ExoBand, A Passive Wearable Device as a Walking Aid in Neuromuscular Patients: First Quantitative Assessment
Abstract
Objective:
Exoband (by Moveo, Padova, Italy) functions as a walking brace, comprising a belt and two leg loops connected by a mechanism that stores energy during the initial phase of the gait cycle and releases it in the subsequent phase. This enhances hip flexor thrust, leading to functional improvement in walking for individuals with conditions characterized by proximal weakness. It has been approved as a passive wearable device for individuals with impaired walking abilities. Objective of this study was to establish a protocol to assess the use of Exoband in patients with various neuromuscular disorders.
Methods:
This exploratory retrospective study includes consecutive patients diagnosed with neuromuscular disorders (CIDP, motor polyneuropathy, MND), exhibiting a proximal involvement and gait abnormalities. The evaluation protocol incorporated specific walking-related outcome measures, the 10-meter walk test (10mWT), Time-up-and-go test (TUG), and 2-minute walking test (2MWT). The assessments were conducted both with and without the Exoband under standard conditions.
Results:
Eight patients (6 males, aged 60–78 years) were tested. An increase in velocity was observed in the 10mWT (median 13.4 sec, IQR 12.0–15.7 vs. 12.2 sec, IQR 11.3–14.2 seconds, p < 0.05) and the TUG (14.0 sec, IQR 13–16.2 vs 13.35 sec, IQR 11–13.8; p < 0.05, by non-parametric Wilcoxon test), and a trend of increase in 2MWT (median 88.2 vs 92.6 m, n.s.). Six out of 8 patients reported subjective benefits from the very first use, including improved walking stability, speed, confidence, and reduced fatigue.
Conclusions:
Our protocol provides a quantitative assessment of Exoband usefulness for patients affected by neuropathies with gait abnormalities. Further investigations are warranted to assess the long-term effects of its regular Exoband use, its efficacy in specific neuromuscular diseases, and its potential role as a rehabilitation device.
INTRODUCTION
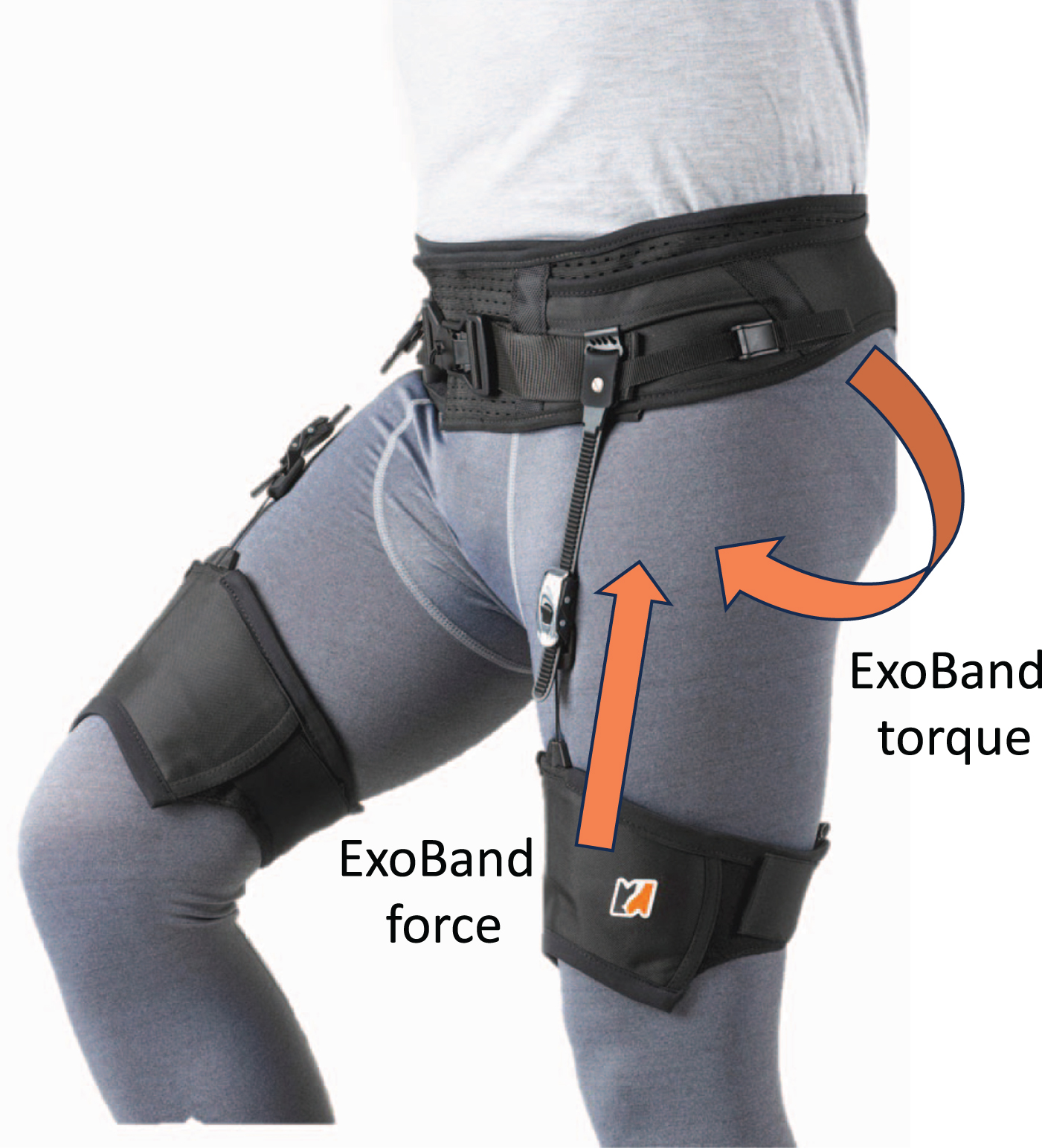
ExoBand (by Moveo, Padova, Italy), a passive wearable device, functions as a walking brace comprising a belt and two leg loops (Fig. 1). It is equipped with a mechanism that stores energy generated during the initial phase of the gait cycle, releasing it in the subsequent phase. This mechanism enhances hip flexor thrust, resulting in functional improvement in walking [1, 2]. The device has received approval for individuals with impaired walking abilities, demonstrating increased walked distance and improved gait pattern in patients with multiple sclerosis, Parkinson disease, stroke sequelae [3]. However, there is no formal study in patients with proximal weakness due to neuromuscular diseases or peripheral nervous system involvement. Aim of this exploratory study was to define a clinical protocol to be used for the quantitative assessment of ExoBand on motor function in patients with neuromuscular diseases (chronic inflammatory demyelinating polyneuropathy, CIDP; motor neuropathy; Kennedy’s disease), and for the qualitative evaluation of the patients’ impressions after using the device
Fig. 1
The device ExoBand by Moveo srl (Padova, Italy).
MATERIALS AND METHODS
Medical authorities of Padova mandate a clinical evaluation and quantitative analysis of gait and motor function for each patient applying for ExoBand, to qualify for reimbursement. We, therefore, conducted a retrospective review of the clinical records of the patients who sought consultation at the Neurology Unit of S. Antonio Hospital, University Hospital of Padova, between January and March 2023. Selection criteria included patients diagnosed with neuromuscular diseases exhibiting proximal muscle weakness (4 + /5 on hip flexion or below measured with Manual muscle testing scored with MRC scale) accompanied by gait abnormalities clinically defined by a neurologist (waddling gait, instability, fatigability). Focus of this review was on those individuals for whom prescription of the ExoBand was considered.
The evaluation protocol was administered by a trained evaluator and encompassed the following walking-related outcome measures:
∘ 10-meter walk test (10mWT)
∘ Time-up-and-go (TUG) test
∘ 2-minute walk test (2MWT)
The last test was introduced later in the observational period, following a specific request of the local medical authorities, and had been applied only in the last three evaluated patients without any clinical selection.
All tests were conducted without and with ExoBand, under standard conditions, on the same day, with sufficient interval between tests to ensure adequate rest.
Following the ExoBand trial, all patients underwent interviews to gather their first impressions, and free-form responses were recorded after the questions: “How do you feel with the device? What is your first impression about it?”.
Statistical analysis to compare matched test results conducted without and with the device used non-parametric Wilcoxon signed-rank test, because of the low numerosity of the study population. Values are expressed as median and interquartile range (IQR). Significance was set p < 0.05.
RESULTS
Eight patients (6 males and 2 females, aged 60–78 years) were evaluated. Five patients were diagnosed with chronic inflammatory demyelinating polyneuropathy (CIDP), while three had other neuromuscular conditions (motor polyneuropathy, Kennedy’s disease, post-infectious motor polyradiculopathy). Four patients consistently used walking aids.
The timed tests were performed after 15 minutes familiarization with the device, and the results are shown in Fig. 2. Significant and consistent improvements were observed in 10mWT (median 13.4 sec without the device, IQR 12.0–15.7, vs. 12.2 sec wearing the device, IQR 11.3–14.2, p < 0.05, Fig. 2a) and TUG test (14.0 sec without the device, IQR 13.0–16.2, vs 13.35 sec wearing the device, IQR 11.0–13.8, p < 0.05, Fig. 2b).
Fig. 2
a-c. Timed test performed in standard conditions without the device (w/o, left) or wearing ExoBand (device, right). Each red line corresponds to one patient; in black, mean and standard deviation. 2d. Patients’ impressions after the trial with the device. Each line corresponds to one patient. In green are positive or very positive impressions, in yellow uncertain ones.
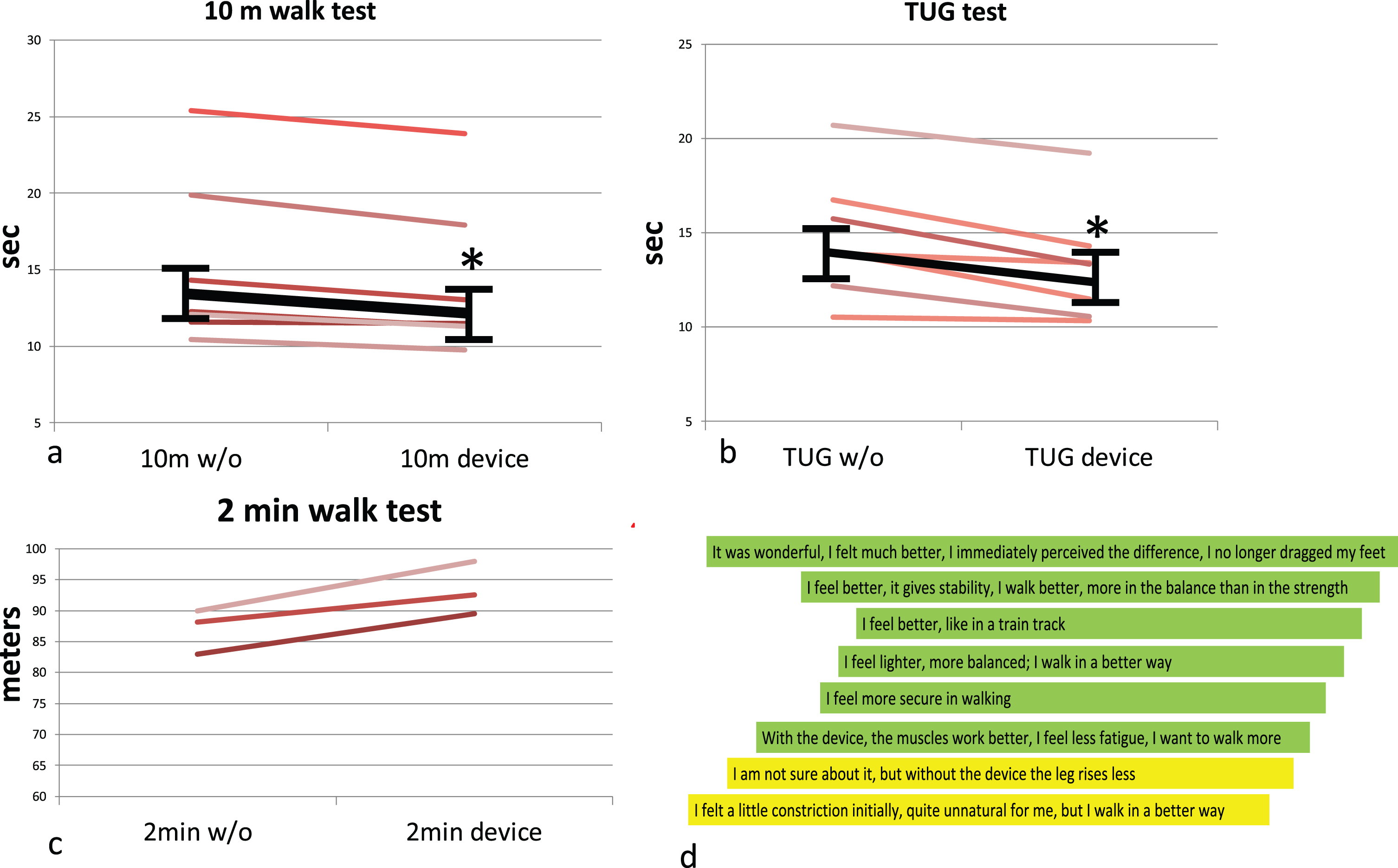
2MWT also showed an increase of walked distance in all patients (median 88.2 vs 92.6 m, n.s., Fig 2c).
Figure 2d illustrates the personal impressions of patients immediately after first use, with each line corresponding to one patient. Six patients reported subjective benefits, noting improvements in walking stability, speed, confidence, and reduced fatigue (green lines in Fig. 1d). Only two patients (yellow lines) expressed uncertainty regarding the impact of the device on their motor function, attributing it to a perceived alteration in usual walking pattern, even if they perceived some improvement.
DISCUSSION
Walking impairment is a prevalent and prominent challenge in various neuromuscular disorders. It is characterized by altered locomotor performance, gait instability, and pattern changes, and it is variable across different neuromuscular diseases [3]. This study focuses on assessing the impact of ExoBand, a novel passive wearable device designed to enhance walking function in patients with neuromuscular diseases. Our preliminary findings indicate that the ExoBand significantly improves walking ability in these patients from the very first use, with the device confirmed as non-harmful and generally well-tolerated by patients [4].
In neuromuscular disorders muscle weakness often results in gait abnormalities, leading to reduced walking distance, increased fatigue, and a high risk of falls [5, 6]. This restricted mobility may also contribute to weight gain, exacerbating motor function. Furthermore, limited mobility significantly curtails social activities, impacting psychological well-being [7].
The introduction of the ExoBand aims to break this vicious cycle by mitigating the effects of muscle weakness.
Walking speed is a valuable tool for tracking progress and health in diverse populations because it reflects overall health and daily living abilities. Because this outcome reflects overall health and daily living abilities, walking speed is a valuable tool for tracking progress and health in diverse populations [8]. The present exploratory study shows that ExoBand exhibits a prompt and measurable impact on walking speed, as indicated by the results of the 10mWT. Additionally, the device demonstrated some efficacy in more complex daily activities, exemplified by improvements in the TUG test. In fact, the TUG test involves a combination of tasks, including rising from a chair, walking in a straight line, and turning around.
During the initial trial with the device, most patients reported an immediate and significant benefit, particularly in terms of balance and stability rather than strength. The ExoBand design, connecting the groin area with the knee, facilitates the correct alignment of hip flexion during the step. This feature also compensates for muscle weaknesses in abductor and/or adductor muscles, reducing overall energy expenditure and providing a heightened sensation of stability.
A study on CIDP patients confirmed substantial limitations in mobility-related activities [5]. According to the current guidelines, orthoses, physiotherapy, and occupational therapy are recommended for CIDP patients [9]. Patients with CIDP are referred to a rehabilitation specialist to consider potential orthoses; this is well known for distal weakness treatment (i.e., ankle-foot orthoses). Now, it can also be considered for proximal weakness that impairs walking ability, thanks to Exoband availability. In fact, the ExoBand offers a potential solution for those with mild-to-moderate disability, potentially improving walking speed and increasing the distance covered, as exemplified with our preliminary tests.
Beyond its measurable physical effects, in the cohort of this analysis, wearing the ExoBand had a positive impact also on psychological aspects: it is interesting to note that some participants after the first use of the device reported feeling more secure, positive, and one expressed a desire to walk more. If these effects persist with prolonged use at home, it could further enhance active patient participation.
While this retrospective exploratory study provides valuable insights, it has limitations such as a small sample size including only patients older than 60 years, disease heterogeneity, the limited number of motor function tests, and the absence of a control group.
Further research is essential to assess the long-term effects of regular ExoBand use at home, including its impact on physical and mental well-being. Additionally, this clinical protocol will help to identify the patient categories that may derive the largest benefit from ExoBand by evaluating the device in specific neuromuscular diseases, both in adults and children with diverse muscle weakness patterns.
CONCLUSIONS
This exploratory study suggests the usefulness of a passive wearable device in patients with CIDP or motor neuropathies presenting proximal weakness and gait abnormalities.
ETHICAL CONSIDERATION
The study complies with the principles stated in the Declaration of Helsinki “Ethical Principles Medical Research Involving ‘Human Subjects”. This retrospective study, in which only aggregate data were analyzed, was communicated to the Ethical Committee of Padova.
ACKNOWLEDGEMENTS
FUNDING
No financial disclosures. The study was non sponsored.
CONFLICT OF INTEREST
CS is employed by Moveo srl, the company that manufactures the Exoband, since September 2023, after the completion of the study. Other authors declare no conflicts of interest.
AUTHORS CONTRIBUTIONS
CS: Conceptualization of the study, data acquisition and analysis, writing & Editing of the draft
MA: Conceptualization of the study, data analysis, Review & Editing of the draft
CA: Conceptualization of the study, data analysis, Review & Editing of the draft
SM: Conceptualization of the study, data analysis, Review & Editing of the draft
FP: Conceptualization of the study, data analysis, writing, review & Editing of the draft
GS: Conceptualization of the study, data analysis, writing, review & Editing of the draft
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author.
REFERENCES
[1] | Panizzolo FA , Bolgiani C , Di Liddo L , Annese E , Marcolin G . Reducing the energy cost of walking in older adults using a passive hip flexion device. J Neuroeng Rehabil. (2019) ;15: (16):117. doi: 10.1186/s12984-019-0599-4. |
[2] | Panizzolo FA , Annese E , Paoli A , Marcolin G . A single assistive profile applied by a passive hip flexion device can reduce the energy cost of walking in older adults. Appl. Sci. (2021) ;11: (6). doi.org/10.3390/app11062851. |
[3] | McDonald CM . Neuromuscular diseases. In: Pediatric rehabilitation: principles and practice. Demos Medical Publishing, New York, 2015. p. 461. |
[4] | Panizzolo FA , Cimino S , Pettenello E , Belfiore A , Petrone N , Marcolin G . Effect of a passive hip exoskeleton on walking distance in neurological patients. Assist Technol. (2022) ;34: (5):527–532. doi: 10.1080/10400435.2021.1880494. |
[5] | Wonink HA , Kruithof WJ , Goedee HS , Beelen A . Chronic inflammatory neuropathies and their impact on activities and participation. Eur J Neurol. (2023) ;30: (7):1928–1936. doi: 10.1111/ene.15815. Epub 2023 Apr 24 |
[6] | Pieterse AJ , Luttikhold TB , de Laat K , Bloem BR , van Engelen BG , Munneke M . Falls in patients with neuromuscular disorders. J Neurol Sci. (2006) ;251: (1-2):87–90. doi: 10.1016/j.jns.2006.09.008. Epub 2006 Nov 9. PMID: 17097113 |
[7] | McCrory MA , Wright NC , Kilmer DD . Nutritional aspects of neuromuscular diseases. Phys Med Rehabil Clin N Am. (1998) ;9: (1):127–43. |
[8] | Middleton A , Fritz SL , Lusardi M . Walking speed: the functional vital sign. J Aging Phys Act. (2015) ;23: (2):314–22. doi: 10.1123/japa.2013-0236. Epub 2014 May 2. PMID: 24812254; PMCID: PMC4254896 |
[9] | Van den Bergh PYK , van Doorn PA , Hadden RDM , Avau B , Vankrunkelsven P , Allen JA , Attarian S , Blomkwist-Markens PH , Cornblath DR , Eftimov F , Goedee HS , Harbo T , Kuwabara S , Lewis RA , Lunn MP , Nobile-Orazio E , Querol L , Rajabally YA , Sommer C , Topaloglu HA . European Academy of Neurology/Peripheral Nerve Society guideline on diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy: Report of a joint Task Force-Second revision. Eur J Neurol. (2021) ;28: :3556–3583. doi: 10.1111/ene.14959. Epub 2021 Jul 30. |




