Sleep Disorders in Early-Onset Parkinsonism: A Population-Based Study
Abstract
Background:
Sleep disturbances are common in parkinsonian disorders; however, whether sleep disorders affect individuals with early-onset parkinsonism and whether they differ from individuals with typical-onset parkinsonism is unknown.
Objective:
To compare the prevalence and incidence of sleep disorders before and after parkinsonian motor symptom onset between individuals with early onset parkinsonism (age ≤50 at motor symptom onset) and typical-onset parkinsonism (age >50 at motor symptom onset).
Methods:
We used a population-based, 1991 to 2015 incident-cohort study of parkinsonism including 38 patients with early-onset and 1,001 patients with typical-onset parkinsonism. Presence or absence and type of sleep disorder as well as the relationship between motor and sleep symptoms were abstracted from the medical records. Rates of sleep disorders before and after onset of parkinsonism were compared with logistic regression and Cox proportional hazards models.
Results:
The prevalence of sleep disorders prior to the onset of parkinsonism in early vs. typical parkinsonism (24% vs. 16% p = 0.19) and incidence of sleep disorders after parkinsonism onset (5.85 cases per 100 person-years vs. 4.11 cases per 100 person-years; HR 1.15 95% CI: 0.74–1.77) were similar between the two groups. Early-onset parkinsonism had a higher risk for developing post-motor insomnia compared with typical-onset parkinsonism (HR 1.73, 95% CI: 1.02–2.93); the risk for developing all other sleep disorders considered was similar between groups.
Conclusion:
Sleep disorders are common in individuals with early-onset parkinsonism and occur with similar frequency to those with typical-onset parkinsonism, except for insomnia, which was more frequent in the early-onset group.
INTRODUCTION
Sleep disorders, including REM sleep behavior disorder (RBD), insomnia, hypersomnia, and sleep-disordered breathing, are common in parkinsonian disorders, negatively affecting the quality of life of patients and caregivers and tend to increase over time as the disease progresses [1–4]. However, the distribution of specific sleep disorders tends to differ among the etiologies of parkinsonism (i.e., RBD tends to be more common in synucleinopathies compared with tauopathies) [5]. Further, sleep disorders may evolve prior to, in conjunction with, or after the development of motor symptoms [4, 6]. To date, most of the research on sleep disorders in parkinsonism has been focused on those with typical-onset degenerative parkinsonism or been age-agnostic in regards to parkinsonism onset, with little research published on sleep disorders in early-onset Parkinson’s disease (PD) or parkinsonism. The relative rarity of early-onset PD compared with typical PD and heterogenous definitions of “early-onset” historically used have limited the ability to study various symptoms, including sleep, in early-onset PD. However, a consensus definition of early-onset parkinsonism as parkinsonism-symptom onset between age 21 and 50 years has recently been proposed to address this limitation [7].
The few studies evaluating sleep disturbances in early-onset PD have used differing age cutoffs for early-onset PD and largely focused on survey measures to assess sleepiness and sleep quality, namely the Epworth Sleepiness Scale (ESS) and Pittsburg sleep quality index (PSQI). Individuals with typical-onset PD tend to be worse sleepers (as denoted by higher scores on PSQI) and have a higher frequency of insomnia but similar levels of daytime sleepiness as measured by the ESS compared with early-onset PD [8, 9]. The frequency of RBD as assessed by the RBDSQ was similar between early-onset PD and typical-onset PD in one study while probable RBD was less frequent in early-onset than typical-onset PD patients in another study [8, 10]. The frequency of restless legs syndrome (RLS) and sleep-disordered breathing has not been studied in early-onset PD nor specifically compared with typical-onset PD.
Given the limited data on sleep disorders in early-onset PD, it is unknown whether frequency and type of sleep disorders differ in those with early-onset parkinsonism compared with those of typical-onset parkinsonism. Further knowledge of sleep disorders impacting those with early-onset PD would be beneficial to provide individualized care and maximize quality of life in patients with early-onset PD [11]. The objective of this study was 1) to compare the type and frequency of sleep disorders between individuals with early-onset parkinsonism (age ≤50 at parkinsonism onset) vs. those with typical-onset parkinsonism in a population-based study in Olmsted County, MN USA; and 2) to determine whether or not the relationship between parkinsonian motor-symptom onset and sleep-symptom onset differed between early- and typical-onsetparkinsonism.
METHODS
2.1Case ascertainment
Parkinsonism patients were identified using a population-based incident cohort in Olmsted County from 1991 to 2015 utilizing the Rochester Epidemiology Project (REP), which has a population catchment rate of >97% of individuals in Olmsted County and allows for the review of life-long medical histories of consenting participants from any medical setting within this county [12, 13]. Early-onset parkinsonism was defined as motor-symptom onset at or before age 50 whereas typical-onset parkinsonism was defined as motor-symptom onset after age 50 [7]. Potential parkinsonism patients were identified using the REP, and all identified cases were reviewed to confirm diagnosis. The date when the presence of 1 of the 4 cardinal features of parkinsonism was first documented in the medical record was defined as the onset of motor symptoms. Details of case ascertainment have been previously reported [12, 13]. Diagnoses included PD, dementia with Lewy bodies (DLB), multiple system atrophy (MSA), progressive supranuclear palsy (PSP), corticobasal degeneration (CBD), amyotrophic lateral sclerosis-parkinsonism, drug-induced parkinsonism, vascular parkinsonism, and surgically induced parkinsonism [12, 13]. Diagnoses were based on the clinical criteria utilized at the time of diagnosis rather than at the time of retrospective chart review. Methodology of case identification and diagnoses has been reported previously [12]. Cases not meeting criteria for a specific type of parkinsonism were classified as unspecified parkinsonism.
A comprehensive medical record review was performed to confirm the presence and onset of sleep disorders including snoring, obstructive sleep apnea (OSA), RLS, probable REM sleep behavior disorder (pRBD), insomnia, narcolepsy, or hypersomnia not meeting narcolepsy criteria were determined by the treating providers documentation [14–16]. Regarding sleep-disordered breathing specifically, those with a diagnosis of OSA were formally diagnosed with OSA whereas those who either noted snoring without confirmatory OSA testing or patients with documented snoring who underwent OSA evaluation without confirmation were considered to have snoring. Diagnoses were made based on the diagnostic criteria at the time of diagnosis and not at the time of chart review with the onset of sleep disorder symptoms determined based on the time of their first mention in the medical record. Pre-motor sleep symptoms were defined as sleep symptoms documented prior to the onset of parkinsonian motor symptoms whereas post-motor sleep symptoms developed after (and were documented after) the onset ofparkinsonism.
Statistical analysis
Continuous characteristics were summarized with means and standard deviations as well as medians with interquartile ranges; categorical features were summarized with frequency counts and percentages. Patient sex and parkinsonism subtype were compared between early-onset and typical-onset parkinsonism patients using two-sided Fisher’s exact tests.
The frequency of pre-motor sleep disorders was compared between early-onset and typical-onset parkinsonism using logistic regression adjusted for patient sex. The risk of developing post-motor sleep disorders was evaluated using Cox proportional-hazards regression adjusted for patient sex and duration of available follow-up. Cox models used the onset of parkinsonism as the reference time point and only included patients without a given sleep disorder at parkinsonism onset. Results were considered statistically significant for p-values<0.05. Exploratory analysis comparing those with suspected early-onset PD due to a degenerative etiology with typical-onset PD due to presumed degenerative etiology are reported descriptively given few subjects in the early-onset subgroup.
Standard protocol approvals, registrations, and patient consents
The Mayo Clinic and Olmsted Medical Center Institutional Review Boards approved this study. The participating patients (or their legally authorized representatives) provided informed written consent for the use of their medical information for research.
RESULTS
Demographics
A total of 1,039 parkinsonism patients were identified with 38 (3.7%) cases meeting early-onset parkinsonism criteria. Average age of parkinsonism onset was 42.6±7.7 years for patients with early onset and 74.9±9.8 years in patients with typical onset. There were no sex differences between early and typical-onset cases of parkinsonism. Table 1 summarizes patient demographics and disease characteristics.
Table 1
Patient demographics and parkinsonism disease characteristics in early- and typical-onset parkinsonism
| Typical-Onset | Early-Onset | Overall | |
| (N = 1,001) | (N = 38) | (N = 1,039) | |
| Age at Disease Onset, y | |||
| Mean (SD) | 74.9 (9.8) | 42.6 (7.7) | 73.7 (11.4) |
| Median (Q1, Q3) | 75.9 (67.7, 81.7) | 44.5 (41.0, 48.7) | 75.4 (67.1, 81.6) |
| Sex, n (%) | |||
| Female | 402 (40.2%) | 15 (39.5%) | 417 (40.1%) |
| Male | 599 (59.8%) | 23 (60.5%) | 622 (59.9%) |
| Parkinsonism Type, n (%) | |||
| Parkinson’s Disease | 435 (43.5%) | 17 (44.7%) | 452 (43.5%) |
| Dementia with Lewy Bodies | 153 (15.3%) | 0 (0%) | 153 (14.7%) |
| Parkinson’s Disease with Dementia | 77 (7.7%) | 0 (0%) | 77 (7.4%) |
| Drug-Induced Parkinsonism | 57 (5.7%) | 16 (42.1%) | 73 (7.0%) |
| Progressive Supranuclear Palsy | 38 (3.8%) | 0 (0%) | 38 (3.7%) |
| Multiple System Atrophy | 21 (2.1) | 2 (5.3%) | 23 (2.2%) |
| Vascular Parkinsonism | 15 (1.5%) | 1 (2.6%) | 16 (1.5%) |
| Corticobasal Syndrome | 7 (0.7%) | 0 (0%) | 7 (0.7%) |
| Surgical Parkinsonism | 1 (0.1%) | 1 (2.6%) | 2 (0.2%) |
| ALS Parkinsonism | 1 (0.1%) | 0 (0%) | 1 (0.1%) |
| Unspecified Subtype | 196 (19.6%) | 1 (2.6%) | 197 (19.0%) |
SD, standard deviation; Q1, 1st quartile, Q3, 3rd quartile; ALS, amyotrophic lateral sclerosis.
Early-onset patients were more commonly diagnosed with drug-induced parkinsonism compared to typical-onset patients (42% vs. 6%; p < 0.001) whereas typical-onset patients were more commonly diagnosed with dementia with Lewy bodies (15% vs. 0%; p = 0.004) and unspecified parkinsonism (20% vs. 3%; p = 0.005). Rates of PD were nearly identical between the two cohorts (early-onset 45%; typical-onset 44%). Frequency of motor symptoms were similar between early-onset and typical-onset parkinsonism patients except impaired postural reflex, which was significantly less common among early-onset patients compared to typical-onset patients (29% vs. 60%; p < 0.001). The duration of follow-up was 9.9 years (IQR 7.4–12.6) for typical-onset patients and 15.1 years (IQR 12.2–19.5) for early-onset patients.
Sleep disorders
Table 2 summarizes the prevalence of pre-motor and post-motor sleep disorders in patients early and typical-onset parkinsonism. There were 158 (16%) typical-onset patients diagnosed with ≥1 pre-motor sleep disorder, compared with 9 (24%) early-onset patients. There were no differences in pre-motor sleep disorders between early-onset and typical-onset patients after adjusting for patient sex (OR = 1.08, 95% CI: 0.96–1.22, p = 0.19). Among patients not diagnosed with a pre-motor sleep disorder, there were 258 (31%) of 843 typical-onset patients who developed ≥1 post-motor sleep disorder compared with 17 (59%) of 29 early-onset patients. The incidence of any post-motor sleep disorders was similar between the groups, with 4.11 cases per 100 person-years for typical-onset patients and 5.85 cases per 100 person-years for early-onset patients, adjusting for duration of available follow-up and patient sex (HR = 1.15, 95% CI: 0.74–1.77, p = 0.54).
Table 2
Frequency of sleep disorders in parkinsonism between early- and typical-onset parkinsonism
| Typical-Onset | Early-Onset | Overall | |
| (N = 1001) | (N = 38) | (N = 1039) | |
| Probable REM Sleep Behavior Disorder, n (%) | |||
| None | 910 (90.0%) | 32 (84.2%) | 942 (90.7%) |
| Pre-Motor Onset | 16 (1.6%) | 0 (0%) | 16 (1.5%) |
| Post-Motor Onset | 75 (7.5%) | 6 (15.8%) | 81 (7.8%) |
| Restless Legs Syndrome, n (%) | |||
| None | 883 (88.2%) | 31 (81.6%) | 914 (88.0%) |
| Pre-Motor Onset | 36 (3.6%) | 0 (0%) | 36 (3.5%) |
| Post-Motor Onset | 82 (8.2%) | 7 (18.4%) | 89 (8.6%) |
| Obstructive Sleep Apnea, n (%) | |||
| None | 871 (87.0%) | 30 (78.9%) | 901 (86.7%) |
| Pre-Motor Onset | 50 (5.0%) | 2 (5.3%) | 52 (5.0%) |
| Post-Motor Onset | 80 (8.0%) | 6 (15.8%) | 86 (8.3%) |
| Snoring, n (%) | |||
| None | 963 (96.2%) | 35 (92.1%) | 998 (96.1%) |
| Pre-Motor Onset | 19 (1.9%) | 1 (2.6%) | 20 (1.9%) |
| Post-Motor Onset | 19 (1.9%) | 2 (5.3%) | 21 (2.0%) |
| Narcolepsy, n (%) | |||
| None | 998 (99.7%) | 38 (100%) | 1036 (99.7%) |
| Pre-Motor Onset | 2 (0.2%) | 0 (0%) | 2 (0.2%) |
| Post-Motor Onset | 1 (0.1%) | 0 (0%) | 1 (0.1%) |
| Insomnia, n (%) | |||
| None | 724 (72.3%) | 16 (42.1%) | 740 (71.2%) |
| Pre-Motor Onset | 89 (8.9%) | 6 (15.8%) | 95 (9.1%) |
| Post-Motor Onset | 188 (18.8%) | 16 (42.1%) | 204 (19.6%) |
| Hypersomnolence, n (%) | |||
| None | 790 (78.9%) | 26 (68.4%) | 816 (78.5%) |
| Pre-Motor Onset | 50 (5.0%) | 2 (5.3%) | 52 (5.0%) |
| Post-Motor Onset | 161 (16.1%) | 10 (26.3%) | 171 (16.5%) |
Insomnia
A total of 89 (9%) typical-onset parkinsonism patients were diagnosed with pre-motor insomnia, compared with 6 (16%) early-onset patients. This did not differ after adjusting for patient sex (OR = 1.07, 95% CI: 0.98–1.18, p = 0.15).
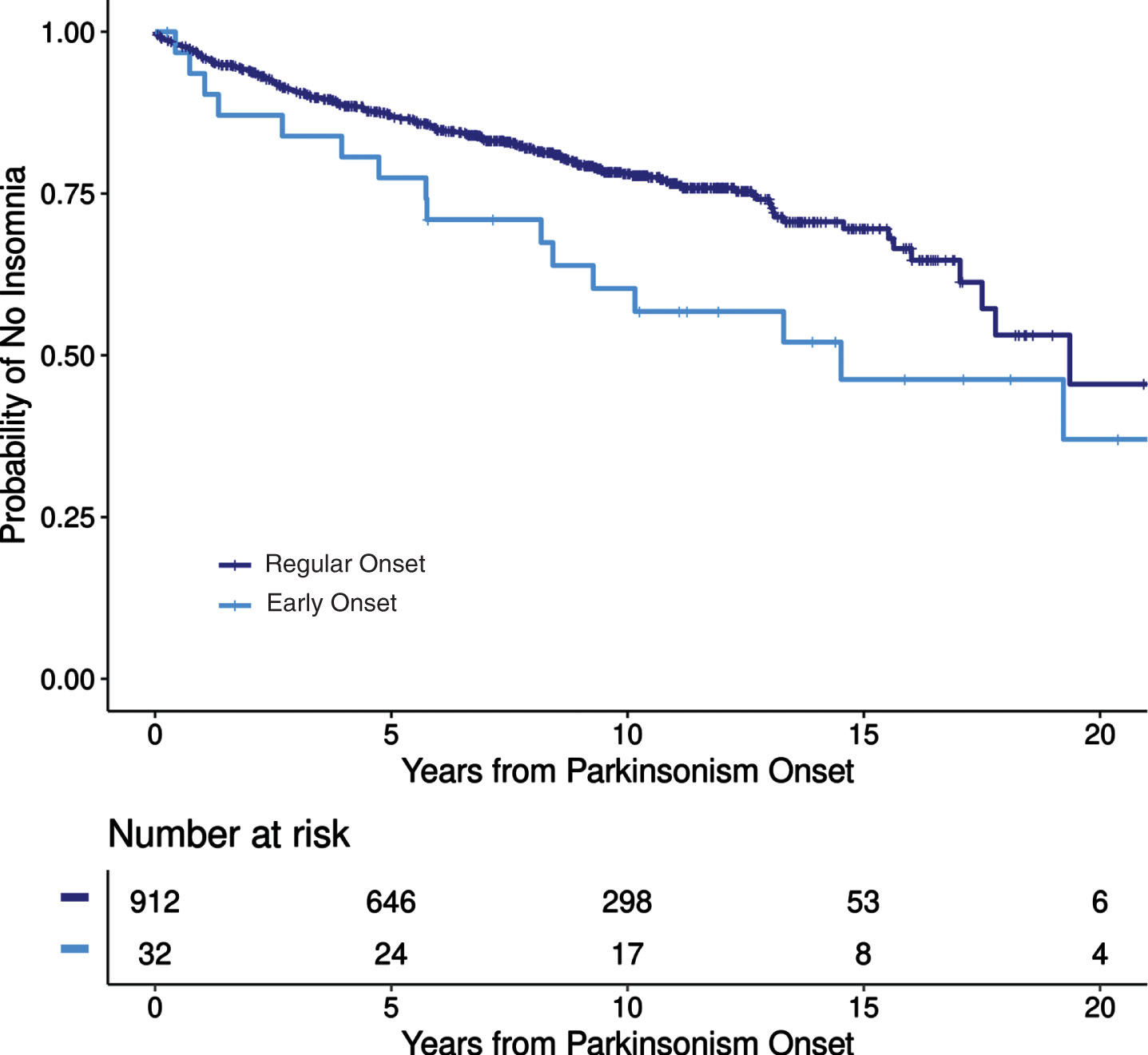
Among patients without pre-motor insomnia, 188 (21%) of 912 typical-onset patients and 16 (50%) of 32 early-onset patients developed post-motor insomnia. Early-onset patients had 73% greater risk of developing post-motor insomnia compared to typical-onset patients after adjusting for length of follow-up and patient sex (HR = 1.73, 95% CI: 1.02–2.93, p = 0.042, Fig. 1).
Fig. 1
Incidence of insomnia after motor symptoms onset in early-onset and typical-onset parkinsonism.
Probable REM sleep behavior disorder
A total of 16 (2%) typical-onset parkinsonism patients were diagnosed with pre-motor pRBD, compared with 0 early onset patients. After adjusting for sex, there was no difference in pre-motor pRBD between the cohorts (OR = 0.98, 95% CI: 0.95–1.02, p = 0.43).
Among patients without pre-motor pRBD, 75 (8%) of 985 typical-onset patients and 6 (16%) of 38 early-onset patients developed post-motor pRBD. Of note, only 1 early-onset pRBD patient underwent overnight polysomnography but did not achieve sufficient REM sleep for a diagnosis of definite RBD. After adjusting for length of follow-up and sex, there was no difference in post-motor pRBD between the cohorts (HR = 0.94, 95% CI: 0.37–2.34, p = 0.89).
Restless legs syndrome
A total of 36 (4%) typical-onset parkinsonism patients were diagnosed with pre-motor RLS, compared with 0 early-onset patients, which was no different after adjusting for patient sex (OR = 0.96, 95% CI: 0.91–1.02, p = 0.24). Among patients without pre-motor RLS, 82 (8%) of 965 typical-onset patients, and 7 (18%) of 38 early-onset patients developed post-motor RLS, which was also similar between groups after adjusting for length of follow-up and sex (HR = 1.40, 95% CI: 0.64–3.09, p = 0.40).
Obstructive sleep apnea and snoring
Fifty (5%) typical-onset parkinsonism patients were diagnosed with pre-motor OSA, compared with 2 (5%) early-onset patients, which was similar between cohorts (OR = 1.00, 95% CI: 0.93–1.08, p = 0.94). A total of 19 (2%) typical-onset parkinsonism patients were diagnosed with pre-motor snoring, compared with 1 (3%) early onset patients, which also did not differ between cohorts (OR = 1.01, 95% CI: 0.96–1.05, p = 0.75).
Among patients without pre-motor OSA, 80 (8%) of 951 typical-onset patients and 6 (17%) of 36 early-onset patients developed post-motor OSA. Among patients without pre-motor snoring, 19 (2%) of 982 typical-onset patients and 2 (5%) of 37 yearly-onset patients developed post-motor snoring. After adjusting for length of follow-up and patient sex, there was no difference in post-motor OSA or snoring between the cohorts (HR = 1.24, 95% CI: 0.53–2.90, p = 0.62 and HR = 2.26, 95% CI: 0.52–9.77, p = 0.28, respectively).
Narcolepsy and hypersomnolence
Two typical-onset parkinsonism patients were diagnosed with pre-motor narcolepsy while only one typical-onset parkinsonism patient was diagnosed with post-motor narcolepsy compared with zero early-onset patients for both pre- and post-motor narcolepsy. Fifty (5%) typical-onset parkinsonism patients were diagnosed with pre-motor hypersomnolence, compared with 2 (5%) early-onset patients., which was similar between groups (OR = 1.00, 95% CI: 0.93–1.08, p = 0.94). Among patients without pre-motor hypersomnolence, 161 (17%) of 951 typical-onset patients and 10 (28%) of 36 early-onset patients developed post-motor hypersomnolence, which was no different after adjusting for length of follow-up and patient sex (HR = 0.99, 95% CI: 0.52–1.91, p = 0.99).
Sleep disorders in early-onset PD and drug-induced parkinsonism
Given the importance of understanding differences in early- and typical-onset neurodegenerative PD we performed additional analysis in the subgroup of 15 early-onset PD and 435 typical-onset PD patients. There were no differences between typical-onset and early-onset PD in pre-motor sleep disorders (13% in each group). However, median latency of pre-motor sleep-disorder symptom onset to motor-symptom onset was shorter in early-onset PD 0.4 (IQR 0.2–0.5) years compared with 4.0 (IQR 1.7–9.5) years for typical-onset patients. A total of 154 (41%) of 378 typical-onset patients developed at least one post-motor sleep disorder compared with 9 (69%) of 13 early-onset patients, although there was no difference after adjusting for sex and follow-up duration. The incidence of any post-motor sleep disorder was 5.43 cases per 100 person-years for typical-onset PD patients and 7.03 cases per 100 person-years for early-onset PD patients.
As drug induced parkinsonism made up the second largest group of early-onset cases we performed additional analysis in this cohort. 10/57 (18%) of typical-onset drug induced parkinsonism patients developed at least one sleep disorder prior to parkinsonism onset compared with 6/16 (38%) early-onset drug-induced patients. After adjusting for patient sex, early-onset drug-induced patients were more likely to have a sleep disorder prior to parkinsonism onset (OR = 1.29, 95% CI: 1.01–1.64, p = 0.043). There were no differences in post-motor sleep disorders between early and late onset drug-induced cases although due to the limited number of patients in the early-onset subgroups limited confident interpretation of this analysis.
There were no differences in the prevalence or incidence of specific pre- and post-motor sleep disorders between early-onset and typical-onset PD. However, insomnia mirrored the results from the entire whole cohort, with 116 (29%) of 403 typical-onset patients and 9 (64%) of 14 early-onset patients developing post-motor insomnia, although this was not significant after adjusting for length of follow-up and patient sex (HR = 1.71, 95% CI: 0.86–3.43, p = 0.13). No early-onset PD patients had pre-motor probable RBD compared with 5 (1%) of typical-onset PD, while 5 (33%) early-onset PD patients developed post-motor probable RBD compared with 46 (11%) typical-onset PD, although these were not statistically different between groups.
DISCUSSION
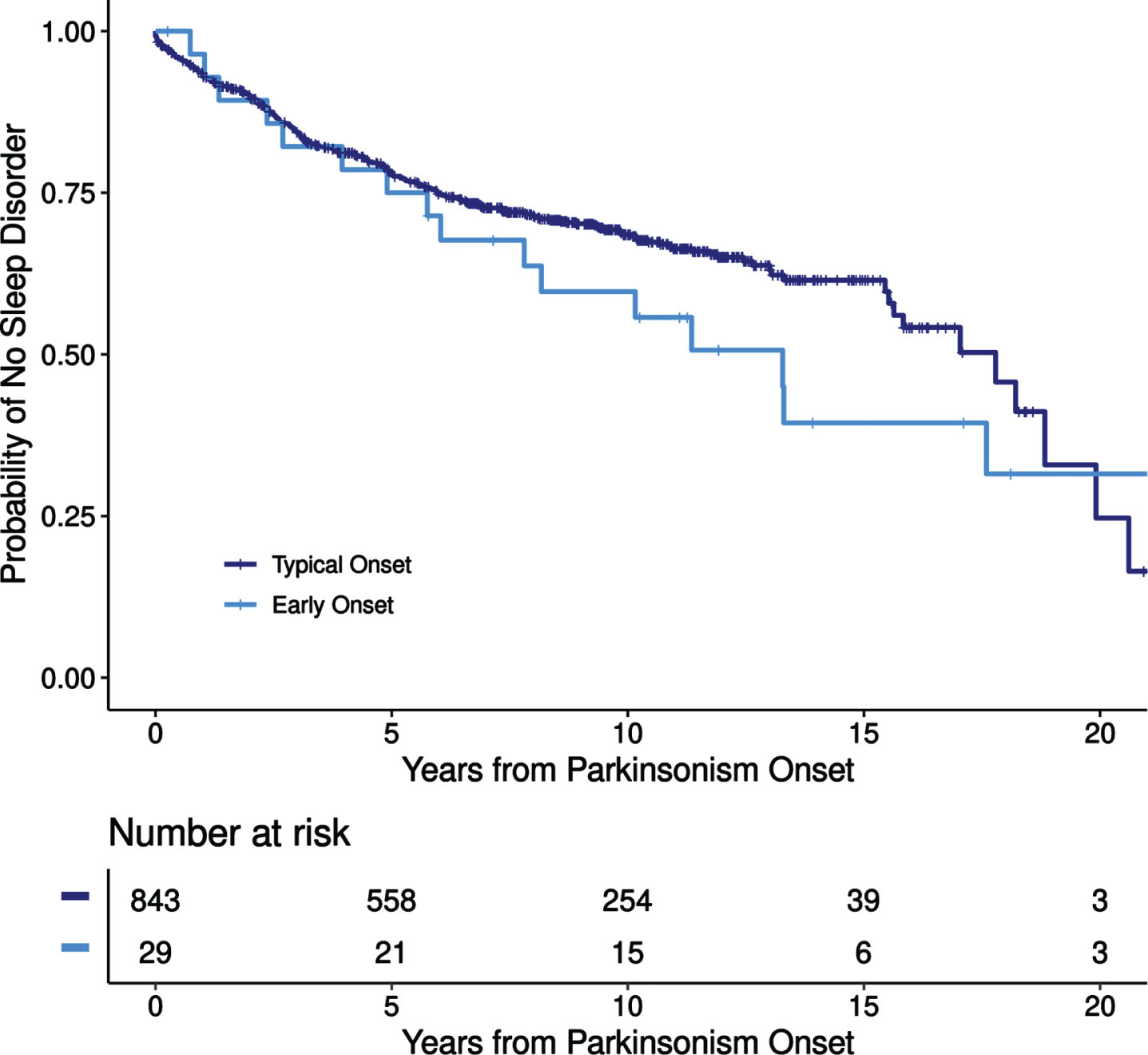
Our study yielded several noteworthy findings about sleep disturbances in early-onset parkinsonism. First, sleep disorders are common in both early- and typical-onset parkinsonism, particularly developing after parkinsonism-symptom onset, with similar overall incidence of post-motor sleep disorders between groups. While RBD is a well-established prodromal alpha-synucleinopathy, particularly in typical cases of parkinsonism, the frequency of other sleep disorders prior to the onset parkinsonism is not well established, particularly for early-onset parkinsonism [17]. Overall, we found that the prevalence of pre-motor sleep disorders were similar between groups (16% vs. 24%; typical-onset vs. early-onset) with a similar incidence in post-motor sleep disorders between groups of 4.11 cases per 100 years in typical-onset patients compared with 5.85 cases per 100 years in early-onset patients with increasing incidence over time (Fig. 2). Our findings of increasing incidence of sleep disorders over time is consistent with prior reports of increasing frequency of sleep disorders with PD disease progression [3, 11]. The reason for increasing incidence of sleep disorders over time is likely multifactorial. First, provider bias based on parkinsonism symptoms likely increased the likelihood of specifically querying about post-motor sleep disorders, especially RBD. Further, medications utilized to treat parkinsonian symptoms, such as carbidopa/levodopa or dopamine agonists, are known to impact sleep and certainly could have contributed to sleep disturbances, along with progression of parkinsonism itself. Additionally, given the relatively high frequency of drug-induced parkinsonism, particularly in the early-onset group, these medications likely also influence sleep and contribute to a high incidence of post-motor sleep disturbance. Further, mood-disorder comorbidities and/or other medications such as antidepressants, which are common in patients with parkinsonism, could have also contributed to sleep disturbances but were not systematically assessed as part of this study.
Fig. 2
Incidence of post-motor sleep disorders in early- and typical-onset parkinsonism.
We found that individuals with early-onset parkinsonism had a 73% greater risk of insomnia after the onset of parkinsonian-motor symptoms compared to individuals with typical-onset parkinsonism. Our results differ from prior studies, which have typically reported higher incidence of insomnia or poorer sleep quality in patients with typical-onset PD [8, 9]. There are multiple explanations for these differences. First, prior studies utilized different definitions of early-onset, with one study using an age cutoff of 40 years; thus, the higher frequency of insomnia in “typical” parkinsonism may have been due to age [9]. Second, our study is a population-based sample compared with patients recruited from neurology clinics; thus inherent differences in patient populations contributed to differences from the current study and previous reports. Additionally, as nearly 50% of our early-onset parkinsonism cases were drug-induced, it is likely that contributions from medications and/or underlying diseases for which medications were required (which were most commonly psychiatric) increased the frequency of insomnia in early-onset parkinsonism [18].
When comparing subgroups of early-onset PD with typical-onset PD due to neurodegenerative etiology, results largely mirrored the results of the entire cohort. There was a trend toward increased incidence of post-motor insomnia in the early-onset PD group indicating overall group differences in insomnia were not purely due to the high frequency of drug-induced parkinsonism in the early-onset group. This suggests an inherent difference in etiologies of insomnia in early-onset PD, although additional studies are needed to understand the potential mechanisms. Some prior studies have reported a higher incidence of rigidity in early-onset PD, which could contribute to difficulties with sleep initiation or maintenance; however, this was not the case in our study, suggesting other factors influenced insomnia in our early-onset cohort [19]. Most importantly, our results highlight the importance of screening patients diagnosed with early-onset PD for insomnia so that appropriate treatment or cognitive behavioral therapy strategies can be implemented. We also found that latency of sleep disorders to motor symptoms was much shorter in early-onset PD compared to typical-onset (0.4 years (IQR 0.2–0.5) vs. 4.0 years (IQR 1.7–9.5), although the significance of this will require replication given few numbers of early-onset PD patients with pre-motor sleep disorders but in part likely reflects the absence of pre-motor pRBD in our early-onset cases. Further studies with a larger cohort of neurodegenerative early-onset PD are needed to confirm our findings.
While there were no statistical differences between groups, given the relevance of REM sleep behavior disorder to parkinsonism, additional commentary is warranted. Little is known about the frequency of RBD in early-onset PD. One study reported a frequency of probable RBD by questionnaire in 28% in early-onset PD (EOPD criteria <50 years) that did not differ from typical PD, although timing of RBD symptoms in relationship to motor symptoms was not reported [7]. Another study found a significantly lower frequency of probable RBD in early-onset PD of 7.8% compared with typical-onset PD. This study used a definition of 20 to 40 years for early-onset PD, possibly explaining differences from our and the prior study [10]. This study also reported that the majority of patients had RBD symptoms after the onset of parkinsonian motor symptoms, similar to what was found in our study, although they did not specify if there were differences between early- and typical- onset cases [10]. We found an overall pRBD frequency of 33% in early-onset PD (only 1 case had PSG but insufficient REM sleep for diagnosis), similar to prior studies using the <50-year definition of early-onset PD. Further, all patients developed RBD after onset of motor symptoms, suggesting RBD may manifest differently in early-onset PD compared with typical-onset PD, where RBD is often a pre-motor manifestation [20]. This highlights the importance of consistent screening of early-onset PD patients for dream-enactment behaviors so that appropriate bedroom-safety counseling and treatment can be implemented in hopes of preventing dream enactment behavior-related injury [21].
Our study has notable limitations. As a retrospective study relying on medical records, ascertainment bias likely influenced our results as sleep symptoms were not uniformly sampled in clinical practice or by survey measures, and some sleep symptoms were not reported by patients or identified by the clinician. Because of this, our study likely underestimates the frequency and incidence of sleep disorders in our cohort, which is supported by the less-than-expected prevalence of both OSA and pRBD in our cohort compared with what has been found in a large population-based study of aging in Olmsted County, MN, although that study formally assessed sleep symptoms by survey, likely explaining differences between that and the current study [22]. However, as sleep symptoms were of sufficient severity to warrant clinical documentation or evaluation, our prevalence estimates likely reflect at least the minimal prevalence and incidence of clinically significant sleep disorders in both pre- and post-motor parkinsonism. The presence of a diagnosis of parkinsonism also likely influenced provider querying about sleep disturbances, particularly RBD, which could also influence results and we did not systematically collect medication use or medical comorbidities which may also influence sleep.
As our study spans multiple decades, awareness of specific sleep disorders (specifically RBD, which was identified in 1986 but became more widely known in the late 2000s) as well as differing diagnostic criteria used during different decades may have influenced our results. Our reliance on clinical medical record documentation for diagnosis also introduces a potential bias due to the use of heterogenous methodologies for arriving at a specific diagnosis. For instance, some provides may diagnose hypersomnia based on symptoms alone without full hypersomnia workup to assess for narcolepsy although this would be expected to impact all patients equally. The lack of polysomnographic evaluation in all patients included in our population-based cohort very likely led to underdiagnosis (particularly for RBD and OSA) of sleep disorders in our cohort, considering that many RBD patients are unaware of their abnormal nocturnal behaviors [23].
The overall relatively small number of patients in the early-onset group limited our ability to compare individual subgroups of parkinsonism confidently, thus further studies are required to better delineate the differences in sleep disorders between specific etiologies of early and late onset parkinsonism. Additionally, as our study is a population-based study with varying etiologies of parkinsonism being diagnosed by different specialists or general practitioners (i.e., Psychiatry may be more likely to encounter drug-induced parkinsonism) the potential for ascertainment bias in identification of motor parkinsonism is possible. Finally, while some genetic etiologies of PD are associated with sleep disorders, such as mutations in the LRRK2 and GBA genes, this was not formally assessed in this study.
Overall, sleep disorders appear to impact patients with early-onset and typical-onset parkinsonism to a similar degree, apart from insomnia, which appears to develop more frequently after parkinsonian motor symptoms in early-onset cases. Mechanisms for the different insomnia frequencies between groups are likely multifactorial and requires further study. However, providers caring for individuals with early-onset parkinsonism should be aware of the increased risk of insomnia in these patients so that appropriate management strategies can be implemented to prevent insomnia-related worsening quality of life. Future well-designed studies consistently utilizing the recently proposed criteria of motor-symptom onset between age 21 and 50 for early-onset parkinsonism with structured sleep assessments and objective testing are needed to replicate and expand upon the findings of the current study [7].
ACKNOWLEDGMENTS
The authors would like to thank Lea Dacy for assistance with editing and manuscript submission.
FUNDING
Institutional research grant support: Alector, Biogen, Transposon, Cognition Therapeutics, GE Healthcare; NIH grant support: P30 AG062677, U19 AG063911, R01 AG038791, U01 NS100620, U19 AG071754, U24 AG056270; foundation support: Lewy Body Dementia Association, American Brain Foundation; philanthropic support: Mayo Clinic Dorothy and Harry T. Mangurian Jr. Lewy Body Dementia Program, the Little Family Foundation, the Ted Turner and Family Functional Genomics Program.
R Savica reports research support from the National Center for Advancing Translational Sciences (NCATS), a component of the National Institutes of Health (NIH), Michael J. Fox Foundation and Acadia Pharmaceuticals Inc.
CONFLICT OF INTEREST
SJ McCarter, E Camerucci, AF Mullan, CD Stang, and P Turcano, report no conflicts of interest.
EK St. Louis reports research funding support from NIH and Spark, Inc.
BF Boeve reports honorarium for SAB activities for the Tau Consortium.
R Savica is an Editorial Board member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review.
DATA AVAILABILITY
All the relevant data have been shared and published in this article; data regarding case ascertainment of parkinsonism and methodology on case identification have been previously published [13].
REFERENCES
[1] | Pandey S , Bajaj BK , Wadhwa A , Anand KS ((2016) ) Impact of sleep quality on the quality of life of patients with Parkinson’s disease: A questionnaire based study. Clin Neurol Neurosurg 148: , 29–34. |
[2] | Perez E , Perrin PB , Lageman SK , Villasenor T , Dzierzewski JM ((2022) ) Sleep, caregiver burden, and life satisfaction in Parkinson’s disease caregivers: A multinational investigation. Disabil Rehabil 44: , 1939–1945. |
[3] | Xu Z , Anderson KN , Saffari SE , Lawson RA , Chaudhuri KR , Brooks D , Pavese N ((2021) ) Progression of sleep disturbances in Parkinson’s disease: A 5-year longitudinal study. J Neurol 268: , 312–320. |
[4] | De Cock VC , Vidailhet M , Arnulf I ((2008) ) Sleep disturbances in patients with parkinsonism. Nat Clin Pract Neurol 4: , 254–266. |
[5] | Boeve BF ((2010) ) REM sleep behavior disorder: Updated review of the core features, the REM sleep behavior disorder-neurodegenerative disease association, evolving concepts, controversies, and future directions. Ann N Y Acad Sci 1184: , 15–54. |
[6] | Xu Z , Anderson KN , Pavese N ((2022) ) Longitudinal studies of sleep disturbances in Parkinson’s disease. Curr Neurol Neurosci Rep 22: , 635–655. |
[7] | Mehanna R , Smilowska K , Fleisher J , Post B , Hatano T , Pimentel Piemonte ME , Kumar KR , McConvey V , Zhang B , Tan EK , Savica R , International Parkinson and Movement Disorder Society Task Force on Early Onset Parkinson’s Disease ((2022) ) Age cutoff for early-onset Parkinson’s disease: Recommendations from the International Parkinson and Movement Disorder Society Task Force on Early Onset Parkinson’s Disease. Mov Disord Clin Pract 9: , 869–878. |
[8] | Liu M , Luo YJ , Gu HY , Wang YM , Liu MH , Li K , Li J , Zhuang S , Shen Y , Jin H , Chen J , Mao CJ , Liu CF ((2021) ) Sex and onset-age-related features of excessive daytime sleepiness and night-time sleep in patients with Parkinson’s disease. BMC Neurol 21: , 165. |
[9] | Mahale R , Yadav R , Pal PK ((2015) ) Quality of sleep in young onset Parkinson’s disease: Any difference from older onset Parkinson’s disease. Parkinsonism Relat Disord 21: , 461–464. |
[10] | Mahale R , Yadav R , Pal PK ((2014) ) Rapid eye movement sleep behaviour disorder in young- and older-onset Parkinson disease: A questionnaire-based study. Sleep Med 15: , 642–646. |
[11] | Liguori C , De Franco V , Cerroni R , Spanetta M , Mercuri NB , Stefani A , Pierantozzi M , Di Pucchio A ((2021) ) Sleep problems affect quality of life in Parkinson’s disease along disease progression. Sleep Med 81: , 307–311. |
[12] | Camerucci E , Stang CD , Hajeb M , Turcano P , Mullan AF , Martin P , Ross OA , Bower JH , Mielke MM , Savica R ((2021) ) Early-onset parkinsonism and early-onset Parkinson’s disease: A population-based study (2010-2015). J Parkinsons Dis 11: , 1197–1207. |
[13] | Savica R , Grossardt BR , Bower JH , Ahlskog JE , Rocca WA ((2013) ) Incidence and pathology of synucleinopathies and tauopathies related to parkinsonism. JAMA Neurol 70: , 859–866. |
[14] | ((1979) ) Diagnostic classification of sleep and arousal disorders. 1979 first edition. Association of Sleep Disorders Centers and the Association for the Psychophysiological Study of Sleep. Sleep 2: , 1–154. |
[15] | (2005) International Classification of Sleep Disorders, 2nd edition. American Academy of Sleep Medicine, Westch- ester, IL. |
[16] | (2005) International Classification of Sleep Disorders, 2nd edition. American Academy of Sleep Medicine, Westch-ester, IL. |
[17] | Stang CD , Mullan AF , Hajeb M , Camerucci E , Turcano P , Martin P , Mielke MM , Josephs KA , Bower JH , St Louis EK , Boeve BF , Savica R ((2021) ) Timeline of rapid eye movement sleep behavior disorder in overt alpha-synucleinopathies. Ann Neurol 89: , 293–303. |
[18] | DeMartinis NA , Winokur A ((2007) ) Effects of psychiatric medications on sleep and sleep disorders. CNS Neurol Disord Drug Targets 6: , 17–29. |
[19] | Mehanna R , Moore S , Hou JG , Sarwar AI , Lai EC ((2014) ) Comparing clinical features of young onset, middle onset and late onset Parkinson’s disease. Parkinsonism Relat Disord 20: , 530–534. |
[20] | St Louis EK , Boeve AR , Boeve BF ((2017) ) REM Sleep behavior disorder in Parkinson’s disease and other synucleinopathies. Mov Disord 32: , 645–658. |
[21] | Howell M , Avidan AY , Foldvary-Schaefer N , Malkani RG , During EH , Roland JP , McCarter SJ , Zak RS , Carandang G , Kazmi U , Ramar K ((2023) ) Management of REM sleep behavior disorder: An American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 19: , 759–768. |
[22] | Molano JR , Boeve BF , Roberts RO , Geda YE , Christianson TJ , Knopman DS , Smith GE , Ivnik RJ , Pankratz VS , Silber MH ((2009) ) Frequency of sleep disorders in the community-dwelling elderly: The Mayo Clinic Study of Aging. Neurology 72: , A107. |
[23] | Fernandez-Arcos A , Iranzo A , Serradell M , Gaig C , Santamaria J ((2016) ) The clinical phenotype of idiopathic rapid eye movement sleep behavior disorder at presentation: A study in 203 consecutive patients. Sleep 39: , 121–132. |




