Musculoskeletal system symptoms in patients with COVID-19 and the impact of these symptoms on quality of life
Abstract
BACKGROUND:
Coronavirus disease 2019 (COVID-19) is a respiratory disease caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and also affects the musculoskeletal system.
OBJECTIVE:
This study was conducted to investigate the musculoskeletal symptoms, type of pain and effect on quality of life in patients presenting with pain after COVID-19.
METHODS:
This prospective, descriptive study included 97 patients aged 18 years or older who were diagnosed with COVID-19 based on a positive polymerase chain reaction test result, with or without musculoskeletal pain prior to COVID-19 infection. Patients who applied to the post-COVID-19 outpatient clinic with the complaint of pain at least 1 month and maximum 1 year after the diagnosis of COVID-19 were included. Patients’ demographic characteristics and musculoskeletal examination findings were evaluated. The patients were examined, and the questionnaire forms were completed. The pain was assessed using the visual analog scale (VAS), the Douleur-Neuropathique-4 (DN-4) questionnaire, while the quality of life was assessed using the Short Form-36 (SF-36) survey. Patients were divided into groups in terms of gender, age, body mass index. Shapiro-Wilk’s test, the independent samples t-test and the Mann-Whitney U test were used for statistical analyses.
RESULTS:
The mean age of the patients was 46.5
CONCLUSIONS:
Arthralgia, myalgia, and neuropathic pain, all of which negatively affect the quality of life, are often observed in the patients infected with COVID-19.
1.Introduction
Coronavirus disease 2019 (COVID-19) is a respiratory disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 is a new type of coronavirus that emerged on December 31
COVID-19 affects the musculoskeletal system in various ways. SARS-CoV-2 enters the cells via the angiotensin-converting enzyme 2 (ACE2) receptors using the transmembrane protease, a serine 2 (TMPRSS2) enzyme. In addition to the respiratory system, ACE2 receptors are also found in the tissues of other systems. COVID-19 gives rise to pathological changes in the musculoskeletal system, i.e., the muscle tissue, the synovium, and cortical bone, which include these receptors [4, 5]. Cytokines and pro-inflammatory molecules released after COVID-19 infection also cause damage to the musculoskeletal system [2, 6]. At least one rheumatic and musculoskeletal symptom was reportedly observed in 74.6% and 43.2% of the COVID-19 patients during three-month and six-month follow-up periods, respectively [7]. On the other hand, studies that investigated either arthralgia or myalgia in COVID-19 patients reported the rate of patients with myalgia between 13.1% and 60.7% and the rate of patients with arthralgia between 5.7% and 43.6% [8, 9, 10, 11, 12, 13]. Additionally, neuropathic pain and paresthesia were reported in 21% and 33% of COVID-19 patients, respectively [14, 15].
Complaints such as fatigue, myalgia, shortness of breath, hot flashes, headache, tachycardia, chest tightness, and anxiety continue even after the acute phase of COVID-19. These prolonged complaints have been evaluated as the symptoms of the post-COVID syndrome and have reportedly affected the patients both physically and psychologically, causing them difficulties in daily life activities, a situation that negatively affects not only the patients but also their social environment [16, 17].
More detailed data for musculoskeletal symptoms will increase our understanding of the effects of COVID-19 and lead to more efficient and better care for those recovering from COVID-19. In this context, the objective of this study is to assess the musculoskeletal symptoms seen in patients who received outpatient or inpatient treatment due to COVID-19 and to determine the impact of these symptoms on the quality of life. The changes observed in these symptoms in COVID-19 patients who already had these symptoms before they were infected with COVID-19 were also investigated, given the limited studies on this subject.
2.Materials and methods
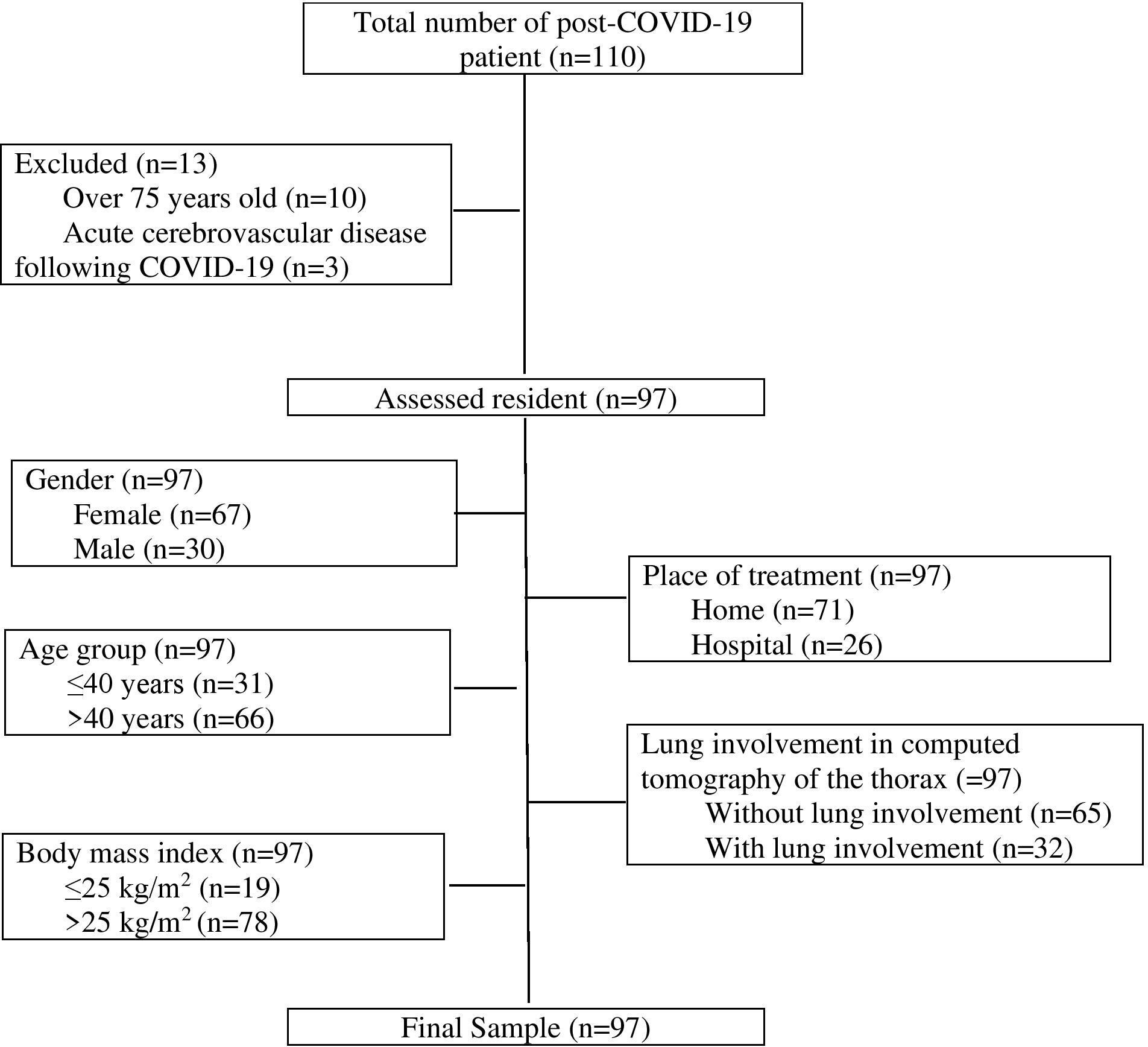
The population of this prospective, descriptive, gender-neutral study included 110 patients aged 18 years and over with or without musculoskeletal pain before COVID-19 infection, who applied to a post-COVID clinic, were diagnosed with COVID-19 based on the positive result of the polymerase chain reaction test using a nasopharyngeal sample, were treated as inpatients or outpatients between May and August 2021, and met the WHO termination criteria for isolation. Ten patients aged over 75 years old and three patients who developed acute cerebrovascular disease following COVID-19 were excluded from the study. In the end, the study sample consisted of 97 patients (Fig. 1).
Figure 1.
Flowchart for the inclusion of the patients and patient grouping.
The study protocol was approved by the Clinical Research Committee of Hitit University (Approval No.: 418, Approval Date: 10/03/2021). The study was conducted in accordance with the principles set forth in the Declaration of Helsinki. Written informed consent was obtained from all patients before the study.
Patients aged between 18 and 75 years, who presented to the post-COVID-19 outpatient clinic with pain, were referred to the Physical Medicine and Rehabilitation outpatient clinic. Inclusion criteria encompassed individuals who had a documented COVID-19 infection within the past year, had been diagnosed with COVID-19 for at least 1 month, and met the aforementioned age range. Exclusion criteria comprised patients who received intensive care unit treatment during their COVID-19 infection, had a history of cerebrovascular disease, exhibited cognitive impairment, suffered from rheumatological disease, joint contracture, neurological disease, or were pregnant women. Patients’ demographic data (age, gender, body mass index [BMI], educational status, marital status, occupation), comorbidities (hypertension, diabetes mellitus, thyroid disease, heart disease, respiratory disease, malignancy, and others), smoking status, presence of lung involvement in computed tomography of the thorax, length of hospital stay, steroid use, and time from COVID-19 diagnosis to assessment (duration of the disease) were recorded. Patients’ muscle pain, joint pain, the type of pain (nociceptive pain, neuropathic pain) they experienced, and changes in pain intensity compared to before COVID-19 were assessed, and superficial sensory examinations (touch, pain-temperature) were performed. In our study, patients underwent examination and completed questionnaire forms upon their presentation to the outpatient clinic.
The pain experienced by patients was assessed using the visual analog scale (VAS). VAS is a standard pain assessment tool used in daily clinical practice. Patients assess their pain by assigning a score between 0 (no pain) and 10 (unbearable pain). A VAS score of 1 to 3 indicates mild pain, 4 to 6 moderate pain, and 7 to 10 severe pain [18].
Patients’ neuropathic pain assessment was carried out using the Douleur Neuropathique 4 (DN-4) questionnaire. The DN-4 questionnaire, which was developed by the French Neuropathic Pain Group, assesses the symptoms and findings related to neuropathic pain in four subscales and ten items. The first two subscales focus on anamnesis, and the remaining two focus on the clinical examination findings. The first subscale queries the character of the pain (burning sensation, sensation of painful cold, electric shock sensation) in three items. The second subscale queries paresthesia/dysesthesia (tingling, pins-and-needles, numbness, itching sensations) in the body site with pain in four items. The third subscale queries the sensory deficit (hypoesthesia to touch, hypoesthesia to pinprick) in the body site where the pain is located in two items. Finally, the fourth subscale queries whether brushing inflicts or aggravates pain in one item. Items are answered as either ‘yes’ (1 point) or ‘no’ (0 points). Patients with a total score of 4 or more are considered to have neuropathic pain [19]. The Turkish adaptation, validity, and reliability studies of the DN-4 questionnaire were carried out by Cevik et al. [20].
The patients’ quality of life and functional status were evaluated using the Short Form-36 (SF-36) survey. This self-assessment survey evaluates the quality of life in the last four weeks by focusing on the physical, social, and spiritual components of health without concentrating on a specific age or disease group. The survey consists of 36 items that are measured in eight subscales: physical function subscale (10 items), social function subscale (2 items), role limitations due to physical problems subscale (4 items), role limitations due to emotional problems subscale (3 items), mental health subscale (5 items), vitality subscale (4 items), pain subscale (2 items), and general perception of health subscale (5 items). The total score that can be obtained from each subscale ranges from 0 to 100. The higher the score, the higher the quality of life [21]. The Turkish adaptation, validity, and reliability studies of the SF-36 survey were carried out by Koçyiğit et al. [22].
Age, gender, obesity, lung involvement, and hospitalization have been cited as factors that affect the severity of COVID-19 [23], which may also have an effect on pain. The patients included in the study group were divided into various subgroups according to gender, place of treatment (home/hospital), age group (
2.1Statistical analysis
Power analysis and sample size calculation were carried out using PASS 11.0 (Power Analysis and Sample Size Software, NCSS LLC, Kaysville, UT, USA). Accordingly, the power of the study with a sample size of 77 COVID-19 patients was calculated as 95% (alpha
Statistical analyses were performed using SPSS version 22.0 (IBM Corp., Armonk, NY, USA) software package. Descriptive statistics obtained from the collected data were expressed as mean and standard deviation values in the case of continuous variables determined to conform to the normal distribution, as median and interquartile range values in the case of continuous variables determined not to conform to the normal distribution, and as frequency (n) and percentage (%) values in the case of categorical variables. Normal distribution characteristics of the continuous variables were analyzed using Shapiro-Wilk’s test. Comparisons of two independent groups were made using the independent samples
3.Results
Table 1
Demographic and clinical characteristics of the patients (
| Parameters | |
|---|---|
| Age (years) | 46.5 |
| Gender (female/male) | 67 (69)/30 (31) |
| BMI (kg/m | 29.0 |
| Educational status | |
| Illiterate | 3 (3.1) |
| Primary school | 50 (51.5) |
| Middle school | 15 (15.5) |
| High school | 18 (18.6) |
| College/university | 11 (11.3) |
| Smoking | 13 (13.8) |
| Disease duration (months) | 4.0 |
| Place of treatment (home/hospital) | 71 (73.2)/26 (26.8) |
| Length of hospital stay (days) | 3.1 |
| Steroid use | 18 (18.6) |
| Lung involvement | 32 (33) |
| Myalgia before COVID-19 | 49 (50.5) |
| Myalgia after COVID-19 | 96 (99) |
| Arthralgia before COVID-19 | 40 (41.2) |
| Arthralgia after COVID-19 | 77 (79.4) |
| New arthralgia developed after COVID-19 | 37 (38.1) |
| New myalgia developed after COVID-19 | 47 (48.5) |
| Type of pain before COVID-19 | |
| Nociceptive/neuropathic | 35 (36.1)/14 (14.4) |
| Type of pain after COVID-19 | |
| Nociceptive/neuropathic | 50 (51.5)/47 (48.5) |
| Increase in pre-COVID pain intensity | 49 (100) |
| Abnormal sensory examination findings | 18 (18.6) |
| VAS score after COVID-19 | 6.9 |
| DN4 score after COVID-19 | 3.5 |
| Neuropathic pain confirmed with DN4 after COVID-19 | |
| 41 (42.3) | |
| SF-36 subscale scores after COVID-19 | |
| Physical function | 60.4 |
| Physical role limitation | 35.8 |
| Emotional role limitation | 36.4 |
| Vitality | 36.9 |
| Mental health | 49.2 |
| Social function | 53.1 |
| Pain | 34.3 |
| General perception of health | 40.3 |
BMI: body mass index, DN4: Douleur Neuropathique 4 questionnaire, SF-36: Short Form-36 survey, VAS: visual analog scale. The data are expressed as mean
The mean age of the 97 patients included in the study, of whom 67 (69.1%) were males, was 46.5
Table 2
Location of arthralgia and myalgia of patients (
| Parameters | |
|---|---|
| Location of arthralgia before COVID-19 | |
| Shoulder | 17 (17.5) |
| Elbow | 11 (11.3) |
| Wrist | 8 (8.2) |
| Hip | 6 (6.2) |
| Knee | 24 (24.7) |
| Foot/ankle | 5 (5.2) |
| Location of arthralgia after COVID-19 | |
| Shoulder | 43 (44.3) |
| Elbow | 26 (26.8) |
| Wrist | 21 (21.9) |
| Hip | 22 (22.7) |
| Knee | 39 (40.2) |
| Foot/ankle | 13 (13.4) |
| Location of myalgia before COVID-19 | |
| Neck-back-lumbar | 27 (27.8) |
| Front trunk | 2 (2.1) |
| Arm | 22 (22.7) |
| Forearm | 22 (22.7) |
| Thigh | 15 (15.5) |
| Calf | 20 (20.6) |
| Location of myalgia after COVID-19 | |
| Neck-back-lumbar | 59 (60.8) |
| Front trunk | 16 (16.5) |
| Arm | 43 (44.3) |
| Forearm | 37 (38.1) |
| Thigh | 32 (33.0) |
| Calf | 44 (45.4) |
The data are expressed as
Sixty percent of the patients were homemakers, and 17% were working actively. Eighty-one percent of the patients were married, and 14.4% were living alone. The most common comorbidities were hypertension (24.7%), diabetes mellitus (14.4%), respiratory disease (10.3%), thyroid disease (8.2%), heart disease (5.2%), and malignancy (2.1%). In addition, 45 (46.4%) patients had no comorbidities, 33 (34%) had only one comorbidity, 14 (14.4%) had two comorbidities, and 5 (5.2%) had three or more comorbidities.
The most common site of arthralgia in patients with arthralgia before the COVID-19 infection was the knee (24.7%), whereas the most common site of arthralgia after the COVID-19 infection was the shoulder (44.3%). On the other hand, both the most common locations of myalgia before COVID-19 infection and after COVID-19 infection were neck/back/lumbar (27.7% and 60.8%, respectively) (Table 2).
The mean VAS scores of the patients who had musculoskeletal pain were 3.2
There were 49 patients with myalgia and 40 with arthralgia before the COVID-19 infection. The comparisons between the patients with and without arthralgia and myalgia before COVID-19 are given in Table 3.
Table 3
Comparison of the demographic and clinical data of the patients according to arthralgia before COVID-19 and myalgia before COVID-19
| Variable | Arthralgia before COVID-19 ( | Myalgia before COVID-19 ( | ||||
|---|---|---|---|---|---|---|
| Not developed ( | Developed ( |
| Not developed ( | Developed ( |
| |
| Age (years) | 44.9 | 48.7 | 0.171 | 44.7 | 48.2 | 0.195 |
| Gender (female/male) | 37 (64.9)/20 (35.1) | 30 (75)/10 (25) | 0.290 | 28 (58.3)/20 (41.7) | 39 (79.6)/10 (20.4) | 0.024 |
| BMI (kg/m | 28.9 | 29.2 | 0.727 | 28.8 | 29.2 | 0.605 |
| Number of comorbidities | 0.7 | 0.9 | 0.443 | 0.6 | 0.9 | 0.324 |
| Disease duration (months) | 4 (3–5.3) | 3 (2–5) | 0.076 | 4 (2–5.4) | 3.5 (2.1–5) | 0.635 |
| Length of hospital stay (days) | 3.3 | 2.9 | 0.829 | 1.9 | 4.3 | 0.249 |
| Place of treatment (home/hospital) | 42 (73.7)/15 (26.3) | 29 (72.5)/11 (27.5) | 0.897 | 37 (77.1)/11 (22.9) | 34 (69.4)/15 (30.6) | 0.392 |
| Smoking | 8 (14) | 5 (12.5) | 0.827 | 9 (18.8) | 4 (8.2) | 0.126 |
| Comorbidities | 29 (50.9) | 23 (57.5) | 0.520 | 25 (52.1) | 27 (55.1) | 0.766 |
| Steroid use | 11 (19.3) | 7 (17.5) | 0.823 | 8 (16.7) | 10 (20.4) | 0.636 |
| Lung involvement | 19 (33.3) | 13 (32.5) | 0.932 | 15 (31.3) | 17 (34.7) | 0.718 |
| Arthralgia | 37 (35.1) | 40 (100) |
| 30 (62.5) | 47 (95.9) |
|
| Myalgia | 57 (100) | 39 (97.5) | 0.231 | 47 (97.9) | 49 (100) | 0.495 |
| Arthralgia developed after COVID-19 | 37 (64.9) | 0 (0) |
| 25 (52.1) | 12 (24.5) | 0.005 |
| Myalgia developed after COVID-19 | 43 (75.4) | 4 (10) |
| 47 (97.9) | 0 (0) |
|
| VAS score | 7 (6–8) | 7 (6–8) | 0.961 | 7 (6–8) | 7 (6–8) | 0.656 |
| DN-4 score | 3 (2–5) | 3 (2–5) | 0.965 | 2 (2–4) | 4 (2–5.5) | 0.023 |
| Neuropathic pain confirmed with DN-4 | 24 (42.1) | 17 (42.5) | 0.969 | 16 (33.3) | 25 (51) | 0.078 |
| SF-36 subscale scores | ||||||
| Physical function | 63.1 | 56.6 | 0.133 | 70 (55–80) | 55 (42.5–72.5) | 0.003 |
| Physical role limitation | 25 (0–75) | 25 (0–50) | 0.152 | 25 (0–75) | 25 (0–50) | 0.151 |
| Emotional role limitation | 33.3 (0–66.7) | 33.3 (0–58.4) | 0.498 | 33.3 (0–91.7) | 33.3 (0–66.7) | 0.646 |
| Vitality | 40 (30–50) | 35 (20–45) | 0.149 | 38.6 | 35.4 | 0.323 |
| Mental health | 50.5 | 47 | 0.300 | 52 | 46.2 | 0.082 |
| Social function | 55 | 50.3 | 0.365 | 58.9 | 47.4 | 0.024 |
| Pain | 35 (22.5–45) | 22.5 (10.6–46.9) | 0.297 | 37.5 (33.1–47.5) | 22.5 (10–45) | 0.003 |
| General perception of health | 40 (35–55) | 40 (26.3–48.8) | 0.308 | 43.9 | 36.9 | 0.047 |
BMI: body mass index, DN4: Douleur Neuropathique 4 questionnaire, SF-36: Short Form-36 survey, VAS: visual analog scale. The data are expressed as mean
Gender-based comparisons of the patients revealed that the mean VAS score of females, as well as the number of female patients who developed myalgia after COVID-19, were significantly higher than those of males (
Table 4
Comparison of the demographic and clinical data of the patients according to gender, age group and body mass index
| Variables | Gender | Age group | Body mass index | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Female ( | Male ( |
|
|
| |||||
| Age (years) | 45.3 | 49.1 | 0.208 | 30.8 | 53.8 |
| 33.6 | 49.6 |
|
| Gender (female/male) | * | * | * | 24 (77.4) | 43 (65.2) | 0.223 | 14 (73.7) | 53 (67.9) | 0.628 |
| 7 (22.6) | 23 (34.8) | 5 (26.3) | 25 (32.1) | ||||||
| BMI (kg/m | 28.8 | 29.6 | 0.437 | 25.7 | 30.5 | 0.001 | 21.7 | 30.8 |
|
| Number of comorbidities | 0.7 | 0.8 | 0.687 | 0.4 | 1.0 | 0.010 | 0.5 | 0.9 | 0.087 |
| Disease duration (months) | 4 (2–5) | 3.8 (2.9–5.9) | 0.475 | 3.5 (2–4.5) | 4 (2–5) | 0.324 | 3 (2–5.5) | 4 (2–5) | 0.383 |
| Length of hospital stay (days) | 2.8 | 3.6 | 0.634 | 1.0 | 4.1 | 0.010 | 1.2 | 3.6 | 0.201 |
| Place of treatment | |||||||||
| Home | 52 (77.6) | 19 (63.3) | 27 (87.1) | 44 (66.7) | 17 (89.5) | 54 (69.2) | 0.089 | ||
| Hospital | 15 (22.4) | 11 (36.7) | 0.142 | 4 (12.9) | 22 (33.3) | 0.034 | 2 (10.5) | 24 (30.8) | |
| Smoking | 2 (3.0) | 11 (36.7) |
| 2 (6.5) | 11 (16.7) | 0.168 | 2 (10.5) | 11 (14.1) | 1.000 |
| Comorbidities | 32 (47.8) | 20 (66.7) | 0.084 | 12 (38.7) | 40 (60.6) | 0.044 | 6 (31.6) | 46 (59) | 0.032 |
| Steroid use | 11 (16.4) | 7 (23.3) | 0.418 | 3 (9.7) | 15 (22.7) | 0.123 | 19 (100) | 18 (23.1) | 0.019 |
| Lung involvement | 19 (28.4) | 13 (43.3) | 0.147 | 6 (19.4) | 26 (39.4) | 0.052 | 3 (15.8) | 29 (37.2) | 0.103 |
| Arthralgia | 55 (82.1) | 22 (73.3) | 0.325 | 21 (67.7) | 56 (84.8) | 0.052 | 13 (68.4) | 64 (82.1) | 0.188 |
| Myalgia | 66 (98.5) | 30 (100) | 1.000 | 30 (96.8) | 66 (100) | 0.320 | 18 (94.7) | 78 (100) | 0.196 |
| Arthralgia developed after COVID-19 | 25 (37.3) | 12 (40.0) | 0.801 | 8 (25.8) | 29 (43.9) | 0.086 | 4 (21.1) | 33 (42.3) | 0.116 |
| Myalgia developed after COVID-19 | 27 (40.3) | 20 (66.7) | 0.016 | 14 (45.2) | 33 (50.0) | 0.657 | 10 (52.6) | 37 (47.4) | 0.684 |
| VAS score | 7 (6–8) | 6 (5–7) | 0.01 | 7 (6–9) | 7 (6–8) | 0.925 | 6 (6–7) | 7 (6–8) | 0.211 |
| DN4 score | 3 (2–5) | 3 (2–5) | 0.70 | 4 (2–5) | 3 (2–5) | 0.177 | 6 (6–7) | 3 (2–5) | 0.909 |
| Neuropathic pain confirmed with DN4 | 29 (43.3) | 12 (40.0) | 0.762 | 17 (54.8) | 24 (36.4) | 0.086 | 8 (42.1) | 33 (42.3) | 0.987 |
| SF-36 subscale scores | |||||||||
| Physical function | 56.4 | 69.2 | 0.005 | 60 (50–75) | 65 (45–75) | 0.750 | 65 (50–75) | 65 (45–70) | 0.913 |
| Physical role limitation | 25 (0–75) | 25 (0–81.3) | 0.471 | 25 (0–75) | 25 (0–56.3) | 0.604 | 25 (0–75) | 25 (0–75) | 0.763 |
| Emotional role limitation | 33.3 (0–66.7) | 33.3 (0–75) | 0.655 | 33.3 (0–66.7) | 33 (0–66.7) | 0.489 | 33.3 (0–66.7) | 33.3 (0–66.7) | 0.996 |
| Vitality | 35.4 | 40.5 | 0.157 | 35.4 | 37.6 | 0.540 | 37.1 | 36.9 | 0.970 |
| Mental health | 47.7 | 52.0 | 0.20 | 47.8 | 49.6 | 0.624 | 48.8 | 49.1 | 0.946 |
| Social function | 51.5 | 56.7 | 0.30 | 50 (37.5–75) | 62.5 (37.5–75) | 0.925 | 62.5 (37.5–75) | 50 (37.5–75) | 0.336 |
| Pain | 29.8 | 44.4 | 0.02 | 35 (20–47.5) | 35 (22.5–45) | 0.491 | 35 (22–45) | 35 (22.5–47.5) | 0.516 |
| General perception of health | 38.0 | 45.5 | 0.05 | 41.6 | 39.7 | 0.624 | 45 (25–50) | 40 (33.7–55) | 0.964 |
BMI: body mass index, DN4: Douleur Neuropathique 4 questionnaire, SF-36: Short Form-36 survey, VAS: visual analog scale. The data are expressed as mean
Additionally, age-based comparisons of the patients revealed that the mean BMI, number of comorbidities, number of patients with comorbidities, number of patients treated as inpatients, and the mean length of hospital stay were significantly higher in patients
BMI-based comparisons of the patients revealed that the mean age, number of patients with comorbidities, and number of patients using steroids were significantly higher in patients with a BMI of
Table 5
Comparison of the demographic and clinical data of the patients according to place of treatment and lung involvement
| Variable | Place of treatment ( | Lung involvement ( | ||||
|---|---|---|---|---|---|---|
| Home ( | Hospital ( |
| Without lung involvement ( | With lung involvement ( |
| |
| Age (years) | 44.7 | 51.3 | 0.031 | 44.6 | 50.2 | 0.057 |
| Gender (female/male) | 52 (73.2)/19 (26.8) | 15 (57.6)/11 (42.4) | 0.142 | 48 (73.8)/17 (26.2) | 19 (59.4)/13 (40.6) | 0.147 |
| BMI (kg/m | 28.3 | 31.0 | 0.022 | 28.0 | 31.7 | 0.003 |
| Number of comorbidities | 0.6 | 1.2 | 0.001 | 0.5 | 1.2 | 0.001 |
| Disease duration (months) | 4 (2.5–5) | 2 (1.5–4.5) | 0.027 | 4 (2.5–5.3) | 4 (2–4.5) | 0.099 |
| Length of hospital stay (days) | NA | 11.6 | NA | 0.1 | 9.3 | 0.001 |
| Place of treatment (home/hospital) | * | * | * | 64 (98.5)/1 (1.5) | 7 (21.9)/25 (78.1) |
|
| Smoking | 9 (12.7) | 4 (15.4) | 0.729 | 8 (12.3) | 5 (15.6) | 0.652 |
| Comorbidities | 30 (42.3) | 22 (84.6) |
| 26 (40.0) | 26 (81.2) |
|
| Steroid use | 1 (1.4) | 17 (65.4) |
| 0 (0) | 18 (56.2) |
|
| Lung involvement | 7 (9.9) | 25 (96.2) |
| * | * | * |
| Arthralgia | 55 (82.1) | 22 (73.3) | 0.576 | 50 (76.9) | 27 (84.4) | 0.394 |
| Myalgia | 70 (98.6) | 26 (100) | 1.000 | 64 (98.5) | 32 (100) | 0.481 |
| Arthralgia developed after COVID-19 | 26 (36.6) | 11 (42.3) | 0.642 | 23 (35.4) | 14 (43.8) | 0.425 |
| Myalgia developed after COVID-19 | 36 (50.7) | 11 (42.3) | 0.464 | 32 (49.2) | 15 (46.9) | 0.827 |
| VAS score | 7 (6–8) | 7 (6–7) | 0.240 | 7 (6–8) | 7 (6–8) | 0.716 |
| DN4 score | 3 (2–5) | 3 (1–4.3) | 0.350 | 3 (2–5) | 3 (1.3–4) | 0.325 |
| Neuropathic pain confirmed with DN4 | 30 (42.3) | 11 (42.3) | 0.996 | 37 (56.9) | 13 (40.6) | 0.818 |
| SF-36 subscale scores | ||||||
| Physical function | 59.2 | 63.6 | 0.355 | 65 (47.5–75) | 62.5 (45–75) | 0.796 |
| Physical role limitation | 25 (0–75) | 25 (0–56.3) | 0.666 | 25 (0–75) | 25 (0–50) | 0.379 |
| Emotional role limitation | 33.3 (0–66.7) | 33.3 (0–66.7) | 0.781 | 33.3 (0–66.7) | 33.3 (0–66.7) | 0.961 |
| Vitality | 35 (20–45) | 40 (33.7–55) | 0.091 | 35 (20–45) | 40 (35–55) | 0.030 |
| Mental health | 47.7 | 52.1 | 0.265 | 48.0 | 51.3 | 0.335 |
| Social function | 62.5 (37.5–75) | 50 (25–62) | 0.466 | 62.5 (37.5–75) | 50 (25–62.5) | 0.072 |
| Pain | 35 (22.5–45) | 40 (22.5–57.5) | 0.253 | 35 (22.3–45) | 35 (22.5–56.9) | 0.424 |
| General perception of health | 40 (25–55) | 40 (33.8–50) | 0.815 | 40.2 | 40.4 | 0.957 |
BMI: body mass index, DN4: Douleur Neuropathique 4 questionnaire, SF-36: Short Form-36 survey, VAS: visual analog scale. The data are expressed as mean
Table 6
Correlation of patients with various variables
| Age | BMI | After COVID-19 VAS | Before COVID-19 VAS | DN-4 | Physical function | Physical role limitation | Emotional role limitation | Vitality | Mental health | Social function | Pain | General perception of health | ||
| Age |
| 1 | ||||||||||||
|
| ||||||||||||||
| BMI |
| 0.458 | 1 | |||||||||||
|
|
| |||||||||||||
| After COVID-19 VAS |
| 0.011 | 0.107 | 1 | ||||||||||
|
| 0.916 | 0.295 | ||||||||||||
| Before COVID-19 VAS |
| 0.240 | 1 | |||||||||||
|
| 0.670 | 0.644 | 0.018 | |||||||||||
| DN-4 |
| 0.214 | 0.201 | 1 | ||||||||||
|
| 0.301 | 0.826 | 0.018 | 0.049 | ||||||||||
| Physical function |
| 1 | ||||||||||||
|
| 0.347 | 0.083 |
| 0.019 |
| |||||||||
| Physical role limitation |
| 0.105 | 0.013 | 0.411 | 1 | |||||||||
|
| 0.307 | 0.899 |
| 0.132 | 0.001 |
| ||||||||
| Emotional role limitation |
| 0.035 | 0.028 | 0.275 | 0.658 | 1 | ||||||||
|
| 0.736 | 0.785 | 0.026 | 0.494 | 0.001 | 0.007 |
| |||||||
| Vitality |
| 0.062 | 0.286 | 0.213 | 0.257 | 1 | ||||||||
|
| 0.544 | 0.959 | 0.035 | 0.004 | 0.001 | 0.005 | 0.038 | 0.012 | ||||||
| Mental health |
| 0.135 | 0.263 | 0.113 | 0.260 | 0.646 | 1 | |||||||
|
| 0.187 | 0.776 | 0.042 | 0.001 | 0.001 | 0.009 | 0.271 | 0.010 |
| |||||
| Social function |
| 0.531 | 0.416 | 0.344 | 0.326 | 0.224 | 1 | |||||||
|
| 0.729 | 0.107 |
| 0.018 |
|
|
| 0.010 | 0.001 | 0.028 | ||||
| Pain |
| 0.066 | 0.052 | 0.437 | 0.360 | 0.319 | 0.356 | 0.242 | 0.563 | 1 | ||||
|
| 0.519 | 0.614 |
| 0.001 |
|
|
| 0.002 |
| 0.018 |
| |||
| General perception of health |
| 0.484 | 0.360 | 0.263 | 0.375 | 0.297 | 0.376 | 0.391 | 1 | |||||
|
| 0.910 | 0.035 |
| 0.020 |
|
|
| 0.009 |
| 0.003 |
|
|
BMI: body mass index, DN4: Douleur Neuropathique 4 questionnaire, VAS: visual analog scale. Significant p values are written in bold.
In terms of the place of treatment, the mean age, mean BMI, number of comorbidities, number of patients with comorbidities, number of patients using steroids, and the number of patients with lung involvement were significantly higher in patients who were treated as inpatients than in patients who were treated as outpatients (
Additionally, the mean BMI, number of comorbidities, number of patients with comorbidities, number of patients treated as inpatients, mean length of hospital stay, number of patients using steroids, and the mean score of the vitality subscale of the SF-36 survey were significantly higher in patients who had lung involvement in the computed tomography of the thorax than in patients who did not (
There was a moderate correlation between BMI and the length of hospitalization in days in the positive direction (rs
4.Discussion
This study featured the musculoskeletal symptoms and their effects on the quality of life in 97 patients who applied to the post-COVID treatment unit due to pain and were treated for COVID-19. In the post-COVID period, myalgia was observed in 48 (49.5%) and arthralgia in 37 (38.1%) patients with no previous pain. Forty-seven (49.5%) patients described numbness, burning, and tingling. In addition, neuropathic pain was identified in 41 (42.3%) patients based on the DN4 questionnaire scores. The pain intensity in all 49 patients with myalgia and/or arthralgia before COVID-19 increased after the infection. In addition, the patients included in the study scored significantly lower than the general Turkish population in all subscales of the SF-36 survey, indicating significantly poorer quality of life [25]. There was a significant relationship between the presence of pain before and after the COVID-19 infection and neuropathic pain. In addition, the quality of life decreased with the increase in pain and BMI values.
Several studies have examined the relationship between COVID-19 and musculoskeletal symptoms [23, 26]. In a review of 35 studies on COVID-19, 18 reported that the most common musculoskeletal symptoms were arthralgia and myalgia [27]. Studies that investigated both arthralgia and myalgia in COVID-19 patients reported the rate of patients with both arthralgia and myalgia between 27.8% and 30% of the COVID-19 patients [10, 15]. In Bakilan et al.’s study [13] conducted with 280 post-acute COVID-19 patients 12 weeks after COVID-19 infection, it was determined that 240 (85.7%) patients had at least one musculoskeletal system symptom that either developed anew or aggravated with COVID-19 infection, whereas that 40 (14.3%) patients did not have any musculoskeletal symptom that developed anew or aggravated with COVID-19 infection. In the said study, 60.7% and 43.6% of the patients had myalgia and arthralgia, respectively. In addition, it was determined that muscle and joint pain developed anew or aggravated in 51.1% and 36.1% of the COVID-19 patients, respectively. In comparison, in this study, 96 (99%) and 77 (79.4%) patients had myalgia and arthralgia 4.0
During the infection, muscle fibers in ischemic muscle experience an increase in sarcoplasmic reticulum proteins such as creatine kinase, lactate, alanine aminotransferase, and aspartate aminotransferase. Simultaneously, there is a decrease in pH and oxygen levels. Among the proinflammatory molecules produced by COVID-19 infection that affect musculoskeletal tissues, interleukin-6, interleukin-1
Studies have reported back pain as the most common (6.8%–43%) complication in the spine after COVID-19 infection [8, 10, 29, 30, 31, 32]. The VAS scores in these patients with pain ranged between 4.8 and 7.2. Bakilan et al. [13] reported that 70.1% of the 280 post-acute COVID-19 patients had spinal pain, which was mostly observed in the back (30.4%), followed by the waist (16.1%), and the neck (11.4%), and that joint pain was mostly observed in the knee (12.1%) followed by the shoulder (5.7%). In a cohort study covering 1.5 to 3 months after COVID-19 diagnosis with a positive PCR test, Oguz-Akarsu et al. [30] determined that 159 (71.6%) of the 222 patients had pain and that the most common type of pain was back pain (41.8%), followed by widespread pain (36.4%) and extremity pain (21.8%). In another study conducted with 1712 COVID-19 patients, the most common site of pain was the head, followed by the neck, back, lower back, and shoulder [33]. Mils et al. reported that pain was detected in half of the patients with COVID-19 infection and that it was most commonly (27%) observed in the lower extremities, followed by the lumbar region (19%), and the upper extremities (13%) [34]. In comparison, the most common (60.8%) site for muscle pain after COVID-19 infection in patients included in this study was the neck-back-waist region, followed by the calves (45.4%) and the arms (44.3%), while the most common (44.3%) site for the joint pain after COVID-19 infection was the shoulder followed by the knee (40.2%). The mean VAS score of these patients after COVID-19 infection was 6.9
The prevalence of neuropathic pain in patients with COVID-19 infection reported in the literature varies between 0.4% and 25% [14, 29, 30, 35, 36], whereas the prevalence of paresthesia varies between 6% and 33% [15, 37]. Miller et al. [36] detected peripheral nerve injury in the upper extremity in 15 of the 114 COVID-19 patients hospitalized in the prone position and treated in the intensive care unit for an average of 32.5 days. The electroneuromyographic assessment revealed neuropathic pain, and motor weakness in all the said 15 patients on, indicating high-grade nerve damage. In another study conducted with 135 COVID-19 patients, sensory impairment was detected in 20 (14.8%) patients, while 29 (25%) patients described numbness, burning, and tingling [38]. In a study conducted with 29 patients that were followed up for an average of 24 days in the intensive care unit for COVID-19 infection, the incidence of neuropathic pain within 4–6 weeks after discharge was reported as 21% [14]. In comparison, in this study, sensory dysfunction was detected in 18 (18.6%) patients, neuropathic pain was detected in 41 (42.2%) patients based on the scores obtained from the DN-4 questionnaire, and 47 (48.5%) patients described numbness, burning, and tingling after the COVID-19 infection. Accordingly, it was determined that the pain before COVID-19 was associated with neuropathic pain.
It has been well-established in the literature that the quality of life of patients with COVID-19 is adversely affected [23, 38, 39, 40, 41]. A study reported that the quality of life was adversely affected even approximately seven months after the infection in 52.2% of COVID-19 patients. In another study, it was determined that all COVID-19 subgroups scored lower than the general population in the SF-36 survey in the 3rd month after discharge [42, 43]. In a systematic review by Malik et al., including twelve studies that assessed the quality of life in COVID-19 patients using the European Quality of Life 5 Dimensions and 5 Levels Scale (EQ-5D-5L), the prevalence of mobility, personal care, usual quality, pain/discomfort, and anxiety/depression was 36%, 8%, 28%, 42%, and 38%, respectively [39]. In a study that assessed the quality of life in 323 healthcare workers using the Sheehan Disability Scale, it was determined that work life, social life, and life at home were adversely affected due to the ongoing symptoms of COVID-19 infection in26%, 15%, and 12% of the workers, respectively [40]. Menges et al. reported that at least one dimension of quality of life, i.e., pain/discomfort in 39% and anxiety/depression in 31% of the patients, was adversely affected in 225 (52.2%) of the 431 patients after an average of 7.2 months after they were diagnosed with COVID-19 [42]. In another study, 110 COVID-19 patients scored significantly lower in all subscales of the SF-36 survey an average of 83 days after they were discharged from the hospital compared to the age-matched general population [43]. In a study that compared patients who received and did not receive corticosteroids during COVID-19 infection in terms of quality of life, it was determined that the patients who received corticosteroids obtained significantly better results from the ’physical pain’ and ’mental health’ subscales of the SF-36 survey and that corticosteroids reduced long-term symptoms and improved quality of life [39]. Rass et al. [38] determined using the SF-36 that the quality of life of 28 (31%) of the 90 COVID-19 patients deteriorated three months after COVID-19 infection. Of these 28 patients, six and 16 obtained significantly lower scores from the physical and mental components of SF-36, respectively. The remaining six received significantly lower scores from both mental and physical components. In comparison, the patients included in this study scored significantly lower compared to the general Turkish population in all subscales of the SF-36 survey [25]. On the other hand, there was no significant difference between the subgroups created according to the place of treatment and age in terms of quality of life. In addition to the rate of hospitalization and steroid use, the vitality subscale score was higher among the patients with lung involvement. Additionally, female COVID-19 patients had significantly lower SF-36 physical function and pain subscale scores. Lastly, the presence of pain severity and neuropathic pain both before and after the COVID-19 infection were found to be associated with quality of life.
It is important to consider that pain may manifest in the musculoskeletal system following a COVID-19 infection, or pre-existing pain may intensify. Patients should be thoroughly evaluated in this regard. Seeking pain treatment in specialized clinics holds significance in improving patients’ quality of life. The presence of neuropathic pain is valuable for clinicians and researchers as it aids in determining the neural involvement of the viral agent.
The relatively limited sample size is the primary limitation of this study. In addition, given that the study sample consisted only of patients who applied to the post-COVID clinic with at least one complaint of myalgia or arthralgia, the study results cannot be generalized to all post-acute COVID-19 patients. Lastly, the treatment and laboratory data of the patients, except for steroid use data as well as long-term musculoskeletal system and quality of life outcomes of the patients, were lacking.
5.Conclusion
Arthralgia, myalgia, and neuropathic pain, which negatively affect the quality of life, were detected in approximately one-third of the patients after COVID-19 infection. Therefore, it is recommended to query COVID-19 patients, especially those whose pain increased after COVID-19 infection, in terms of musculoskeletal symptoms and neuropathic pain in particular, in order to manage the respective symptoms and outcomes.
Funding
The authors report no funding.
Informed consent
An informed consent form was included at the beginning of the survey, which presented the necessary information and recorded the participant’s willingness to participate in the study.
Author contributions
All named authors have made an active contribution to the conception design, analysis, interpretation of the data, drafting of the paper and have critically reviewed its content. All authors read and approved the final version submitted for publication.
Acknowledgments
The authors are grateful to all participants in this study.
Conflict of interest
The authors declare that they have no conflicts of interest.
References
[1] | Wang J, Wang Z. Strengths, Weaknesses, Opportunities and Threats (SWOT) Analysis of China’s Prevention and Control Strategy for the COVID-19 Epidemic. Int J Environ Res Public Health. (2020) ; 17: (7): 2235. doi: 10.3390/ijerph17072235. |
[2] | Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. (2020) ; 395: (10223): 497-506. doi: 10.1016/S0140-6736(20)30183-5. |
[3] | World Health Organization. Weekly Epidemiological Update on COVID-19 – 14-December 2022. Available from: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19—14-december-2022. |
[4] | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. (2020) ; 181: (2): 271-280.e8. doi: 10.1016/j.cell.2020.02.052. |
[5] | Disser NP, De Micheli AJ, Schonk MM, Konnaris MA, Piacentini AN, Edon DL, et al. Musculoskeletal Consequences of COVID-19. J Bone Joint Surg Am. (2020) ; 102: (14): 1197-1204. doi: 10.2106/JBJS.20.00847. |
[6] | Misra DP, Agarwal V, Gasparyan AY, Zimba O. Rheumatologists’ perspective on coronavirus disease 19 (COVID-19) and potential therapeutic targets. Clin Rheumatol. (2020) ; 39: (7): 2055-2062. doi: 10.1007/s10067-020-05073-9. |
[7] | Karaarslan F, Güneri FD, Kardeş S. Long COVID: rheumatologic/musculoskeletal symptoms in hospitalized COVID-19 survivors at 3 and 6 months. Clin Rheumatol. (2022) ; 41: (1): 289-296. doi: 10.1007/s10067-021-05942-x. |
[8] | Karaarslan F, Demircioğlu Güneri F, Kardeş S. Postdischarge rheumatic and musculoskeletal symptoms following hospitalization for COVID-19: prospective follow-up by phone interviews. Rheumatol Int. (2021) ; 41: (7): 1263-1271. doi: 10.1007/s00296-021-04882-8. |
[9] | Graham EL, Clark JR, Orban ZS, Lim PH, Szymanski AL, Taylor C, et al. Persistent neurologic symptoms and cognitive dysfunction in non-hospitalized Covid-19 “long haulers”. Ann Clin Transl Neurol. (2021) ; 8: (5): 1073-1085. doi: 10.1002/acn3.51350. |
[10] | Hoong CWS, Amin MNME, Tan TC, Lee JE. Viral arthralgia a new manifestation of COVID-19 infection? A cohort study of COVID-19-associated musculoskeletal symptoms. Int J Infect Dis. (2021) ; 104: : 363-369. doi: 10.1016/j.ijid.2021.01.031. |
[11] | Carvalho-Schneider C, Laurent E, Lemaignen A, Beaufils E, Bourbao-Tournois C, Laribi S, et al. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin Microbiol Infect. (2021) ; 27: (2): 258-263. doi: 10.1016/j.cmi.2020.09.052. |
[12] | Kayaaslan B, Eser F, Kalem AK, Kaya G, Kaplan B, Kacar D, et al. Post-COVID syndrome: a single-center questionnaire study on 1007 participants recovered from COVID-19. J Med Virol. (2021) ; 93: (12): 6566-6574. doi: 10.1002/jmv.27198. |
[13] | Bakılan F, Gökmen İG, Ortanca B, Uçan A, Eker Güvenç Ş, et al. Musculoskeletal symptoms and related factors in postacute COVID-19 patients. Int J Clin Pract. (2021) ; 75: (11): e14734. doi: 10.1111/ijcp.14734. |
[14] | Mateo Rodríguez E, Puchades Gimeno F, Ezzeddine Angulo A, Asensio Samper J, Saiz Ruiz C, López Alarcón MD. Postintensive care syndrome in COVID-19. Unicentric pilot study. Calm does not come after the storm. Med Clin (Engl Ed). (2022) ; 159: (7): 321-326. doi: 10.1016/j.medcle.2021.11.013. |
[15] | Pinzon RT, Wijaya VO, Jody AA, Nunsio PN, Buana RB. Persistent neurological manifestations in long COVID-19 syndrome: A systematic review and meta-analysis. J Infect Public Health. (2022) ; 15: (8): 856-869. doi: 10.1016/j.jiph.2022.06.013. |
[16] | Davido B, Seang S, Tubiana R, de Truchis P. Post-COVID-19 chronic symptoms: a postinfectious entity? Clin Microbiol Infect. (2020) ; 26: (11): 1448-1449. doi: 10.1016/j.cmi.2020.07.028. |
[17] | Salamanna F, Veronesi F, Martini L, Landini MP, Fini M. Post-COVID-19 Syndrome: The Persistent Symptoms at the Post-viral Stage of the Disease. A Systematic Review of the Current Data. Front Med (Lausanne). (2021) ; 8: : 653516. doi: 10.3389/fmed.2021.653516. |
[18] | Leith S, Wheatley RG, Jackson IJ, Madej TH, Hunter D. Extradural infusion analgesia for postoperative pain relief. British Journal of Anaesthesia. (1994) ; 73: (4): 552-558. doi: 10.1093/bja/73.4.552. |
[19] | Bouhassira D, Attal N, Alchaar H, Boureau F, Brochet B, Bruxelle J, et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain. (2005) ; 114: (1–2): 29-36. doi: 10.1016/j.pain.2004.12.01. |
[20] | Unal-Cevik I, Sarioglu-Ay S, Evcik D. A comparison of the DN4 and LANSS questionnaires in the assessment of neuropathic pain: Validity and reliability of the Turkish version of DN4. J Pain. (2010) ; 11: (11): 1129-1135. doi: 10.1016/j.jpain.2010.02.003. |
[21] | Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. (1992) ; 30: (6): 473-483. |
[22] | Koçyiğit H, Aydemir Ö, Ölmez N, Memiş A. Kisa form 36 (KF36)’nın Türkçe versionunun güvenilirligi ve geçerliligi [Validation and reliability of SH-36]. İlaç ve Tedavi Dergisi. (1999) ; 12: : 102-06. |
[23] | Shanbehzadeh S, Tavahomi M, Zanjari N, Ebrahimi-Takamjani I, Amiri-Arimi S. Physical and mental health complications post-COVID-19: Scoping review. J Psychosom Res. (2021) ; 147: : 110525. doi: 10.1016/j.jpsychores.2021.110525. |
[24] | Cohen J. Statistical power analysis for behavioral science (2nd ed.). Hillsdale, New Jersey: Lawrance Erlbaum Associates, Inc., Publisher; (1988) . |
[25] | Demiral Y, Ergor G, Unal B, Semin S, Akvardar Y, Kivircik B, et al. Normative data and discriminative properties of short form 36 (SF-36) in Turkish urban population. BMC Public Health. (2006) ; 6: : 247. doi: 10.1186/1471-2458-6-247. |
[26] | Fernández-de-Las-Peñas C, Navarro-Santana M, Plaza-Manzano G, Palacios-Ceña D, Arendt-Nielsen L. Time Course Prevalence of Post-COVID Pain Symptoms of Musculoskeletal Origin in Patients Who Had Survived to SARS-CoV-2 Infection. Pain. (2021) ; 163: (7): 1220-1231. doi: 10.1097/j.pain.0000000000002496. |
[27] | Khoja O, Silva Passadouro B, Mulvey M, Delis I, Astill S, Tan AL, et al. Clinical Characteristics and Mechanisms of Musculoskeletal Pain in Long COVID. J Pain Res. (2022) ; 15: : 1729-1748. doi: 10.2147/JPR.S365026. |
[28] | Veronesi F, Contartese D, Martini L, Visani A, Fini M. Speculation on the pathophysiology of musculoskeletal injury with COVID-19 infection. Front Med (Lausanne). (2022) ; 9: : 930789. doi: 10.3389/fmed.2022.930789. |
[29] | Jena D, Sahoo J, Barman A, Gupta A, Patel V. Musculoskeletal and neurological pain symptoms among hospitalized COVID-19 patients. Am J Phys Med Rehabil. (2022) ; 101: (5): 411-416. doi: 10.1097/PHM.0000000000001969. |
[30] | Oguz-Akarsu E, Gullu G, Kilic E, Dinç Y, Ursavas A, Yilmaz E, Pandemic Study Team, et al. Insight into pain syndromes in acute phase of mild-to-moderate COVID-19: Frequency, clinical characteristics, and associated factors. Eur J Pain. (2022) ; 26: (2): 492-504. doi: 10.1002/ejp.1876. |
[31] | Şahin T, Ayyildiz A, Gencer-Atalay K, Akgün C, Özdemir HM, Kuran B. Pain Symptoms in COVID-19. Am J Phys Med Rehabil. (2021) ; 100: (4): 307-312. doi: 10.1097/PHM.0000000000001699. |
[32] | Murat S, Dogruoz Karatekin B, Icagasioglu A, Ulasoglu C, İçten S, Incealtin O. Clinical presentations of pain in patients with COVID-19 infection. Ir J Med Sci. (2021) ; 190: (3): 913-917. doi: 10.1007/s11845-020-02433-x. |
[33] | Aratel M, Bulut ZI, Sari EO, Pelin Z, Yakut Y. The profile of musculoskeletal pain and its associations with sleep quality and depression during the COVID-19 in Turkey. Korean J Pain. (2022) ; 35: (1): 78-85. doi: 10.3344/kjp.2022.35.1.78. |
[34] | Mills G, Briggs-Price S, Houchen-Wolloff L, Daynes E, Singh S. The prevalence and location of musculoskeletal pain following COVID-19. Musculoskeletal Care. (2022) ; 20: (4): 972-976. doi: 10.1002/msc.1657. |
[35] | Di Stefano G, Falco P, Galosi E, Di Pietro G, Leone C, Truini A. A systematic review and meta-analysis of neuropathic pain associated with coronavirus disease 2019. Eur J Pain. (2023) ; 27: (1): 44-53. doi: 10.1002/ejp.2055. |
[36] | Miller C, O’Sullivan J, Jeffrey J, Power D. Brachial Plexus Neuropathies During the COVID-19 Pandemic: A Retrospective Case Series of 15 Patients in Critical Care. Phys Ther. (2021) ; 101: (1): pzaa191. doi: 10.1093/ptj/pzaa191. |
[37] | Eitner L, Maier C, Brinkmann F, Schlegtendal A, Knoke L, Enax-Krumova E, Lücke T. Somatosensory abnormalities after infection with SARS-CoV-2 – A prospective case-control study in children and adolescents. Front Pediatr. (2022) ; 10: : 977827. doi: 10.3389/fped.2022.977827. |
[38] | Rass V, Beer R, Schiefecker AJ, Kofler M, Lindner A, Mahlknecht P, et al. Neurological outcome and quality of life 3 months after COVID-19: A prospective observational cohort study. Eur J Neurol. (2021) ; 28: (10): 3348-3359. doi: 10.1111/ene.14803. |
[39] | Malik P, Patel K, Pinto C, Jaiswal R, Tirupathi R, Pillai S, et al. Post-acute COVID-19 syndrome (PCS) and health-related quality of life (HRQoL) – A systematic review and meta-analysis. J Med Virol. (2022) ; 94: (1): 253-262. doi: 10.1002/jmv.27309. |
[40] | Havervall S, Rosell A, Phillipson M, Mangsbo SM, Nilsson P, Hober S, et al. Symptoms and functional impairment assessed 8 months after mild COVID-19 among health care workers. JAMA. (2021) ; 325: (19): 2015-2016. doi: 10.1001/jama.2021.5612. |
[41] | Hansel TC, Saltzman LY, Melton PA, Clark TL, Bordnick PS. COVID-19 behavioral health and quality of life. Sci Rep. (2022) ; 12: (1): 961. doi: 10.1038/s41598-022-05042-z. |
[42] | Menges D, Ballouz T, Anagnostopoulos A, Aschmann HE, Domenghino A, Fehr JS, et al. Burden of post-COVID-19 syndrome and implications for healthcare service planning: A population-based cohort study. PLoS One. (2021) ; 16: (7): e0254523. doi: 10.1371/journal.pone.0254523. |
[43] | Arnold DT, Hamilton FW, Milne A, Morley AJ, Viner J, Attwood M, et al. Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: results from a prospective UK cohort. Thorax. (2020) ; 76: (4): 399-401. doi: 10.1136/thoraxjnl-2020-216086. |




