sequencing and RNA seq was performed. Data underwent subsequent bio informatic analysis. The patient is well and is subject to active surveillance for the remaining tumors in the right kidney.
Results: Light microscopical evaluation of the tumor tissue favors a diagnosis of CCPRCC. This diagnosis is partially supported by immunohistochemistry. Analysis of whole genome and RNA sequencing data however point to a hybrid form of RCC with clear cell histology.
Conclusion: This case illustrates a patient with multifocal bilateral renal tumors. Histopathological evaluation favored the diagnosis CCPRCC, a benign tumor. We note however histological and marker anomalies with diagnostic impact. In depth genomic analysis yielded results indicating features not commensurate with CCPRCC. Patients diagnosed with newly defined RCC types with unusual clinic or discordant pathological findings might benefit from a low threshold to reevaluate diagnosis since it may have impact on follow-up and treatment.
13Body composition and outcomes to Immune-Oncology agents in 165 patients with metastatic clear cell renal cell carcinoma
First Author: Ged, Yasser
Co-Authors: Patil, Sujata (; Duzgol, Cihan; Petruzella, Stacey; Stein, Emily; Mourtzakis, Marina; Paris, Michael; Sanchez, Alejandro; Chaim, Joshua; Akin, Oguz; Carlo, Maria; Xenakis, Marybeth; Hakimi, A.Ari; Motzer, Robert; Furberg, Helena; Voss, Martin
Authors Company: Memorial Sloan Kettering Cancer Center, University of Waterloo, Westchester Public Schools
Background: Obesity was recently reported to be associated with improved clinical outcomes to Immune-Oncology (I/O) agents in metastatic melanoma (McQuade, Lancet Onc, 2018) and clear cell renal cell carcinoma (mccRCC) (De Giorgi, CCR, 2019). This may be due to a state of chronic inflammation. However, body mass index (BMI) determined by height and weight alone is a crude measure of body size, limiting the interpretation and clinical application of such findings. Herein, we used a large retrospective mccRCC cohort to study the association between IO outcomes and different radiographically-assessed body composition parameters, in addition to BMI.
Methods: Baseline clinical features and therapeutic outcomes were retrospectively collected for patients with mccRCC treated with I/O agents at Memorial Sloan Kettering Cancer Center. Three body size variables were determined at the start of I/O therapy: BMI, visceral tissue adiposity index (VATI) and skeletal muscle index (SMI). VATI and SMI were derived from computed tomography images taken at the level of the L3 vertebra using Sliceomatic software. High vs. Low VATI categories were based on the gender-specific median values, while high vs. low SMI (i.e., sarcopenia) categories were based on the International Consensus of Cancer Cachexia criteria (<35 cm2/m2 in females and <55 cm2/m2 in males. The relationship between each size variable with overall survival (OS), progression free survival (PFS), and objective response rate (ORR) was evaluated using the log-rank test and multivariable Cox regression for the time-to-event outcomes, and the Chi-Square test for binary outcomes.
Results: A total of 165 I/O treated patients were included in the analysis. 113 patients (68%) were overweight or obese as determined by BMI. 99 patients (60%) were classified as sarcopenic. Both higher VATI and SMI values were significantly associated with higher BMI values (Each P<0.001). Sarcopenia was significantly associated with poor IMDC risk score (P<0.001). In univariable analysis, overweight and obese patients had prolonged OS compared to normal weight patients (Obese vs. normal [HR 0.54, 95% CI: 0.31, 0.95] P=0.032). However, this association became non-significant after adjusting for IMDC score (adjusted HR 0.72, 95% CI: 0.39, 1.31; P=0.287). There was no association between VATI category and OS. Sarcopenia was significantly associated with adverse OS (HR 2.73, 95% CI: 1.63, 4.59, P<0.001). In multivariate analyses adjusting for IMDC scores, number of lines, and receipt of ipilimumab/nivolumab combination, there was an independent association between sarcopenia and inferior OS (adjusted HR 1.8, 95% CI: 1.04, 3.13, P=0.036). We found no association between any body size features (BMI, VATI and SMI) with PFS or ORR.
Conclusions: In this observational cohort of mccRCC patients treated with I/O, we did not find an independent association with I/O outcomes, neither for BMI/obesity nor for visceral tissue adiposity. However, sarcopenia was independently associated with inferior OS in multivariable analysis. Further research is needed to determine the underlying mechanism for this association and whether low skeletal muscle can be modified to improve clinical outcomes among mccRCC patients.
14BONSAI trial: a prospective trial evaluating a first line treatment with cabozantinib in metastatic collecting duct carcinoma.
First Author: Sepe, Pierangela
Co-Authors: Verzoni, Elena; Mennitto, Alessia; Claps, Melanie; De Braud, Filippo; Procopio, Giuseppe
Authors Company: Fondazione IRCCS Istituto Nazionale Tumori
Background: Collecting duct carcinoma (CDC) is a rare and aggressive disease with poor prognosis. Different from other renal cell carcinoma (RCC) subtypes, our knowledge on genetic and metabolic alterations is very poor and treatment choice, including chemotherapy or TKIs, is unfortunately based on retrospectives data. Cabozantinib is a multi-kinase inhibitor showing a strong activity in clear cell RCC. The possibility of blocking multiple pathways combined with the likely high mutational burden of CDC make cabozantinib a promising candidate for CDC treatment. Patients and methods: This is a prospective, monocentric, single arm phase II trial, evaluating a first line treatment with cabozantinib in patients (pts) with untreated locally advanced or metastatic CDC. Cabozantinb is given orally at dose of 60 mg/day until disease progression defined by RECIST 1.1 or unacceptable toxicity. Main inclusion criteria: previously untreated advanced or metastatic CDC, presence of measurable disease by RECIST v1.1 criteria. A central review of tumor tissue samples is mandatory before study entry. Primary endpoint: objective response rate. Secondary endopoints: progression free survival, overall survival and treatment tolerability. Exploratory objectives: identification of somatic mutation profiles on tissue samples by NGS and RNA sequencing; monitoring of plasma and viable peripheral blood mononuclear cell (PBMC) for immune-phenotyping to assess the modulating activity of cabozantinib on local and systemic tumor immunity. The study design is based on a Simon’s two stage optimal design: in the first step at least 2 responses in 9 pts enrolled are needed to go to the second stage of the study (14 additional pts). Fourteen pts have been enrolled thus far.Clinical trial information: NCT03354884
15Brain Metastasis from Renal Cell Carcinoma: an Institutional Study
First Author: Suarez-Sarmiento, Jr., Alfredo
Co-Authors: Shuch, Brian; Nguyen, Kevin; Syed, Jamil; Hurwitz, Michael; Chiang, Veronica; Kluger, Harriet
Author Company: Nova Southeastern University, UCLA, Yale
Introduction and Objectives: Brain Metastases (BM) are frequently observed in advanced renal cell carcinoma (RCC). Historically these individuals have been excluded from clinical trials, but recently with better local control, many can receive aggressive therapy after treatment. We evaluate our single institutional experience over various treatment eras.
Methods: Patients undergoing evaluation for RCCBM (RCC brain metastases) from 2001-2018 were identified from our institutional database. Clinical notes, demographics, comorbidities, histology, central nervous system (CNS) treatments, systemic therapy, and outcomes were reviewed. Overall survival (OS) and CNS recurrence-free survival (RFS) were evaluated with the Kaplan-Meier method. Cumulative incidence was evaluated using a competing risk model.
Results: We identified 158 patients with RCCBM, of which 94.4% had clear cell RCC, and 90.6% had extracranial metastases at diagnosis. Of these patients, 94 individuals (60%) developed RCCBM over time, while 46 patients (29.1%) had RCCBM upon initial presentation. Clinical symptoms were noted in 81.9% of patients. Number of lesions and not largest lesion size correlated with symptoms. Ninety (56.9%) patients received systemic therapy after RCCBM treatment. The median OS after diagnosis of RCCBM was 8.4 months with a three-year OS of 28.2%. The median OS after RCCBM was not different by mode of presentation. The median CNS RFS was 8.5 months overall, however, those with 1 and >1 lesion had median CNS RFS of 12.4 months and 6 months, respectively (P <0.001). Patients diagnosed within the most recent years (2011-2018) demonstrated the most favorable median OS (p=0.0028, log-rank test).
Conclusions: The majority of RCC patients with BM are symptomatic and had prior metastatic disease that progressed to the brain. Those with a solitary RCCBM are less likely to develop CNS recurrence after local therapy and are ideal candidates for enrollment into clinical trials. A subset of patients can have extended survival, and while the overall prognosis remains poor, we demonstrate improvement in outcome.
16Choosing First-Line Treatment for Metastatic Renal Cell Carcinoma (mRCC) in the Immuno-oncology (IO) Era: Systematic Review and Network Meta-Analysis
First Author: Sabino, Fernando
Co-Authors: Debiasi, Márcio; Cauduro, Carolina; Schutz, Fabio; Sasse, Andre; Soares, Andrey; Mendes, Gabriela; Bastos, Diogo; Maluf, Fernando Fay, Andre;
Authors Company:
Latin American Cooperative Oncology Group
Hospital Santa Lucia, Brasilia, Brazil
Hospital Universitário de Brasilia, Brasilia, Brazil
BP – A Beneficência Portuguesa de São Paulo, São Paulo, Brazil
Grupo Sonhe, Campinas, Brazil
Centro Paulista de Oncologia, São Paulo, Brazil
Hospital Albert Einstein, São Paulo, Brazil
Hospital Sirio-Libanes, Sao Paulo, Brazil
Introduction: In recent years the tyrosine kinase inhibitor (TKI) cabozantinib and other IO combinations (i.e. IO-IO and IO-TKI) have improved clinical outcomes compared to sunitinib in first-line mRCC treatment. This analysis compare and rank different strategies to identify the best first-line regimen associated with longer overall survival (OS).
Methods: PubMed, Embase and Cochrane Databases were searched for randomized clinical trials comparing sunitinib with cabozantinib or IO combinations for first-line treatment in mRCC. The primary outcome was OS and secondary outcomes were progression free survival (PFS), overall response rate (ORR) and toxicity. A Bayesian network meta-analysis (NMA) was conducted to multiple treatment comparison (MTC), and treatment regimens were ranked according to their relative comparisons and confidence intervals.
Results: Among 5 clinical trials (n=3,915), 1,958 patients received sunitinib as control arm, while 79, 550, 432, 442 and 454 patients received cabozatinib (cabo), ipilimumab+nivolumab (ipi+nivo), pembrolizumab+axitinib (pembro + axi), avelumab + axitinib (avelu+axi) and atezolizumab + bevacizumab (atezo+bev), respectively. HRs for OS and PFS (ITT population) are described in Table 1. In favorable-risk (FR) patients, no significant differences in OS were observed across the regimens. However, in intermediate-risk (IR) and high-risk (HiR) patients both ipi+nivo and pembro+axi combinations were found to be superior to sunitinib, with no significant difference between them. In the ranking for OS, pembro+axi had the highest probability of being the most effective first-line treatment in FR and IR/HiR patients (79.4% and 84.9%, respectively).
Conclusions: In this indirect comparison, superiority in OS was found only for IR/HiR patients with ipi+nivo and pembro+axi compared to sunitinib, with no significant difference between them. Detailed results for PFS and ORR comparisons, as well as toxicity profile, will be presented.
| ITT - OS | ||||||
| ITT - PFS | ATEZO + BEV | 1.19 (0.81-1.76) | 1.16 (0.74-1.85) | 1.31 (0.99-1.73) | 1.75 (1.19-2.59) | 0.93 (0.76-1.14) |
| 1.20 (0.93-1.56) | AVELU + AXI | 0.98 (0.57-1.66) | 1.10 (0.75-1.61) | 1.47 (0.92-2.35) | 0.78 (0.56-1.09) | |
| 1.73 (1.09-2.74) | 1.44 (0.89-2.31) | CABO | 1.13 (0.71-1.78) | 1.51 (0.89-2.57) | 0.80 (0.53-1.21) | |
| 0.98 (0.78-1.22) | 0.81 (0.63-1.05) | 0.57 (0.36-0.89) | IPI + NIVO | 1.34 (0.92-1.97) | 0.71 (0.59-0.86) | |
| 1.20 (0.93-1.55) | 1.00 (0.76-1.32) | 0.70 (0.43-1.12) | 1.23 (0.97-1.57) | PEMBRO + AXI | 0.53 (0.38-0.74) | |
| 0.83 (0.70-0.98) | 0.69 (0.56-0.85) | 0.48 (0.31-0.75) | 0.85 (0.73-0.98) | 0.69 (0.57-0.84) | SUNITINIB | |
NA-Not Available
17Chromosome 8 Deficient Clear Cell RCC: An Analysis of Clinicopathologic Characteristics
First Author: Valdez, Tyler
Co-Authors: Mendhiratta, Neil; Sanchez, Desiree; Cone, Brian; Sajed, Dipti; Kang, Sung-Hae; Pooli, Aydin; Lebacle, Cedric; Pantuck, Allan; Ye, Huihui; Shuch, Brian
Author Company: David Geffen School of Medicine at UCLA
Background: Renal cell carcinomas subtypes can be often distinguished by their histological features, immunohistochemical differences, and clinical characteristics. The most common subtype is clear cell renal cell carcinoma (ccRCC), of which, over 80% of well-annotated cases are characterized by loss of chromosome 3p and inactivation of VHL by mutation or promotor hypermethylation However, a subset of ccRCC does not have loss of 3p/VHL. Recently, a new class of ccRCC was found with recurrent alterations with loss of chromosome 8/TCEB1 within this subset. In a small report, this new group of tumors has distinct morphologic features with less aggressive clinical characteristics. In this study, we aimed to identify and characterize the clinical and molecular behavior of TCEB1-mutated tumors using a large cytogenetic database.
Methods: We queried a prospective cytogenetics database of resected renal tumors at the University of California, Los Angeles (UCLA) from 1999 to 2017 to identify clear cell renal tumors. Karyotypes were reviewed with a molecular geneticist (SK) to identify tumors without loss of 3p but loss of chromosome 8. These cases were reviewed by two genitourinary pathologists (DS, HY) to evaluate morphology. Clinical characteristics and demographics were reviewed from the medical records.
Results: From 1209 patients, we identified 783 ccRCC tumors (64.7%). Of these cases, a total of 16 tumors (2%) had loss of chromosome 8 but still possessed chromosome 3p. The mean and median age of the cohort was respectively 57 years and 61 years, with men accounting for 75% of the cohort. Mean and median tumor size was 3.4 cm ± 2.4 and 2.9 cm respectively. There were 15 tumors (93.75%) identified as pT1 and 1 tumor (6.25%) which was pT3. With respect to tumor grade, one of the tumors (6.25%) was Fuhrman grade 1, 10 tumors (62.5%) were Fuhrman grade 2, and 5 tumors (31/25%) were Fuhrman grade 3. A total of 4 patients (25%) had metastatic disease at presentation. Reported pathologic characteristics such as fibromuscular stroma and clear cell cytology with voluminous cytoplasm were commonly observed. Initial immunohistochemistry results showed diffuse box-like staining in CAIX staining, and patchy positivity with CK7 staining.Conclusion: We report the largest experience with tumors with loss of chromosome 8, most often associated with TCEB1-mutated ccRCC. Similar to the available literature, our cohort of chromosome 8 deficient ccRCC was found in 2% of cases. Unlike in the prior cohorts, these tumors were shown to exhibit variable behavior, with the potential to act aggressively as metastatic disease was observed. Furthermore, not all cases have classic morphologic patterns, so molecular characterization may be required. Immunohistochemistry staining and genetic profiling of identified cases are ongoing to confirm TCEB1 mutations.
18Circulating Tumor DNA (ctDNA) results in 110 Patients with Advanced Renal Cell Carcinoma
First Author: Kotecha, Ritesh
Co-Authors: Gedvilaite, Erika; Murray, Samuel; Foster, Ashley; Motzer, Robert; Tsui, Dana; Voss, Martin
Authors Company: Memorial Sloan Kettering Cancer Center
Background: Circulating tumor DNA (ctDNA) profiling is a non-invasive approach to genomically interrogate solid tumors. As a novel tool increasingly used in other solid tumors, key benchmarks are required to evaluate how best to apply this in the management of metastatic renal cell carcinoma (RCC) patients. To assess the utility and fidelity of ctDNA, we performed a large cohort analysis using a comparative approach correlating oncogenomics of primary tissue and matched ctDNA in patients with metastatic ccRCC.
Methods: Patients with prior tissue mutational profiles generated via next generation sequencing (NGS) from nephrectomy or metastatic site specimens underwent a single-time point plasma collection for cfDNA extraction. Targeted NGS sequencing using MSK-IMPACT was performed on tumor and ctDNA, with bi-directional cross genotyping using Waltz 2.0. All patients had matched germline comparison from peripheral blood. Liberal (1-2 reads) and stringent (=3 reads) filters were applied, with a cut-off of <30% allele frequency to remove germline mutations. Clinical data was extracted from the medical record to determine relevant parameters for correlation with ctDNA load.
Results: 110 metastatic clear-cell RCC patients, of whom available IMDC-risk was favorable (25%), intermediate (45%), or poor (4%) were included for analysis. 106/110 (96%) of patients had undergone nephrectomy prior to ctDNA collection, and most patients were heavily pre-treated with an average of 3 systemic therapies (R:0-8). 18/110 (16%) of patients included had sarcomatoid features, and 35% of patients had metastatic disease at presentation. The median time from diagnosis to ctDNA collection was 22.1 months (R: 2.3-215), and the median time difference from primary tissue to ctDNA collection was 23.8 months (R: 1-177). In primary tissue sequencing, 587 mutations were identified across the entire cohort, 58% from primary kidney tumors and 42% from metastatic sites, with a median of 5 alterations per patient (R:1-23). Using “liberal” criteria, 72 patients were found to have at least 1 alteration in ctDNA, with a total of 182 alterations across the entire cohort. Applying “stringent” criteria, 24 patients were found to have at least 1 alteration in ctDNA with a total of 43 alterations across the entire cohort with mean 0.4 mutations/patients (R:0-5). For RCC-specific alterations, VHL alterations were identified in primary tissue and ctDNA (both liberal and stringent) in 102, 31 and 13 alterations, respectively.Conclusions: This large cohort of matched genomics from primary tumor and ctDNA, while demonstrating comparable mutational profiles for RCC-specific genomic alterations, highlights relevant challenges of developing liquid biopsy approaches in this disease. Further efforts that integrate algorithm-based approaches to improve the fidelity of ctDNA and integrate clinical parameters for timepoint selection may help address some of these limitations and will be of critical importance in for the developing this promising new technology for clinical application.
19Clinical correlates of oncogenic events in translocation renal cell carcinoma
First Author: Marcon, Julian
Co-Authors: Sanchez, Alejandro; Di Natale, Renzo G.; Kotecha, Ritesh; Gupta, Sounak; Kuo, Fengshen; Sandhu, Amar; Mano, Roy; Silagy, Andrew W.; Blum, Kyle A.; Nassau, Daniel E.; Benfante, Nicole E.; Ortiz, Michael V.; Carlo, Maria I.; Motzer, Robert J.; Voss, Martin H.; Coleman, Jonathan A.; Russo, Paul; Reuter, Victor E.; Hakimi, A. Ari; Reznik, Ed
Authors Company: Memorial Sloan Kettering Cancer Center
Introduction: Translocation renal cell carcinoma (tRCC) is an aggressive RCC variant. The role of molecular features as biomarkers of disease aggressiveness has not been fully explored. We therefore investigated the association between somatic alterations and clinical outcomes in two independent cohorts of tRCC.
Material and Methods: We included 22 cytogenetically diagnosed tRCCs (MSK-IMPACT cohort), all underwent targeted sequencing of cancer-associated genes. A subset of MSK-IMPACT cases was recaptured by exome-sequencing and then combined with prior whole-exome data of 14 tRCC cases from The Cancer Genome Atlas (TCGA). We explored the data for recurrent somatic mutations, tumor mutation burden (TMB), copy number variations (CNVs) and fraction of copy number altered genome (FCNAg). In TCGA cases, we carried out neoantigen prediction and immune cell deconvolution using RNA sequencing and exome data. We explored the associations between molecular events and clinical outcomes using non-parametric hypothesis testing. Survival estimates were computed using the Kaplan-Meier method and the log rank test. We measured time-on-treatment (TOT) for 14 patients who underwent systemic therapy and explored genomic profiles in patients with a TOT >12 months. Statistical significance was defined by a p-value <0.05.
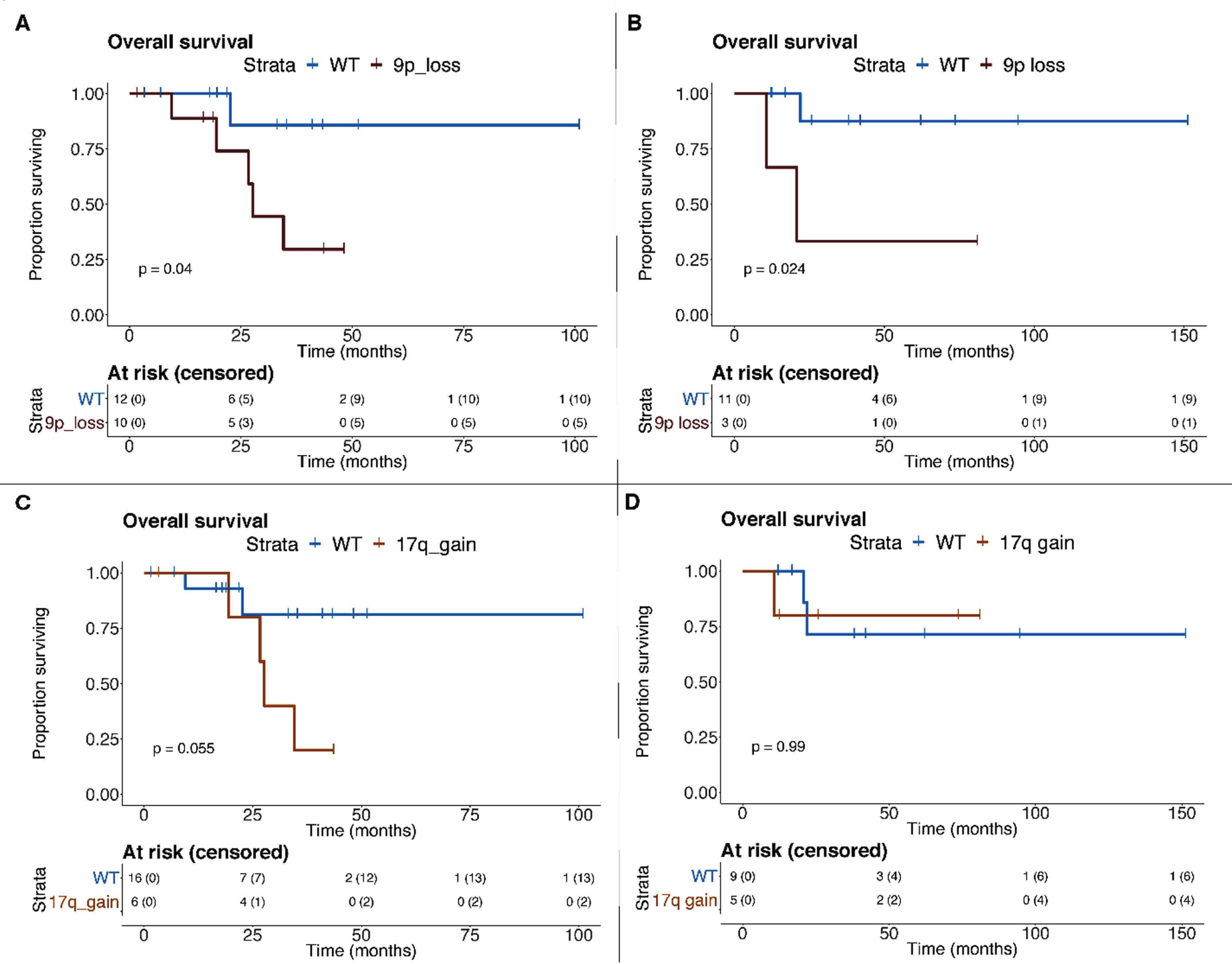
Results: Clinical and molecular features of the MSK-IMPACT cohort are displayed in Fig. 1. A loss of chromosome 9p or gain of 17q was associated with poor overall survival (p=0.04 and p=0.055, respectively), loss of 9p was further associated with poor survival in TCGA cases (p=0.024, Fig. 2). A higher FCNAg was associated with higher AJCC stage in both cohorts (p=0.03 and p=0.019, respectively). TMB and single nucleotide variations were not significantly associated with outcome. Patients with a TOT >12 months showed a non-significantly higher FCNAg (p=0.23), but no other genomic correlates. We found a lower FCNAg (p=0.04) in pediatric patients compared to adults. 7/14 TCGA-exome cases showed a predicted affinity to an HLA-epitope. TCGA-exome cases showed distinct profiles of angiogenesis and PD-L1 gene expression compared to other TCGA-RCC histologies.
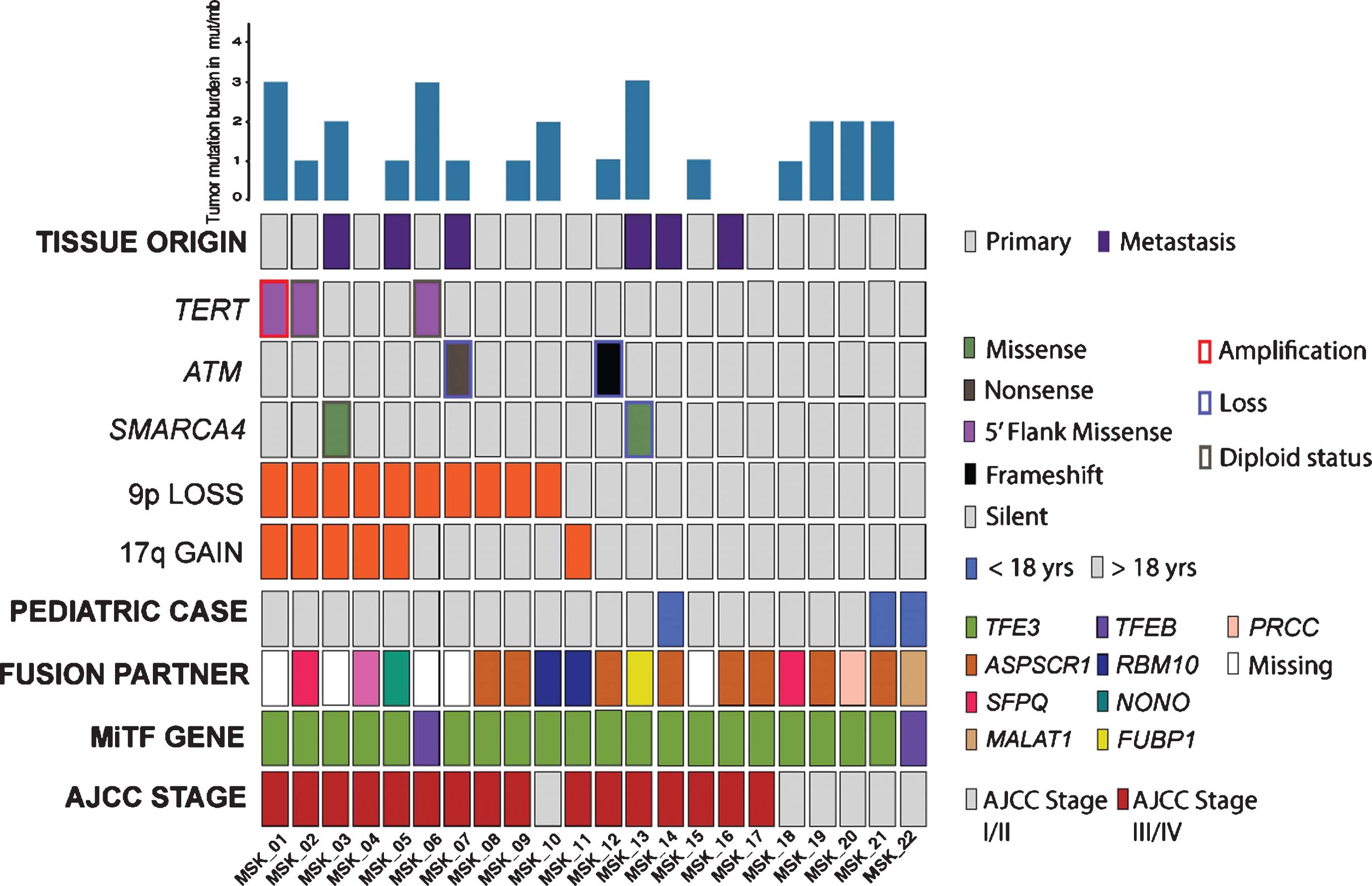
Conclusion: Both CNVs in 9p and 17q and a higher FCNAg were associated with poor outcome in patients with tRCC. An increased load of genomic events in adult patients suggests a more aggressive disease course. Our preliminary findings suggest immunogenicity in a subset of tRCCs, which warrants further exploration.
20Combination cabozantinib and nivolumab treatment in patients with refractory metastatic renal cell carcinoma (mRCC)
First Author: Kinsey, Emily
Co-Authors: Brown, Landon; Kao, Chester; Healy, Patrick; Armstrong, Andrew; McNamara, Megan; Ramalingham, Sundhar; Harrison, Michael; George, Daniel; Zhang, Tian
Authors Company: Duke Medical Center
Background: The treatment landscape has drastically changed in mRCC, moving from anti-VEGF therapies to an immunotherapeutic approach in the first line setting for IMDC intermediate or poor risk mRCC. Combining anti-VEGF and anti-PD-1 or anti-PDL-1 therapies have shown survival improvements in mRCC, leading to approvals for first-line axitinib-pembrolizumab and axitinib-avelumab [1,2]. Cabozantinib and nivolumab (cabo/nivo) is also a safe option in phase 1 trials with some durable responses [3]. We evaluated the outcomes of patients who received cabo/nivo for mRCC refractory to immunotherapy alone.
Methods: A retrospective analysis was performed of patients with mRCC treated with ipilimumab and nivolumab (ipi/nivo) and subsequently with cabo/nivo at Duke Cancer Center between September 2017 and February 2019. Patient outcomes were collected including demographic information, treatment details, responses, and frequency of adverse events. The cohort of patients treated with the cabo/nivo combination is presented here.Results86 patients were treated with ipi/nivo for mRCC and of these patients, 34 patients also received cabozantinib, either alone or in combination with nivolumab. Nine patients received cabozantinib prior to ipi/nivo, and 14 patients received cabozantinib after ipi/nivo, and 10 patients received combination therapy with cabo/nivo. Of the ten patients who received combination cabo/nivo, 2 were favorable risk, 6 were intermediate risk, and 2 were poor risk. One patient had progressive disease, 2 patients had stable disease, 3 patients had a partial response (50% responses), and 4 patients were unevaluable due to insufficient follow up. Eight of 10 patients had treatment ongoing at the time of data collection.
Conclusions: Progression on immunotherapy alone did not appear to confer resistance to cabo/nivo treatment for five of the six patients who had a disease response assessment. Phase III studies COSMIC-313 and Alliance A031704 (PDIGREE) are ongoing to evaluate cabozantinib in combination or in sequence to ipilimumab immunotherapy.
References
[1] Motzer RJ, Penkov K, Haanen J, Rini B, Albiges L, Campbell MT, et al. Avelumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med [Internet]. 2019;380(12):1103–15.
[2] Rini BI, Plimack ER, Stus V, Gafanov R, Hawkins R, Nosov D, et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med [Internet]. 2019;NEJMoa1816714.
[3] Nadal R, Mortazavi A, Stein M, Pal SK, Davarpanah N, Parnes HL, et al. Final results of a phase I study of cabozantinib (cabo) plus nivolumab (nivo) and cabonivo plus ipilimumab (Ipi) in patients (pts) with metastatic urothelial carcinoma (mUC) and other genitourinary (GU) malignancies. Ann Oncol. 2017;28(supplement 5):846O.
21Combining immunotherapy and VEGFR inhibition improves the outcomes of elderly and favorable risk patients with metastatic renal cell carcinoma
First Author: Varkaris, Andreas
Co-Authors: Xu, Wenxin; Davis, Roger; Healy, Brian; McDermott, David
Authors Company: Beth Israel Deaconess Medical Center, Harvard T.H. Chan School of Public Health
Background: Recently, two randomized controlled trials reported the efficacy of combining immunotherapeutics with VEGFR inhibition in patients with metastatic renal cell carcinoma (RCC). When combined with axitinib (a VEGFR inhibitor), both pembrolizumab (a PD-1 inhibitor) and avelumab (a PD-L1 inhibitor) were superior to sunitinib (a VEGFR inhibitor) alone in overall response rate and progression free survival (PFS) for the overall population. Subgroup analysis showed benefit among all patients. However, analyses of elderly patients (aged >65 years) and favorable risk patients (based on IMDC score) did not reach statistical significance in one or both studies.
Methods: We performed a meta-analysis to evaluate the efficacy of immunotherapy-VEGFRi combinations compared to sunitinib in elderly patients (age >65 years) and patients with favorable risk disease.
Results: A random effects model meta-analysis demonstrated that PFS was significantly prolonged with combination therapy compared to sunitinib in patients with age >65 years (HR: 0.66, 95% CI 0.52-0.84, p=0.001). The PFS in favorable risk disease was improved with combination therapy compared to sunitinib but the difference was not statistically significant. (HR: 0.68, 95% CI: 0.46-1.01, p=0.055).
Conclusion: Our results indicate that elderly patients and patients with favorable risk mRCC benefit from combining PD-1 or PD-L1 and VEGFR inhibition
22Connections between BAP1 and the Type I Interferon Pathway in ccRCC
First Author: Langbein, Lauren
Co-Authors: Yang, Haifeng
Authors Company: Thomas Jefferson University
Abstract: BRCA1-associated protein 1 (BAP1) is a deubiquitinase that is mutated in 10-15% of clear cell renal cell carcinoma (ccRCC). Despite the association between BAP1 loss and aggressive disease, the specific role of BAP1 in disease development and/or progression remains unclear. In this study, we found that BAP1 regulates the expression of STAT2 and IRF9, components of the interferon stimulated gene factor 3 (ISGF3) transcription factor involved in Type I interferon signaling. BAP1 enhances ISGF3 levels and downstream target expression, which depends on its deubiquitinase activity, and may promote IFNB1 transcription. Suppression of IFNAR1 abrogates the effect of BAP1 on ISGF3, suggesting that autocrine IFN signaling through the interferon a/ß receptor is necessary for this pathway. Additionally, preliminary results in a xenograft model suggest BAP1 indeed functions as a tumor suppressor in ccRCC. Together, our results indicate that BAP1 loss in ccRCC reduces ISGF3 function, which may influence several aspects of ccRCC tumor biology.
23Cytoreductive Nephrectomy for Symptomatic Primary Tumors: Important Therapeutic Endpoint in the Management of mRCC
First Author: Bilotta, Alyssa
Co-Authors: McFarland, Suzie; Sexton, Wade; Poch, Michael; Manley, M.D., Brandon; Spiess, Philippe
Authors Company: Moffitt Cancer Center, USF Morsani College of Medicine
Background: Cytoreductive nephrectomy (CN) has held an important role in the management of appropriately selected patients with metastatic renal cell carcinoma (mRCC). Recent advances in systemic therapies have provided more treatment options for mRCC. Recent studies have found comparable survival rates for patients exclusively treated with targeted therapy alone. However, other factors including symptom burden were not specifically evaluated as treatment endpoints in these trials. In patients with symptomatic primary tumors, quality of life could potentially be improved with CN versus systemic therapy alone. The purpose of this study is to determine if there is a symptomatic benefit to undergoing CN for mRCC patients and define the specific subset of patients who may benefit most from undergoing upfront CN.
Methods: An IRB-approved retrospective analysis on 167 patients diagnosed with mRCC who underwent CN at Moffitt Cancer Center between 2009 and 2019 was conducted. Statistical analysis was performed using IBM SPSS version 25, defining P<0.05 as statistically significant. Kaplan-Meier survival analysis was used to calculate survival and log-rank test were used to compare survival curves. Point-biserial correlation was run to determine the relationship between the following variables: age, tumor size and symptomatic presentation.
Results: For the total patient population, the median age was 62 years (IQR, 57-69 years). The median tumor size was 10.0 cm (IQR 7.3-11.9 cm). 57.5% of patients were symptomatic at presentation with median age 61 years (IQR, 56-68 years), median tumor size 10.5 cm (IQR 8.4-13.0 cm), 73% male. For these patients, 11.5% had a pathologic stage T1-T2, and 88.5% had T3-T4. 42.5% of patients were asymptomatic with median age 63 years (IQR, 58-70 years), median tumor size was 9.0 cm (IQR 6.0-10.5 cm), 69% male. For these patients, 29.6% had a pathologic stage T1-T2, and 70.4% had T3-T4.
There was a positive correlation between tumor size and symptomatic presentation, which was statistically significant (rpb = 0.323, n = 167, p < .001). The asymptomatic patient group had an older median age. No statistically significant correlation between age and symptomatic presentation exists (rpb = -0.079, n = 167, p = 0.310).
The most common symptoms were: ipsilateral flank/ abdominal pain (60.6%), hematuria (48.5%), weight loss (37.1%) and fatigue (26.8%). Four patients had no follow-up after surgery, three of those due to patient death. 87.7% of patients had their pain resolved after CN. 100% of patients had their hematuria resolved after CN.
The mean overall survival (OS) for symptomatic patients was 36.0 months (95% CI, 29.6- 42.5 months). The mean OS for asymptomatic patients was 48.6 months (95% CI, 40.5- 56.7 months). There is significant evidence of a difference in OS for symptomatic and asymptomatic patients (p = 0.020).
Conclusions: Although recent research has shown that systemic therapy alone has comparable survival outcomes to CN with systemic therapy, CN may still have a role in reducing a patient’s symptomatic burden. Our findings suggest that among patients with mRCC, undergoing CN may improve the quality of life for those with larger, symptomatic primary tumors.
Figure 1.
Kaplan Meier Analysis for Overall Survival by Symptomatic Presentation
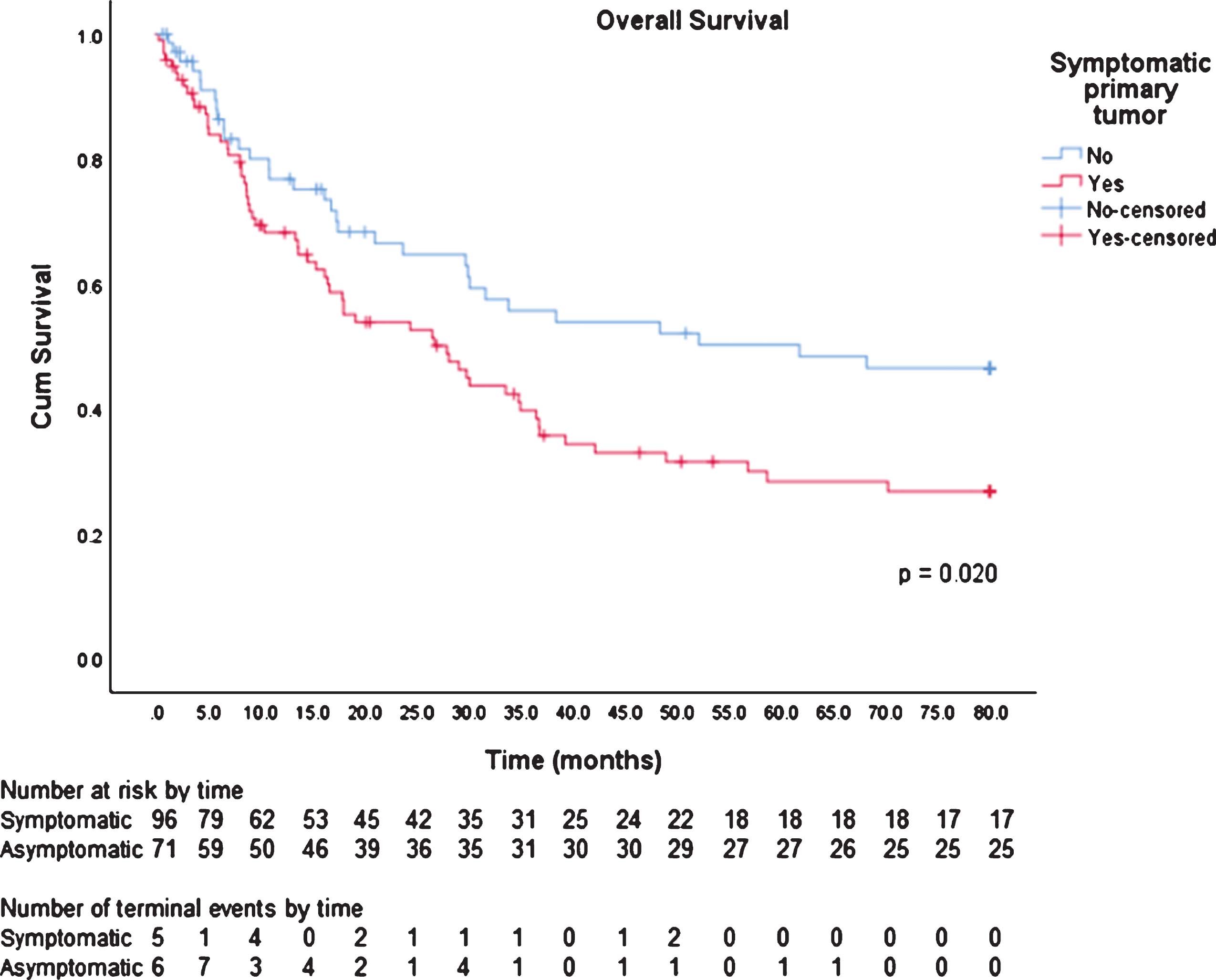
24Development and validation of a risk score based on patient characteristics to predict major complications after partial nephrectomy
First Author: Huynh, Melissa
Co-Authors: Wang, Ye; Krasnow, Ross; Chung, Benjamin; Chang, Steven
Authors Company: Brigham and Women’s Hospital, MedStar Health, Stanford University Medical Center
Introduction: Partial nephrectomy has become the gold standard in the treatment of clinical T1a renal masses. While tumour characteristics have been shown to correlate with complication rates in some studies, fewer studies have focused on the impact of patient comorbidities. We, therefore, sought to develop and validate a risk score to predict the probability of major complications following partial nephrectomy based on patient characteristics.
Methods: The Premier Healthcare Database (Premier Inc., Charlotte, NC, U.S.) was used to identify patients who had undergone elective partial nephrectomies. Through review of available ICD9 codes, we identified comorbidities and major surgical complications (Clavien grade 3–5). We used half of the set as the training cohort to develop our risk score and the other half as a validation cohort. Covariates with a p<0.20 in the univariate analysis were included as candidate variables in the multivariable logistic regression to identify predictors of major complications.
Results: From 2003–2015, 25 451 partial nephrectomies were performed. The overall rate of major complications for the whole cohort was 4.9%. The final risk score consisted of 10 predictors (Table 1) and stratifies patients into low-, intermediate-, high-, and very high-risk categories. In the training cohort, the area under the receiver-operator characteristic curve (AUC) was 0.75 (95% confidence interval [CI] 0.73–0.78) for major complications, while the AUC for the validation cohort was 0.73 (95% CI 0.70–0.75) (Fig. 1). The predicted probabilities of major complication in patients in the low-risk (=10 points), intermediate-risk (11–20 points), high-risk (21–30 points), and very high-risk (>30 points) categories were 3% (95% CI 2.6–3.2), 8% (95% CI 7.2–9.2), 24% (95% CI 20.5–27.8), and 41% (95% CI 34.5–47.8), respectively.
Table 1.
Scoring scheme for predicted risk of complications following partial nephrectomy based on patient characteristics
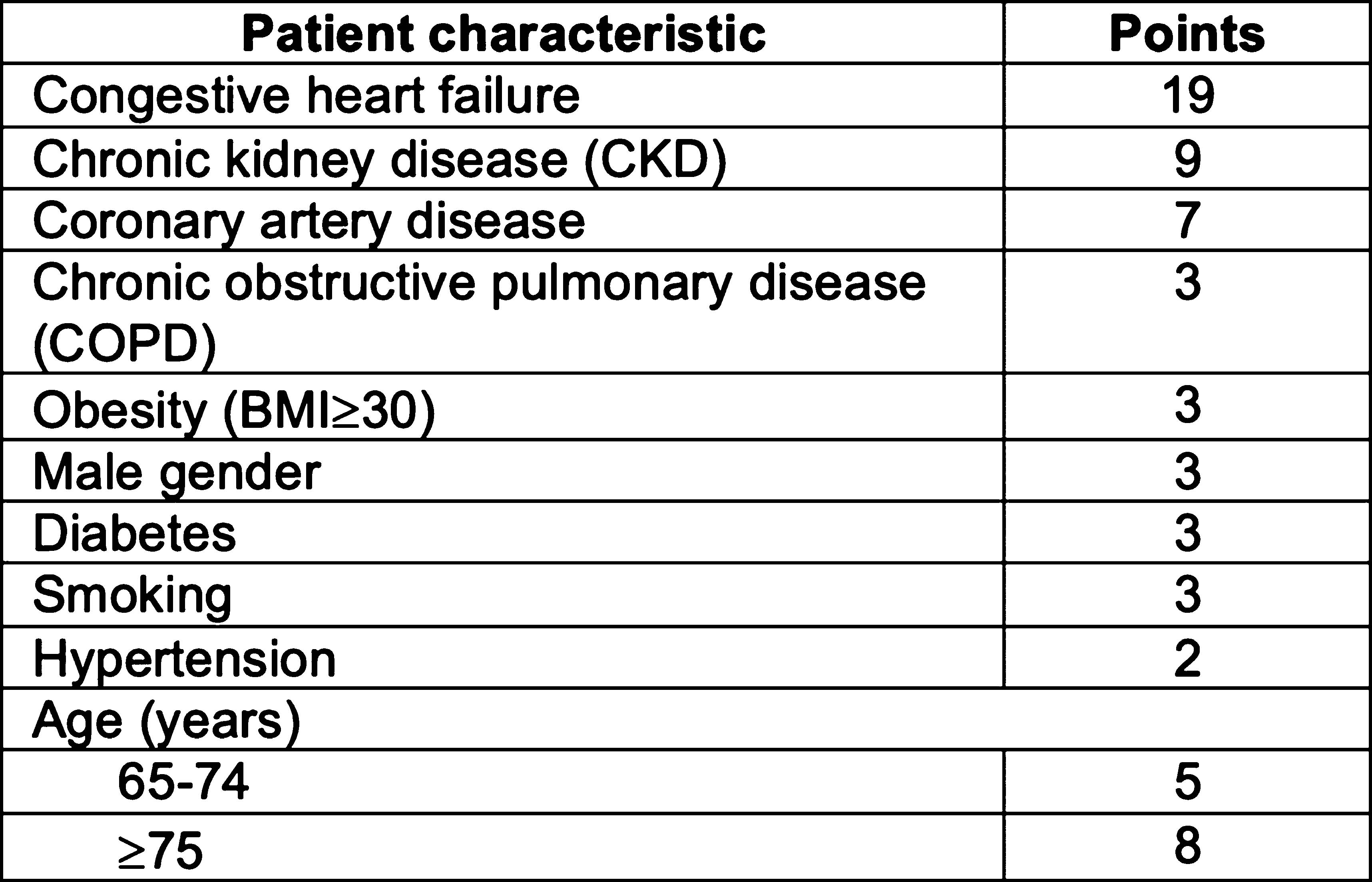 |
Figure 1.
Receiver-operator characteristic curve for the developed risk score in training and validation cohorts.
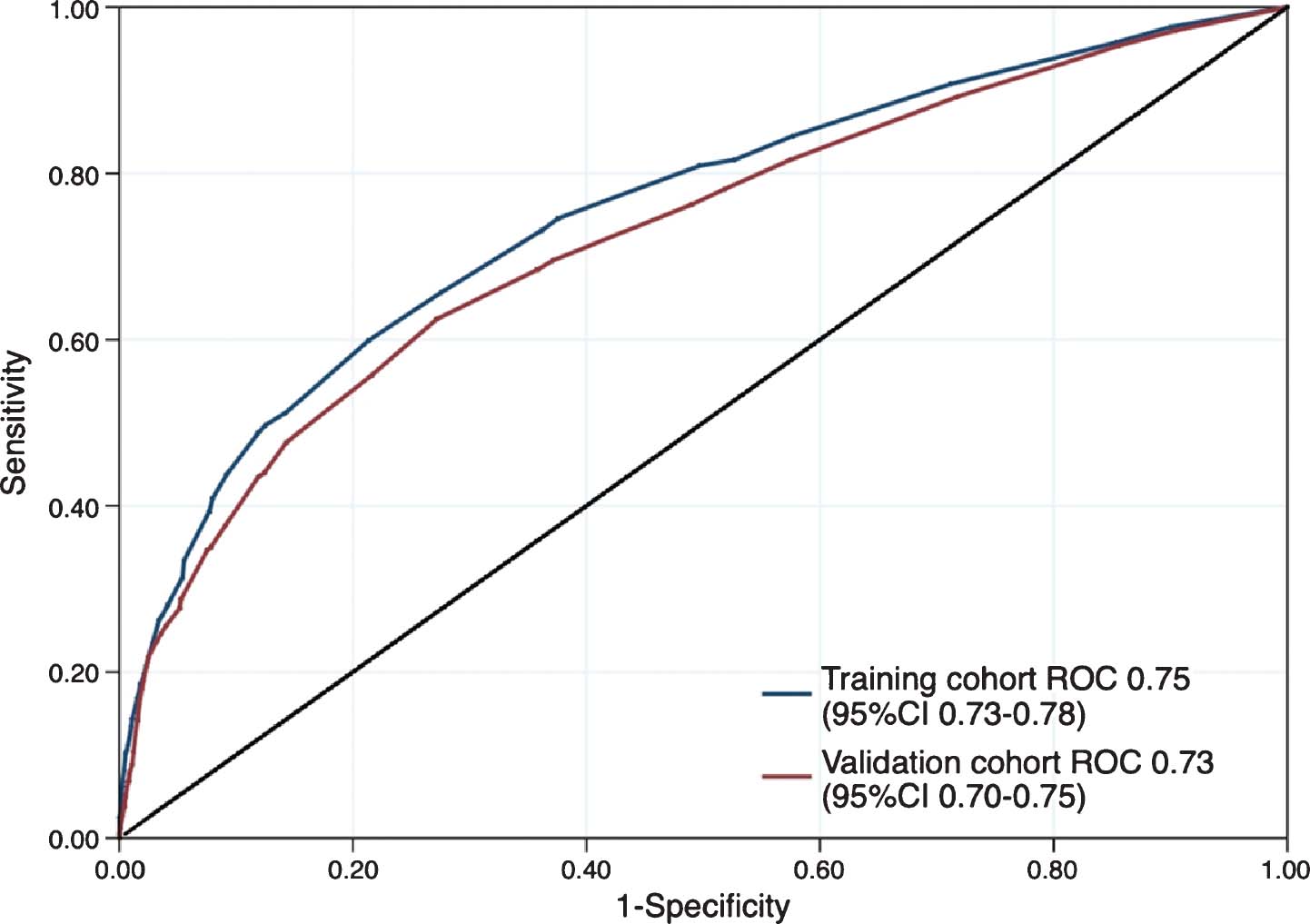
Conclusions: We developed and validated a risk score to predict the risk of complications following partial nephrectomy based on patient characteristics. Calculation of a risk score can enhance the informed consent process for those planning to undergo partial nephrectomy for the management of a renal mass.
25DNA hypomethylating agents activate ERV expression in kidney cancer to potentially enhance response to immunotherapies
First Author: de Cubas, Aguirre
Co-Authors: Gyan Bhanot, Shridar Ganesan, and W. Kimryn Rathmell
Authors Company: Vanderbilt University Medical Center
Abstract: Recently, we reported that expression of endogenous retroviruses (ERVs), a class of transposable element, is associated with response to immune checkpoint blockade (ICB) in renal cell carcinoma (RCC). Aberrant expression of ERVs can activate host antiviral responses, as well as produce neoantigens. ERV expression is repressed by DNA methylation and can be activated by DNA hypomethylating agents. Here, we investigate whether Decitabine, a DNA hypomethylating agent, can activate ERV expression and host antiviral defenses in RCC to potentially enhance response to ICB. Decitabine induced expression of ERV3-2 and ERV4700 in RCC cells lines, which was accompanied by activation of host antiviral defense genes and increased secretion of inflammatory cytokines. This effect was attenuated in non-cancerous transformed kidney cells. We validated this effect in patient-derived RCC cells. Bioinformatic analysis of RNAseq showed the Decitabine-induced gene signature could be associated with increased CD8 infiltration and response to ICB. Conditioned media from decitabine treated RCC cells was capable of inducing host anti-viral defense in naïve RCC cells and could modestly improve activation of T-cells from healthy donors. In a small retrospective cohort of metastatic RCC patients treated with single-agent PD-1/PD-L1 blockade, activation of some host antiviral defense genes was significantly higher in responders compared with nonresponders. Thus, modulation of ERV expression by Decitabine to activate host antiviral defenses could represent a novel therapeutic strategy to enhance RCC patient response to immune checkpoint blockade.
26Effects of immune checkpoint inhibitors on the morphology of renal cell carcinoma
First Author: Setoodeh, Sasan
Co-Authors: Elias, Roy; Brugarolas, James; Kapur, Payal
Authors Company: Univrsity of Texas Southwestern
Background: Immune checkpoint inhibitors (ICI), including the programmed cell death protein 1 (PD1) inhibitor, Nivolumab, alone or in combination with the cytotoxic T-lymphocyte-associated antigen 4 (CTLA4) inhibitor, Ipilimumab, are increasingly being used in advanced renal cell carcinoma (RCC). The goal of this study was aimed to evaluate their histologic effects. Design12 patients with post-ICI nephrectomy, including 8 conventional clear cell RCC (CCRCC), 1 sarcomatoid CCRCC, 1 papillary RCC (PRCC), 1 TFE-B rearranged RCC (TFE-B RCC), and 1 with complete response were selected from the University of Texas Southwestern Medical Center Kidney Cancer Program database and were compared to 10 random nephrectomy cases without neo-adjuvant therapy. Clinical characteristics were comparable to non-ICI patients. Viable tumors and non-neoplastic background kidney sections were evaluated for several histopathologic features as tabulated in table 2.
Table 2.
Histologic findings
 |
 |
Sarc: Sarcomatoid, L: lympohocytes, P: plasma cells, N: neutrophils, H: histiocytes
Results: Three patients had 12 or more cycles of ICI and had prior therapies (tyrosine kinase inhibitor or mTOR pathway inhibitor). 9 cases had only 1-2 cycle of ICI before nephrectomy. 7 cases had received only Nivolumab and 5 cases had received combination of Nivolumab and Ipilimumab. One case (case 1) showed complete response with no residual viable tumor. The most striking findings observed in CCRCCs were: 1) reduction/shrinkage of tumor cell cytoplasmic and nucleus (reminiscent of ISUP/WHO nucleolar grade 1), 2) prominent fibrin deposition in vessel walls with capillary endothelial hyperplasia, endothelialitis and fibrosis, 3) prominent fibrous scaring and collagenization suggesting therapy effect. These features were not observed in any of the ICI naive nephrectomies. PRCC and TFE-B RCC cases did not show the morphologic changes seen in most of CCRCCs. The sarcomatoid RCC case showed prominent dense fibrosis, inflammation, vascular fibrin deposition and endothelialitis. The most frequent findings in the non-neoplastic kidney were marked interstitial chronic inflammation, interstitial fibrosis and tubular atrophy, features not specific for response to ICIs.
Conclusion: This is the first study to show the effects of ICI on renal cell carcinoma histopathology. Interestingly, the most specific findings for ICI treated CCRCCs were vascular changes including fibrin deposition in vessel walls and capillary endothelial hyperplasia. These findings are particularly of interest considering the recent observation of better responses to ICI and VEGF inhibitors in combination. Due to the shrinkage in nuclear size, the tumors will likely be under graded post ICI therapies. Use of tumor histologic characteristics may help better patient selection for higher response rates.
27Efficacy of Nivolumab (NIVO) plus Ipilimumab (IPI) in metastatic Renal Cell Carcinoma (RCC) and brain metastases.
First Author: Gul, Anita
Co-Authors: Kimberly Allman, Moshe C. Ornstein, Allison Martin , Laura Wood, Jorge A. Garcia, Brian I. Rini
Authors Company: Cleveland Clinic Taussig Cancer Institute
Methods: Patients (pts) with metastatic RCC and brain metastases treated with NIVO+IPI were retrospectively reviewed. All pts had brain imaging (CT or MRI) at baseline per treating MD discretion. Baseline pt characteristics, outcome to therapy and adverse effects as per CTCAE v5.0 were collected from the electronic medical record.
Results: A total of 12 pts were identified (8 men and 4 women). The median age was 61 years (range, 54-67). Eleven pts had clear cell histology and one patient had chromophobe histology. Eight pts had nephrectomy before initiation of NIVO+IPI. Ten pts had ECOG PS 1, one had ECOG PS 2, and one pt had ECOG PS 3. IMDC risk group at the time of NIVO+IPI initiation was favorable risk (5 pts), intermediate risk (4 pts), and poor risk (3 pts). Eight pts received NIVO+IPI as 1st line therapy, two pts had NIVO+IPI as 2nd line treatment, and two pts (17%) had NIVO+IPI as 5th line treatment. Three pts presented with lower extremity weakness, two pts with seizures, one pts with anomic aphasia, one pt presented with dizziness, and five pts were asymptomatic at the time of initial presentation of brain metastases. Five pts had solitary brain metastasis and seven pts had multiple (range, 2-5) brain metastases at the time of initial presentation. The median size of metastatic brain lesion on initial presentation was 1 cm (range, 0.2-3.9). All pts received gamma knife surgery (GKS) for brain metastases. Eight pts (66%) had stable brain metastases and 4 pts (33%) developed new brain metastases outside of GKS fields during treatment with NIVO+IPI. Median time on NIVO+IPI was 4.5 months (range, 1-14). The overall survival (at the time of last follow up) since initiation of NIVO+IPI was 12 months (range, 2-14). Maintenance nivolumab was held in one pt due to asymptomatic grade 4 immune-mediated hepatotoxicity requiring steroids and mycophenolate mofetil.
Conclusions: NIVO+IPI is feasible and safe in pts with metastatic RCC and brain metastases.
28Efficacy of Systemic Treatment After Cabozantinib Failure in Metastatic Renal Cell Carcinoma
First Author: Di Nunno, Vincenzo
Co-Authors: Alves Costa Silva, Carolina; Benchimol, Axelle; Colomba, Emeline; Guida, Annalisa; Escudier, Bernard; Albiges, Laurence
Author Company: S. Orsola Malpighi - Sant’Orsola, Gustave Roussy Cancer Campus, University of Modena
Background: Cabozantinib is a potent multi-tyrosine kinases inhibitor, which has demonstrated overall survival (OS) benefit over everolimus in patients previously treated patients with VEGFR TKI for metastatic Renal Cell Carcinoma (mRCC). Cabozantinib is now considered as standard of care in VEGR –TKI and nivolumab/ICB failure. Few data are available after cabozantinib failure in advanced lines of the disease.
Methods: We conducted a retrospective analysis of patients who received systemic treatment after cabozantinib failure in late line of mRCC. We assessed progression free survival (PFS), OS and objective response rate (ORR) in patients who have received subsequent systemic therapy beyond cabozantinib. Long-rank test have been used.
Result: Of 150 patients treated with cabozantinib for mRCC in our institution, 43 (28.7%) received subsequent systemic therapy beyond cabozantinib and were selected for this analysis (Table 1). Subsequent line after Cabozantinib failure was administered in third (44.2%) and fourth line (25.6%). Axitinib was the subsequent treatment in 21 (48.8%) patients followed by immune-checkpoint inhibitors (34.8%), everolimus (34.8% ), other VEGF inhibitors (13.9% ) and other treatment (6.9%). Interestingly, 11 patients received 2 lines or more after cabozantinib (25.5%). PFS and OS from subsequent systemic therapy start after cabozantinib failure were 4 months (95%CI 2.8-5.2) and 7 months (95%CI 3.6-10.4), respectively (Table 2). Axitinib was the only drug in our series to provide significant clinical benefit.
Table 1
Baseline characteristics ( n=43) at start of subsequent Therapy
| Sex | M= 27 (62.8%) |
| F= 16 (37.2%) | |
| Pathology | ccrCC = 29 (67.4%) |
| nccRCC = 14 (32.6%) | |
| Number of prior line (including cabozaninib) | 1, n=1 (2.3%) |
| 2, n=19 (44.2%) | |
| 3, n=11 (25.6%) | |
| 4 or more, n=12 (27.9%) | |
| IMDC | Good = 8 (18.6%) |
| Intermediate = 15 (34.9%) | |
| Poor = 6 (14%) | |
| Missed= 14 (32.6%) | |
| Duration of CABOZANTINIB | <6mo = 16 (37.2%) |
| >6 mo = 27 (62.8%) |
Table 2
Results, n= 43 patients.
| Overall (n=60) | Axitinib (n=21) | Nivolumab (n=15) | mTOR (n=15) | Others (n=9) | |
| PFS | 4 months (95% CI 2.8 – 5.2) | 7 months (95%CI 3.7-10.2) | 3 months (95%CI 1.9-4.1) | 3 months (95%CI 1.6-4.4) | 3 months (95%CI 2.2-3.7) |
| OS | 7 months (95%CI 3.6 – 10.4) | 10 months (95% CI 2.1-17.9) | 7 months (95%CI 4.3-9.6) | 7 months (95% CI 5.2-8.8) | 4 months (95%CI 1.5-6.5) |
| RR | 2 (3.3%) | 1 (4.8%) | 1 (6.7%) | 0 (0%) | 0 (0%) |
| SD | 18 (30%) | 8 (38.09%) | 3 (20%) | 4 (26.6%) | 3 (33.3%) |
| PD | 27 (45%) | 7 (33.3 %) | 8 (53.4%) | 8 (53.4%) | 4 (44.4%) |
| Not Assessed | 13 (21.6%) | 5 (23.8%) | 3 (20.0%) | 3 (20%) | 2 (22.2%) |
Conclusion: Outcome of patients after cabozantinib failure in mRCC still remains poor. However, in our series, axitinib was associated with longer PFS, OS and better disease control rate and it may represent a treatment option in patients with previously treated metastatic RCC after cabozantinib failure.
29Exploratory Analysis of Adherence to Anticancer Therapies in Metastatic Renal Cell Carcinoma in the United States (US)
First Author: Kim, Ruth
Co-Authors: Bhanegaonkar, Abhijeet; Krulewicz, Stan; Polson, Michael; Kangethe, Anne
Authors Company: Pfizer, Inc., EMD Serono, Magellan Method
Objectives: With the increasing variety of treatments for metastatic renal cell carcinoma (mRCC), understanding patient adherence to various agents will help illustrate products with better adherence and potentially better outcomes. This study sought to determine patient adherence to medical treatment of mRCC.
Methods: This is a retrospective analysis of patients enrolled in commercial and Medicare plans covering the continental US using administrative claims from Magellan Rx incurred between 01/01/2013 – 06/30/2018. Patients were included if they were >18 years old on index date and had >1 medical claim with a diagnosis code for RCC, >2 medical claims for metastasis on or after initial RCC diagnosis date, evidence of any mRCC systemic therapy on or after initial mRCC diagnosis date, and continuous enrollment for >6 months prior to the index date until >6 months after the index date.
Results: Various therapeutic classes (TCs) of anticancer agents were used among 903 mRCC patients treated. There were differences in adherence between each selected therapeutic class, which included mechanistic target of rapamycin (mTOR) inhibitors (n=185), tyrosine kinase inhibitors (TKI) (n=409), immune-oncology (IO) agents (n=140), and others (n=324). Using proportion of days covered methodology, which assesses the number of days of supplied therapy for the first 6 months of therapy, resulted in adherence of 0.50 for mTOR, 0.61 for TKI, and 0.71 for IO. Days supply information was taken directly from submitted ambulatory pharmacy claims and inferred from the labeling information for products billed via the medical benefit. Patients who switched classes in the first 6 months were counted in each class.
Conclusion: Differences in adherence exist between TCs used for the treatment of mRCC, and IO showed a 10%-21% greater adherence than other TCs. Understanding patient adherence may help to guide choice in therapy toward agents with better real world adherence and potentially better outcomes.
30Fine Needle Aspiration Based Immune Organoids Recapitulate TME in ccRCC Cancer
First Author: Wolf, Melissa
Co-Authors: Brad I. Reinfeld, Matthew Z. Madden, W. Kim Rathmell, Vivian L. Weiss
Authors Company: Vanderbilt University
Abstract: Replication of the tumor microenvironment (TME) is a major limitation in organoid development as a model system for cancer research. Studies aimed at understanding the TME are typically performed in an oversimplified 2D cell culture system, or utilizing complexmouse models. Not only are in vivo mouse studies time consuming and costly, significant differences in the mouse immune system and TME may hinder the applicability of the results in a translational paradigm. Here, we show that human specimens obtained via fine needle aspiration (FNA) from cancerous clear cell renal cell (ccRCC) and thyroid tumors can grow and be maintained as immune organoids harboring a diverse lymphoid and myeloid population in a matrigel-based culture model. This system provides several advantages over cocultured cell lines with primary immune cells. In our FNA based method, the immune cell population is obtained directly from the TME, reflecting a realistic representation of tumor-associated exhaustive and/or regulatory immune cells. In addition, these studies provide a framework for future drug development and allow for the evaluation of a 3D organization of tumor cells with immune cells that could recapitulate physiological cancer processes. Ultimately, there is a translational gap between 2D cell culture and animal model testing; therefore, evaluation of the 3D organization of tumor cells within an ideal TME could recapitulate physiological cancer processes.
31Genomic sequencing of high-grade unclassified renal cell carcinoma allows molecular stratification and improves prognostication
First Author: DiNatale, Renzo G
Co-Authors: Marcon, Julian; Jayakumaran, Gowtham; Ghanaat, Mazyar; Kotecha, Ritesh; Makarov, Vladimir; Reznik, Ed; Ling, Lilan; Carlo, Marie; Brannon, Rose; Al-Ahmadie, Hikmat; Fine, Samson W; Gopalan, Anuradha; Sirintrapun, S Joseph; Tickoo, Satish K; Arcila, Maria E; Motzer, Robert J; Coleman, Jonathan; Russo, Paul; Chan, Timothy A; Reuter, Victor E; Hakimi, A Ari; Chen, Yingbei
Authors Company: MSKCC
Introduction: High grade unclassified renal cell carcinoma (uRCC) constitutes a heterogeneous group of non-clear cell kidney neoplasms with variable histopathologic features, disease course and aggressiveness. Due to the absence of well-defined parameters to inform prognosis, management of uRCC can be particularly challenging. Our group previously described distinctive molecular features that characterize these tumors, however, their prognostic significance has not been fully elucidated and a systematic approach to their assessment is still lacking. Therefore, we profiled an expanded high-grade uRCC cohort using next-generation sequencing (NGS) to identify potential genomic biomarkers. We aimed to leverage molecular features, genomic and clinical information to develop a combined prognostic model for this poorly-understood group of non-clear cell RCCs.
Methods: After IRB approval, we retrospectively identified 134 high-grade uRCC cases. All samples were reviewed by experienced genitourinary pathologists using the World Health Organization (WHO) criteria. A total of 160 samples (10 multiregional) were sequenced using our institutional NGS assay (MSK-IMPACT®). Mutations and copy-number (CN) aberrations were assessed using our previously-validated pipeline. Additionally, we leveraged mutation data, tumor purity and CN at each locus to estimate the number of cancer cells bearing each mutation (cancer-cell fraction). Clonal variants deemed to be oncogenic (according to OncoKB®) were grouped by the molecular pathway affected (10 pathways total). The primary outcome was overall survival and the Kaplan-Meier method was used to compute estimates. Cox regressions were used to assess potential survival predictors. Finally, a multivariable analysis including clinical, molecular and genomic data was used to produce a combined prognostic model.
Results: The overall mutational frequencies wereconsistent with our previous report. Of the 134 cases, 22 were identified as newly-defined or emerging WHO entities. The integrative pathway analysis was performed in the remaining 112 cases, and supported the presence of four major molecular subsets according to the pathway affected: HIPPO/NF2, PI3K/MTOR, TP53/Cell-Cycle/DNA damage repair and chromatin remodeling (Figure 1a). Notably, HIPPO (NF2) pathway alterations were found to be mutually-exclusive with PI3K (MTOR) alterations (p<0.001) and to co-occur with aberrations in chromatin-remodeling genes (p=0.01). These molecular subsets were also found to have different levels of genomic instability (p=0.01), with the PI3K/MTOR representing the lowest-end of the spectrum. Tumors with PI3K/MTOR alterations had better prognosis (HR:0.38 [0.15-0.97], p=0.04); whereas those with HIPPO or DDR/TP53/RB1 alterations were associated with significantly-worse OS (HR:2.6, p=0.004 and HR:2.9, p=0.004, respectively). Additionally, a higher number of clonal oncogenic mutations was also associated with poor prognosis (HR:1.4 [1.2-1.6], p<0.001). On multivariable analysis (n=104), both molecular and genomic features were found to have independent prognostic value (Figure 1b).
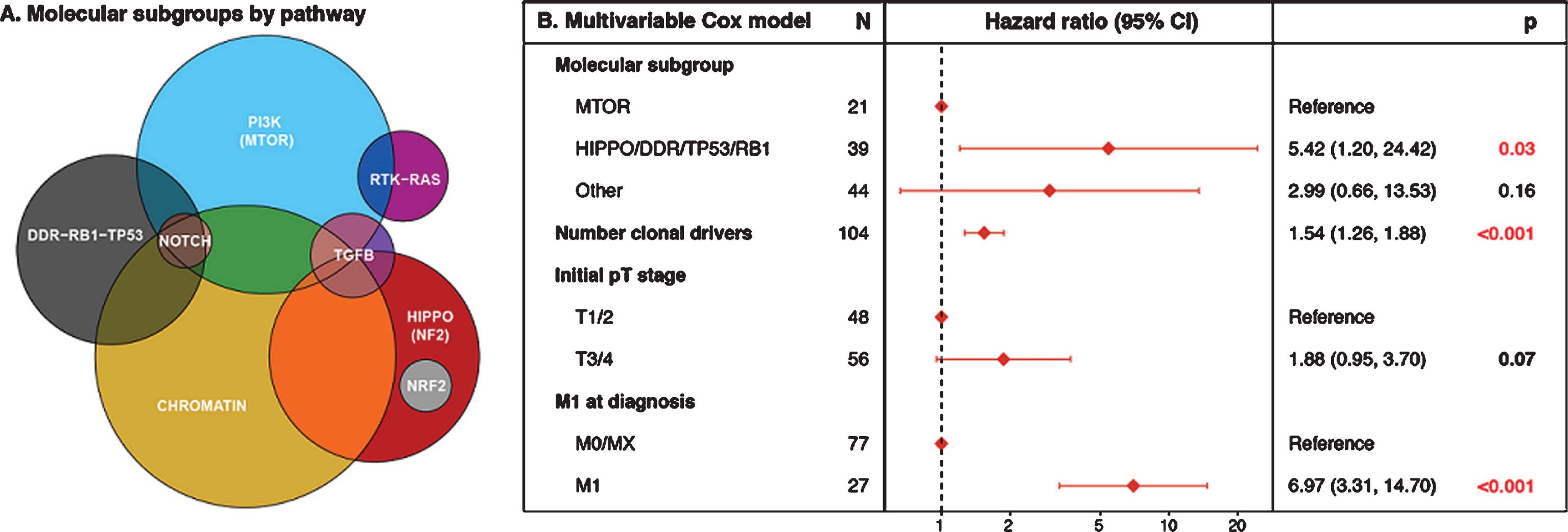
Conclusion: High grade uRCC can be defined by distinct molecular subtypes with prognostic value independent of currently-used clinical parameters. NGS assays can provide additional predictive ability as well as a standardized way to assess the tumor biology.
32Identification of Oncocytic Neoplasms with Cyclin D1 Immunohistochemistry
First Author: Mendhiratta, Neil
Co-Authors: Adebowale Adeniran , Jamil Syed, Tyler Valdez, Dipti Sajed, Peter Humphrey, Sung-Hae Kang, Huihui Ye, Brian Shuch
Authors Company: UCLA School of Medicine, Yale
Background: Renal oncocytoma and chromophobe renal cell carcinoma (chRCC) are difficult to distinguish on biopsy. Due to this concern, biopsies are not often performed and most patients and proceed to surgery with ultimately 20% of small renal masses found to be benign. Recent investigation has identified a subset of oncocytomas characterized by CCND1 rearrangement with overexpression of Cyclin D1, a cell cycle regulator and oncoprotein. We hypothesized that immunohistochemistry (IHC) staining for Cyclin D1 would be a highly specific marker of oncocytoma to be used in conjunction with a renal mass biopsy.
Method: We retrospectively identified 70 consecutive oncocytic renal tumors resected from 2000-2010 at Yale New Haven Hospital. All cases were reviewed by two expert genitourinary pathologists (PH/AA). A tissue microarray was constructed with 3 punches from identified tumor samples. IHC staining was performed on the tissue microarray with Cyclin D1 antibody (1:250, ab6152, Abcam, Cambridge, MA). IHC results were evaluated by a separate blinded expert genitourinary pathologist (HY) to determine presence of Cyclin D1 overexpression, which was then compared to the original histopathologic classification (Figure 1).
Figure 1.
Examples of immunohistochemistry staining of punched tissue specimen with the Cyclin D1 antibody demonstrating (A) normal expression, (B) overexpression, (C) normal expression, and (D) overexpression of Cyclin D1.
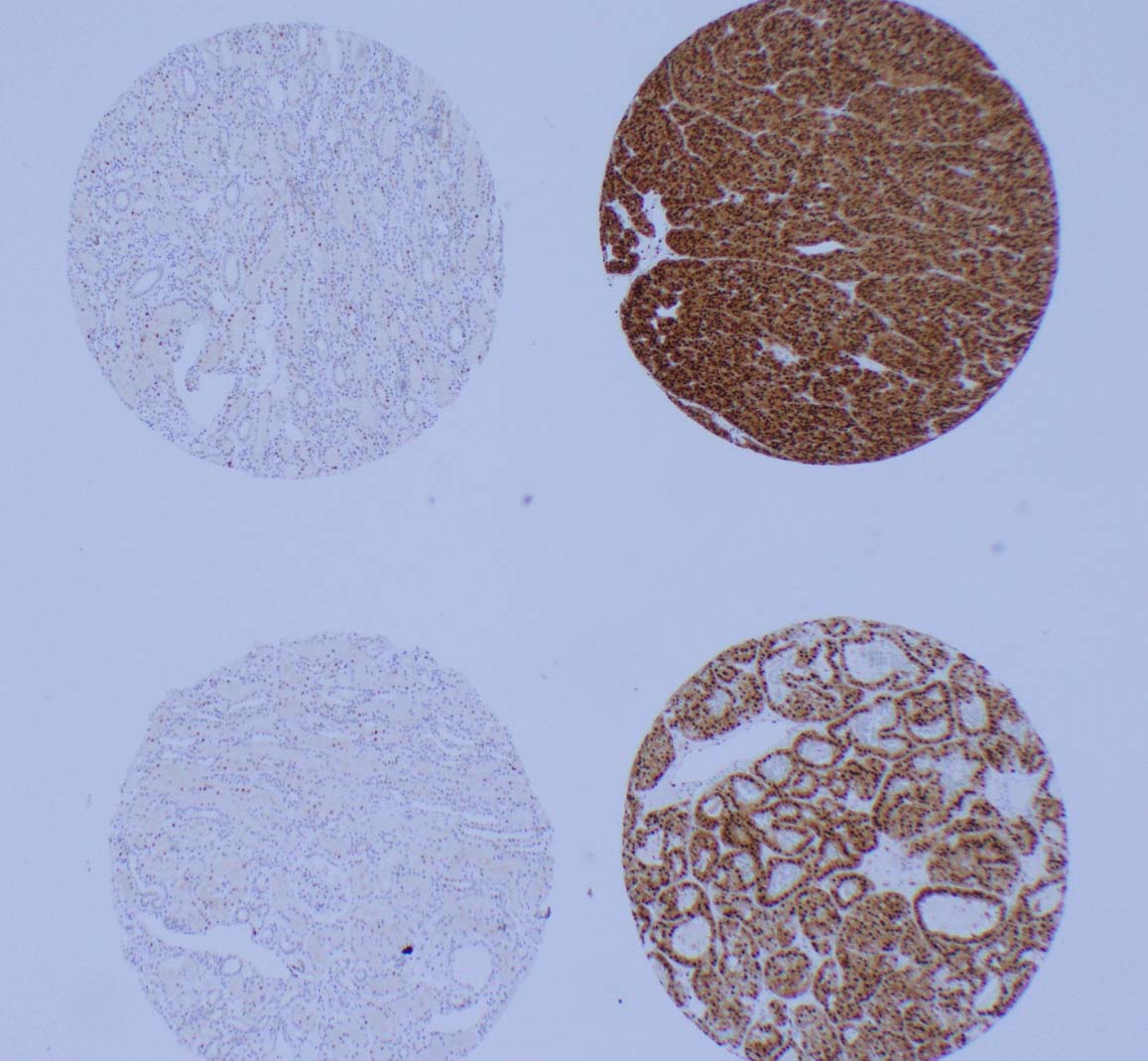
Results: A total of 38 oncocytomas and 32 chRCC tumors were identified. For the oncocytoma and chRCC cohorts, the mean age was 63.9 years vs 58.0 years (p = 0.053) and mean tumor size was 4.3cm vs 5.3cm (p = 0.246). Overexpression of Cyclin D1 was identified in 16 of 38 (42%) oncocytomas and 1 of 32 (3%) of chRCC tumors. Cytoplasmic and nuclear expression was fairly uniform through the sample (Figure 1). The sensitivity for Cyclin D1 IHC staining for oncocytoma was 42.1% and specificity was 96.9%.
Conclusions: Cyclin D1 IHC staining is a highly specific test to distinguish oncocytoma from morphologically similar chromophobe renal cell carcinoma. Clinically this technique may be applied to oncocytic tumors identified on biopsy to identify the subset of oncocytomas characterized by this alteration which may reduce the amount of unnecessary surgery performed for indolent disease.
33Immunologic correlates of previously-treated metastatic renal cell carcinoma (mRCC) patients (pts) treated on a phase II trial of intermittent nivolumab therapy
First Author: Ornstein, Moshe C.
Co-Authors: Diaz-Montero, Claudia; Rayman, Patricia A.; Wood, Laura S.; Wei, Wei; Allman, Kimberly; Martin, Allison; Garcia, Jorge A.; Gilligan, Timothy D.; Rini, Brian I.
Author Company: Cleveland Clinic Taussig Cancer Institute
Background: A recent prospective phase II clinical trial demonstrated that intermittent nivolumab (Nivo) monotherapy in previously-treated mRCC patients (pts) is feasible and does not compromise clinical outcomes (Ornstein MC JITC 2019; NCT03126331). Data regarding serial blood-based immunologic correlates of patients on this trial are presented.
Methods: Pts enrolled on this trial were consented for serial blood draws at all staging time points. MDSCs were enumerated in fresh unfractionated blood (WB) and in peripheral blood mononuclear cells (PBMC). Total (T-) myeloid derived suppressor cells (MDSC) were defined as CD33+HLADR-, of which the subsets of monocytic (M)-MDSC (CD14+CD15-), polymorphonuclear (PMN)-MDSC (CD14-CD15+), and immature (I)-MDSC (CD14-CD15-) were isolated. MDSC populations were presented as % of live nucleated blood cells and as absolute numbers from WB. PD1, PDL1, and V-domain Ig suppressor of T cell activation (VISTA) expression on MDSCs and cytotoxic T lymphocytes (CTL; CD8+CD4-) from PBMC was quantified by flow cytometry. MDSCs were summarized by time point and intermittent eligibility status, and compared using Wilcoxon rank sum test.
Results: Fourteen pts were enrolled; 13 (93%) male, median age 65. Five (36%) pts met the criteria (=10% decrease in tumor burden) for the treatment free interval (TFI) phase. With a median f/u of 48 wks, one pt restarted therapy, and the other four pts have a sustained response for a median of 34 weeks (range, 16–53) off therapy. The most frequent pre-treatment WB and PBMC MDSC subsets in all patients was PMN-MDSC (67% and 83%, respectively). Pre-treatment PD-1, PD-L1, and VISTA expression was highest on M-MDSCs, while PD-L1 expression was highest on PMN-MDSC. The five pts eligible for a TFI had significantly higher WB M-MDSC (median 0.0046 vs. 0.0011, p=0.048), higher % of PD1+ PB PMN MDSCs (median 4.8 vs. 3.4, p=0.02), and higher % of VISTA+ PB UC MDSCs (median 17.2 vs. 8.1, p=0.03) than those of ineligible patients at the prior to their TFI. There were no statistically significant changes in immune parameters in while in the TFI.
Conclusion: In this small prospective clinical trial of intermittent nivolumab in mRCC, higher WB M-MDSC, % of PD1+ PB PMN MDSCs, and % of VISTA+ PB UC MDSCs was noted in patients who met pre-specified criteria for a TFI. Further investigation in larger cohorts and longer follow-up is necessary to develop biomarkers for identification of patients who can benefit from prolonged TFI. Additional analyses for this trial are ongoing and will be updated for the conference.
34Improved response rate of Cabozantinib after immune checkpoint therapy in patients with metastatic Renal Cell Carcinoma
First Author: Alves Costa Silva, Carolina
Co-Authors: Le Teuff, Gwénaël; Hirsch, Laure; Benchimol-Zouari, Axelle; Guida, Annalisa; Colomba, Emeline; Derosa, Lisa; Escudier, Bernard; Albiges Laurence
Authors Company: Gustave Roussy Institute, University of Modena, Institut National de la Santé Et de la Recherche Médicale
Background: Cabozantinib (CABO), a potent tyrosine kinase inhibitor (TKI), was approved for the treatment of metastatic Renal Cell Carcinoma (mRCC) patients. The results from retrospective cohorts suggest improved efficacy of CABO after progressive disease (PD) to immune checkpoint therapy (ICT).
Methods: Retrospective unicentric study of consecutive patients treated with CABO for mRCC from January 2014 to February 2019. Computed tomography images were centrally reviewed by a trained radiologist for RECIST evaluations. Clinical data were collected from IGRECC (Institut Gustave Roussy REnal Cell Carcinoma) database and medical record. Overall response rate (ORR), defined as XX, was measured and a logistic regression was used to evaluate the effect of IMDC (poor, intermediate, good), histology (clear cell, non-clear cell), CABO line and previous CABO treatment (ICT versus TKI and others). Kaplan Meier estimation was used to estimate median progression-free survival (mPFS) and median overall survival (mOS) from CABO start.
Results: Ninety-nine patients with a median age of 61 (range 22-78) years and a median-follow-up of 16 months from CABO start were included. Forty-three patients received ICT and 56 received TKI or others as previous line just before CABO. Table 1 describes the patients’ characteristics by type of previous treatment and overall. In ICT group, 37 patients had cc and, for those, ORR was 54% and 5.4% PD. For the 6 patients with non-cc, ORR was 16.6% and 16.6% had PD. In TKI and others group, 34 patients had cc for which ORR was 26.5% and 5.9% PD and 22 patients had non-cc with ORR of 13.6% and 18.2% PD. ORR was significantly higher when CABO was given after ICT versus TKI and others (OR 3.614 95%CI 1.158-11.277, p=0.0269). Cc histology had more benefit than non-cc (OR 3.091, 95%CI 0.877-10.898, p=0.0791) from CABO post ICT. Overall, mPFS was 9.27 months (95%CI 8.572-11; 79.8% events) and mOS was 20.35 months (95%CI 13.98-17.31; 49.5% deaths) (Figure 1, left). Both endpoints were not associated to previous treatment before Cabozantinib start (Figure 1, right).
Table 1
Patients’ characteristics
| Groups Characteristics | Cabozantinib post-ICT (N=43) | Cabozantinib post-TKI (N=56) | Overall (N=99) |
| Histology | |||
| Clear cell (cc) | 37 (86%) | 34 (60.7%) | 71 (71.7%) |
| Non-cc | 6 (14%) | 22 (39.3%) | 28 (28.3%) |
| IMDC risk group | |||
| Good | 7 (16.3%) | 5 (9%) | 12 (12.2%) |
| Intermediate | 29 (67.4%) | 33 (60%) | 62 (63.3%) |
| Poor | 7 (16.3%) | 17 (31%) | 24 (24.5%) |
| Missing | 1 | 1 | |
| Number of previous lines | |||
| 2nd | 2 (4.6%) | 30 (53.6%) | 32 (32.3%) |
| 3rd | 19 (44.2%) | 8 (14.3%) | 27 (27.3%) |
| ≥4th line | 22 (51.2%) | 18 (32.1%) | 40 (40.4%) |
| ORR | 21 (48.8%) | 12 (21.4%) | 33 (33.3%) |
| SD | 19 (44.2%) | 38 (67.9%) | 57 (57.6%) |
| PD | 3 (7%) | 6 (10.7%) | 9 (9.1%) |
Conclusion: Our data reinforces that Cabozantinib display higher response rate when given after ICT, especially for ccRCC.
35Incidence of Occult Brain Metastases Detected at Study Screening in Patients with Advanced Renal Cell Carcinoma
First Author: Kotecha, Ritesh
Co-Authors: Flippot, Ronan; Nortman, Taylor; Guida, Annalisa; Foster, Ashley; Escudier, Bernard; Motzer, Robert; Albiges, Laurence; Voss, Martin
Authors Company: Memorial Sloan Kettering Cancer Center, Gustave Roussy
Ritesh R. Kotecha1, Ronan Flippot2, Taylor Nortman1, Annalisa Guida2,3, Ashley Foster1, Bernard Escudier2, Robert J. Motzer1, Laurence Albiges2, Martin H. Voss1
Affiliations: 1Genitourinary Oncology Service, Memorial Sloan Kettering Cancer Center, New York, USA, 2Department of Cancer Medicine, Gustave Roussy, Paris Sacaly University, Villejuif, France, 3Department of Oncology and Hematology, University of Modena and Reggio Emilia, Modena, Italy
Background: Current guidelines for metastatic renal cell carcinoma (RCC) management only recommend brain imaging if clinically indicated, and the rate of occult brain metastases is unknown. Early detection of brain metastases may allow early intervention to limit morbidity associated with brain progression. We investigated the rate of occult brain metastasis in a large international cohort of metastatic RCC patients.
Methods: A retrospective review of patients screened for completed and actively accruing metastatic RCC clinical trials at Gustave Roussy and Memorial Sloan Kettering Cancer Center was performed. Asymptomatic patients identified at screening with mandatory brain imaging were included for analysis, and patient demographics, clinical course and treatment history were collected. Patients with neurologic symptoms identified by chart review were excluded, and descriptive statistics were applied to analyze findings across the cohort.
Results: Across 68 clinical trials which required baseline brain imaging conducted in patients with metastatic RCC between 2001-2019, 72/1597 (4.6%) of patients screened were found to have occult brain metastasis. Of these individual patients, the median age at diagnosis of metastatic disease was 56.1 years, available IMDC risk at enrollment was favorable (26%), intermediate (61%), poor (13%), and 43/72 (60%) presented with de-novo metastatic disease. 32% of patients were un-treated, 43% were screening for second-line therapy, and 25% had received more than 2 lines of therapy. 14% of patients had only one other site of metastatic disease, while 86% of patients had 2 or more sites of extra-cranial disease. Brain metastases were multi-focal in 38% of patients, sub-centimetric in 55% of patients, and had a median size of 1 cm at largest lesion diameter (range 0.3-5.1 cm). From available follow-up, 57/61 (93%) underwent brain directed treatment with either surgery (10%), radiation (80%) or both (3%), with a median time to treatment of 24 days (R: 4-153). Median overall survival by Kaplan-Meier method from metastatic disease diagnosis and from brain metastasis diagnosis was 35.9 months (IQR: 19.1-73.5), and 11.3 months, (IQR: 4.85-30.3) respectively.
Conclusions: This large, international, multi-institutional cohort identifies that the rate of occult brain metastasis occurs in approximately 5% of advanced RCC patients. These findings may inform standardized screening guidelines for disease assessment and allow for early intervention for asymptomatic patients.
36Interim results of a phase 2 study of sapanisertib in patients with previously treated advanced renal cell carcinoma
First Author: McKay, Rana
Co-Authors: Lillian Werner, Walter M. Stadler, Yousef Zakharia, Ajjai Alva, M. Dror Michaelson, Elaine T. Lam, Shilpa Gupta, Amin Nassar, David J. Kwiatkowski, Bradley A. McGregor, Toni K. Choueiri, Lauren C. Harshman
Authors Company: University of California San Diego, Dana-Farber Cancer Institute, University of Chicago, University of Iowa, University of Michigan, Massachusetts General Hospital, University of Colorado, University of Minnesota, Brigham and Women’s Hospital
Background: The mammalian target of rapamycin (mTOR) pathway regulates tumor cell proliferation, angiogenesis, and metabolism, providing rationale for targeting this pathway in renal cell carcinoma (RCC). The approved rapalogs, everolimus and temsirolimus, are allosteric inhibitors of mTOR complex (mTORC) 1 and cause only partial mTORC1 inhibition. The clinical benefit of these two rapalogs is modest with only a two month improvement in progression-free survival over placebo. Sapanisertib (TAK-228) is a novel, highly selective, ATP-competitive inhibitor of both mTORC1 and mTOR2, developed to address the incomplete inhibition by rapalogs.
Methods: We performed a phase 2, open-label, single-arm two-stage study of sapanisertib in patients with refractory metastatic RCC. Patients with clear cell histology must have progressed or been intolerant to at least one anti-angiogenic agent and at least one programmed death-1 pathway blocking agent unless contraindicated. Patients with non-clear cell histology must have progressed on at least one anti-cancer therapy. Prior therapy with rapalogs was allowed. Eligible patients were treated with sapanisertib 30 mg orally once per week and continued treatment until radiographic progression, toxicity or withdrawal. The primary endpoint was objective response rate (ORR) as assessed by Response Evaluation Criteria In Solid Tumors version 1.1. Target enrollment was 40 patients, with an interim analysis to assess safety and futility after the first 20 patients based on a Simon design with null hypothesized ORR of 5% and alternative of 20%, and a of 0.05 with 90% power. Secondary endpoints included progression-free survival, overall survival, safety, and biomarkers of response and resistance.
Results: We present results from the interim analysis. Most patients had clear cell histology (n=14) and five patients had sarcomatoid differentiation. Five patients had received a prior mTOR inhibitor. The most common treatment-related toxicities were nausea (n=10), vomiting (n=9), fatigue (n=8), mucositis (n=7), anorexia (n=4), diarrhea (n=4), and hyperglycemia (n=4). Seven patients required a dose modification. There were no grade 4 or 5 adverse events. There was one confirmed partial response in a patient with clear cell histology (duration of response 5.5 months), and thus the study proceeded to the second stage. Table 1 denotes response by histology and prior mTOR status. The median progression-free survival for the cohort was 2.3 months (95% CI 1.7-5.4 months).
Table 1.
| Clear Cell | Non-Clear Cell | No Prior mTOR | Prior MTOR | |
| N | N | N | N | |
| Partial Response | 1 | 0 | 0 | 1 |
| Stable Disease | 5 | 1 | 4 | 2 |
| Progressive Disease | 6 | 2 | 7 | 1 |
| Unknown | 2 | 3 | 4 | 1 |
Conclusions: This interim analysis demonstrates that the toxicity of sapanisertib was manageable and anti-tumor activity was worthy of further exploration. Currently, 34/40 patients have been enrolled. Biomarker analyses profiling the mTOR pathway to identify predictors of response to therapy are ongoing.
37Is anesthesia associated with oncological outcomes after nephrectomy?
First Author: Silagy, Andrew
Co-Authors: Hannum, Margaret; Mano, Roy; Attalla, Kyrollis; DiNatale, Renzo; Marcon, Julian; Tan, Kay See; Coleman, Jonathan; Russo, Paul; Fischer, Gregory; McCormick, Patrick; Hakimi, A Ari; Mincer, Joshua
Authors Company: Memorial Sloan Kettering Cancer Center
Background: Emerging data suggests that intraoperative opioids may impact oncological outcomes in various malignancies, however this association has not been tested in kidney cancer. We aimed to determine whether there is an association between anesthesia and cancer-specific outcomes for kidney cancer.
Methods: We retrospectively reviewed 2981 adult patients with malignant histology treated at our institution with a partial or radical nephrectomy. Total intraoperative morphine milligram equivalent (MME) dosage of opioids and the administration of ketamine, as a binary variable, were determined from the anesthetic charts. Baseline patient characteristics and postoperative follow up were pulled from our prospectively maintained nephrectomy database. We performed univariate and multivariate Cox regression (adjusting for TNM stage, histology and tumor size) to look for associations between opioid exposure and ketamine administration with recurrence-free survival (RFS) for localized patients, and cancer-specific survival (CSS) and overall survival (OS) for the entire cohort.
Results: The cohort consisted of 2775 localized and 206 metastatic patients, with a median age of 60 (52, 68). There were 188 patients who received ketamine, these patients received significantly less intraoperative opioids (60 vs 70 MME, respectively) and were higher TNM stage. There were 152 recurrences and 286 deaths during the follow up period. An increase of 10 MME (100mcg of Fentanyl) was associated with adverse RFS (HR 1.06, 1.03 – 1.10, p<0.001), CSS (HR 1.09, 1.06 – 1.13, p<0.001) and OS (HR 1.07, 1.04 – 1.09, p<0.001) on univariate analysis, but this was not significant on multivariate analysis. Conversely, ketamine administration was associated with improved RFS on multivariate analysis (HR 0.28, 0.11 – 0.70, p=0.007) (Table 1), but not CSS and OS.
Table 1.
RFS Adjusted Cox Proportional Hazard1
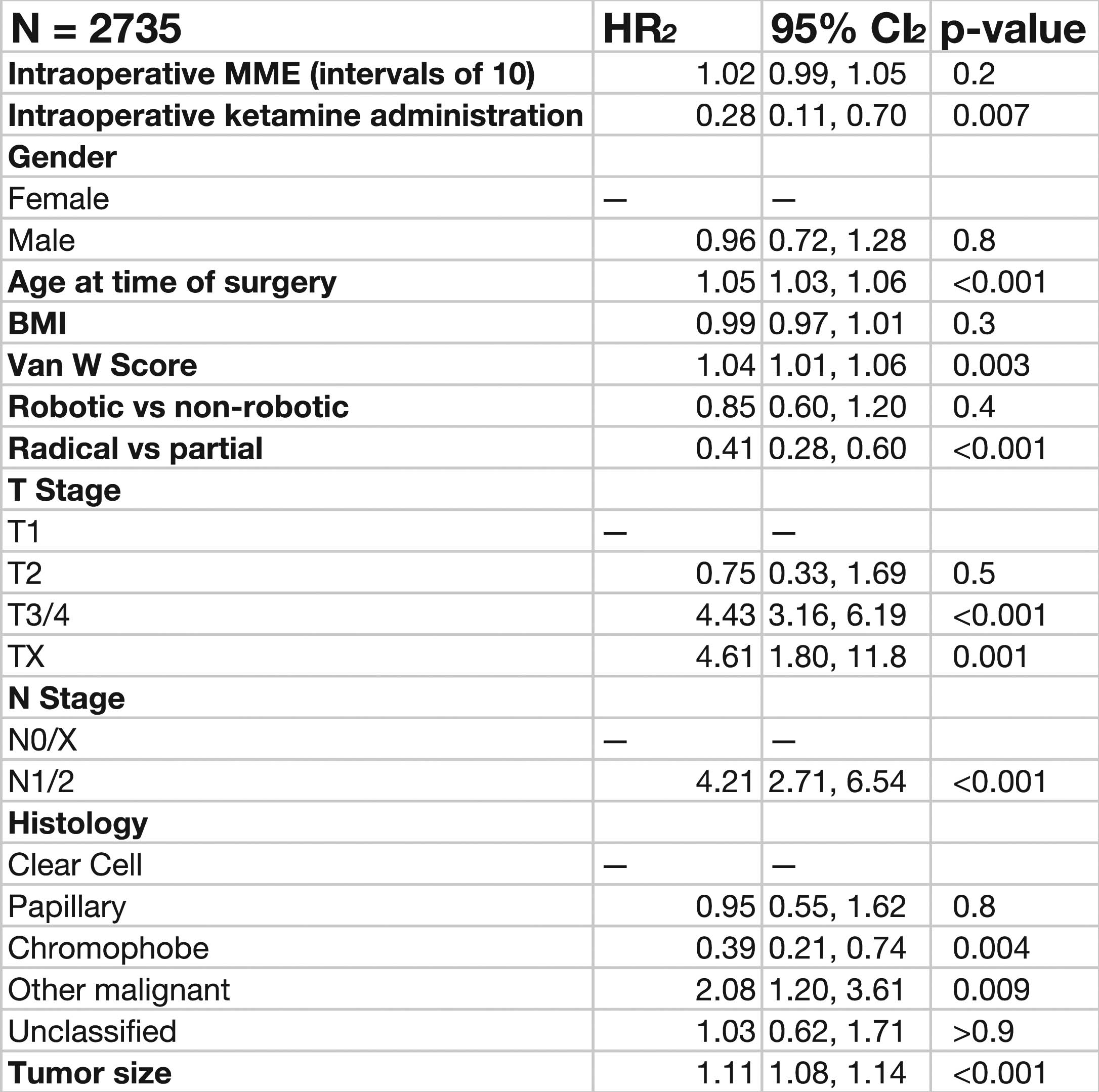 |
Conclusions: Preliminary results suggest that ketamine and opioid administration may impact oncological outcomes in patients with kidney cancer. Further research with subgroup analyses based on histology and exploration of other anesthetic agents is currently being undertaken.
38Local and Regional Recurrences of Clinically Localized Renal Cell Carcinoma: A 15 Year Institutional Experience with Prognostic Features and Oncologic Outcomes
First Author: Ray, Shagnik
Co-Authors: Cheaib, Joseph; Pineault, Kevin; Biles, Michael; Pierorazio, Phillip
Authors Company: The James Buchanan Brady Urological Institute, The Johns Hopkins University School of Medicine, Baltimore, MD
Introduction and Objectives: Local recurrence (LR) of clinically localized (cT1/cT2) renal cell carcinoma (RCC) without concurrent systemic metastasis occurs rarely (1-5%) after surgical treatment. LR may be a harbinger of poor outcome and the best management of these patients is unclear. The purpose of our study was to evaluate outcomes for patients with LR.
Methods: We retrospectively reviewed patients surgically treated for clinically localized RCC with subsequent LR (in the partial or radical nephrectomy bed) and/or regional recurrence (RR; in the abdomen distant from the direct site of surgery)without concurrent metastasis from our institutional RCC database (2004-2018). Patient outcomes were analyzed based on recurrence management type.
Results: Out of 2953 total patients, 1962 had clinically localized RCC, with 25 patients (1.29%) identified as having isolated LR/RR. Median time to recurrence was 24 months (IQR:16-35). For the 21 patients treated surgically, 13 (52%) recurred over a median follow-up time of 26 months (IQR:18-45) from the first recurrence’s management date. The 1-year and 2-year secondary recurrence-free survival are 59% and 45% respectively. =2 sites of locoregional recurrence had a hazard of 4.34(p=0.030) for secondary recurrence/metastasis after surgical management for local recurrence.
Conclusions: Our results suggest that aggressive surgical management for patients who are surgical candidates with surgically resectable disease can be appropriate, with 38% of such patients remaining alive without any recurrence of disease. Additionally, the number of sites of LR and/or RR can be used to better select patients that will benefit from surgery or systemic versus combination therapy. This work provides a framework onto which further studies regarding surgery and LR and/or RR of clinically localized RCC can be performed.
39Long-Term Follow-Up of Nivolumab Versus Everolimus in Patients With Advanced Renal Cell Carcinoma (aRCC): The Phase 3 CheckMate 025 Trial
First Author: Motzer, Robert
Co-Authors: Tykodi, Scott; Escudier, Bernard; Carducci, Michael; Oudard, Stéphane; Hammers, Hans; McDermott, David; George, Saby; Castellano, Daniel; Sosman, Jeffrey; Choueiri, Toni; Alva, Ajjai; Richardet, Martin; Chevreau, Christine; Plimack, Elizabeth; Srinivas, Sandhya; Procopio, Giuseppe; Donskov, Frede; Gurney, Howard; Peltola, Katriina; Wagstaff, John; Fukasawa, Satoshi; Tomita, Yoshihiko; Gauler, Thomas; Schutz, Fabio; Kollmannsberger, Christian; Larkin, James; Cella, David; McHenry, Brent; Maurer, Matthew; Tannir, Nizar
Authors Company: Memorial Sloan Kettering Cancer Center, University of Washington and Fred Hutchinson Cancer Research Center, Gustave Roussy, Johns Hopkins Medicine, Sidney Kimmel Comprehensive Cancer Center, Hôpital Européen Georges Pompidou, AP-HP, UT Southwestern Kidney Cancer Program, Beth Israel Deaconess Medical Center, Beth Israel Deaconess Medical Center, Dana-Farber/Harvard Cancer Center, Roswell Park Cancer Institute, Oncologia Medica, Hospital Universitario, Northwestern University Medical Center, University of Michigan, Fundacion Richardet Longo, Instituto Oncologico de Cordoba, IUCT-O Institut Claudius Regaud, Fox Chase Cancer Center, Stanford University Medical Center, Fondazione Istituto Nazionale Tumori, Aarhus University Hospital, Westmead Hospital and Macquarie University, Helsinki University Hospital, South West Wales Cancer Institute and Swansea University College of Medicine, Chiba Cancer Center, Niigata University Graduate School of Medical and Dental Sciences, University Hospital Essen of University of Duisburg, Beneficencia Portuguesa de São Paulo, Vancouver Cancer Centre, Royal Marsden Hospital NHS Foundation Trust, Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Feinberg School of Medicine, Bristol-Myers Squibb, MD Anderson Cancer Center
Background: CheckMate 025 demonstrated superior efficacy with nivolumab versus everolimus in previously treated patients with aRCC (N Engl J Med 2015); nivolumab treatment also resulted in rapid and sustained health-related quality of life (HRQoL) improvement compared with everolimus after 14 months minimum follow-up (Lancet Oncol 2016). Here, we report an updated analysis of CheckMate 025 with an extended minimum follow-up of 51 months as long-term updates critically inform the benefit-to-risk profile of immunotherapeutic regimens.
Methods: Adults with clear cell aRCC that progressed after 1-2 antiangiogenic therapies were randomized (1:1) to nivolumab 3 mg/kg intravenously every 2 weeks or everolimus 10 mg orally once daily until progression (or beyond progression if patients tolerated therapy and exhibited investigator-assessed clinical benefit) or unacceptable toxicity. The primary endpoint was overall survival (OS). Secondary endpoints included investigator-assessed objective response rate (ORR) and progression-free survival (PFS) per Response Evaluation Criteria in Solid Tumors v1.1, safety, and patient-reported HRQoL.
Results: Overall, 410 versus 411 patients were randomized to nivolumab versus everolimus, respectively (20 vs 2 patients remain on treatment). With a minimum follow-up of 51 months, improved OS was maintained with nivolumab versus everolimus (hazard ratio [HR] 0.72; 95.45% confidence interval [CI], 0.61-0.85; P<0.0001), with 48-month OS probabilities of 30% versus 23%, respectively. Investigator-assessed ORR was consistent with previous analyses: 25.6% (nivolumab) and 5.6% (everolimus) (odds ratio 5.79; 95% CI, 3.60-9.32; P<0.0001). Median (95% CI) duration of response was 12.9 (9.2–18.2) months for nivolumab responders (n=105) and 11.3 (5.4–18.7) months for everolimus responders (n=23). Ongoing response was noted in 14.3% (15/105) of nivolumab responders versus 4.3% (1/23) of everolimus responders. Investigator-assessed PFS favored nivolumab versus everolimus with extended follow-up (HR 0.85; 95% CI, 0.72-0.99; P=0.0355). In total, 274 patients (67%) in the nivolumab arm and 295 patients (72%) in the everolimus arm received subsequent systemic anticancer therapy. The median (range) time from last study drug dose to subsequent systemic therapy was 7.7 weeks (0.1–251.3) with nivolumab and 5.3 weeks (0.1–265.4) with everolimus. The most common subsequent systemic therapies in the nivolumab arm were everolimus (33.7%), axitinib (33.2%), cabozantinib (12.4%), and pazopanib (11.7%). In the everolimus arm, the most common subsequent therapies were axitinib (40.9%), nivolumab (25.3%, independent of crossover), pazopanib (18.2%), sorafenib (10.7%), and sunitinib (10.5%). No new safety signals or treatment-related deaths emerged with long-term follow-up. The most common any-grade treatment-related adverse events were fatigue (34.5%), pruritus (15.5%), and nausea (15.0%) with nivolumab, and fatigue (34.3%), stomatitis (29.5%), and anemia (24.4%) with everolimus. Patients treated with nivolumab experienced sustained or improved HRQoL from baseline, whereas HRQoL deteriorated from baseline with everolimus.
Conclusions: In this 4-year update of CheckMate 025, nivolumab continued to demonstrate a durable survival benefit versus everolimus, comprising the longest follow-up of a phase 3 trial of immune checkpoint inhibitor therapy reported to date in previously treated patients with aRCC. No new safety signals were detected, and the previously observed notable improvement in HRQoL with nivolumab was sustained.
40Loss of SETD2 drives mitochondrial dysfunction in clear cell renal cell carcinoma
First Author: Haake, Scott
Co-Authors: Hapke, Robert; Beckermann, Katherine; Rathmell, W. Kimryn
Authors Company: Vanderbilt University Medical Center
Background: SET domain-containing protein 2 (SETD2) is among the most commonly mutated genes in clear cell renal cell carcinoma (ccRCC). Classically, SETD2 modulates chromatin structure via the methylation of lysine 36 on histone H3. However, histone-independent functions of SETD2 are emerging. We sought to discover novel SETD2-dependent changes to the cellular lysine methylation landscape during kidney cancer tumorigenesis.
Methods: In this study, we used HKC human proximal tubule kidney cell lines, the likely nephrogenic region of origin for ccRCC. The proteomes of wild type (WT) and SETD2-knock out (KO) cells were labeled using stable isotope labeling by amino acids in cell culture (SILAC). Proteins were trypsinized and lysine-methylated peptides were immunoprecipitated. Relative abundance of lysine-methylated peptides and total peptides were calculated in the WT and SETD2-KO using nano-liquid chromatography/tandem mass spectrometry (LC/MS-MS).
Results: >30,000 peptides were quantified, including >50 lysine methylated peptides. We observed decreased lysine methylation of eukaryotic elongation factor 1A (eEF1A), including K165 trimethylation (K165me3) and K318 monomethylation (K318me1). eEF1A functions include delivery of aminoacyl-tRNA to the ribosome, and its function is modulated via the lysine methyltransferases eEF1AKMT1-4. eEF1AKMT2 is responsible for methylation of K318, and eEF1AKMT3 is responsible for trimethylation of K165. We observe decreased expression of both eEF1AKMT2 and eEF1AKMT3 in SETD2-KO cells as well as global changes in protein translation and translational regulation. Expression of eEF1AKMT2-3 can be rescued with re-expression of wild type SETD2 and SETD2 with SRI-domain mutation. However, re-expression of SETD2 with SET-domain mutations fail to rescue eEF1AKMT2-3, suggesting their expression is dependent on the methyltransferase activity of SETD2.
Summary: Our data suggests a previously unidentified role for SETD2 in regulating protein translation in ccRCC.
41Loss of tumor suppressor SETD2 alters identity of centromeric chromatin and promotes generation of neocentromeres
First Author: Mason, Frank
Co-Authors: Kounlavong, Emily; Rathmell, W. Kimryn
Authors Company: Vanderbilt University Medical Center
Background: Intratumor heterogeneity (ITH) is known to underlie resistance to therapy in many tumor types, but direct molecular mechanisms that cause ITH are largely unknown. In clear cell Renal Cell Carcinoma (ccRCC), tumors with the most ITH harbor inactivating mutations in the methyltransferase SETD2. SETD2 is the sole enzyme responsible for trimethylation of Histone 3 at Lysine 36 (H3K36me3) and binds RNA polymerase II. As H3K36me3 correlates with active gene transcription, the prevailing hypothesis is that loss of SETD2 causes altered gene transcription and thus genomic instability. Yet, it is unclear how dysregulation of gene transcription, in and of itself, could drive aneuploidy and ITH. We recently identified that loss of SETD2 causes aneuploidy through chromosome segregation errors in mitosis, independent of its role in transcription. We sought to identify how loss of H3K36me3 promotes mitotic errors in SETD2 mutant cells.
Methods: Using immunofluorescence microscopy and quantitative image analyses in control and SETD2 mutant cell lines, we assayed for disruptions in microtubule-chromosome interactions during mitosis, either of which can result in genome instability. We also assessed whether there were structural changes in mitotic chromosome architecture via metaphase spreads in control and SETD2 mutant cells.
Results: We have identified that mitotic errors in SETD2 mutant cells are caused by dysregulation of centromeres, which are specified by the centromeric Histone 3-variant CENP-A and are the site of microtubule attachment to chromosomes during mitosis. Loss of SETD2 increased levels of CENP-A not only at centromeres but also ectopically across chromosome arms to generate neo-centromeres. Neocentromeres have been shown to cause inappropriate microtubule-chromosome attachment, chromosome mis-segregation during mitosis, DNA damage, replication stress, and are epigenetically inherited.
Conclusions: Loss of SETD2 causes genomic instability, at least in part, through dysregulation of centromeric identity. We propose that generation of neocentromeres in SETD2 mutant cells could contribute to the increased clonality and heterogeneity in ccRCC tumors.
42Mitotic defects and delays are characteristic features of SETD2 loss
First Author: Kounlavong, Emily
Co-Authors: Mason, Frank; Norris, Stephen; Verhey, Kristen; Walker, Cheryl; Rathmell, Kimryn
Authors Company: Vanderbilt University Medical Center, University of Michiagn Medical Center, Baylor College of Medicine
Background: Chromosomal instability (CIN) is a hallmark of cancer with potential for therapeutic targeting. While CIN is known to originate from mitotic errors in chromosome segregation, precise molecular mechanisms promoting CIN in different tumor types are largely unknown. Renal cell carcinoma (RCC) exhibits genomic instability, which may be attributed to chromosome segregation errors. Loss of the methyltransferase SETD2, a tumor suppressor, is correlated with an increase in genomic instability and intratumor heterogeneity in RCCs. SETD2functions to regulate transcription through tri-methylation of Lysine 36 on histone 3 (H3K36me3) and methylates alpha tubulin on lysine 40, yet it is unclear whether loss of histone or tubulin methylation is the primary driver of CIN. Tubulin post-translational modifications (PTMs), such as methylation, regulate tubulin polymer structure and function. Consequently, changes in tubulin PTMs can contribute to CIN by changing microtubule dynamics, leading to improper chromosome attachment and/or segregation in mitosis. Here we investigate the role that SETD2 plays in tubulin PTMs and dynamics during mitosis and the subsequent mitotic complications leading to genomic instability.
Methods: We have identified in cultured cells that SETD2 loss causes delays in mitosis, formation of micronuclei, and aneuploidy through immunofluorescence microscopy and live imaging. A comet assay was also performed on control and SETD2 mutant cells to asses MT growth changes.
Results: We found that loss of tubulin methylation in SETD2 depleted cells correlated with changes in several tubulin PTMs during mitosis. These PTM changes also correlated with increased MT assembly rates in interphase and mitosis, 1.27- and 1.23- fold respectively. The increase in MT assembly rates and polymer stability are known to drive CIN in other solid tumor models.
Conclusion: SETD2 loss promotes changes in microtubule modifications and dynamics, which promotes chromosome segregation errors during mitosis. We propose these microtubule defects may be a significant contributor to genome instability in RCC.
43Molecular chaperones Hsp70 and Hsp90 stabilize and maintain HIF2a activity in ccRCC
First Author: Smith, Britannia
Co-Authors: Woodford, Mark; Backe, Sarah; Sharpiro, Oleg; Mollapour; Bratslavsky, Gennady
Authors Company: SUNY Upstate Medical University
Abstract: Hypoxia Inducible Factor-2a (HIF2a) is a transcription factor responsible for upregulation of genes that facilitate cellular adaptation to hypoxia. In normal cells under normoxia, HIF2a is recognized by the tumor suppressor Von Hippel-Lindau (VHL) and subsequently ubiquitinated and marked for proteasomal degradation. Mutation and inactivation of VHL is the key molecular event associated with the most common type of kidney cancer, clear cell Renal Cell Carcinoma (ccRCC). Stabilization of HIF2a as a result of VHL inactivation leads to transcriptional upregulation of genes that drive ccRCC. The molecular chaperones Heat Shock Protein 70 (Hsp70) and Heat Shock Protein 90 (Hsp90) are involved in the folding, stability, and activation of numerous client proteins involved in tumor growth. Previous work has shown that Hsp90 stabilizes HIF1a, however the influence of Hsp90 on HIF2a remains elusive. Here we show that Hsp70 and Hsp90 work cooperatively to maintain the stability and function of the new client HIF2a in ccRCC. Pharmacologic inhibition of Hsp70 or Hsp90 leads to ubiquitination and degradation of HIF2a in the proteasome. These new insights into the molecular mechanism of HIF2a stabilization are crucial to understanding potentially novel therapeutic options for ccRCC management.
44Multicenter Randomized Phase III Trial of Deferred Cytoreductive Nephrectomy in Synchronous Metastatic Renal Cell Carcinoma Receiving Checkpoint Inhibitors: a DaRenCa and NoRenCa Trial Evaluating the Impact of Surgery or No Surgery. The NORDIC-SUN-Trial
First Author: Donskov, Frede
Co-Authors: Overby, Anders; Ljungberg, Börje; Nielsen, Tommy; Bex, Axel; Møller, Anne Kirstine; Beisland, Christian
Authors Company: Aarhus University Hospital, Umeaa University, The Royal Free London NHS Foundation Trust, Herlev Hospital, Bergen University Hospital
Background: For patients with synchronous metastatic renal cell carcinoma (mRCC), cytoreductive nephrectomy (CN) is controversial. The CARMENA and SURTIME trials were challenged by low accrual rate; data point towards a survival benefit for CN in intermediate risk patients, and deferred CN patients. The deferred CN approach ensures systemic therapy for all patients, avoid systemic treatment delay, and spare surgery in patients with progressive disease. However, data on the role of CN in the era of checkpoint immunotherapy is lacking.The purpose of the study is to evaluate the effect of deferred CN compared with no surgery following nivolumab combined with ipilimumab. The primary objective is overall survival (OS). Secondary objectives include safety of surgery, objective response rate (ORR) and progression free survival (PFS) per RECIST1.1. Exploratory endpoints include tumor genome, circulating tumor DNA, immune activity in blood and tumor tissue, and microbiome characteristics, correlated with OS, PFS and ORR.
Methods: This trial is an investigator-initiated, open, randomized, multicenter, phase III trial. Major inclusion criteria include age 3 18, synchronous mRCC, IMDC intermediate or poor risk, RCC histology, no autoimmune disease requiring steroids, and no antibiotics for 14 days.Outline of the trial: All patients will receive checkpoint immunotherapy nivolumab combined with ipilimumab immediately after inclusion. After 3 months or 4 cycles, whichever comes first, tumor assessment by CT and blood samples will be obtained; the patient will be discussed for resectability at the multidisciplinary meeting (MDT). Whether the patient is eligible for CN is at the discretion of the urologist at the local MDT. Patients with =3 IMDC risk factors and deemed suitable for CN will then undergo randomization. Patients randomized to CN should have this procedure performed within 4 weeks (immune AEs treated with prednisolone may delay nephrectomy until the patient is clinically stable and prednisolone dose =10 mg daily), followed by maintenance nivolumab. Patients randomized to no nephrectomy will receive maintenance nivolumab without surgery. Patients deemed not suitable for surgery or have > 3 IMDC risk features at the 3-month evaluation continue systemic therapy for additional 3 months, followed by evaluation CT and blood test. Patients with =3 IMDC risk factors and deemed suitable for cytoreductive nephrectomy will then undergo randomization. Patients randomized to CN should have this procedure performed within 4 weeks (immune AEs treated with prednisolone may delay nephrectomy until the patient is clinically stable and prednisolone dose is =10 mg daily), followed by maintenance nivolumab. Patients randomized to no nephrectomy continue nivolumab without surgery. Patients deemed not suitable for surgery or have > 3 IMDC risk features at the 6-month evaluation continue systemic therapy. Nivolumab may continue until unacceptable toxicity, progression, or total treatment length of 2 years from inclusion. Patients with unequivocal progression at the 3-months or 6-months evaluation should not proceed to randomization and will exit the study.The planned size for the study is 400. The trial is registered at ClinicalTrials.gov: NCT03977571Status: Not yet recruiting
45Need for Sex Disaggregated Data in Outcomes Reporting for the Treatment of Renal Cell Carcinoma with Immunotherapy
First Author: Sterling, Joshua
Co-Authors: Patel, Hiren; Shinder, Brian; Sadimin, Evita; Mayer, Tina; Saraiya, Biren; Jang, Thomas; Kim, Isaac; Singer, Eric
Authors Company: Robert Wood Johnson University Hospital, CINJ
Introduction: There are biologic, behavioral, psychologic, and environmental exposure differences between males and females that can potentially affect the immune response to both foreign and self-antigens. Significant sex-related dimorphisms in both innate and adaptive immune systems have been reported. Emerging data also indicate that a subset of kidney cancers express the androgen receptor, a finding seen in both men and women. Additionally, studies have shown increased androgen receptor expressivity is associated with worse prognosis in patients with localized disease; suggesting androgen signaling pathways may play a role in disease progression. The last decade has seen the rapid development of immune-oncology therapies for the treatment of RCC, specifically immune check point inhibitors. However, gender differences have been almost completely ignored with respect to the advent, development, and implementation of immunotherapy checkpoint inhibitors. This study is to highlight that gap in our scientific knowledge.
Methods: A systematic search of PubMed and Google Scholar for clinical trials that used immune checkpoint inhibitors for the treatment of RCC was performed. Abstracts, duplicate entries, non-English publications, and publication dates prior to 2000 were removed. The remaining publications were reviewed for any sub-group analysis based on sex. A review of currently recruiting clinical trials in the US was also performed to see if any listed differences based on sex as a primary or secondary outcome.
Results: 153 publications were identified and reviewed. Of those, only 1 publication (the results of the CheckMate 025 trial by Motzer et al) included a subgroup analysis based on sex. They found that nivolumab therapy resulted in a statistically significant overall survival benefit compared to everolimus for males but not for females. Currently, there are 32 clinical trials using at least one immunotherapy agent for patients with RCC that are either actively recruiting patients or will begin enrollment shortly. None of these studies list gender targets in terms of enrollment or planned analyses by sex.
Conclusion: Immune checkpoint inhibitors have been shown to improve survival for patients with advanced RCC, but this benefit maybe influenced by patient sex. Ongoing immuno-oncology studies should collect and publish sex disaggregated data, and future studies should consider sex and gender when setting enrolment goals, so we can further elucidate their impact on the efficacy and toxicity of checkpoint inhibition for RCC.
46Neutrophil-to-lymphocyte ratio in unclassified renal cell carcinoma is associated with outcome and differs between molecular subgroups
First Author: Marcon, Julian
Co-Authors: Di Natale, Renzo; Weng, M.S., Stanley (; Ghanaat, Mazyar; Silagy, Andrew; Mano, Roy; Blum, Kyle; Attalla, Kyrollis; Reznik, Ed; Carlo, Maria; Motzer, Robert; Coleman, Jonathan; Russo, Paul; Hakimi, A. Ari; Chen, M., Ying-Bei
Authors Company: Memorial Sloan-Kettering Cancer Center
Introduction: Unclassified renal cell carcinoma (uRCC) is a rare non-clear cell RCC variant. Next-generation sequencing studies have reported molecular subgroups based on pathways associated with better (MTOR/PI3K) and worse (HIPPO/TP53/DNA damage repair) prognosis. High neutrophil-to-lymphocyte ratio (NLR) is considered to be a marker of inflammatory response and has been found to be associated with worse clinical outcomes. We aimed to investigate what role NLR plays in this setting and also explored the differences in NLR among uRCC molecular subgroups.
Material and methods: After IRB approval we queried our internal database. A total of 112 patients with uRCC, who had undergone molecular profiling, were identified. 85 patients with complete demographic data, a preoperative complete blood count obtained at the time of diagnosis and comprehensive follow-up data were included. NLR was calculated by dividing the absolute neutrophil count by the absolute lymphocyte count (in K/μl). We used non-parametric hypothesis testing with a pre-rejection alpha of 0.05 in all the analyses. Cancer-specific survival (CSS) was calculated from the time of initial pathologic diagnosis to the time of death or last follow-up. Survival estimates were computed using uni- and multivariable Cox regression models.
Results: The study cohort included a total of 51 men and 34 women, the median age was 60.5 years (IQR: 51.5-70). The median follow-up time was 68.8 months. The median NLR of the entire cohort was 3.05 K/μl (IQR: 2.04-4.2). Patients with higher NLR showed poor CSS (Cox p<0.0001, HR 1.43, 95%CI: 1.26-1.62). After accounting for pT and M stage, molecular subgroups and the number of driver mutations at multivariable analysis, higher NLR remained significantly associated with poor CSS (p=0.003, HR 1.32, CI: 1.11-1.57 – Fig.1). MTOR/PI3K pathway-related subgroups were identified in 17 (20.0%) patients, while 30 cases showed mutations in genes of other major pathways (HIPPO/TP53/DNA damage repair/cell cycle). Moreover, there was a significant difference in NLR between genomic subgroups (Kruskal-Wallis p=0.002, Fig. 2).
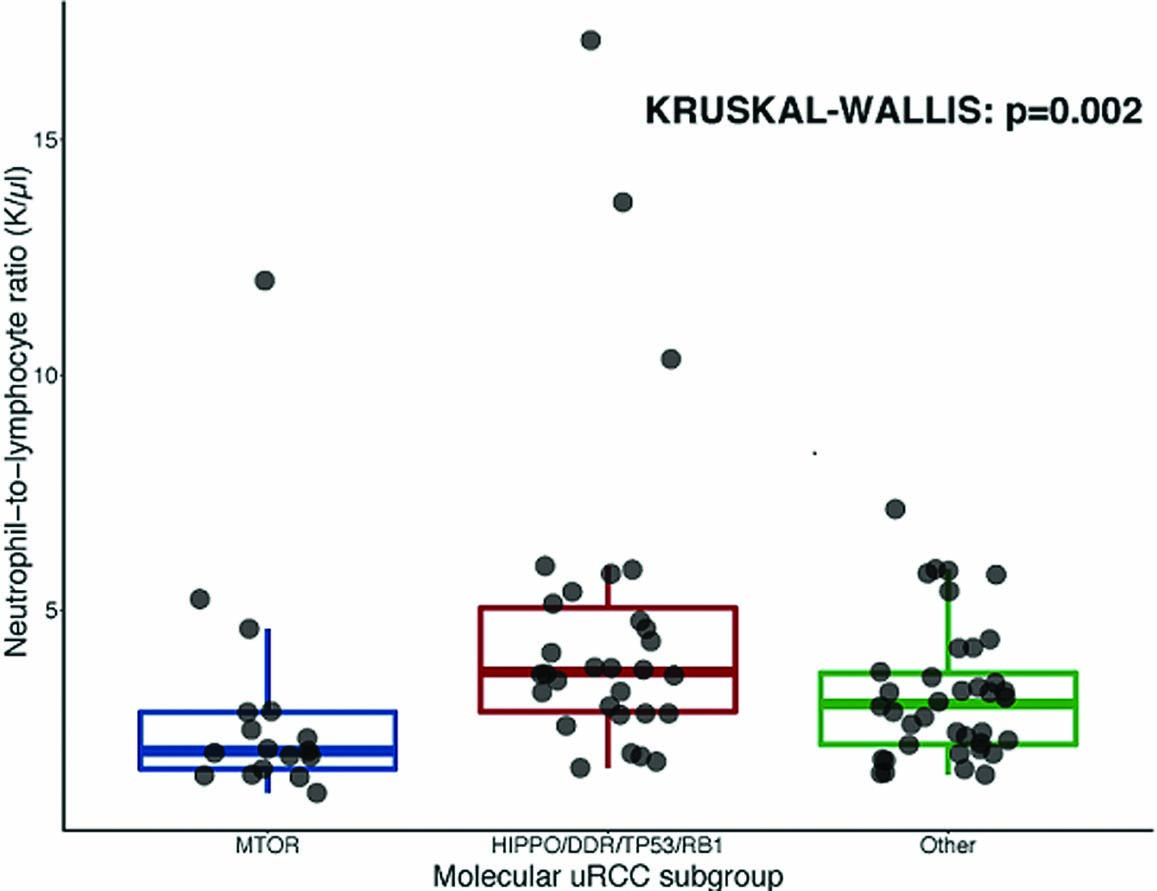
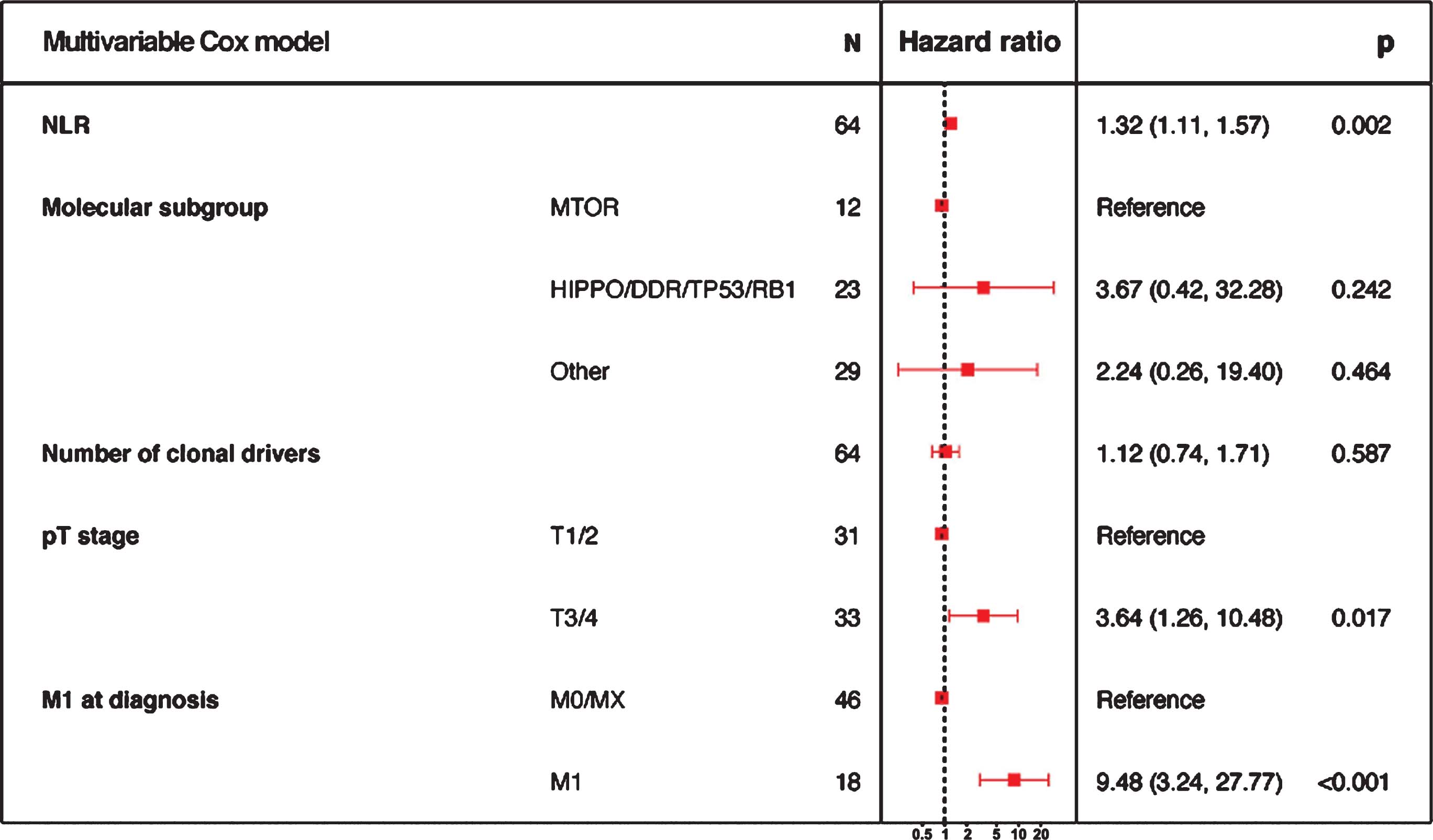
Conclusion: We show that NLR is significantly associated with clinical outcomes in uRCC, its use as a biomarker of disease aggressiveness should be evaluated in further studies. Furthermore, distinct genomic subgroups of uRCC differ considerably in relationship to this parameter.
47Nuances in surgical selection for cytoreductive nephrectomy among IMDC intermediate-poor risk patients
First Author: Silagy, Andrew
Co-Authors: Iosepovici, Skylar; Mano, Roy; Attalla, Kyrollis; DiNatale, Renzo; Marcon, Julian; Motzer, Robert; Coleman, Jonathan; Russo, Paul; Hakimi, A Ari
Authors Company: Memorial Sloan Kettering Cancer Center
Introduction: Retrospective studies of surgical cohorts are limited because patients are preselected for surgery. Therefore, we aimed to evaluate preoperative decision-making that determines patient selection for cytoreductive nephrectomy.
Methods: We reviewed 284 IMDC intermediate-poor risk patients with metastatic renal cell carcinoma (RCC) who were referred to MSKCC urologists for consideration of a cytoreductive nephrectomy between 2009 and 2019. We compiled baseline patient and tumor characteristics, including the sites of metastases. We compared patients that were and were not selected for cytoreductive nephrectomy. For patients not operated on, we performed an iterative thematic analysis of the reasons cited for recommending non-surgical management, illustrating this as a Euler diagram. ResultsThere were 211 patients who received a cytoreductive nephrectomy and 73 patients managed non-operatively. Non-operative patients had more metastatic disease sites (p = 0.007), specifically bone metastases (p = 0.016), and a higher proportion of poor risk IMDC status (p = 0.003). There was no significant difference between the patient groups regarding smoking history, eGFR at encounter, diabetes, and primary tumor size. (Table 1)There were seven distinct themes for recommending non-operative management. These were a combination of patient and tumor factors. (Figure 1)
Table 1
| Non-operative | Operative | p-value | |
| Cohort | 73 | 211 | |
| Male gender (%) | 53 (72.6) | 158 (74.9) | 0.756 |
| Age (median [IQR]) | 64.00 [56.00, 71.00] | 61.00 [53.00, 69.00] | 0.169 |
| Patient race (%) | 0.647 | ||
| Asian | 2 (2.7) | 5 (2.4) | |
| Black | 5 (6.8) | 8 (3.8) | |
| Other | 2 (2.7) | 5 (2.4) | |
| Patient Refused/Unknown | 6 (7.2) | 11 (5.2) | |
| White | 58 (79.5) | 182 (86.3) | |
| Smoking history (%) | 0.517 | ||
| Current | 12 (16.4) | 30 (14.2) | |
| Former | 30 (41.1) | 86 (40.8) | |
| Never | 30 (41.1) | 83 (39.3) | |
| Unknown | 1 (1.4) | 12 (5.7) | |
| eGFR (median [IQR]) | 72.63 [54.22, 93.00] | 73.11 [59.21, 89.49] | 0.665 |
| Diabetes (%) | 15 (20.5) | 43 (20.4) | 1 |
| Radiographic tumor size (median [IQR]) | 7.50 [5.20, 8.90] | 7.20 [5.40, 9.20] | 0.6 |
| No. of metastatic organs (median [IQR]) | 2.00 [1.00, 3.00] | 2.00 [1.00, 2.00] | 0.007 |
| IMDC Risk Profile (%) | 0.003 | ||
| Intermediate | 40 (54.8) | 156 (73.9) | |
| Poor | 33 (45.2) | 55 (26.1) | |
| Visceral metastases (%) | 52 (71.2) | 161 (76.3) | 0.434 |
| Bone metastases (%) | 36 (49.3) | 69 (32.7) | 0.016 |
| Nodal metastases (%) | 32 (43.8) | 76 (36.0) | 0.264 |
| Other sites of metastases (%) | 9 (12.3) | 17 (8.1) | 0.345 |
Figure 1.
The reasons a patient was considered suitable for nonoperative management.
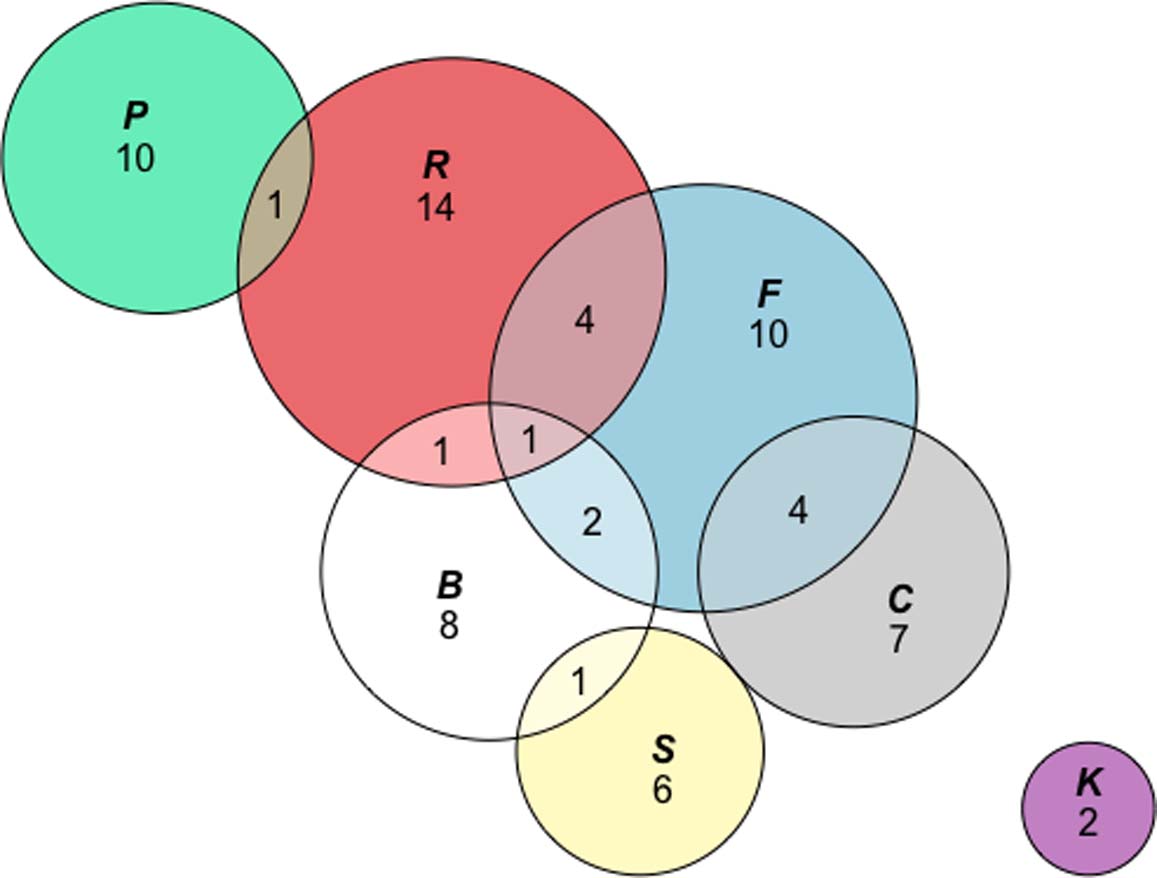
Conclusion: While IMDC poor risk group patients were less likely to be selected for surgery, this was not an exclusive categorization for patient selection. Operative selection is a nuanced process, with detailed consideration given to evaluating every individual patient.
48Patient-reported experience of diagnosis, management, and burden of renal cell carcinomas: North American Results from a global patient survey in 43 countries
First Author: Jonasch, Eric
Co-Authors: Deborah Maskens, Rachel H Giles, Robert Bick, Robin Martinez, Sara Jane Maclennan, Malcolm Packer, Daniel Yick Chin Heng, James M. G. Larkin, Axel Bex, Michael A.S. Jewett, Eric Jonasch
Authors Company: International Kidney Cancer Coalition
Purpose: The International Kidney Cancer Coalition (IKCC) is a federation of 44 affiliated patient organizations representing over 1.2 million patients worldwide, committed to reduce the global burden of kidney cancer. A large-scale global survey of RCC patients to capture real world experiences has never been undertaken.
Methods: The 35-question survey was designed to identify geographic variations in patient education, experience and awareness, access to care, best practices, quality of life and unmet psychosocial needs. It was completed online, by telephone, or in paper form by kidney cancer patients and /or their caregivers (Sept-Oct 2018). The survey was translated into 14 languages (including English UK and US, French and Canadian French, Portuguese and Brazilian Portuguese) and was disseminated with the support of 30 kidney cancer patient organizations worldwide.
Results: 1,983 responses were recorded from 43 countries and were analysed independently of the IKCC by market research experts. Globally, analysis revealed that at diagnosis, 43% of all respondents had no understanding of their RCC sub-type; RCC patients < 45 years old reported nearly twice as many barriers to treatment as patients > 45 at diagnosis. Females reported longer delays to diagnosis than males. With regard to clinical trials, 70% of patients surveyed had never been asked to participate in a clinical trial, but of those 89% said it was fairly likely they would have participated had they been asked (45% of these patients were being treated in a major cancer center). Shared decision making remains aspirational: globally 29% of all patients reported no involvement in their treatment decision, responding ‘my doctor decided for me’. While 96% of respondents reported psychosocial impacts, surprisingly, only 50% disclosed them to their healthcare team. Most difficult times experienced by patients varied by gender and by country. Specifically in North America, results from Canada (n=246), United States (n=233) and Mexico (n=144) were separately analysed and reported with variances from global data noting best practices in each country, along with areas requiring specific focus to improve patient experience across all stages of RCC.
Conclusion and clinical implications: This research sheds light on the unmet needs in the RCC patient experience and highlights improvements to the design and delivery of supportive care. Specifically in North America, this research demonstrates the heterogeneity of patient experiences across North America and opportunities to improve RCC patient experience in Canada, the United States, and Mexico. This first-ever global survey serves as a benchmark for longitudinal data collection and could be used to inform future disease-specific quality of life instruments.
49PDIGREE: An adaptive phase 3 trial of PD-inhibitor nivolumab and ipilimumab (IPI-NIVO) with VEGF TKI cabozantinib (CABO) in metastatic untreated renal cell cancer (Alliance A031704)
First Author: Zhang, Tian
Co-Authors: Ballman, Karla; Choudhury, Atish; Chen, Ronald; Watt, Colleen; Wen, Yujia; Zemla, Tyler; Emamekhoo, Hamid; Vaishampayan, Ulka; Morris, Michael; George, Daniel; Choueiri, Toni
Authors Company: Duke Cancer Institute, Weill Cornell Medical College, Dana Farber Cancer Institute, UNC Chapel Hill, Alliance for Clinical Trials in Oncology, University of Wisconsin, Karmanos Cancer Institute, Memorial Sloan Kettering Cancer Center
Background: First-line treatment of mRCC has rapidly changed to include IPI-NIVO or cabozantinib, with clinical benefit of each based on the Checkmate 214 and CABOSUN trials. Combination immunotherapy with VEGF therapies have shown benefit over sunitinib in the JAVELIN 101 and KEYNOTE 426 trials. It is yet unclear which patients (pts) benefit most from combination immunotherapy-VEGF inhibitors, and the optimal sequence of drugs.
Methods: In an adaptive, randomized, multicenter, phase 3 trial (Alliance A031704, PDIGREE), pts will start treatment with induction IPI 1mg/kg and NIVO 3mg/kg intravenously (IV) once every 3 weeks. Key inclusion criteria include clear cell mRCC, IMDC intermediate or poor risk, Karnofsky performance status >70, and no prior treatments for mRCC. Based on 3-month radiographic assessment (after completing IPI-NIVO combination), pts with complete responses (CR) will undergo maintenance NIVO 480mg IV every 4 weeks, pts with progression of disease (PD) will switch to CABO 60mg oral daily, and pts with non-CR/non-PD will be randomized to NIVO 480mg IV every 4 weeks versus NIVO 480mg IV every 4 weeks with CABO 40mg oral daily. Randomization will be stratified by IMDC risk criteria and presence of bone metastases. The primary endpoint of the study is overall survival (OS). We hypothesize that 3-year OS rate will improve to 70% for NIVO-CABO compared to 60% for NIVO alone; to achieve 85% power with a two-sided alpha of 0.05 and exponential distribution, 696 patients will be randomized. Accounting for 30% patients with either CR or PD, and 5% dropout from toxicity, up to 1046 pts will be enrolled. Key secondary endpoints include PFS, 12-month CR rate, ORR based on RECIST 1.1 and irRECIST criteria, and toxicity profiles. Quality of life will be assessed based on the FKSI-19, PROMIS-fatigue, and EQ5D-5L questionnaires. Biomarkers associated with CR and association of IL-6 with treatment benefit will be assessed. Other tissue-based and plasma-based biomarkers are planned. Enrollment will begin this year.
Support from UG1CA189823, U24CA196171; ClinicalTrials.gov Identifier: NCT03793166; https://acknowledgments.alliancefound.org
50Pembrolizumab Plus Axitinib vs Sunitinib as First-Line Therapy for Metastatic Renal Cell Carcinoma: Outcomes by IMDC Risk and Sarcomatoid Subgroups of the Phase 3 KEYNOTE-426 Study
First Author: Soulières, Denis
Co-Authors: Plimack, Elizabeth R.; Rini, Brian I.; Stus, Viktor; Gafanov, Rustem; Hawkins, Robert; Nosov, Dmitry; Pouliot, Frédéric; Melichar, Bohuslav; Vynnychenko, Ihor; Azevedo, Sergio J.; Borchiellini, Delphine; McDermott, Raymond S.; Bedke, Jens,; Tamada, Satoshi; Wan, Shuyan M.; Perini, Rodolfo F.; Chen, Mei; Atkins, Michael B.; Powles, Thomas
Authors Company: Centre Hospitalier de l’Universitaire de Montréal, Fox Chase Cancer Center, Cleveland Clinic Taussig Cancer Institute, Dnipropetrovsk Medical Academy of Ministry of Health of Ukraine, Russian Scientific Center of Roentgenoradiology, The Christie NHS Foundation Trust, Central Clinical Hospital With Outpatient Clinic, CHU de Québec and Université Laval, Palacky University Medical School and Teaching Hospital, Sumy State University, Sumy Regional Oncology Center, Hospital de Clínicas de Porto Alegre, Centre Antoine Lacassagne, Université Côte d’Azur, Adelaide and Meath Hospital and University College Dublin, Eberhard-Karls University Tübingen, Osaka City University Hospital, Merck & Co., Inc., Georgetown Lombardi Comprehensive Cancer Center, Barts Health and the Royal Free NHS Trusts
Background: A phase 1b study showed that the combination of the PD-1 inhibitor pembrolizumab and the VEGFR tyrosine kinase inhibitor axitinib had substantial antitumor activity and manageable safety in patients with previously untreated metastatic renal cell carcinoma (mRCC). In the randomized, multicenter, phase 3 KEYNOTE-426 study, pembrolizumab+axitinib significantly improved overall survival (OS; HR, 0.53; P<0.0001), progression-free survival (PFS; HR, 0.69; P=0.0001), and objective response rate (ORR; 59.3% vs 35.7%; P<0.0001) vs sunitinib as first-line therapy for mRCC (NCT02853331), with manageable toxicity. We present data for the combined International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) intermediate-/poor-risk group and for patients with sarcomatoid features.Methods: 861 patients with clear cell mRCC, no prior systemic therapy, and Karnofsky Performance Status score =70% were randomly assigned 1:1 to receive pembrolizumab 200 mg IV Q3W (maximum 35 cycles) plus axitinib 5 mg orally BID (N=432) or sunitinib 50 mg orally QD (4 weeks on/2 weeks off) (N=429). Primary end points were OS and PFS (RECIST v1.1, blinded, independent central review [BICR]). ORR (RECIST v1.1 by BCIR) was the key secondary end point. Analysis by IMDC risk groups (intermediate-/poor-risk vs favorable) was prespecified; sarcomatoid group was exploratory.
Results: Median follow-up was 12.8 months (range, 0.1-22.0). 42% of patients with =1 postbaseline evaluable imaging assessment in the pembrolizumab+axitinib arm and 16% in the sunitinib arm had a =60% decrease in the number of target lesions. 592 (68.8%) of all randomized patients were IMDC intermediate-/poor-risk (294, pembrolizumab +axitinib; 298, sunitinib). 269 (31.2%) patients had favorable risk (138, pembrolizumab + axitinib;131, sunitinib). Pembrolizumab + axitinib improved OS (HR, 0.52; 95% CI, 0.37-0.74; 12-month rate, 87.3% vs 71.3%), PFS (HR, 0.67; 95% CI, 0.53-0.85; median 12.6 vs 8.2 months) and ORR (55.8% [95% CI, 49.9-61.5] vs 29.5% [24.4-35.1]) in patients with intermediate-/poor-risk; CR rates were 4.8% (95% CI, 2.6-7.9) vs 0.7% (0.1-2.4). In patients with favorable risk, 12-month OS rates were 95.0% vs 94% (HR, 0.64; 95% CI, 0.24-1.68) and 12-month PFS rates were 68.0% vs 60% (HR, 0.67; 95% CI, 0.53-0.85) for pembrolizumab+axitinib vs sunitinib, respectively. ORR was 55.8% [95% CI, 49.9-61.5] with pembrolizumab+axitinib and 29.5% [95% CI, 24.4-35.1] with sunitinib; CR rates were 4.8% (95% CI, 2.6-7.9) vs 0.7% (0.1-2.4), respectively. Of 578 patients with known status, 105 (18.2%) had sarcomatoid features (51, pembrolizumab+axitinib; 54, sunitinib). Pembrolizumab+axitinib improved OS (HR, 0.58; 95% CI, 0.21-1.59; 12-month rate, 83.4% vs 79.5%), PFS (HR, 0.54; 95% CI, 0.29-1.00; median not reached vs 8.4 months), and ORR (58.8% [95% CI, 44.2-72.4] vs 31.5% [19.5-45.6]) in patients with sarcomatoid features; CR rates were 11.8% (95% CI, 4.4-23.9) vs 0% (0.0-6.6). For patients with sarcomatoid features, 54% of patients in the pembrolizumab+axitinib arm and 16% in the sunitinib arm had a =60% decrease in the number of target lesions
Conclusions: Pembrolizumab+axitinib provides benefit in the combined population of patients with IMDC intermediate- or poor-risk and in patients whose tumors had sarcomatoid features. Observed benefits were consistent with those in the total population.
51Perspective Matters: Patient-reported ECOG performance status (PS) is lower than oncologist-reported PS and is associated with worse psychosocial outcomes
First Author: Bergerot, Cristiane
Co-Authors: Bergerot, Paulo Gustavo; Philip, Errol J.; Hsu, JoAnn; Dizman, Nazli; Salgia, Meghan; Battle, Dena; Vaishampayan, Ulka; Loscalzo, Matthew; Dale, William; Pal, Sumanta K.
Authors Company: City of Hope Comprehensive Cancer Center, University of California San Francisco, Kidney Cancer Research Alliance (KCCure), Wayne State University/Karmanos Cancer Institute
Background: Patient-reported outcomes (PROs) has been used to assess effectiveness of treatments, and to actively engage patients in their disease management. Evidence indicates a benefit of assessing and monitoring PROs with standardized measures, such as performance status (PS), however questions remain as to whether clinician or patient ratings provide the greatest insight across certain domains. We sought to describe PS as reported by clinicians and self-reported by patients, and to determine the association between PS and psychosocial outcomes (quality of life and distress).
Methods: We assessed patients with advanced renal cell carcinoma, receiving treatment at a single institution, for PS (self-reported version of the Eastern Cooperative Oncology Group, ECOG), QOL (FACT-G) and emotional well-being (PROMIS-Anxiety and Depression). Clinical data, including ECOG PS by clinicians rating, was extracted from medical records. Multivariate analysis was used to determine the association between PS, QOL and psychological symptoms.Results: Among 85 patients screened (27.1% female and 72.9% male; median age of 63, range from 25-87), 47% were receiving first line therapy, and 21% second line. Clinicians were more likely to classify patients as ECOG PS 0 compared with patients themselves (94% vs 68%, P=0.03). Worse ECOG PS score reported by either clinician or patient was associated with poorer QOL (P=0.001). Importantly, self-reported ECOG PS score, but not clinician rating, was also associated with poorer emotional well-being (P<0.05).
Conclusions: The current study suggests that clinicians may tend to overestimate ECOG PS compared to patients own self-rating. These findings underscore the potential role of self-reported ECOG PS, especially given its association with both QOL and emotional well-being. Self-reported ECOG PS could serve as a useful eligibility criterion. Future studies are needed to examine their association with treatment decision making, adherence and disease outcomes.
52PET-CT as predictor of response in the clear-cell metastatic renal cell carcinoma (mRCC) treatment
First Author: Moraes Lages, M. D., Paulo Sergio
Co-Authors: Prado, Luciano; Soares, Andrey; Schutz, Fabio; Prado, Leonardo; Lages, Luciana; Barra, Renato
Authors Company: Oncovida , Lacog, Imeb, Brasilia, (Centro Paulista De Oncologia, Beneficencia Portuguesa De Sao Paulo
Background: Clear-cell mRCC is highly dependent of VEGF(R) pathway. VEGF tyrosine kinase inhibitors (TKI) are among the cornerstones of treatment. Some patients treated with VEGFR TKI have long term duration of response. There is evidence suggesting different gene signature profiles in clear-cell mRCC: angiogenesis and T-effector gene signatures (Rini B, et al. ESMO 2018). These subgroups have distinct benefit to VEGFR TKI and immune checkpoint blockers (ICB). FDG-PET correlates to glucose hypermetabolism and inflammatory infiltrate. PSMA-PET is a new molecular image that besides being used mostly in prostate cancer, also correlates to angiogenesis. Little is known about the usefulness of these PET images in clear-cell mRCC. We hypothesized that FDG- and PSMA-PET could help predicting response to VEGFR TKI.
Methods: We included six clear-cell mRCC patients treated with VEGFR TKI (sunitinib or pazopanib) in the first or second-line. FDG- and PSMA PET were performed prior to therapy initiation in all patients. Patients were then categorized in 2 groups: 1) PSMA High/FDG Low (Angiogenesis profile); and, 2) PSMA Low/FDG High (Immune profile).
Results: Three patients had PSMA high/FDG low PET scans (Angiogenesis profile), and all of them had long-term partial responses to VEGFR TKI or Imune Checkpoint blockers. Three patients had PSMA low/FDG high (Immune profile), and all of them had a poor response to VEGFR-TKI.
Conclusion: Currently, there is no good predictive factor for mRCC treatment. Gene signature profiles usually have limited access and are not readily available. PSMA and FDG PET may identify different clear-cell mRCC profiles (Angiogenesis or Immune profiles) and help selecting different treatment strategies (VEGFR TKI or ICB). This hypothesis lead LACOG-GU to propose a prospective trial to validate the correlation between gene signatures, molecular images with PET and response to therapy in clear-cell mRCC patients.
53Phase 2 study of lenvatinib plus pembrolizumab for disease progression after PD-1/PD-L1 immune checkpoint inhibitor in metastatic clear cell renal cell carcinoma (mccRCC): results of an interim analysis
First Author: Lee, Chung-Han
Co-Authors: Amishi Y. Shah2, Vicky Makker1, Matthew Taylor3, David Shaffer4, James J. Hsieh5, Allen L. Cohn6, Chris DiSimone7, Alvaro Pinto Marin8, Drew Rasco9, Sara Gunnestad Ribe10, Donald A. Richards11, Daniel E. Stepan12*, Corina E. Dutcus12, Jane Wu12, Emmett V. Schmidt13, Rodolfo Perini13, Robert Motzer1
Authors Company: 1Memorial Sloan Kettering Cancer Center,; 2MD Anderson Cancer Center, University of Texas; 3Oregon Health and Science University; 4New York Oncology Hematology; 5Washington University School of Medicine; 6Rocky Mountain Cancer Center; 7Arizona Oncology Associates; 8Hospital Universitario La Paz; 9South Texas Accelerated Research Therapeutics; 10Sorlandet Hospital; 11Texas Oncology, US Oncology Research; 12Eisai Inc; 13Merck & Co. Inc..
*Formerly of Eisai Inc
Background: Lenvatinib (LEN) is a multikinase VEGFR inhibitor approved for use in combination with everolimus to treat advanced RCC after prior VEGF-targeted therapy. Pembrolizumab (PEMBRO) is an anti-PD-1 antibody. Here, we report results of an interim analysis of the RCC cohort of a phase 2 trial of LEN + PEMBRO, specifically in patients who progressed with prior immune checkpoint inhibitor (ICI) therapy.
Methods: This is a per-protocol interim analysis of a multicenter, open-label study for patients with mccRCC, =1 prior therapy, RECIST disease progression on or following an ICI regimen (confirmed = 4 weeks later), measurable disease, and Eastern Cooperative Oncology Group performance status = 1. Patients received LEN 20 mg/d PO plus PEMBRO 200 mg intravenously Q3W until disease progression or toxicity occurred. Tumor assessments were performed every 6 weeks (until week 24), then every 9 weeks.
Results: At data cutoff (March 29, 2019), the first 33 patients enrolled were followed for =12 weeks for response evaluation, and 24 (73%) patients were still on study treatment. The objective response rate was 52%, the disease control rate was 94%, and most patients had tumor shrinkage. Median follow-up time for progression-free survival was 4.2 months. Patient characteristics and efficacy outcomes are summarized in the table. The most common treatment-related adverse events were fatigue (49%), dysphonia (36%), diarrhea (33%), stomatitis (30%), hypertension (24%), dry mouth, nausea, proteinuria, and hand-foot syndrome (21% each). Three (9%) patients discontinued treatment due to adverse events.
Table
Summary of Patient Characteristics and Efficacy Outcomes
| Patient Characteristics, n (%) | LEN + PEMBRO (n = 33) |
| ECOG performance status | |
| 0 | 18 (54.5) |
| 1 | 15 (45.5) |
| Prior anticancer regimens | |
| 1 prior regimen | 14 (42%) |
| >1 prior regimen | 19 (58%) |
| Prior VEGF-targeted therapy | 26 (78.8%) |
| Prior ICI therapy | 33 (100%) |
| PD-1/PD-L1 monotherapy | 14 (42.4%) |
| with VEGF agents | 9 (27.7%) |
| nivolumab + ipilimumab | 7 (21.2%) |
| with other agents | 2 (6.1%) |
| PD-L1 positive | 12 (36.4)* |
| Investigator assessment of efficacy outcomes by irRECIST | LEN + PEMBRO (n = 33) |
| ORR, n (%) | 17 (51.5) |
| 95% CI | 33.5–69.2 |
| Median PFS (95% CI), months | NR (5.6–NR) |
| PFS rate, % (95% CI) at: | |
| 3 months | 93.4 (76.1–98.3) |
| 6 months | 73.8 (45.7–88.9) |
| 9 months | 64.6 (34.5–83.5) |
| Median DOR, (95% CI) months | NR (4.2–NR) |
| Median follow-up time for DOR, months | 6.0 (1.4–7.0) |
| Response duration ≥6 months, n (%)** | 80.8 (42.3–94.9) |
*30.3% were negative and 33.3% were not available.
**Response duration is based on Kaplan-Meier estimation, with 95% CI based on the Greenwood formula using log-log transformation.
CI, confidence interval; DOR, duration of response; ECOG, Eastern Cooperative Oncology Group; ICI, immune checkpoint inhibitor; irRECIST, immune-related Response Evaluation Criteria In Solid Tumors; NR, not reached; ORR, objective response rate; PD-1, programmed cell death protein-1; PD-L1, programmed cell death ligand-1; PFS, progression-free survival; VEGF, vascular endothelial growth factor.
Conclusions: For the subset of mccRCC patients with disease progression during or following ICI therapy, LEN + PEMBRO demonstrated promising antitumor activity. No new safety signals were detected. The study will continue to full cohort expansion.
54Phase II Trial of Intermittent Therapy in Patients (pts) with Metastatic Renal Cell Carcinoma (mRCC) Treated with Front-line Ipilimumab and Nivolumab (Ipi/Nivo)
First Author: Ornstein, Moshe C.
Co-Authors: Wood, Laura S.; Wei, Wei; Allman, Kimberly D.; Martin, Allison; Garcia, Jorge A.; Gilligan, Timothy D.; Rini, Brian I.
Authors Company: Cleveland Clinic Taussig Cancer Institute, Auston
Background: The combination of ipilimumab and nivolumab (Ipi/Nivo) is approved for patients (pts) with treatment-naïve intermediate- and poor-risk metastatic renal cell carcinoma (mRCC). However, duration of therapy and safety/efficacy of Ipi reinduction at progression is unknown. A phase II trial of intermittent Ipi/Nivo with Ipi re-induction at progression was thus designed and is currently accruing (NCT03126331).
Methods: Pts with treatment-naïve mRCC treated with induction Ipi/Nivo followed by 24 weeks (wks) of maintenance Nivo and achieve stable disease (SD), complete response (CR), or partial response (PR) are eligible for inclusion. Patients who achieve SD will continue with Nivo maintenance per standard of care. Pts who achieve a PR or CR enter a treatment-free observation period. All pts will be restaged every 12 wks. Upon disease progression (PD), patients will be re-challenged with 2 doses of Ipi/Nivo every 3 wks followed by CT imaging 6 weeks post-reinduction. Per physician discretion, pts may receive 1 or 2 more doses of re-induction ipilimumab and nivolumab depending on response to first 2 re-induction doses. Following re-induction, pts with PD will be removed from the trial, SD pts will receive maintenance Nivo, and pts who achieve a CR/PR will enter an observation phase. Blood and tissue correlates will be collected at enrollment and other pre-planned time-points to investigate biomarkers of response and resistance.The study objectives are (a) to estimate success rate of observation in pts who achieve a CR/PR (success is defined by 50% of CR/PR pts who sustain a treatment-free interval of at least 9 months), and (b) to estimate toxicity rate (i.e. rate of grade = 3 treatment-related AEs) in patients undergoing re-induction. Forty pts will be enrolled such that the half-width of 95% confidence interval (CI) around success rate will not exceed 0.23. Early termination rules for toxicity are integrated. Summary statistics of success status and toxicity will be provided in frequencies and percentages. Toxicity and success rates will be estimated along with 95% CIs. Logistic regression model will be used to explore effects of patient and tumor characteristics on success status and toxicity. Five pts are currently enrolled. Updated enrollment will be presented.
55Physician Rationale for Prescribing Tyrosine Kinase Inhibitors and Immuno-Oncology Treatments in the First-Line Treatment of Advanced Renal Cell Carcinoma in the United States: Interim Results from a Cross-Sectional Real-World Survey
First Author: Zanotti, Giovanni
Co-Authors: Kim, Ruth; Krulewicz, Stan; Hall, Jennifer; Leith, Andrea; Bailey, Abigail; Piercy, James; Liu, Frank X.; Kearney, Mairead
Authors Company: Pfizer inc., Adelphi Real World, EMD Serono, Inc., Merck KGaA
Background: In 2018, 65,340 renal cell carcinoma (RCC) cases were diagnosed in the United States. Tyrosine kinase inhibitors (TKIs) have been the standard first-line (1L) RCC treatment. Recently approved innovative treatments for 1L RCC include new targeted agents and immuno-oncology (IO) therapies. We examine physician comfort with/rationale for prescribing 1L TKI or IO therapy regimens and treatment patterns in patients with advanced RCC (aRCC).
Methods: Preliminary data were drawn from the Adelphi RCC Disease-Specific ProgrammeTM, a point-in-time study conducted between February and June 2019 with medical oncologists (MDs), nephrologists, and urologists in clinical practice. Physicians completed detailed patient record forms (PRFs) providing demographic, clinical, and treatment patterns for the next 8 consecutive adult patients with a confirmed diagnosis of aRCC plus an online survey providing their own demographics, treatment usage/awareness, and reasons for choice of treatment.
Results: 73 physicians completed the survey (36 MDs, 12 nephrologists, 25 urologists); 51% from a nonacademic setting. Across all physicians, a mean of 39% of 1L patients were stage IV; by specialty: MDs, 61%; nephrologists, 20%; urologists, 14%. Physicians completed PRFs for 399 patients, of whom 270 received a heterogenous mix of 1L treatments, including IO monotherapy in 23 patients; IO/IO combination in 61; TKI monotherapy in 153; TKI/IO combination in 9; other combination in 24.
When ranking the top 5 of 24 attributes, physicians reported the most important attributes for 1L systemic treatments as overall survival (OS; 84%), progression-free survival (PFS; 64%), and high response rate (HRR; 56%). Duration of response (DOR) was more important for MDs (72%) and urologists (52%) than nephrologists (33%). Low incidence of serious events/severe side effects (SEs) was more important for urologists (48%) and nephrologists (42%) than MDs (31%).
In practice (PRF data), physicians included OS, PFS, HRR, and manageable SEs as the most common reasons influencing choice of 1L TKI monotherapy (OS, 46%; PFS, 54%; HRR, 42%; SE, 55%), IO monotherapy (OS, 39%; PFS, 43%; HRR, 48%; SE, 43%), or IO/IO combination (OS, 43%; PFS, 39%; HRR, 44%; SE, 31%). Other important characteristics were method of administration (29%) for TKIs, DOR (26%) and performance status (26%) for IOs, and effective combination (30%) for IO/IO combination. Important characteristics for TKI/IO combination were OS (78%), manageable SEs (44%), and maintaining quality of life (44%).
In general, MDs and nephrologists were comfortable prescribing oral TKI and intravenous IO medications while corresponding figures for urologists were lower: IO 56% and TKI 80%. On average, MDs reported that they had more patients currently receiving a targeted therapy or IO drug (30%/30%, respectively) than nephrologists (13%/9%, respectively) or urologists (15%/13%, respectively).
Conclusions: In general, all physicians consider OS, PFS, and DOR as the most important attributes when prescribing a new treatment regimen for aRCC. When selecting treatments for individual patients, HRR and SE profile are also important. Due to the recent introduction of newly approved TKI/IO combination therapies, the proportion of patients receiving these regimens is low, so further research is needed to determine how they are being incorporated in routine clinical practice.
56Pilot study to evaluate the biologic effect of the probiotic CBM588 in combination with nivolumab/ipilimumab for patients with mRCC
First Author: Bergerot, Paulo
Co-Authors: Philip, Errol J.; Bergerot, Cristiane Decat; Dizman, Nazli; Salgia, Meghan; Salgia, Nicholas; Hsu, JoAnn; Pal, Sumanta K.
Authors Company: City of Hope Comprehensive Cancer Center, University of California San Francisco
Background: For patients with intermediate- to high-risk disease clear cell renal cell carcinoma (RCC), nivolumab/ipilimumab (nivo/ipi) is an FDA approved treatment option. Although data are encouraging for the strategy of dual checkpoint inhibition, less than half of patients will exhibit a partial or complete response to nivo/ipi, and approximately 20% of patients will progress through this regimen. Recent studies suggest that the gut microbiome may play a key role in modulating response to immunotherapy (Routy et al Science 2018). Our preliminary data examining patients receiving immunotherapy for mRCC suggests that certain gut bacteria (e.g., Bifidobacterium spp) may predispose patients to response (Caitano et al ASCO GU 2017). We therefore propose assessing CBM588 in combination with nivo/ipi in this patient cohort. CBM588 is a strain of Clostridium butyricum, a Gram-positive bacillus and human gut commensal. Evidence suggests it can promote the growth of certain gut bacteria like Bifidobacterium, Lactobacillus and Bacteroides spp, each of which has been shown to enhance anti-tumor immune responses. This is related to the production of short-chain fatty acids, such as lactic and butyric acids. Butyric acid protects the mucin layer of the intestinal epithelia and has immunomodulatory effects, promoting Th1 cell IL-10 production to maintain intestinal homeostasis. Emerging research shows that modulation of the gut microbiota may represent a novel and important adjunct to current anti-cancer therapeutic modalities. This study sought to identify the biologic effect of CBM588 with nivo/ipi.
Methods: This phase I/II trial will be conducted at a single cancer center, with patients randomized to one of two treatment arms. The control arm will receive the standard nivo/ipi (3 mg/kg / 1 mg/kg) treatment. One cycle of therapy will constitute an every-3-week regimen of nivo/ipi for the first 12 weeks followed by nivolumab administered on a monthly schedule. The experimental arm will follow the same schedule and doses for of nivo/ipi and will also be given the experimental agent CBM588. CBM588 will be administered orally at a starting dose of 60 mg orally three times per day. Patients are eligible for the trial if they are age 18 or older, have metastatic histologically confirmed mRCC, have planned treatment with nivo/ipi, and have no prior therapy with immune checkpoint inhibition. The primary objective is to determine the biologic effect of CBM588 in combination with nivo/ipi in modulation of the gut microbiome in patients with mRCC. We will assess changes in Bifidobacterium composition as well as the Shannon index (a measure of microbial diversity) of stool samples at baseline and week 13 of therapy. Our secondary objectives include clinical efficacy (progression free survival by RECIST 1.1) and to determine the effect of CBM588 on systemic immune-modulation through the comparison of the proportion of circulating Tregs, MDSCs, and circulating cytokines/chemokines. Stool samples and serum correlatives will be collected on weeks 1 and 13 of treatment. Change in the Bifidobacterium from baseline to week 12 will be assessed using a two-group t-test with a one-sided type I error of 0.05. Clinical trial information: NCT03829111.
57Prognostic significance and immune correlates of CD73 expression in renal cell carcinoma (RCC)
First Author: Tripathi, Abhishek
Co-Authors: Lin, Edwin; Xie, Wanling; Flaifel, Abdallah; Steinharter, John; Stern-Gatof, Emily; Jennings, Rebecca; Bouchard, Gabrielle; Fleischer, Justin; Martinez Chanza, Nieves; Gray, Connor; Mantia, Charlene; Thompson, Linda; Wei, Xiao; Giannakis, Marios; McGregor, Bradley; Choueiri, Toni; Agarwal, Neeraj; McDermott, David; Signoretti, Sabina; Harshman, Lauren
Authors Company: University of Oklahoma Stephenson Cancer Center, University of Utah Huntsman Cancer Institute, Dana-Farber Cancer Institute, Brigham And Women’s Hospital, Beth Israel Deaconess Medical Center, Oklahoma Medical Research Foundation
Background: CD73 (encoded by NT5E) mediates conversion of adenosine monophosphate to adenosine which promotes immunosuppression in the tumor microenvironment. We investigated the prognostic significance of tumor CD73 expression in an institutional cohort of patients with localized and de novometastatic RCC. We performed a complementary analysis of gene expression data from The Cancer Genome Atlas (TCGA) to evaluate the correlation of NT5EmRNA expression with infiltrating immune cell subsets and markers of angiogenesis, myeloid inflammation and effector T cell response.
Methods: We assessed CD73 protein expression using immunohistochemistry (Cell Signaling Technology, D7F9A, 1:25) on RCC tumor tissue from patients with localized orde novo metastatic RCC treated with nephrectomy at Dana-Farber/Harvard Cancer Centers.CD73 expression was quantified using a combined score (CS: intensity x percentage of cells staining positive). CD73 positivity was defined as any CD73 expression on tumor cells (CS>0) and patients were categorized into CD73 negative (CS=0), low (<median CS) and high (= median CS) groups. Multivariable Cox regression analysis adjusted for stage and nuclear grade evaluated association of CD73 expression with disease-free and overall survival (DFS, OS). In the TCGA cohort, patients were split into low, intermediate, and high NT5EmRNA expression groups (= -1, -1 to 1 and =1 standard deviation from the overall mean). Abundance of infiltrating immune cell subsets was calculated based on expression of canonical marker genes. Gene expression signatures reflecting angiogenesis, myeloid inflammation and effector T cell response were calculated based on expression of previously validated genes. The Mann-Whitney U test assessed statistical significance. Bonferroni correction controlled for false discovery rate.
Results: Of the 138 patients included, any CD73 expression (CS>0) was noted in 23% (25/107) and 55% (17/31) of tumors from patients with localized and de novo metastatic disease respectively. High CD73 expression (CS> median) was significantly associated with nuclear grade =3 (P=0.035), advanced T stage (P=0.004) and presence of sarcomatoid histology (<0.001). In patients with localized disease, patients with high tumor CD73 expression had significantly worse 5-yr DFS (42% vs. 75%; adjusted HR: 2.7 (1.3-5.9); P=0.01) and 10-yr OS (22% vs. 64%; adjusted HR: 2.6 (1.2-5.8); P=0.02) compared to CD73 negative group after adjusting for nuclear grade and stage. In the TCGA cohort, high NT5EmRNA expression correlated with significantly decreased expression of CD56 bright NK and Th17 T cell markers and increased expression of markers of immunosuppressive Type 2 helper and regulatory T cells. There was no association of NT5E with angiogenesis, myeloid inflammation or effector T cell signatures.
Conclusions: CD73 expression was found in 30% of RCC samples analyzed and was associated with adverse pathologic features, advanced stage and poor prognosis along with an increase in immunosuppressive T cells and decrease in NK and Th17 cells in the tumor microenvironment. Our results provide strong rationale for targeting the CD73-adenosine pathway in advanced RCC.
58Prognostic Values of Pretreatment Lymphocite-to-Monocite Ratio in Patients with in Non-Metastatic Renal Cell Carcinoma: A Systematic Review and Meta-Analysis
First Author: Garcia-Rojo, Dario
Co-Authors: Prera, Angel; Muñoz-Rodriguez, Jesus; Centeno, Clara; Dominguez, Arturo; Abad, Carlos; Vicente, Eduardo; Gonzalez-Sala, Jose Luis; Hannaoui, Naim; Capdevila, Marta; de Verdonces, Leticia; Rosado, Mario; Planelles, Paula; Ferran, Anna; Pla, Carmina; Azuara, Paula; Prats, Juan
Authors Company: Parc Tauli Hospital Universitari
Background: The prognostic value of pretreatment lymphocyte to monocyte ratio (LMR) in patients with renal cell carcinoma and, especially, in non-metastatic patients remains controversial. Therefore, we herein conducted a meta-analysis to systematically assess the prognostic value of LMR in patients with non-metastatic renal cell carcinoma.
Methods: A systematic review of the literature was performed using the PICO method (patient, intervention, comparison and outcome), with the aim of answering the following clinical question: “Is pretreatment low LMR a prognostic factor in non-metastatic renal cell carcinoma?”. PubMed was used to search for articles published from January 1965 to July 2019 using the key words “renal cancer”, “lymphocyte to monocyte ratio” and “prognosis”. No restrictions to date, language, or article type were applied. Cohort or observational studies in patients with non-metastatic cell carcinoma histolopathologically confirmed, with hazard ratios (HR) and corresponding 95% confidence intervals (CI) that assessed association between LMR and overall survival (OS), cancer-specific survival (CSS), recurrence-free survival (RFS), and disease-free survival (DFS) were analyzed. The quality of the studies was evaluated by the New Castle Ottawa scale. Data were combined using random effect models. The Cochrane Chi-square (Cochrane Q) statistic and the I-square test were used to analyze heterogeneity. The publication bias was graphically explored through funnel plot, and Duval and Tweedie’s “trim-and-fill” test was used to correct possible publication bias. Statistics were done using R 3.5.0 (R Core Team, 2018) and the meta (v4.1-5; Schwarzer, Guido, 2019) package.
Results: Among 29 articles, 18 were selected to be fully read. The selection process yielded only seven studies with eligibly criteria for analysis, that gave information on 4666 patients with non-metastatic renal cell carcinoma. Of these studies, 5 reported the results of OS, 3 results of DFS, 2 results of PFS and 4 results of CSS. The synthesized analyses showed that a low LMR was associated with poorer CSS (fix-effect model, HR 3.04, 95% CI 2.05-4.51, P < 0.05). We obtained a heterogeneity chi-squared value Q exp=0. (p=0.82) (I-square = 0%). Funnel plot not suggested a possible publication bias in the analysis. However, the association between and low LMR and OS (fix effect model, HR 0.86, 95% CI 0.68-1.08, p>0.05), DFS (fix effect model, HR 0.66 95% CI 0.50-0.89, p>0.05) and RFS (fix effect model HR 0.97, 95% CI 0.68–1.39, P >0.05) did not obtain significance.
Fig 1.
The synthesized HR assessing the prognostic value of the pretreatment LMR for CSS.
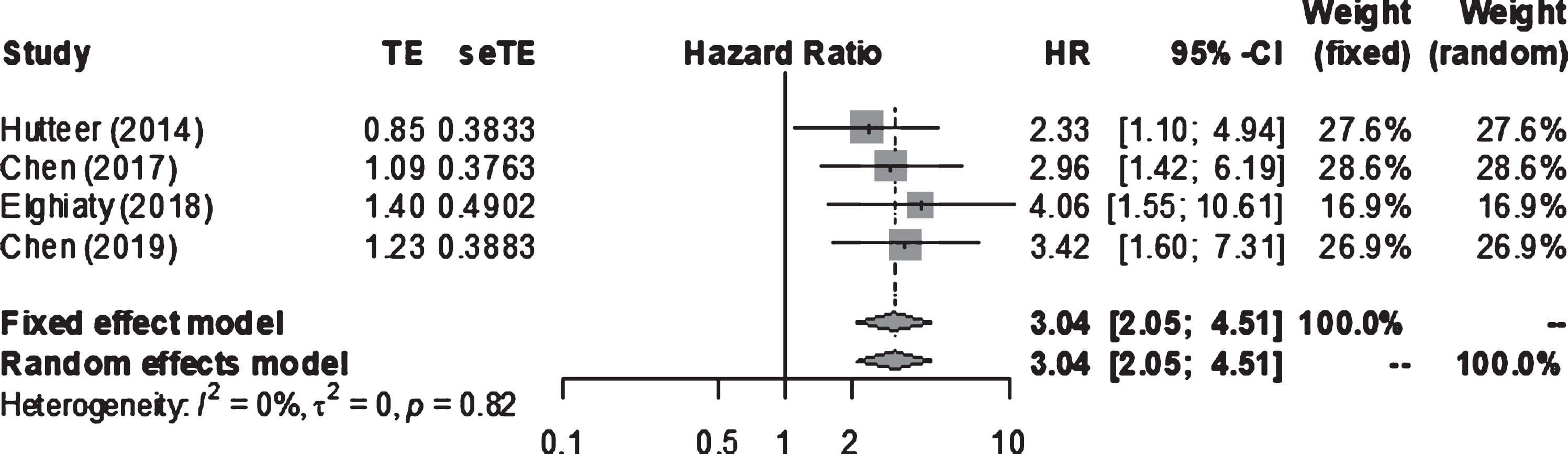
Fig 2.
The funnel plot test for the publication bias assessment of the synthesized HR assessing the prognostic value of treatment LMR for CSS in non-metastatic renal cell carcinoma.
Conclusion: This meta-analysis demonstrated that a low pretreatment LMR was associated with poorer CSS, but non with poorer OS, DFS or RFS in patients with non-metastatic renal cell carcinoma. Nevertheless, more prospective studies with large samples are required to further confirm our findings before it is applied for daily clinical decision making.
59PROSPER: A phase III randomized study comparing perioperative nivolumab vs. observation in patients with renal cell carcinoma (RCC) undergoing nephrectomy (ECOG-ACRIN 8143)
First Author: Harshman, Lauren C.
Co-Authors: Allaf, Mohamad
Author Company: Dana-Farber Cancer Institute, Harvard Medical School, Johns Hopkins
PROSPER: A phase III randomized study comparing perioperative nivolumab vs. observation in patients with renal cell carcinoma (RCC) undergoing nephrectomy (ECOG-ACRIN 8143)Lauren C Harshman, Maneka Puligandla, Mohamad Allaf, David F. McDermott, Charles G. Drake, Sabina Signoretti, David Cella, Rajan T Gupta, Brian M. Shuch, Primo Lara, Anil Kapoor, Daniel Yick Chin Heng, Bradley Leibovich, M Dror Michaelson, Toni K. Choueiri, Viraj Master, Michael A.S. Jewett, Deb Maskens, Naomi B. Haas, Michael Anthony Carducci, On Behalf of the PROSPER RCC Investigators
Background: The anti-PD-1 antibody nivolumab improves overall survival in metastatic RCC and is well tolerated. There is no standard adjuvant systemic therapy that increases overall survival over surgery alone for non-metastatic RCC. Priming the immune system prior to surgery with anti-PD-1 improves survival compared to a pure adjuvant approach in mouse solid tumor models. Multiple phase 2 studies in bladder, lung and breast cancers have shown remarkable pathologic responses with neoadjuvant PD-1 blockade. Two ongoing phase 2 studies of perioperative nivolumab in M0 RCC patients are showing preliminary feasibility and safety with no surgical delays (NCT02575222; NCT02595918). With PROSPER RCC (NCT03055013), we aim to improve clinical outcomes by priming the immune system with neoadjuvant nivolumab prior to nephrectomy followed by continued engagement with adjuvant blockade in patients with high risk RCC compared to standard of care surgery alone.
Methods: This global, unblinded, phase 3 National Clinical Trials Network study is currently accruing patients with clinical stage =T2 or TanyN+ RCC of any histology planned for radical or partial nephrectomy. Select oligometastatic disease is permitted if the patient can be rendered ‘no evidence of disease’ within 12 weeks of nephrectomy (=3 metastases: no brain, bone or liver). To enhance accrual and patient quality of life we amended the study to extend nivolumab dosing to 480mg q4 weeks and to require the baseline tumor biopsy only in the nivolumab arm. The investigational arm receives 1 dose of nivolumab prior to surgery followed by 9 adjuvant doses. The control arm undergoes nephrectomy followed by observation, as is a standard of care. There is no placebo. Randomized patients are stratified by clinical T stage, node positivity, and M stage. Accrual of 805 patients provides 84.2% power to detect a 14.4% absolute benefit in recurrence-free survival (RFS) at 5 years assuming the ASSURE historical control of ~56% to 70% (HR = 0.70). The study is powered to evaluate a significant increase in OS (HR 0.67). Critical perioperative therapy considerations such as safety, feasibility, and quality of life endpoints have been integrated. PROSPER RCC embeds a wealth of translational work to investigate the contribution of the baseline immune milieu, the changes induced by neoadjuvant anti-PD-1 priming, and how both may predict clinical outcomes.
60Protocol for a feasibility study of a cohort embedded randomised controlled trial comparing NEphron Sparing Treatment (NEST) for small renal masses
First Author: Neves, Joana B.
Co-Authors: Cullen, David; Grant, Lee; Walkden, Miles; Bandula, Steve; Patki, Prasad; Barod, Ravi; Mumtaz, Faiz; Aitchison, Michael; Pizzo, Elena; Ranieri, Veronica; Williams, Norman; Wildgoose, William; Gurusamy, Kurinchi; Emberton, Mark; Bex, Axel; Tran, Maxine G. B.
Authors Company: University College London, Royal Free London NHS Foundation Trust
Introduction: Small renal masses (SRM;<=4cm) account for two thirds of new diagnoses of kidney cancer, the majority of which are incidental findings. The natural history of the SRM seems largely indolent. There is increasing concern regarding surgical overtreatment and the associated health burden in terms of morbidity and economy. Observational data supports the safety and efficacy of percutaneous cryoablation but there is an unmet need for high quality evidence on non-surgical management options and a head-to-head comparison with standard of care is lacking. Historical interventional trial recruitment difficulties demand novel study conduct approaches. We aim to assess if a novel trial design, the cohort embedded randomized controlled trial (RCT), will enable carrying out such a comparison.Methods and analysis: Single centre prospective cohort study of adults diagnosed with SRM (n=200) with an open label embedded interventional RCT comparing nephron sparing interventions. Cohort participants will be managed at patient and clinicians’ discretion and agree with longitudinal clinical data and biological sample collection, with invitation for trial interventions and participation in comparator control groups. Cohort participants with biopsy-proven renal cell carcinoma eligible for both percutaneous cryoablation and partial nephrectomy will be randomly selected (1:1) and invited to consider percutaneous cryoablation (n=25). The comparator group will be robotic partial nephrectomy (n=25). The primary outcome of this feasibility study is participant recruitment. Qualitative research techniques will assess barriers and recruitment improvement opportunities. Secondary outcomes are participant trial retention, health-related quality of life, treatment complications, blood transfusion rate, Intensive Care Unit admission and renal replacement requirement rates, length of hospital stay, time to return to pre-treatment activities, number of work days lost, and health technologies costs.Ethics: Ethical approval has been granted (UK HRA REC 19/EM/0004) Trialregistration: ISRCTN18156881 Funding: Facing up 2 Kidney Cancer, National Institute for Health Research (NIHR)
61Quality of Life in Previously Untreated Patients With Advanced Renal Cell Carcinoma (aRCC) in Checkmate 214: Updated Results
First Author: Cella, David
Co-Authors: Escudier, Bernard; Ivanescu, Cristina; Maurer, Matthew; Lord-Bessen, Jennifer; Gooden, Kyna
Authors Company: Northwestern University Feinberg School of Medicine, Gustave Roussey, IQVIA, Zuidoost, Bristol-Myers Squibb
Background: The primary health-related quality of life (HRQoL) ana Gustave Roussy lysis from CheckMate 214 has been published (Cella et al. Lancet Oncol 2019). Nivolumab plus ipilimumab (NIVO+IPI) led to superior overall survival (hazard ratio [HR], 0.63; P<0.001) and more favorable HRQoL than sunitinib (S) as first-line treatment for intermediate/poor-risk patients with aRCC. Here, we report HRQoL analyses from the 30-month follow-up.
Methods: Patients were randomized 1:1 to receive NIVO 3 mg/kg + IPI 1 mg/kg every 3 weeks for 4 doses, then NIVO 3 mg/kg every 2 weeks or S 50 mg/day orally for 4 weeks (6-week cycle). HRQoL was assessed on day 1 of weeks 1 and 4 of the first 2 cycles, on day 1 of weeks 1 and 5 of the next 2 cycles, and on day 1 of week 1 of subsequent cycles. An exploratory HRQoL analysis was conducted using the Functional Assessment of Cancer Therapy-Kidney Symptom Index (FKSI-19), Functional Assessment of Cancer Therapy-General (FACT-G), and EQ-5D instruments. The analyses included mixed-model repeated measures (MMRM) for change from baseline at 145 weeks (while on treatment) and time to deterioration (TTD).
Results: 1096 patients were randomized to NIVO+IPI (intermediate/poor risk, 425; favorable risk, 125) and S (intermediate/poor risk, 422; favorable risk, 124). HRQoL assessment completion rates were >78% in the first 145 weeks. In the total and intermediate/poor-risk populations, NIVO+IPI patients reported improved FKSI-19 total scores over time to week 145, while a decrease was observed with S. At 145 weeks ( Table), the difference in change from baseline between arms for FKSI-19 total, disease-related symptoms (DRS)-physical, and treatment side effects scores significantly favored NIVO+IPI versus S. TTD was statistically significantly longer with NIVO+IPI for most domains in both populations. Similar results were observed for FACT-G and EQ-5D change from baseline and TTD.
Table
MMRM analysis (treatment differences at week 145) and time to deterioration for FKSI-19
| Domain | LSM difference | NIVO+IPI vs S (95% CI) | TTD, months | HR (95% CI) | ||||
| All | Intermediate/Poor risk | All | Intermediate/Poor risk | |||||
| Total | 2.99 | (0.92-5.06)a | 4.24 | (1.38-7.09)a | 0.54 | (0.47-0.63)a | 0.54 | (0.46-0.63)a |
| DRS | 0.83 | (-0.15 to 1.82) | 1.18 | (-0.20 to 2.56) | 0.64 | (0.55-0.74)a | 0.66 | (0.56-0.79)a |
| DRS-Physical | 1.69 | (0.33-3.05)a | 2.49 | (0.58-4.40)a | 0.57 | (0.49-0.67)a | 0.58 | (0.49-0.69)a |
| DRS-Emotional | 0.23 | (-0.04 to 0.49) | 0.10 | (-0.26 to 0.46) | 0.90 | (0.74-1.09) | 0.90 | (0.73-1.13) |
| Treatment side effects | 0.73 | (0.26-1.20)a | 1.01 | (0.36-1.65)a | 0.42 | (0.36; 0.49)a | 0.45 | (0.38-0.53)a |
| Functional well-being | 0.47 | (-0.23 to 1.18) | 0.75 | (-0.22 to 1.72) | 0.76 | (0.66-0.88)a | 0.77 | (0.66-0.91)a |
aP<0.05.
CI, confidence interval; LSM, least squares mean.
Positive LSM or HR<1 favors NIVO+IPI vs S.
Conclusions: NIVO+IPI significantly improved overall survival versus S without worsening HRQoL. NIVO+IPI sustained long-term good HRQoL and significantly delayed TTD in both the total and intermediate/poor-risk populations.Originally presented at the European Society for Medical Oncology (ESMO) Congress; September 27-October 1, 2019; Barcelona, Spain.
62Radiomic signatures of CD8+ T cell infiltration and PD-L1 expression in clear-cell renal cell carcinoma (ccRCC)
First Author: Zahoor, Haris
Co-Authors: Sali, Akash; Aron, Manju; Siddiqui, Imran; Varghese, Bino; Cen, Steven; Lei, Xiaomeng; D’Souza, Anishka; Fullenkamp, Austin; Desai, Mihir; Gill, Inderbir; Quinn, David; Duddalwar, Vinay
Authors Company: USC Norris Comprehensive Cancer Center
Background: CD8+ T cell infiltration and programmed death-1 ligand (PD-L1) expression have been associated with enhanced treatment response to immune checkpoint inhibitors (ICI). However, these pathologic-based markers are associated with challenges of pathologic specimen requirement (biopsy), tumor heterogeneity and sampling variability. Radiomics, defined as translation of standard medical imaging data into quantitative data, may complement these markers and potentially overcome these limitations. We investigated computed tomography (CT)-based radiomic signatures and their association with CD8+ T cell infiltration and PD-L1 expression in patients with clear-cell renal cell carcinoma (ccRCC).
Methods: A retrospective analysis of patients with localized ccRCC who underwent surgical resection of their primary renal tumors between 2009-2018 was done. Patients with preoperative multiphase CT scan and available tumor tissue from the nephrectomy samples were included. Immunohistochemistry (IHC) for CD8+ T cells (Leica Bond-III automated IHC platform using Bio-Rad anti-CD8 monoclonal antibody clone 4B11) and PD-L1 (Leica Bond-III automated IHC platform using Abcam anti-PD-L1 monoclonal antibody clone 28-8) were performed. Manually segmented whole lesions from the multiphase CT acquisitions were evaluated using a radiomics panel comprising of 1708 metrics. Radiomic signatures were created from voxel-wise data analysis code developed in Matlab and tumor segmentation in Synapse 3D. Area under the curve (AUC) based on predicted probability from 48 iterations of leave-one-out cross-validation (LOOCV) testing data was used to assess robust discrimination accuracy.
Results: Fifty patients were included in this analysis. Of 50 patients, 16 (32%) were PD-L1 positive and 34 (68%) were PD-L1 negative based on tumor proportion score with a cut-off of =1% tumor cells. A total of 48/50 patients were evaluable for CD8 staining. Of these 48 patients, 25 (52%) were CD8 high and 23 (48%) were CD8 low based on a cut-off of 80 lymphocytes per high power field. A radiomic signature of CD8+ T cells was developed which was able to discriminate CD8 high and CD8 low tumors with an AUC of 0.9 (95% CI, 0.8 to 1). While we cannot rule out discovery by chance due to a small sample size, distribution of predicted probability using histogram showed a complete separation between CD8 high and CD8 low tumors around 0.5. Similarly, a radiomic signature of PD-L1 expression was developed which was able to discriminate PD-L1 positive and PD-L1 negative tumors with an AUC of 0.67 (95% CI, 0.5 to 0.84).
Conclusions: CT-based radiomic signatures can predict CD8+ T cell infiltration and PD-L1 expression in ccRCC. Further studies to validate these findings are being performed.
63Real-World Clinical Outcomes Of Patients With Metastatic Renal Cell Carcinoma (mRCC) Receiving First-Line (1L) Therapy In The United States (US) Veteran Population
First Author: Phatak, Hemant
Co-Authors: Zheng, Ying; Bhanegaonkar, Abhijeet; Kim, Ruth; Pandya, Shivani; Wang, Li; Krulewicz, Stan; Kasturi, Vijay
Author Company: EMD Serono, Inc., Pfizer Inc, STATinMED Research,
Background: Despite the expansion of the treatment options for patients (pts) with mRCC in the past decade, the survival rate among mRCC pts has plateaued, with most mRCC pts being treated with monotherapies in the 1L setting until early 2018. The current study aims to evaluate real-world clinical outcomes associated with 1L monotherapy in mRCC pts using US Veterans Health Administration (VHA) claims data and to quantify the unmet medical need.
Methods: This retrospective study identified newly diagnosed and newly treated adult pts with mRCC who initiated =1 line of therapy from Oct2013-Dec2017 (index date=1L therapy start date) and had continuous enrollment from =6 months prior to the mRCC diagnosis until =3 months post–index date. Second-line (2L) therapy was defined as switch to any non-index therapy. The Kaplan-Meier method was used to estimate the median overall survival (OS), time to treatment failure (TTF; time from index date to 1L discontinuation or death), and time to next treatment (TNT; time from index date to 2L start or death) in pts receiving tyrosine kinase inhibitor (TKI), mechanistic target of rapamycin (mTOR), immune checkpoint inhibitor (ICI), or other therapy.
Results: Of 809 mRCC pts, 85.0%, 7.7%, 4.7%, and 2.6% were treated with TKI, mTOR, ICI, or other therapy in 1L, respectively. Pt characteristics and clinical outcomes in each cohort are summarized in the table below.
Conclusions: The current descriptive non-comparative analyses provide new insights on the real-world clinical outcomes of mRCC pts in the VHA treated with 1L monotherapy. High unmet medical needs continue to persist as measured by limited TTF, TNT, and OS, despite the usage of the 4 monotherapy treatment classes. More efficacious combination treatment regimens (newly approved or in development) may address the medical needs and further improve the outcomes of mRCC pts.
| TKI (N=688) | mTOR(N=62) | ICI (N=38) | Others (N=21) | |
| Patient Characteristics | ||||
| Age (mean; years) | 68.7 | 67.1 | 67.0 | 65.1 |
| Quan CCI (mean) | 9.1 | 9.6 | 8.4 | 8.8 |
| Outcomes (median; months) | ||||
| OS | 25.5 | 10.6 | 19.1 | 37.3 |
| TNT | 8.6 | 3.9 | 13.3 | 8.5 |
| TTF | 3.5 | 1.9 | 2.9 | 0.7 |
| 1-year survival rate (%) | 68% | 48% | 63% | 76% |
64Real-world results from one year of therapy with tivozanib
First Author: Staehler, M.D., Michael
Co-Authors: Annabel K. Spek, Severin Rodler, Melanie Schott, Jozefina Casuscelli, Lena Mittelmeier, Marcus Schlemmer
Authors Company: University of Munich
Real-world results from one year of therapy with
Introduction: Tivozanib was approved in Europe in November 2017. We report on our initial experience of the use of Tivozanib in metastatic Renal Cell Carcinoma (RCC).
Material and Methods: N=23 patients undergoing Tivozanib therapy were included in this retrospective analysis from a prospective database at the Multidisciplinary Center on Renal Tumors, Department of Urology, University of Munich between Nov. 2017 and Oct. 2018.
Results: Median age was 69.1 years (range 42.7-83.8) n=8 patients were started on tivozanib in first line (34.8%) and n=15 (75.2% in later line therapy (2nd-6thline). Tumor response according to RECIST criteria was PR in 39.1%, SD in 52.2% and PD in 8.7% of the patients. Median progression free survival (PFS) was 14.9 months (95% CI 5.1-24.8). Median overall survival (OS) was not reached so far. Although statistically not significant there was a difference in median PFS for first line patients with 30.3 months versus later line patients with 8.6 months (CI 5.1-12.2) (p=0.291). Patients experiencing a PR had a median PFS of 30.2 months (CI NR-NR) and with SD it was 7.5 months (CI NR-NR) (p=0.266). Major side effects consisted of diarrhea, hypertension, fatigue and hoarseness. 34.8% of the patients had grade II side effects, namely diarrhea and fatigue and 21.7% had grade 3 adverse events, mostly diarrhea, leading to treatment discontinuation in 3 patients (13%).
Conclusion: Tivozanib is a highly active tyrosine kinase inhibitor even in later lines of therapy. The side effects are well tolerable and no new safety signals were detected.
65Real-World Treatment Patterns Among Patients With Metastatic Renal Cell Carcinoma Receiving First-Line Therapy in the United States Veteran Population
First Author: Bhanegaonkar, Abhijeet
Co-Authors: Kim, Ruth; Pandya, Shivani; Wang, Li; Krulewicz, Stan; Pennock, Gregory; Phatak, Hemant
Authors Company: EMD Serono, Inc., Pfizer, Inc. STATinMED Research
Background: To assess treatment patterns in first-line (1L) setting among US Veterans Health Administration patients with metastatic renal cell carcinoma (mRCC).
Methods: This retrospective study identified newly diagnosed adult patients with mRCC who initiated =1 line of therapy from 01Oct2013-31Dec2017 (index date=1L therapy start date) and had continuous health plan enrollment from =6 months prior to the mRCC diagnosis until =3 months post–index date. Second-line (2L) therapy was defined by switch to any non-index therapy. Duration of 1L therapy was measured from index date to the earliest of last prescription’s end date, 2L therapy start date, or follow-up end. Univariate Poisson regression was used to compare patients treated with tyrosine kinase inhibitor (TKI) therapy vs mechanistic target of rapamycin (mTOR), immuno-oncology (IO) therapy, or other therapy, separately, based on rates (in 100-person years) of continuation, permanent discontinuation, interruption, and switch to 2L therapy.
Results: Of 809 patients (mean age, 68.4y; 98.4% male; 79.9% white), 85.0%, 7.7%, 4.7%, and 2.6% were treated with TKI, mTOR, IO, or other therapy in 1L, respectively. Patients in the IO cohort were more likely to have baseline COPD (31.6% vs 16.7%; p=0.022) a higher continuation rate (47.7 vs 14.5; p<0.0001) and a lower rate of switching to 2L therapy (13.0 vs 50.3; p=0.0073) vs TKI cohort. Patients received mTOR had a higher rate of permanent discontinuation (49.0 vs 31.0; p=0.0334) and interruption (221.0 vs 122.9; p=0.0002). Duration of 1L therapy (in days) was similar for TKI vs. IO (153.3 vs133.2, p=0.3372), longer for TKI vs. mTOR (153.3 vs 96.9, p=0.0055) and TKI vs. other therapy (153.3 vs 42.9, p<0.0001).
Conclusions: Among patients with mRCC, although 1L TKI therapy showed similar duration vs IO, IO therapy demonstrated higher continuation rates vs TKI. Future research should focus on understanding which patients would benefit from IO+TKI.
66Renal cell carcinoma induced by end stage renal disease: A population-based study in Sweden
First Author: Åkerlund, John
Co-Authors: Holmberg, Erik; Lindblad, Per; Ljungberg, Börje; Stendahl, Maria; Torstenson, Andreas; Lundstam, Sven
Authors Company: Sahlgrenska University Hospital, Örebro University, Umeå University, Swedish Renal Registry, Karolinska Institute
Introduction: End stage renal disease (ESRD) is a known risk factor for renal cell carcinoma (RCC) and for benign renal tumors in the native kidneys. It is unknown to what extent RCC developed in patients with ESRD differs from RCC among the general population without ESRD.
Material and Methods: 9,416 patients with RCC identified in the National Swedish Kidney Cancer Register from 2005 to 2014 and 94,159 matched controls, were linked to the Swedish Renal Registry, the National Patient Register and the Cause of Death Register. ESRD was defined as Chronic Kidney Disease 5 (CKD 5) or treatment with hemodialysis 0–40 (median 9) years before the diagnosis of RCC.
Results: 115 patients with ESRD and subsequent RCC were identified and compared with 9,087 patients with RCC but without ESRD before the diagnosis of RCC. Median age at diagnosis was 64 vs. 67 years. Median tumor diameter was smaller (4.8 vs. 6 cm), lesser were high stage (64 vs. 53% were T1; 10 vs. 15% T2; 18 vs. 27% T3–T4 and 13 vs. 19% were M1).
Tumor histopathology revealed 57 vs. 79% clear cell RCC, 25 vs. 11% papillary and 8 vs. 5% chromophobe RCCs.
Radical nephrectomy was performed in 71 vs. 67% and partial nephrectomy in 4 vs. 16% of patients. Sixty-one percent of the ESRD patients received a renal transplant.
There was a statistically significant higher risk for RCC (p < 0.001), with odds ratio 5.5 (95% CI: 4.4–6.9), when ESRD patients were compared with those without ESRD.
The 5- and 10-year overall survival was 50% and 26% respectively in patients with ESRD before the diagnosis of RCC and 63% and 47% respectively in patients with RCC without ESRD (p < 0.05).
Conclusion: End stage renal disease is a risk factor for RCC. Compared to RCC patients without ESRD the tumors are smaller, have a lower stage and are less often metastatic at the time of detection. Furthermore, there were less frequently clear cell RCCs and more frequent papillary and chromophobe RCCs. In spite of having less advanced tumors at diagnosis, the overall survival in ESRD patients with RCC is lower compared to patients with RCC without ESRD.
67Resectable non-clear cell renal cell carcinoma: 25 years of single-center experience
First Author: Mukhomediarova, Albina
Co-Authors: Alekseev, Boris; Vorobiev, Nikolay; Kaprin, Andrey; Kalpinskiy, Alexey
Authors Company: Moscow Hertzen Research Oncology Institute – filial of FGBI “National Medical Research Center of Radiology” of the Russian Federation Ministry of Heal
Introduction & objectives: Non-clear cell renal cell carcinoma (nccRCC) is a heterogeneous group of rare renal tumors with limited survival data and lack of well-known predictors of survival. The aim was to assess progression-free survival (PFS) and cancer-specific survival (CSS) of patients with different types of nccRCC and evaluate the association between tumor pathology characteristics and survival.
Materials & Methods: A single-institute retrospective review of renal cancer database was conducted. Newly diagnosed patients with localized and locally-advanced nccRCC who underwent surgical resection between 1993 -2018 were included in analysis. Postoperative pathology reports were reviewed and tumor characteristics were collected. Spearman’s correlation analysis was performed. Survival was analyzed with the Kaplan-Meier method. Results: A review of 2040 patients yielded 265 (13%) nccRCC patients. Mean patient age was 57.3 (16-76). Of 265 included patients, 120 (6.7%) had papillary tumor, 102 (5.7%) had chromophobe tumor, 4 (0.2%) had collecting-duct carcinoma, 18 (1 %) had mixed type, and 10 (0.6%) had other rare variants of nccRCC. There was following pathology stage distribution: 126 (47.4%) patients with pT1a, 67 (25.2%) with pT1b, 17 (6.4%) with pT2a, 6 (2.2%) with pT2b, 28 (10.5%) with pT3a, and 3 (1.1%) with pT3b. Seventeen (6.4%) patients had pN+. Median follow-up was 26 months (1-258mo). 5-year PFS of patients with papillary type 1 patients was 93.1%, papillary type 2 - 70.3%, and with chromophobe tumor – 93.4% ( p=0.01). 5-year CSS for the same tumor subtypes was 93.5%, 77.4%, 97.1%, respectively (p=0.02). There was statistically significant correlation between probability of disease progression and pT stage (R= 0.27), tumor size (R= 0.27), vascular invasion (R= 0.44), Fuhrman grade (R=0.15), stage pN+ (R = 0.19), and type of nccRCC (R = 0.17)p <0.05. Probability of death due to RCC progression had low correlation with pT stage (R = 0.16), tumor size (R = 0.12), vascular invasion (R = 0.25), stage pN + (R = 0.14), and Fuhrman grade (R = 0.18), p <0.05.
Conclusions: PFS and CSS vary between different pathology types of nccRCC with the most unfavorable pattern in papillary type 2 tumors. Classical pathology parameters, such as pT, lymph node status, tumor size, presence of vascular invasion, and Fuhrman grade have low correlation with progression rate and the probability of death from nccRCC. The further investigation of survival predictors is needed.
68Safety and efficacy of nivolumab plus ipilimumab (NIVO+IPI) in patients with advanced renal cell carcinoma (aRCC) with brain metastases: interim analysis of CheckMate 920
First Author: Emamekhoo, Hamid
Co-Authors: Carthon, Bradley; Olsen, Mark; Drakaki, Alexandra; Percent, Ivor; Molina, Ana; Cho, Daniel; Bendell, Johanna; Gordan, Lucio; Rezazadeh-Kalebasty, Arash; George, Daniel; Hutson, Thomas; Lee, Richard; Young, Tina; Johansen, Jennifer; Tykodi, Scott
Authors Company: University of Wisconsin School of Medicine and Public Health, Emory University, Winship Cancer Institute, Oklahoma Cancer Specialists and Research Institute, University of California Los Angeles, Florida Cancer Specialists South/Sarah Cannon Research Institute, Weill Cornell Medicine, New York University Langone Health, Sarah Cannon Research Institute/Tennessee Oncology, Norton Cancer Institute, Duke University Medical Center, Texas A&M College of Medicine, Bristol-Myers Squibb, University of Washington and Fred Hutchinson Cancer Research Center
Background: Previous clinical trials in aRCC, including CheckMate 214, have mostly excluded patients with brain metastases. However, antitumor activity in patients with brain metastases has been observed in patients with melanoma treated with NIVO 1 mg/kg + IPI 3mg/kg and patients with non-small cell lung cancer treated with NIVO 240 mg + IPI 1mg/kg. CheckMate 920 is an ongoing, phase 3b/4 trial of NIVO+IPI in patients with aRCC, a population with high unmet medical need. We present interim safety and efficacy results for the brain metastases cohort.
Methods: Patients with previously untreated aRCC of any histology, with asymptomatic brain metastases (not on corticosteroids or receiving radiation), and Karnofsky performance status = 70% were assigned to NIVO 3 mg/kg + IPI 1 mg/kg Q3W for 4 doses, followed by NIVO 480 mg Q4W. Patients were treated until disease progression or unacceptable toxicity, or for a maximum of 2 years. The primary endpoint was incidence of high-grade (= grade 3) immune-mediated adverse events (IMAEs). Key secondary endpoints included progression-free survival (PFS) and objective response rate (ORR) by RECIST v1.1 per investigator. Exploratory endpoints included additional safety analyses and overall survival (OS).
Results: Overall, 28 patients were enrolled in the brain metastases cohort. With a minimum follow-up of 6.47 months, 6 cases of grade 3–4 IMAEs were reported within 100 days of last dose. Grade 3–4 IMAEs observed in =1 patient were diarrhea, colitis, diabetic ketoacidosis, immune-mediated hepatitis, hypophysitis, and rash of any type (n=1 each). No treatment-related grade 5 IMAEs were reported. ORR by RECIST v1.1 per investigator in all treated subjects was 28.6% (95% CI, 13.2–48.7). Median PFS in all treated patients was 9.0 months (95% CI, 2.9–not estimable [NE]). Median OS has not been reached (95% CI, 13.1 months–NE).
Conclusions: In patients with aRCC and brain metastases a population often excluded from clinical trials, the safety profile of NIVO+IPI was consistent with previous reports of this dosing regimen, with encouraging antitumor activity.Reused with permission from the American Society of Clinical Oncology (ASCO). This abstract was previously presented at the 2019 ASCO Annual Meeting. All rights reserved.
69Safety and Feasibility of Surgery Following Preoperative Immunotherapy in Patients with Metastatic Renal Cell Carcinoma
First Author: Attalla, Kyrollis
Co-Authors: DiNatale, Renzo; Silagy, Andrew; Marcon, Julian; Weng, Stanley; Chen, Yingbei; Reznik, Ed; Dalbagni, Guido; Coleman, Jonathan; Lee, Chung-Han; Carlo, Maria; Voss, Martin; Motzer, Robert; Russo, Paul; Hakimi, A. Ari
Authors Company: Memorial Sloan Kettering Cancer Center
Introduction: Despite theoretical and demonstrated preclinical benefits of immune checkpoint inhibition (ICI) in the preoperative setting, the role of preoperative ICI in patients with metastatic renal cell carcinoma (mRCC) is not well defined. We report the safety and feasibility of surgery following preoperative administration of ICI in patients with mRCC.
Methods: A retrospective review was performed on an institutional database identifying patients with mRCC who received ICI and were subsequently selected for cytoreductive nephrectomy.
Results: A total of 10 patients were identified. Of these, 8 received combination ipilimumab/nivolumab and 2 received nivolumab monotherapy. Complete (R0) resection was achieved in 9/10 patients. Estimated blood loss was 353 mL and length of stay was 2.2 days. There were no 30-day readmissions, mortality, or operative morbidity. Grade =3 ICI related adverse events prior to surgery were seen in 3/10 patients, 2 patients developing hepatitis and 1 patient with pulmonary toxicity. A pathologic response in the primary tumor was seen in 4/10 patients, all of whom received combination therapy; 2/10 patients achieved a complete pathologic response. One patient achieved a near-complete response with greater than 99% of the primary tumor necrotic and only a single microscopic focus of viable tumor present, and another patient achieved a partial pathologic response, with pre-nephrectomy biopsy demonstrating sarcomatoid features, of which none was seen on final pathologic examination.
Conclusions: We demonstrate that surgery can be performed safely in mRCC patients receiving preoperative ICI, emphasizing the need for further prospective investigations into the efficacy and optimal treatment regimens of ICI in this setting. Pre- and post-nephrectomy molecular characterizations are underway.
70Treatment-Free Survival, With and Without Toxicity, as a Novel Outcome Applied to Immuno-Oncology Agents in Advanced Renal Cell Carcinoma
First Author: Regan, Meredith M.
Co-Authors: Atkins, Michael; Powles, Thomas; Werner, Lillian; Mantia, Charlene; Yang, Shuo; Johansen, Jennifer; Rao, Sumati; Gooden, Kyna; McDermott, David F.
Authors Company: Dana-Farber Cancer Institute, Georgetown-Lombardi Comprehensive Cancer Center, Barts Cancer Institute, Beth Israel Deaconess Medical Center, Bristol-Myers Squibb
Background: Conventional measures such as median progression-free survival may suboptimally characterize the full impact of immuno-oncology (I-O) agents versus other systemic anticancer therapies. Patients discontinuing I-O agents may experience periods of disease control without needing subsequent systemic anticancer therapy but may still experience toxicity. Treatment-free survival (TFS) ± toxicity can simultaneously characterize disease control and toxicity for this off-treatment period.
Methods: Data were analyzed from all patients initiating therapy on the randomized phase 3 CheckMate 214 trial of nivolumab plus ipilimumab (NIVO+IPI) versus sunitinib (SUN) for treatment-naïve predominantly clear cell advanced renal cell carcinoma. TFS was defined as the area between Kaplan–Meier curves for 2 conventional time-to-event endpoints defined from randomization: time to protocol therapy cessation and time to subsequent therapy or death. TFS was subdivided as TFS with and without toxicity by defining a third endpoint: time to cessation of therapy and toxicity. Toxicity was defined as grade =3 treatment-related adverse events. Area under each Kaplan–Meier curve was estimated by the 36-month restricted mean time to event.
Results: In total, 1082 patients initiated therapy (547 patients in the NIVO+IPI arm and 535 patients in the SUN arm). At 36 months, 60% of NIVO+IPI and 51% of SUN patients were alive, 15% of NIVO+IPI and 9% of SUN patients remained on original therapy, and 34% of NIVO+IPI and 19% of SUN patients were surviving free of subsequent therapy. The 36-month restricted mean TFS was 6.7 versus 2.9 months for all NIVO+IPI versus SUN patients, respectively (6.4 vs 2.8 months TFS without toxicity). The Table shows time by TFS subdivision and International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) risk.
Conclusions: NIVO+IPI provides longer survival and delayed time to subsequent therapy versus SUN. More importantly, NIVO+IPI provides longer TFS without toxicity, during which patients do not require treatment and are free from toxicity. Given the durability of I-O responses relative to SUN after treatment cessation, it will be of interest to measure TFS over time. Originally presented at the European Society for Medical Oncology (ESMO) Congress; September 27–October 1, 2019; Barcelona, Spain.
| IMDC risk group | All | Favorable | Intermediate/Poor | ||||||
| NIVO+IPI N=547 | SUN N=535 | Difference | NIVO+IPI N=124 | SUN N=119 | Difference | NIVO+IPI N=423 | SUN N=416 | Difference | |
| OS, months | 28.0 | 25.6 | 32.0 | 32.9 | 26.9 | 23.5 | |||
| Time on protocol therapy, months | 13.4 | 12.7 | 13.5 | 20.1 | 13.4 | 10.6 | |||
| TFS, months(95% CI) | 6.7 | 2.9 | 3.8 (2.4-5.3) | 9.8 | 2.7 | 7.2 (4.4-10.0) | 5.8 | 3.0 | 2.8 (1.7-4.0) |
| TFS with toxicity | 0.4 | 0.1 | 0.2 (0.05-0.4) | 0.5 | 0.1 | 0.4 (0.2-0.6) | 0.3 | 0.2 | –0.1 (–0.03 to 0.3) |
| TFS without toxicity | 6.4 | 2.8 | 3.6 (2.3-5.0) | 9.4 | 2.6 | 6.8 (4.1-9.5) | 5.5 | 2.8 | 2.7 (1.6-3.8) |
| Survival after subsequent therapy initiation, months | 7.9 | 10.0 | 8.7 | 10.2 | 7.7 | 10.0 | |||
CI, confidence interval; OS, overall survival.




