Psychometric Evaluation of the A-NKS: A Diagnostic Instrument to Assess Impairment of Activities of Daily Living in Mild and Major Neurocognitive Disorder According to DSM-5
Abstract
Background:
Activities of daily living (ADL) functioning are important in the diagnosis of neurocognitive disorders (NCD), yet no standardized and validated instrument exist based on international classification systems.
Objective:
We aimed to psychometrically evaluate the differentiated assessment of ADL and instrumental ADL (IADL) impairments due to NCD according to DSM-5 criteria (Instrument für die Erfassung von Alltagsbeeinträchtigungen bei Neurokognitiven Störungen; A-NKS).
Methods:
We conducted a pilot study involving 92 participant-informant dyads of participants with mild or major NCDs, cognitively healthy individuals, and an informant, to test acceptability, internal consistency, and convergent validity with similar measures.
Results:
Both A-NKS versions demonstrated excellent internal consistency (α= 0.95 –0.99) and correlate with other instrumental ADL instruments (participant [informant]: Barthel Index: rs = –0.26, p≤0.05 [rs = –0.30, p≤0.01]; Amsterdam IADL: rs = 0.59, p≤0.01 [rs = 0.48, p≤0.01]; SIDAM ADL: rs = 0.46, p≤0.001 [rs = 0.47, p≤0.001]). Additionally, there are correlations with the scale autonomy of the WHOQOL-OLD (rs = -0.50, p≤0.001 [rs = –0.37, p≤0.001]) and physical, as well as cognitive activities (rs = -0.39, p≤0.001 [rs = –0.50, p≤0.001]). They were well-accepted by participants and informants.
Conclusions:
The A-NKS is an instrument with acceptable psychometric properties to assess ADL due to neurodegenerative decline in healthy individuals, and those with mild or major NCD. Further research is needed to confirm reliability and validity and investigate the factor structure.
INTRODUCTION
Recently, there are over 50 million individuals worldwide living with dementia, a number expected to reach 152 million by 2050 [1]. In Germany, the corresponding figure is 1.7 million individuals, with a projected increase to 2.2 million by 2030 [2]. Dementia is a syndrome characterized by a progressive decline in cognitive functioning, leading to a corresponding progressive decline in the ability to execute activities of daily living (ADL). Impairment in ADL functioning is critical for differentiating between mild and major neurocognitive disorder (NCD). The diagnosis of NCD in DSM-5 (Diagnostic and Statistical Manual of Mental Disorders, 5th edition) differs significantly from that in DSM-IV [3, 4]. While the DSM-IV categorized dementia as a distinct diagnostic group, including amnestic disorders and delirium, the DSM-5 no longer uses the term dementia. Instead, all forms of cognitive impairment are grouped under the term neurocognitive disorders (NCDs), which are differentiated by severity (mild or major), etiology (e.g., Alzheimer’s type, vascular, Lewy body), and associated symptoms (e.g., behavioral disturbances) [4]. The diagnosis of NCD requires neuropsychological testing with standardized measures and a comparison with age- and education-normed values. NCDs are also intended to capture early stages of dementia, which were previously referred to as mild cognitive impairment (MCI) [5, 6], a term not previously listed in the DSM but categorized as mild cognitive disorder in the International Statistical Classification of Diseases and Related Health Problems in its 10th edition (ICD-10) [7].
ADL and instrumental ADL (IADL) can be seen as different concepts: ADL refers to basic self-care tasks such as bathing and dressing, and IADL to more complex tasks such as managing finances and using transportation. A mild NCD diagnosis requires mildly impaired IADL, while for a major NCD diagnosis a more severe impairment requiring support is defined as diagnostic criterion [8]. In contrast, basic ADL remain stable over a longer period and are only severely impaired in major NCD, necessitating assistance [9]. Despite the diagnostic importance of ADL impairment for differentiating between mild and major NCD, how to identify this is not specified in diagnostic guidelines, leaving flexibility and uncertainty for the assessment of ADL functioning for clinicians [10]. This is also true for the diagnosis of dementia compared to mild cognitive disorder according to ICD-10 [7] since only a few examples of ADL impairments that may be associated with cognitive impairments in the context of dementia are provided [7]. The current German guidelines for the diagnosis of dementia [11] also recommend the collection of a detailed self, third-party, family, and social assessment of ADL impairments, but there is also a lack of specifications on how exactly ADL impairments should be assessed (e.g., how many and which ADLs must be impaired and in which way).
The DSM-5 offers more detailed guidelines in this regard [12]. For both mild and major NCDs the DSM-5 provides comprehensive examples of possible ADL impairments corresponding to differentiated six specified neurocognitive domains (complex attention, learning and memory, perceptual-motor functions, language, social cognition, and executive functioning). However, it remains unclear to what extent these examples are based on empirical findings. The DSM-5 also does not specify an instrument to assess ADL impairments. The latter also applies to the assessment of ADL impairments in the context of dementia diagnosis according to the ICD-10 criteria. The instruments listed in the German guidelines for the diagnosis of dementia [11] primarily refer to psychological and behavioral symptoms. To specifically capture ADL, two instruments, the Disability Assessment for Dementia (DAD) [13] and the instrumental IADL scale by Lawton and Brody (1969) [14] are stated. Both instruments, however, only allow an assessment of overall (I)ADL such as bathing, dressing, toileting, continence, feeding or mobility.
To enable a more reliable, standardized, and differentiated assessment of ADL impairments in the context of a dementia-related process of cognitive decline, we therefore recently developed an instrument for the assessment of impairments in activities of daily living due to neurocognitive disorders in German language (Instrument für die Erfassung von Alltagsbeeinträchtigungen bei Neurokognitiven Störungen/A-NKS) [15]. This followed a rigorous development process, described below, which included literature research, expert interviews and expert workshops to promote content validity. Furthermore, the A-NKS has not been examined in a larger sample regarding its psychometric properties or factor structure.
In this study, our aim is to address the psychometric evaluation of the German version of the A-NKS in terms of acceptability, internal consistency, and convergent validity. Our second aim is to provide a first English version of the A-NKS for potential international use.
METHODS
Ethics
This work was approved by the Ethics Committees of the Medical Faculty of the University of Leipzig (ref: 217/21-ek) and of the University Hospital Jena (responsible for the University of Applied Sciences Nordhausen; ref: AZ 2020-1709-Bef & 2020-1709_1-Bef). All participants provided written informed consent before participating in the study.
The instrument development was carried out in cooperation of the Nordhausen University of Applied Sciences (Prof. Dr. rer. med. habil. Tobias Luck, principal investigator) with the University of Leipzig (Prof. Dr. med. Steffi G. Riedel-Heller, MPH, cooperation partner).
A-NKS
The items of the A-NKS were generated in four steps by Funke et al. (2022) [15]:
1. A literature search was conducted in (inter-)national professional databases to gather relevant information and generate a potential item pool for the A-NKS.
2. Existing survey instruments were analyzed to further enrich and refine the item pool.
3. An expert consultation was conducted through a standardized postal survey of 20 clinically practicing diagnosticians (dementia and cognition researchers, psychiatrists, and neurologists with expertise in neurocognitive disorders). During the expert consultation, participants were asked to select five items from the overall item pool of each neurocognitive domain that they deemed particularly suitable. This process aimed to determine any preferences for specific items and assess their differentiating ability by asking participants to indicate whether any items should be assigned to a different domain than originally intended. Quantitative analysis was conducted based on the participants’ ratings of the suitability of the questionnaire for assessing the six neurocognitive domains, as well as a proposed alternative item presentation. Additionally, qualitative analysis was performed on the participants’ free-text comments. The item reduction, following the revision based on expert opinions, resulted in a reduction of approximately 52% (from 92 to 44 items).
4. The fourth step was based on the item pool developed in steps 1 to 3. Eight expert workshops were conducted, involving a total of 24 participants, including elderly individuals, relatives of elderly individuals with dementia, care and health professionals, and diagnosticians. The workshops were semi-structured, and each workshop was documented based on a conversation guide. Two project members facilitated each workshop, with one serving as the moderator and the other as the note-taker. During the workshops, the penultimate version of the A-NKS was reviewed for its usability, completeness, and comprehensibility. The feedback obtained was primarily qualitative, complemented by quantitative frequency counts of similar responses. The revision process resulted in a further reduction of approximately 11% of the items, resulting in a preliminary final instrument comprising 39 items.
Through expert consultation and workshops, the content validity of the A-NKS was also assessed. An overview is provided in Fig. 1.
The A-NKS includes 39 items and comprises six subscales according to the six specified neurocognitive domains in DSM-5 : 1) complex attention; 2) learning and memory; 3) perceptual-motor functions; 4) language; 5) social cognition and 6) executive functioning. The questions are answered with 0 “no difficulties”, 1 “minor difficulties”, 2 “medium difficulties”, 3 “major difficulties”, 4 “I/she/he am/is not able to execute this activity at all” and 5 “does not apply to me/her/him” [15].
Psychometric testing
Recruitment and participants
Recruitment was conducted in the public local community of Leipzig, Germany, e.g., via posters in supermarkets, garden clubs, cafés, libraries or senior citizens’ offices and memory outpatient clinics. In addition, existing networks were used (e.g., inviting subjects of completed studies). Participants aged≥60 years were included and divided into three groups: 1) Cognitively healthy individuals, 2) Individuals with mild NCD, and 3) Individuals with major NCD. Diagnosis was made using the DSM-5 [12] for NCD (see Table 1) or made by a specialist, as described below.
Table 1
DSM-5 criteria for mild and major neurocognitive disorder
| Mild Neurocognitive Disorder | Major Neurocognitive Disorder |
| Criterion A | |
| Evidence of modest cognitive decline from a previous level of performance in one or more cognitive domains (complex attention, executive function, learning and memory, language, perceptual-motor, or social cognition) based on: | Evidence of significant cognitive decline from a previous level of performance in one or more cognitive domains (complex attention, executive function, learning and memory, language, perceptual-motor, or social cognition) based on: |
| 1. Concern of the individual, a knowledgeable informant, or the clinician that there has been a mild decline in cognitive function; and | 1. Concern of the individual, a knowledgeable informant, or the clinician that there has been a significant decline in cognitive function; and |
| 2. A modest impairment in cognitive performance, preferably documented by standardized neuropsychological testing or, in its absence another quantified clinical assessment. | 2. A substantial impairment in cognitive performance, preferably documented by standardized neuropsychological testing or, in its absence, another quantified clinical assessment. |
| Criteria B | |
| The cognitive deficits do not interfere with capacity for independence in everyday activities (i.e., complex instrumental activities of daily living such as baying bills or managing medications are preserved but greater effort, compensatory strategies, or accommodation may be required). | The cognitive deficits interfere with independence in everyday activities (i.e., at a minimum, requiring assistance with complex instrumental activities of daily living such as paying bills or managing medications). |
| Criteria C | |
| The cognitive deficits do not occur exclusively in the context of a delirium. | |
| Criteria D | |
| The cognitive deficits are not better explained by another mental disorder (e.g., major depressive disorder, schizophrenia) | |
| Reference: American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders [12] | |
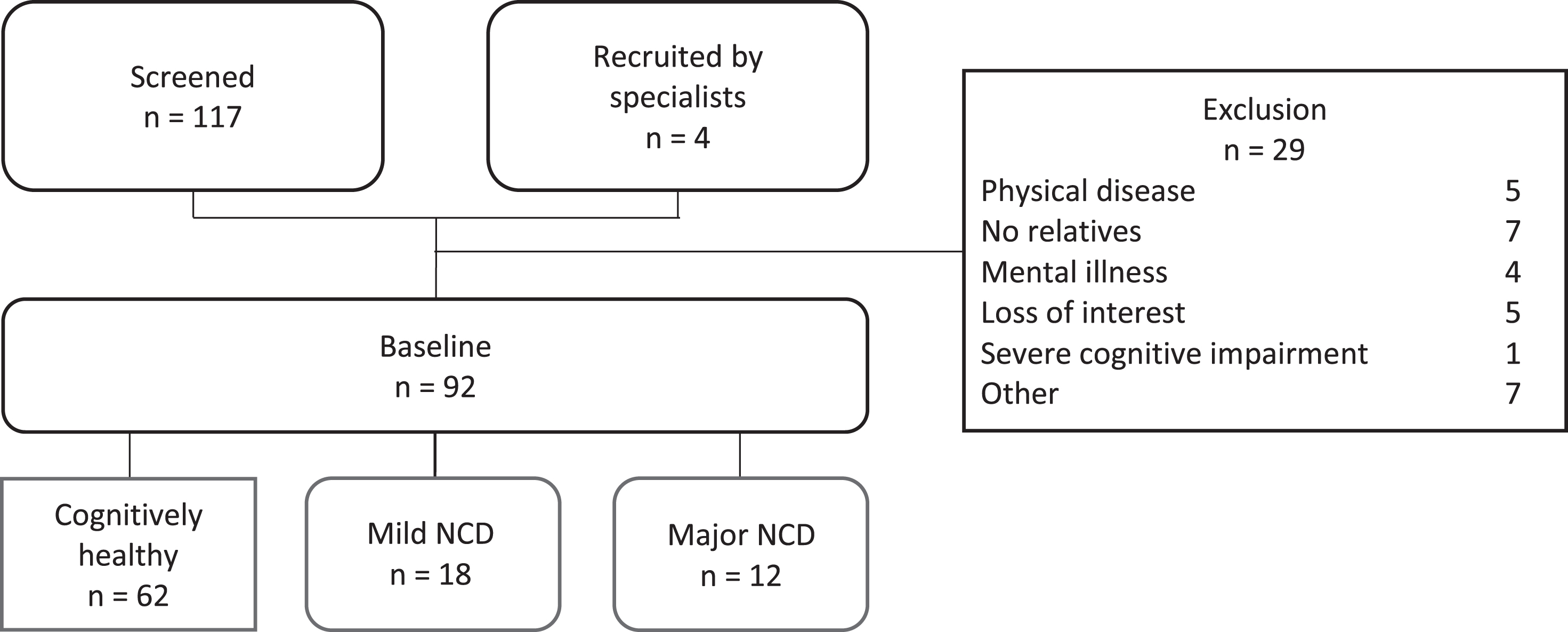
We excluded those with other mental disorders or severe physical illness (e.g., cancer, neurological disease, substance use, current depression, current anxiety). For each participant in the study, including individuals without NCD, mild NCD and major NCD, we recruited a German-speaking informant who was in contact with them at least once a week and aged 18≥years, to obtain an accurate assessment of their ADL function. Of the 118 participants screened, 92 were included and agreed to participate in the study (see Fig. 2). The details regarding the screening process can be found in the next section.
Fig. 2
Flowchart –recruitment and analytic sample selection. NCD, neurocognitive disorder.
The recruitment period was from July 2021 to March 2022, with face-to-face data collection from August 2021 to April 2022.
Procedures
Structured face-to-face interviews were conducted with several researchers. Interviews included a standardized questionnaire on sociodemographic data, the A-NKS and additional assessments of ADL and IADL for comparison, assessments of cognitive abilities, as well as questions related to the usability A-NKS, detailed below.
Once enrolled in the study, participant-rating and informant-rating methods were used to collect data. The assessments were conducted at our institute and included face-to-face interviews and self-administered questionnaires. The assessment conducted with the participants was almost identical to the one conducted with their informants, but included a more extensive neurocognitive evaluation. A detailed description of the instruments used will follow.
Screening
All participants recruited from the general population underwent telephone screening to determine their eligibility for the study and to assign them to the appropriate group (without NCD, mild NCD or major NCD). Participants recruited from memory clinics were exempted from this screening process, as they had already been assessed and diagnosed by a specialist using a gold-standard process that considers various sources of information, and an additional appointment for screening could have been an additional burden.
Screening measures. The telephone screening included the telephone version of the Montreal Cognitive Assessment (T-MoCA), questions about subjective cognitive decline (SCD), physical and mental well-being.
The T-MoCA is a cognitive test battery that assesses aspects of memory, attention, language, abstraction, delayed recall, and orientation, with a maximum score of 22 [16]. Higher scores indicated higher cognitive functioning. Scores more than 18 points indicate no cognitive impairment. Scores ranging from 13 to 17 suggest mild cognitive impairment and scores between 7 and 12 indicates moderate cognitive impairment. Scores below 7 are indicative of severe cognitive impairment, which resulted in exclusion from the study [16].
To exclude participants with other significant mental health problems/mental disorders, screening questions from the Structured Clinical Interview for DSM-5 Axis I [17] were used to identify indications of the current presence of anxiety, trauma, and compulsive spectrum disorders. In addition, questions of the Patient Health Questionnaire Version 2 (PHQ-2) [18] were used for affective disorders. Questions about addictions (including non-substance-related addictions) and physical health problems (such as severe physical illnesses and specific pre-existing conditions, including Parkinson’s disease, stroke, traumatic brain injury, heart disease, hypertension, or diabetes) were added, as well as a question about current or previous treatment of mental health problems. In addition, participants were asked if they were currently suffering from a severe physical illness. Those with presence/indication of the above stated mental and physical problems/disorders were excluded from participating in the study.
Measures for all dyads. Face-to-face and self-administered questionnaires were used to conduct participant- and informant interviews. the questionnaires covered a range of topics, including sociodemographic data, the A-NKS as participant- and informant-rating, cognitive and functional assessments.
Trained interviewers conducted the face-to-face interviews in a private and quiet room at our institute. The self-administered questionnaires were completed on site, and at their own pace.
Sociodemographic data included age, sex, marital status, education, and living situation, among others. To assess SCD in participants, we asked the following yes/no question: “Do you feel like your/his/her memory is getting worse?”.
Depressive symptoms were assessed using the German 15-item version of the Geriatric Depression Scale (GDS) [19]. It includes a total of 15 questions about depressive symptoms answered with “yes” or “no”. Item values are summed up to a total score. The total score ranges from 0 and 15, with higher values indicating a higher probability of depression.
Assessment of acceptability
To assess the acceptability of the A-NKS, participants were asked to rate its clarity, structure, and length on a Likert scale (possible answers: very acceptable, acceptable, unacceptable, and very unacceptable) and its clarity, structure, and length (of the single items and in total; possible answers: yes/no; comments) in participants and their informants. There were also invited to provide general feedback on the instrument. It should be noted that content validity was initially established through the development of the instrument in conjunction with patients and experts [15], as described above.
Assessment of convergent validity
Measuring ADL and IADL is important to determine the convergent validity of the A-NKS because we hypothesized these to be correlated. In this study, ADL were assessed using the Barthel Index (BI) and IADL were assessed using the short form of the Amsterdam IADL Questionnaire and Structured Interview for the Diagnosis of Dementia of the Alzheimer Type, Multi-Infarct Dementia, and Dementias of Other Etiology According to ICD-10 and DSM-IV (SIDAM IADL), in participants and their informants. These instruments are used to assess the convergent validity of the A-NKS.
The BI [20] is a questionnaire for systematically assessing ADL which asks about 10 basic ADLs (for example, personal hygiene, food intake, and toilet use). The maximum score of 100 indicates most independence, with a score of 0 indicating complete dependence in performing basic ADLs.
The short form of the Amsterdam IADL Questionnaire detects cognitive IADL problems that are common in the early stages of NCD by covering a broad range using 30 questions. On a five-point Likert scale ranging from “no difficulty in performing this task” to “no longer able to perform this task”, difficulty in performing IADL is rated [21]. The higher the score, which ranges from 0 to 120, the more difficulties in performing IADL due to cognitive difficulties.
In addition, the IADL scale from the SIDAM was used to assess psychosocial functioning in daily life. It asks whether it is possible to independently perform each of the 14 activities, with responses to “yes” and “no” answers [22]. The total score ranges from 0 to 14, with higher scores indicating fewer limitations in IADL functioning.
Autonomy was assessed using the WHOQOL-OLD scale of the same name. The scale comprises 4 questions, each rated on a five-point Likert scale (1 “not at all” to 5 “an extreme amount”). Higher scores indicated more autonomy. [23]
We chose to use two out of the five EQ-5D items, as they specifically relate to ADL and are therefore conceptually similar to the A-NKS measure. The questions two and three ask about general activities and self-care, which are both important aspects of ADL [24]. Both questions were answered using a five-point Likert scale (1 “no problems” to 5 “not able”).
Cognitive and physical activities were evaluated following the methodology of Verghese et al. (2003), with slight adaptations [25]. Participants reported their engagement in activities over the past four weeks using a five-point ordinal scale indicating frequency (0 “never” to 4 “daily”). The assessment included seven items for physical activities, such as bicycling, walking, swimming, gymnastics, chores/gardening, and a category for other physical activities (e.g., bowling, dancing, bicycling, walking, or golfing). Cognitive activities comprised eight items, including crossword puzzles, memory training/brainteasers, games (card games, board games, or individual games), reading, writing, and playing a musical instrument. Sum score was computed for analysis. Higher scores indicated a higher level of activity.
Neurocognitive assessment for participants
Neurocognitive function was assessed in participant-rated interviews using neuropsychological tests of the Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) test battery were conducted [26], including the Trail making test (TMT) parts A and B, Word list memory/learning, Verbal fluency to test semantic fluency and Constructional praxis [27]. These instruments were used not only to obtain data to validate the A-NKS, but also to determine cognitive function. Notably, in the TMT A and TMT B, a higher score indicates greater deterioration.
Social cognition was assessed using the short form of the “Reading the Mind in the Eyes Test” (RMET) [28]. The test contained 18 pictures presenting the eye region of human faces displaying different facial expressions. Participants were instructed to identify what the person in the picture is thinking or feeling by selecting the most appropriate adjective out of the four displayed around the picture.
Statistical analysis
Differences in sociodemographic, cognitive and health characteristics between cognitively healthy individuals, individuals with mild NCD and major NCD were assessed using Kruskal-Wallis-Test (H-test) for continuous variables and Chi square tests (χ2) for categorical variables. For the A-NKS, we assessed internal consistency using Cronbach’s α. Spearman’s rank coefficient was calculated to determine item-total and item-item correlation. On single item level, Cohen’s kappa was calculated to determine the participant-informant agreement, as a measure of convergent validity [29]. Moreover, we calculated the Intraclass Correlation Coefficient (ICC) for agreement in total scores between participant- and informant-rating.
To investigate whether the A-NKS specifically captures ADL impairments related to the six neurocognitive domains of the DSM-5, descriptive statistics were computed for each subscale. In addition, a group-specific descriptive analysis of the A-NKS scores was performed to examine whether the A-NKS scores were associated with mild and severe NCD, with the aim of testing the criterion validity. The Kruskal-Wallis test was used to test the significance of differences between diagnostic groups.
Convergent validity
To establish the convergent validity, we calculated the Spearman’s rank correlation coefficient based on the correlation between A-NKS scores and (I)ADL performance measured by other widely used instruments assessing (I)ADLs (BI, Amsterdam IADL, SIDAM IADL). Additionally, we correlated A-NKS scores with cognitive performance in the six cognitive domains specified by the DSM-5 cognitive assessments mentioned above (TMT A and B, word list memory/learning, verbal fluency, constructional praxis, and RMET). TMT A was used to validate the “complex attention” subscale, TMT B for the “executive functions” subscale, the word list memory/learning test for the “learning and memory” subscale, the verbal fluency test for the “language” subscale, and constructional praxis test for the “perceptual-motor function” subscale. The RMET scale was used to test the convergent validity of the A-NKS subscale “social cognition” (see Table 2).
Table 2
Instruments used to estimate construct and criterion validity
| A -NKS subscale | Participants | Informants |
| A-NKS total | Barthel-Index; | Barthel-Index; |
| Amsterdam IADL; | Amsterdam ADL; | |
| SIDAM ADL; | SIDAM ADL; | |
| WHOQOL-OLD subscale “autonomy”; | WHOQOL-OLD subscale “autonomy”; | |
| EQ2, EQ3; | EQ2, EQ3; | |
| physical and cognitive activities | physical and cognitive activities | |
| 1. Complex attention | CERAD subscale TMT A | |
| 2. Executive functions | CERAD subscale TMT B | |
| 3. Learning and memory | CERAD subscale word list recall | |
| 4. Language | CERAD subscale verbal fluency animals | |
| 5. Perceptual-motor function | CERAD subscale constructive practice | |
| 6. Social cognition | RMET |
Amsterdam IADL, Amsterdam Instrumental Activities of Daily living [21]; A-NKS, Pilot version of the Instrument for the Assessment of Daily Living Impairments due to Neurodegenerative Decline [15]; Barthel Index [20]; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease [26]; EQ2 and EQ3, Item 2 and Item 3 from the Quality of Life Questionnaire EQ-5D [24]; GDS, Geriatric Depression Scale [19]; RMET, Reading the Mind in the Eyes – Short Form [28]; SIDAM-IADL, Structured Interview for the Diagnosis of Dementia of the Alzheimer’s Type of Multiinfarct Dementia and Dementias of Other Etiologies according to DSM-III-R and ICD-10. Manual [22]; T-MoCA, Telephone Montreal Cognitive Assessment [16]; TMT A, Trail Making Test A from CERAD [26]; TMT B, Trail Making Test B from CERAD [26]; WHOQOL-OLD Autonomy, Quality of Life in Aging – subscale autonomy [23].
Moreover, we hypothesized that the A-NKS score, reflecting ADL performance, would be correlated with more autonomy, better quality of life, and better cognitive performance (WHOQOL OLD autonomy subscale, EQ-5D).
IBM SPSS 27 and STATA 16 were used for statistical analysis [30]. All analyses employed an α-level for statistical significance of 0.05 (two-tailed).
RESULTS
Sample characteristics
Table 3 shows sociodemographic and clinical characteristics of the 92 dyads included in the analysis. Participants had a mean age of 71.7 (SD = 8.2; cognitively healthy individuals: M = 69.1, SD = 7.3; individuals with mild NCD: M = 77.6, SD = 7.9 and individuals with major NCD: M = 76.3, SD = 7.4) years, and 68.5% were women. The majority were married at the time of the interview (60.9%). The majority were highly educated (50.0%). The mean T-MoCA score was 17.4 (SD = 3.2).
Table 3
Sample characteristics (N = 92)
| Participants | Informants | |||||||||
| Total (n = 92) | CH (n = 62) | mild NCD (n = 18) | major NCD (n = 12) | p | Total (n = 92) | CH (n = 62) | mild NCD (n = 18) | major NCD (n = 12) | p | |
| Sex, n (%) | 0.470 | 0.329 | ||||||||
| Female | 63 (68,5) | 45 (72.6) | 11 (61.1) | 7 (58.3) | 51 (55.4) | 32 (51.6) | 10 (55.6) | 8 (72.7) | ||
| Male | 29 (31.5) | 17 (72.4) | 7 (38.9) | 4 (71.7) | 41 (44.6) | 30 (48.4) | 8 (44.4) | 3 (27.3) | ||
| Age, M (SD) | 71.71 (8.23) | 69.11 (7.26) | 77.56 (7.91) | 76.33 (7.38) | ≤0.001 | 61.51 (16.86) | 59.23 (18.10) | 67.44 (13.51) | 64.42 (12.43) | 0.266 |
| Marital status, n (%) | 0.848 | 0.807 | ||||||||
| Married | 56 (60.9) | 36 (58.1) | 12 (66.7) | 8 (66.7) | 60 (65.2) | 40 (64.5) | 12 (66.7) | 8 (66.7) | ||
| Partnership | 4 (4.4) | 4 (6.5) | 0 (0.0) | 0 (0.0) | ||||||
| Single | 3 (3.3) | 2 (3.2) | 1 (5.6) | 0 (0.0) | 21 (22.8) | 13 (21.0) | 5 (27.8) | 3 (25.0) | ||
| Divorced | 20 (21.7) | 16 (25.8) | 2 (11.1) | 2 (16.7) | 5 (5.4) | 3 (4.8) | 1 (5.6) | 1 (8.3) | ||
| Widowed | 13 (14.1) | 8 (12.9) | 3 (16.7) | 2 (16.7) | 2 (2.2) | 2 (3.2) | 0 (0.0) | 0 (0.0) | ||
| Living situation, n (%) | 0.049 | |||||||||
| Alone in private household | 31 (33.7) | 25 (40.3) | 3 (16.7) | 3 (25.0) | ||||||
| In private household with spouse | 55 (59.8) | 36 (58.1) | 11 (61.1) | 8 (66.7) | ||||||
| In private household with other relatives | 3 (3.3) | 1 (1.6) | 2 (11.1) | 0 (0.00) | ||||||
| In private household with others | 1 (1.1) | 0 (0.0) | 1 (5.6) | 0 (0.00) | ||||||
| Assisted living | 2 (2.2) | 0 (0.0) | 1 (5.6) | 1 (8.3) | ||||||
| Retirement homes | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||||||
| Nursing home | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||||||
| Education, n (%)* | 0.176 | 0.545 | ||||||||
| Low | 4 (4.4) | 4 (6.5) | 0 (0.0) | 0 (0.0) | 3 (3.3) | 2 (3.4) | 0 (0.0) | 1 (8.3) | ||
| Middle | 42 (45.7) | 25 (40.4) | 8 (44.4) | 9 (75.0) | 46 (51.7) | 28 (47.5) | 10 (55.6) | 8 (66.7) | ||
| High | 46 (50.0) | 33 (53.2) | 10 (55.6) | 3 (25.0) | 40 (44.9) | 29 (49.2) | 8 (44.4) | 3 (25.0) | ||
| Relationship to informant, n (%) | 0.558 | |||||||||
| Spouse/partner | 46 (50.0) | 29 (46.8) | 9 (50.0) | 7 (58.3) | ||||||
| Child/child-in-law | 25 (27.2) | 18 (29.0) | 5 (27.8) | 3 (25.0) | ||||||
| Other relative | 15 (16.3) | 8 (12.9) | 4 (22.2) | 2 (16.7) | ||||||
| Friend/acquaintance | 6 (6.5) | 7 (11.3) | 0 (0.0) | 0 (0.0) | ||||||
CH, cognitively healthy; M, mean; NCD, neurocognitive disorder; SD, standard deviation; * Informants: missing values n = 3 (3.3%).
Half of the informants were spouses/partners of the participant (48.9%) and the remainder were child or child-in-law (27.2%), friends or acquaintances (6.5%) or other (16.3). Their mean age was 61.5 (SD = 16.9) years, 55.4% were females and 65.9% were married.
Regarding cognitive measures, healthy individuals and those with mild NCD differed from those with major NCD in terms of overall cognitive functioning, TMT A, TMT B, word list recall, constructive praxis, verbal fluency and social cognition significantly from each other.
The same was evident in the ADL and IADL data collected, using the BI, Amsterdam IADL, and SIDAM IADL. Here, the cognitively healthy individuals consistently showed less ADL and IADL impairments than individuals with mild or major NCD.
Individuals with major NCD also showed less physical and cognitive activities compared to the other two groups of individuals. Those with mild NCD had less impairment in activities, assessed by two items of the EQ-5D. Depressive symptoms were also found to be lowest in cognitively healthy individuals and highest in individuals with major NCD. This finding was also reflected in autonomy. See Table 4.
Table 4
Cognitive and behavioral parameters
| Total (n = 92) | Cognitively healthy (n = 18) | Mild NCD (n = 12) | Major NCD (n = 62) | p | |
| Cognitive status (T-MoCA), M (SD) | 17.86 (3.19) | 19.08 (2.3) | 16.00 (2.72) | 12.63 (2.77) | ≤0.001 |
| Missing (n (%)) | 4 (4.3) | 0 (0.0) | 0 (0.0) | 4 (33.3) | |
| CERAD TMT A, M (SD) | 42.60 (22.59) | 35.05 (11.49) | 46.89 (16.89) | 78.09 (39.13) | ≤0.001 |
| Missing (n (%)) | 1 (1.1) | 0 (0.0) | 0 (0.0) | 1 (8.3) | |
| CERAD TMT B, M (SD) | 110.07 (66.61) | 88.76 (46.83) | 111.44 (32.61) | 239.70 (72.12) | ≤0.001 |
| Missing (n (%)) | 2 (2.2) | 0 (0.0) | 0 (0.0) | 2 (16.7) | |
| CERAD word list recall, M (SD) | 5.92 (3.25) | 7.53 (2.022) | 3.33 (2.97) | 1.50 (1.20) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| CERAD constructive practice, M (SD) | 9.75 (1.57) | 10.18 (1.05) | 9.89 (1.13) | 7.33 (2.23) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| CERAD verbal fluency, M (SD) | 22.08 (6.36) | 23.97 (5.16) | 21.89 (5.10) | 12.58 (5.30) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Social cognition (RMET), M (SD) | 0.63 (0.16) | 0.68 (0.15) | 0.60 (0.11) | 0.42 (0.13) | ≤0.001 |
| Missing (n (%)) | 1 (1.1) | 0 (0.0) | 0 (0.0) | 1 (8.3) | |
| Barthel Index, M (SD) | 98.15 (7.59) | 99.67 (1.24) | 99.17 (1.92) | 87.75 (18.72) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Amsterdam IADL, M (SD) | 4.07 (11.08) | 1.10 (2.03) | 3.67 (3.87) | 21.45 (25.82) | 0.003 |
| Missing (n (%)) | 2 (2.2) | 1 (1.6) | 0 (0.0) | 1 (8.3) | |
| SIDAM ADL, M (SD) | 0.45 (1.24) | 0.11 (0.37) | 0.44 (0.70) | 2.17 (2.72) | ≤0.001 |
| Missing (n (%)) | 1 (1.1) | 1 (1.6) | 0 (0.0) | 0 (0.0) | |
| Autonomy (WHOQOL-OLD), M (SD) | 12.40 (2.78) | 12.97 (2.41) | 11.71 (2.28) | 10.42 (4.08) | 0.012 |
| Missing (n (%)) | 3 (3.3) | 1 (1.6) | 1 (5.6) | 1 (8.3) | |
| Depressive symptoms (GDS), M (SD). | 1.71 (2.10) | 0.93 (1.32) | 2.76 (1.71) | 4.83 (3.61) | ≤0.001 |
| Missing (n (%)) | 16 (17.4) | 11 (17.7) | 3 (16.7) | 2 (18.2) | |
| Activities – general activities (EQ-02), M (SD) | 1.33 (0.58) | 1.19 (0.44) | 1.47 (0.62) | 1.83 (0.83) | 0.004 |
| Missing (n (%)) | 5 (5.4) | 4 (6.5) | 1 (5.6) | 0 (0.0) | |
| Activities – self-caring (EQ-03), M (SD) | 1.11 (0.52) | 1.03 (0.26) | 1 (0.0) | 1.67 (1.15) | ≤0.001 |
| Missing (n (%)) | 5 (5.4) | 4 (6.5) | 1 (5.6) | 0 (0.0) | |
| Activities (EQ-02 + EQ-03), M (SD) | 2.45 (0.92) | 2.22 (0.62) | 0.47 (0.62) | 3.50 (1.57) | ≤0.001 |
| Missing (n (%)) | 5 (5.4) | 4 (6.5) | 1 (5.6) | 0 (0.0) | |
| Physical and cognitive activities, M (SD) | 36.68 (11.26) | 41.43 (8.55) | 31.67 (8.93) | 19.67 (6.04) | ≤0.001 |
| Missing (n (%)) | 10 (10.9) | 5 (8.1) | 5 (27.8) | 0 (0.0) |
Amsterdam IADL, Amsterdam Instrumental Activities of Daily living [21]; Barthel Index [20]; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease [26]; EQ2 and EQ3, Item 2 and Item 3 from the Quality of Life Questionnaire EQ-5D [24]; GDS, Geriatric Depression Scale [19]; RMET, Reading the Mind in the Eyes – Short Form [28]; SIDAM-IADL, Structured Interview for the Diagnosis of Dementia of the Alzheimer’s Type of Multiinfarct Dementia and Dementias of Other Etiologies according to DSM-III-R and ICD-10. Manual [22]; T-MoCA, Telephone Montreal Cognitive Assessment [16]; TMT A, Trail Making Test A from CERAD [26]; TMT B, Trail Making Test B from CERAD [26]; WHOQOL-OLD Autonomy, Quality of Life in Aging – subscale autonomy [23]; M, mean; NCD, neurocognitive disorder; SD, standard deviation.
A-NKS descriptive results
Range of responses on A-NKS
Participants used the full range of possible responses on 9 of the 39, while informants used the full range of possible responses on 31 of the A-NKS. Overall, 83 dyads (90.2%) reported at least one impairment on the A-NKS (A-NKS total score > 0). ADL impairments were most frequently reported in both participants and informants for questions 2, 7, and 9 (participants: 2: n = 59 (64.1%), 7 and 9: n = 52 (56.5%); informants: 2: n = 61 (66.3%), 7: n = 51 (55.4%), 9: n = 55 (59.8%)). ADL impairments in question 13 and 29 were also frequently reported in participant-rated A-NKS, but not in informant rated A-NKS (13: n = 54 (58.7%), 29: n = 44 (47.8%)). Informants most frequently mentioned ADL impairments related to questions 6 and 32 (6: n = 54 (58.7%), 32: n = 47 (51.1%)). For questions 15 and 34, few ADL impairment were reported in both A-NKS versions (15: participants: n = 5 (5.4%); informants: n = 10 (10.9%); 34: participants: n = 6 (6.5%); informants: n = 12 (13.0%)). Overall, for each question, at least one participant reported an impairment of ADL in participant-rated A-NKS.
Subscales analysis
When considering the subscales of the A-NKS, significant differences in all subscales between the groups are evident for both A-NKS versions. ADL impairments in the subscale 2 learning and memory were mentioned most frequently, as reflected in the mean impairment score (participants: M = 4.6; SD = 4.6; informants: M = 6.2; SD = 7.7). This was followed by subscale 1 complex attention (participants: M = 3.3; SD = 3.7; informants: M = 4.41; SD = 5.3). In subscale 6 executive functioning, the lowest impairment was reported in the participant-rated A-NKS (M = 1.4; SD = 3.0). In the informant-rated A-NKS, however, it was one of the subscales in which the most ADL impairments were reported (M = 3.25; SD = 6.3). These are followed in ascending order by subscales 3 perceptual-motor functions (participants: M = 1.5; SD = 2.2; informants: M = 2.5; SD = 4.3), 4 language (participants: 1.72; SD = 2.7; informants: M = 2.09; SD = 4.4) and 5 social cognition (participants: M = 2.1; SD = 2.4; informants: M = 3.2; SD = 4.6). Tables 5–7 provide an overview of the results of the subscales and their respective items.
Table 5
Means and standard deviations of the A-NKS with subscales
| Participants | Informants | |||||||||
| M±SD (range) | Total (n = 92) | CH (n = 62) | mild NCD (n = 18) | major NCD (n = 12) | p | Total (n = 92) | CH (n = 62) | mild NCD (n = 18) | major NCD (n = 12) | p |
| Complex attention (0–24) | 3.29±3.67 (0–19) | 1.90±1.90 (0–7) | 5.61±3.47 (1–14) | 6.83±6.24 (0–19) | ≤0.001 | 4.17±5.45 (0–11) | 2.43±3.16 (0–18) | 5.23±4.26 (0–14) | 15.27±4.08 (7–20) | ≤0.001 |
| Missing (n (%)) | 1 (1.1) | 1 (1.6) | 0 (0.0) | 0 (0.0) | 3 (3.3) | 2 (3.2) | 0 (0.0) | 1 (8.3) | ||
| Learning and memory (0–32) | 4.61±4.57 (0–23) | 2.87±2.57 (0–12) | 7.56±3.28 (4–16) | 9.47±8.23 (0–23) | ≤0.001 | 6.16±7.66 (0–28) | 2.45±2.69 (0–12) | 8.23±6.66 (0–23) | 21.58±5.05 (15–28) | ≤0.001 |
| Missing (n (%)) | 2 (2.2) | 1 (1.6) | 0 (0.0) | 1 (8.3) | 2 (2.2) | 2 (3.2) | 0 (0.0) | 0 (0.0) | ||
| Perceptual-motor functions (0–24) | 1.46±2.25 (0–11) | 0.64±1.33 (0–8) | 2.56±1.89 (0–6) | 4.16±3.77 (0–11) | ≤0.001 | 2.48±4.30 (0–18) | 0.68±0.95 (0–5) | 2.31±2.48 (0–9) | 11.87±4.76 (1–18) | ≤0.001 |
| Missing (n (%)) | 2 (2.2) | 1 (1.6) | 0 (0.0) | 1 (8.3) | 1 (1.1) | 1 (1.6) | 0 (0.0) | 0 (0.0) | ||
| Language (0–24) | 1.72±2.68 (0–18) | 1.03±1.56 (0–7) | 2.72±2.42 (0–10) | 3.77±5.30 (0–18) | ≤0.001 | 2.09±4.36 (0–21) | 0.61±1.25 (0–8) | 1.06±1.16 (0–4) | 12.00±5.46 (2–21) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2 (2.2) | 1 (1.6) | 0 (0.0) | 1 (8.3) | ||
| Social cognition (0–24) | 2.06±2.42 (0–12) | 1.54±1.93 (0–7) | 2.61±2.09 (0–8) | 3.92±3.90 (0–12) | ≤0.001 | 3.18±4.59 (0–21) | 1.83±2.22 (0–11) | 2.19±1.70 (0–6) | 12.22±7.00 (2–21) | ≤0.001 |
| Missing (n (%)) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 3 (3.3) | 2 (3.2) | 0 (0.0) | 1 (8.3) | ||
| Executive function (0–28) | 1.39±3.04 (0–16) | 0.51±0.89 (0–4) | 1.31±1.55 (0–5) | 5.95±6.35 (0–16) | ≤0.001 | 3.25±6.34 (0–26) | 0.72±0.97 (0–3) | 3.28±5.47 (0–19) | 17.24±6.81 (3–26) | ≤0.001 |
| Missing (n (%)) | 1 (1.1) | 1 (1.6) | 0 (0.0) | 0 (0.0) | 2 (2.2) | 1 (1.6) | 0 (0.0) | 1 (8.3) | ||
| A-NKS total score (0–156) | 14.09±15.70 (0–92) | 8.54±7.78 (0–33) | 22.37±12.27 (6–59) | 31.94±31.43 (0–92) | ≤0.001 | 21.76±30.70 (0–131) | 8.61±8.60 (0–42) | 22.30±19.25 (0–64) | 90.28±29.76 (36–131) | ≤0.001 |
| Missing (n (%)) | 5 (5.4) | 3 (4.8) | 0 (0.0) | 2 (16.7) | 5 (5.4) | 4 (6.5) | 0 (0.0) | 1 (8.3) | ||
A-NKS, pilot version of the Instrument for the Assessment of Daily Living Impairments due to Neurodegenerative Decline [15]; CH, cognitively healthy; NCD, neurocognitive disorder.
Table 6
Prevalence of the 39 A-NKS items assessed by participant-rated A-NKS (N = 92)
| 0 | 1 | 2 | 3 | 4 | Total > 0 | Missing | ||||||||
| Lately, I’ve been having difficulties ... | n | % | n | % | n | % | n | % | n | % | n | % | n | % |
| Complex attention | ||||||||||||||
| 1. ... concentrating on the content when reading, watching TV or listening to the radio. | 73 | 79.3 | 16 | 17.4 | 2 | 2.2 | 1 | 1.1 | 0 | 0.0 | 19 | 20.7 | 0 | 0.0 |
| 2. ... remembering new information when I get distracted. | 33 | 35.9 | 43 | 46.7 | 9 | 9.8 | 6 | 6.5 | 1 | 1.1 | 59 | 64.1 | 0 | 0.0 |
| 3. ... concentrating for a longer period of time. | 50 | 54.3 | 26 | 28.3 | 14 | 15.2 | 1 | 1.1 | 1 | 1.1 | 42 | 45.7 | 0 | 0.0 |
| 4. ... following a thought to the end. | 66 | 71.7 | 19 | 20.7 | 3 | 3.3 | 3 | 3.3 | 0 | 0.0 | 25 | 27.2 | 1 | 1.1 |
| 5. ... continuing an activity after I was briefly interrupted. | 67 | 72.8 | 20 | 21.7 | 3 | 3.3 | 1 | 1.1 | 0 | 0.0 | 24 | 26.1 | 1 | 1.1 |
| 6. ... doing two things at the same time. | 45 | 48.9 | 32 | 34.8 | 5 | 5.4 | 6 | 6.5 | 3 | 3.3 | 46 | 50.0 | 1 | 1.1 |
| Total for domain | 25 | 27.2 | 67 | 72.8 | 0 | 0.0 | ||||||||
| Learning and memory | ||||||||||||||
| 7. ... remembering new things (e.g., a new PIN code, the names of new people). | 40 | 43.5 | 39 | 32.6 | 14 | 15.2 | 6 | 6.5 | 1 | 1.1 | 51 | 55.4 | 1 | 1.1 |
| 8. ... taking medication correctly (at the right time and the right quantity). | 73 | 79.3 | 10 | 10.9 | 2 | 2.2 | 1 | 1.1 | 0 | 0 | 13 | 14.1 | 6 | 6.5 |
| 9. ... remembering engagements or appointments without any aids (e.g., using a calendar or taking notes). | 40 | 43.5 | 31 | 33.7 | 16 | 17.4 | 2 | 2.2 | 2 | 2.2 | 51 | 55.4 | 1 | 1.1 |
| 10. ... remembering recent events (e.g., the activities of the previous day). | 56 | 60.9 | 23 | 25.0 | 6 | 6.5 | 6 | 6.5 | 0 | 0.0 | 35 | 38.0 | 1 | 1.1 |
| 11. ... remembering the names of familiar people (e.g., relatives, friends, acquaintances). | 56 | 60.9 | 28 | 30.4 | 6 | 6.5 | 2 | 2.2 | 0 | 0.0 | 36 | 39.1 | 0 | 0.0 |
| 12. ... remembering where I put things (e.g., my apartment keys, glasses, wallet). | 58 | 63.0 | 27 | 29.3 | 6 | 6.5 | 1 | 1.1 | 0 | 0.0 | 34 | 37.0 | 0 | 0.0 |
| 13. ... remembering what I wanted to say or do. | 38 | 41.3 | 46 | 50.0 | 6 | 6.5 | 2 | 2.2 | 0 | 0.0 | 54 | 58.7 | 0 | 0.0 |
| 14. ... remembering the content of recent conversations. | 61 | 66.3 | 21 | 22.8 | 9 | 9.8 | 1 | 1.1 | 0 | 0.0 | 31 | 33.7 | 0 | 0.0 |
| Total for domain | 17 | 17.4 | 75 | 81.5 | 1 | 1.1 | ||||||||
| Perceptual-motor function | ||||||||||||||
| 15. ... finding my way around in my familiar surroundings (e.g., house, close neighborhood). | 87 | 94.6 | 4 | 4.3 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 4 | 4.3 | 1 | 1.1 |
| 16. ... finding my way around outside of my familiar surroundings. | 73 | 79.3 | 11 | 12.0 | 3 | 3.3 | 0 | 0.0 | 2 | 2.2 | 16 | 17.4 | 3 | 3.2 |
| 17. ... doing previously familiar activities (e.g., driving, cycling, using public transport). | 71 | 77.2 | 12 | 13.0 | 3 | 3.3 | 3 | 3.3 | 1 | 1.1 | 19 | 20.7 | 2 | 2.2 |
| 18. ... doing manual activities or hobbies (e.g., gardening, crafting, repairing things, doing needlework or handicraft). | 70 | 76.1 | 16 | 17.4 | 2 | 2.2 | 0 | 0.0 | 0 | 0.0 | 18 | 19.6 | 4 | 4.3 |
| 19. ... recognizing faces. | 68 | 73.9 | 12 | 13.0 | 7 | 7.6 | 3 | 3.3 | 0 | 0.0 | 22 | 23.9 | 2 | 2.2 |
| 20. ... estimating gaps or distances correctly (e.g., in traffic, when climbing stairs). | 78 | 84.8 | 10 | 10.9 | 2 | 2.2 | 0 | 0.0 | 1 | 1.1 | 13 | 14.1 | 1 | 1.1 |
| Total for domain | 47 | 47.8 | 45 | 48.9 | 3 | 3.3 | ||||||||
| Language | ||||||||||||||
| 21. ... not faltering while speaking. | 69 | 75.0 | 21 | 22.8 | 1 | 1.1 | 1 | 1.1 | 0 | 0.0 | 23 | 25.0 | 0 | 0.0 |
| 22. ... formulating longer sentences in conversations. | 71 | 77.2 | 16 | 17.4 | 2 | 2.2 | 1 | 1.1 | 1 | 1.1 | 20 | 21.7 | 1 | 1.1 |
| 23. ... finding the right words in conversations. | 52 | 56.5 | 32 | 34.8 | 7 | 7.6 | 1 | 1.1 | 0 | 0.0 | 40 | 43.5 | 0 | 0.0 |
| 24. ... understanding the content of questions directed to me. | 78 | 84.8 | 11 | 12.0 | 3 | 3.3 | 0 | 0.0 | 0 | 0.0 | 14 | 15.2 | 0 | 0.0 |
| 25. ... following longer sentences in conversations. | 74 | 80.4 | 14 | 15.2 | 3 | 3.3 | 1 | 1.1 | 0 | 0.0 | 18 | 19.6 | 0 | 0.0 |
| 26. ... understanding the content of what is said on television or radio. | 80 | 87.0 | 10 | 10.9 | 0 | 0.0 | 2 | 2.2 | 0 | 0.0 | 12 | 13.0 | 0 | 0.0 |
| Total for domain | 42 | 45.7 | 50 | 54.3 | 0 | 0.0 | ||||||||
| Social cognition | ||||||||||||||
| 27. ... maintaining social contacts. | 81 | 88.0 | 8 | 8.7 | 1 | 1.1 | 2 | 2.2 | 0 | 0.0 | 11 | 12.0 | 0 | 0.0 |
| 28. ... understanding feelings of other people (e.g., interpreting a facial expression). | 71 | 77.2 | 17 | 18.5 | 3 | 3.3 | 1 | 1.1 | 0 | 0.0 | 21 | 22.8 | 0 | 0.0 |
| 29. ... responding to the opinions of other people in conversations. | 77 | 83.7 | 10 | 10.9 | 5 | 5.4 | 0 | 0.0 | 0 | 0.0 | 15 | 16.3 | 0 | 0.0 |
| 30. ... having my feelings under control. | 71 | 77.2 | 16 | 17.4 | 5 | 5.4 | 0 | 0.0 | 0 | 0.0 | 21 | 22.8 | 0 | 0.0 |
| 31. ... grasping the intentions of other people. | 48 | 52.2 | 37 | 40.2 | 5 | 5.4 | 0 | 0.0 | 1 | 1.1 | 43 | 46.7 | 1 | 1.1 |
| 32. ... keeping patience. | 51 | 55.4 | 34 | 37.0 | 5 | 5.4 | 2 | 2.2 | 0 | 0.0 | 41 | 44.6 | 0 | 0.0 |
| Total for domain | 31 | 32.6 | 61 | 66.3 | 1 | 1.1 | ||||||||
| Executive function | ||||||||||||||
| 33. ... making decisions. | 61 | 66.3 | 21 | 22.8 | 9 | 9.8 | 1 | 1.1 | 0 | 0.0 | 31 | 33.7 | 0 | 0.0 |
| 34. ... preparing meals or hot drinks (e.g., coffee) independently. | 86 | 93.5 | 4 | 4.3 | 0 | 0.0 | 0 | 0.0 | 1 | 1.1 | 5 | 5.4 | 1 | 1.1 |
| 35. ... planning my grocery shopping. | 82 | 89.1 | 2 | 2.2 | 0 | 0.0 | 0 | 0.0 | 3 | 3.3 | 5 | 5.4 | 5 | 5.4 |
| 36. ... operating technical devices (e.g., (mobile) telephone or television). | 78 | 84.8 | 12 | 13.0 | 1 | 1.1 | 0 | 0.0 | 0 | 0.0 | 13 | 14.1 | 1 | 1.1 |
| 37. ... paying in cash or electronically in shops (e.g., with a debit or credit card). | 82 | 89.1 | 1 | 1.1 | 1 | 1.1 | 1 | 1.1 | 1 | 1.1 | 4 | 4.3 | 6 | 6.5 |
| 38. ... managing my own financial affairs independently (e.g., making bank transfers). | 83 | 90.2 | 3 | 3.3 | 0 | 0.0 | 0 | 0.0 | 3 | 3.3 | 6 | 6.5 | 3 | 3.3 |
| 39. ... planning/ structuring my day. | 78 | 84.8 | 7 | 7.6 | 4 | 4.3 | 1 | 1.1 | 1 | 1.1 | 13 | 14.1 | 1 | 1.1 |
| Total for domain | 51 | 54.3 | 41 | 44.6 | 1 | 1.1 | ||||||||
0 = No difficulties; 1 = Minor difficulties; 2 = Medium difficulties; 3 = Major difficulties; 4 = I am not able to execute this activity at all. Total > 0 = minor or more severe difficulties.
Table 7
Prevalence of the 39 A-NKS items assessed by proxy-rated A-NKS (N = 92)
| 0 | 1 | 2 | 3 | 4 | Total > 0 | Missing | ||||||||
| Lately she/he has been having difficulties ... | n | % | n | % | n | % | n | % | n | % | n | % | n | % |
| Complex attention | ||||||||||||||
| 1. ... concentrating on the content when reading, watching TV or listening to the radio. | 64 | 69.6 | 13 | 14.1 | 9 | 9.8 | 4 | 4.3 | 0 | 0.0 | 26 | 28.3 | 1 | 1.1 |
| 2. ... remembering new information when she/he is getting distracted. | 30 | 32.6 | 37 | 40.2 | 14 | 15.2 | 7 | 7.6 | 2 | 2.2 | 60 | 65.2 | 1 | 1.1 |
| 3. ... concentrating for a longer period of time. | 51 | 55.4 | 23 | 25.0 | 8 | 8.7 | 9 | 9.8 | 0 | 0.0 | 40 | 43.5 | 0 | 0.0 |
| 4. ... following a thought to the end. | 58 | 63.0 | 13 | 14.1 | 14 | 15.2 | 5 | 5.4 | 0 | 0.0 | 32 | 34.8 | 2 | 2.2 |
| 5. ... continuing an activity after she/he was briefly interrupted. | 64 | 69.6 | 9 | 9.8 | 10 | 10.9 | 5 | 5.4 | 2 | 2.2 | 26 | 28.3 | 2 | 2.2 |
| 6. ... doing two things at the same time. | 38 | 41.3 | 27 | 29.3 | 8 | 8.7 | 10 | 10.9 | 5 | 5.4 | 50 | 54.3 | 4 | 4.4 |
| Total for domain | 19 | 20.7 | 70 | 76.1 | 1 | 1.1 | ||||||||
| Learning and memory | ||||||||||||||
| 7. ... remembering new things (e.g., a new PIN code, the names of new people). | 41 | 44.6 | 24 | 26.1 | 9 | 9.8 | 10 | 10.9 | 6 | 6.5 | 49 | 53.3 | 2 | 2.2 |
| 8. ... taking medication correctly (at the right time and the right quantity). | 68 | 73.9 | 5 | 5.4 | 5 | 5.4 | 3 | 3.3 | 5 | 5.4 | 18 | 19.6 | 6 | 6.6 |
| 9. ... remembering engagements or appointments without any aids (e.g., using a calendar or taking notes). | 37 | 40.2 | 27 | 29.3 | 8 | 8.7 | 10 | 10.9 | 5 | 5.4 | 50 | 54.3 | 5 | 5.5 |
| 10. ... remembering recent events (e.g., the activities of the previous day). | 55 | 59.8 | 16 | 17.4 | 7 | 7.6 | 9 | 9.8 | 3 | 3.3 | 35 | 38.0 | 2 | 2.2 |
| 11. ... remembering the names of familiar people (e.g., relatives, friends, acquaintances). | 68 | 73.9 | 12 | 13.0 | 5 | 5.4 | 6 | 6.5 | 1 | 1.1 | 24 | 26.1 | 0 | 0.0 |
| 12. ... remembering where she/he put things (e.g., my apartment keys, glasses, wallet). | 46 | 50.0 | 28 | 40.4 | 13 | 14.1 | 4 | 4.3 | 1 | 1.1 | 46 | 50.0 | 0 | 0.0 |
| 13. ... remembering what she/he wanted to say or do. | 53 | 57.6 | 21 | 22.8 | 11 | 12.0 | 6 | 6.5 | 1 | 1.1 | 39 | 42.4 | 0 | 0.0 |
| 14. ... remembering the content of recent conversations. | 55 | 59.8 | 19 | 20.7 | 8 | 8.7 | 7 | 7.6 | 3 | 3.3 | 37 | 40.2 | 0 | 0.0 |
| Total for domain | 15 | 16.3 | 73 | 79.3 | 4 | 4.3 | ||||||||
| Perceptual-motor function | ||||||||||||||
| 15. ... finding her/his way around in her/his familiar surroundings (e.g., house, close neighborhood). | 82 | 89.1 | 7 | 7.6 | 2 | 2.2 | 1 | 1.1 | 0 | 0.0 | 10 | 10.9 | 0 | 0.0 |
| 16. ... finding her/his way around outside of her/his familiar surroundings. | 61 | 66.3 | 15 | 16.3 | 7 | 7.6 | 5 | 5.4 | 3 | 3.3 | 30 | 32.6 | 1 | 1.1 |
| 17. ... doing previously familiar activities (e.g., driving, cycling, using public transport). | 71 | 77.2 | 10 | 10.9 | 4 | 4.3 | 2 | 2.2 | 5 | 5.4 | 21 | 22.8 | 0 | 0.0 |
| 18. ... doing manual activities or hobbies (e.g., gardening, crafting, repairing things, doing needlework or handicraft). | 66 | 71.7 | 8 | 8.7 | 8 | 8.7 | 2 | 2.2 | 3 | 3.3 | 21 | 22.8 | 5 | 5.4 |
| 19. ... recognizing faces. | 68 | 73.9 | 14 | 15.2 | 7 | 7.6 | 1 | 1.1 | 1 | 1.1 | 21 | 22.8 | 1 | 1.1 |
| 20. ... estimating gaps or distances correctly (e.g., in traffic, when climbing stairs). | 70 | 76.1 | 13 | 14.1 | 4 | 4.3 | 2 | 2.2 | 1 | 1.1 | 23 | 25.0 | 2 | 2.2 |
| Total for domain | 37 | 40.2 | 51 | 55.4 | 4 | 4.3 | ||||||||
| Language | ||||||||||||||
| 21. ... not faltering while speaking. | 79 | 85.9 | 5 | 5.4 | 5 | 5.4 | 2 | 2.2 | 0 | 0.0 | 12 | 13.0 | 1 | 1.1 |
| 22. ... formulating longer sentences in conversations. | 76 | 82.6 | 6 | 6.5 | 6 | 6.5 | 2 | 2.2 | 1 | 1.1 | 15 | 16.3 | 1 | 1.1 |
| 23. ... finding the right words in conversations. | 66 | 71.7 | 13 | 14.1 | 8 | 8.7 | 3 | 3.3 | 1 | 1.1 | 25 | 27.2 | 1 | 1.1 |
| 24. ... understanding the content of questions directed to her/him. | 72 | 78.3 | 12 | 13.0 | 3 | 3.3 | 4 | 4.3 | 0 | 0.0 | 19 | 20.7 | 1 | 1.1 |
| 25. ... following longer sentences in conversations. | 73 | 79.3 | 10 | 10.9 | 3 | 3.3 | 4 | 4.3 | 2 | 2.2 | 19 | 20.7 | 0 | 0.0 |
| 26. ... understanding the content of what is said on television or radio. | 71 | 77.2 | 12 | 13.0 | 5 | 5.4 | 3 | 3.3 | 0 | 0.0 | 20 | 21.7 | 1 | 1.1 |
| Total for domain | 52 | 56.5 | 38 | 41.3 | 2 | 2.2 | ||||||||
| Social cognition | ||||||||||||||
| 27. ... maintaining social contacts. | 72 | 78.3 | 11 | 12.0 | 2 | 2.2 | 2 | 2.2 | 4 | 4.3 | 19 | 20.7 | 1 | 1.1 |
| 28. ... understanding feelings of other people (e.g., interpreting a facial expression). | 70 | 76.1 | 11 | 12.0 | 3 | 3.3 | 4 | 4.3 | 2 | 2.2 | 20 | 21.7 | 2 | 2.2 |
| 29. ... responding to the opinions of other people in conversations. | 58 | 63 | 20 | 21.7 | 5 | 5.4 | 4 | 4.3 | 1 | 1.1 | 30 | 32.6 | 4 | 4.5 |
| 30. ... having her/his feelings under control. | 56 | 60.9 | 25 | 27.2 | 6 | 6.5 | 4 | 4.3 | 1 | 1.1 | 36 | 39.1 | 0 | 0.0 |
| 31. ... grasping the intentions of other people. | 55 | 59.8 | 25 | 27.2 | 4 | 4.3 | 3 | 3.3 | 2 | 2.2 | 34 | 37.0 | 3 | 3.3 |
| 32. ... keeping patience. | 45 | 48.9 | 32 | 34.8 | 9 | 9.8 | 5 | 5.4 | 0 | 0.0 | 46 | 50.0 | 1 | 1.1 |
| Total for domain | 22 | 23.9 | 68 | 73.9 | 2 | 2.2 | ||||||||
| Executive function | ||||||||||||||
| 33. ... making decisions. | 57 | 62.0 | 18 | 19.6 | 5 | 5.4 | 7 | 7.6 | 3 | 3.3 | 33 | 35.9 | 2 | 2.2 |
| 34. ... preparing meals or hot drinks (e.g., coffee) independently. | 80 | 87.0 | 3 | 3.3 | 2 | 2.2 | 3 | 3.3 | 1 | 1.1 | 9 | 9.8 | 3 | 3.3 |
| 35. ... planning her/his grocery shopping. | 74 | 80.4 | 4 | 4.3 | 1 | 1.1 | 4 | 4.3 | 5 | 5.4 | 14 | 15.2 | 4 | 4.5 |
| 36. ... operating technical devices (e.g. (mobile) telephone or television). | 59 | 64.1 | 15 | 16.3 | 11 | 12.0 | 5 | 5.4 | 1 | 1.1 | 32 | 34.8 | 1 | 1.1 |
| 37. ... paying in cash or electronically in shops (e.g., with a debit or credit card). | 77 | 83.7 | 3 | 3.3 | 2 | 2.2 | 3 | 3.3 | 4 | 4.3 | 12 | 13.0 | 3 | 3.3 |
| 38. ... managing her/his own financial affairs independently (e.g., making bank transfers). | 73 | 79.3 | 4 | 4.3 | 1 | 1.1 | 3 | 3.3 | 7 | 7.6 | 15 | 16.3 | 4 | 4.3 |
| 39. ... planning/ structuring her/his day. | 71 | 77.2 | 9 | 9.8 | 2 | 2.2 | 7 | 7.6 | 2 | 2.2 | 20 | 21.7 | 1 | 1.1 |
| Total for domain | 43 | 46.7 | 48 | 52.2 | 1 | 1.1 | ||||||||
0 = No difficulties; 1 = Minor difficulties; 2 = Medium difficulties; 3 = Major difficulties; 4 = She/he is not able to execute this activity at all. Total > 0 = minor or more severe difficulties.
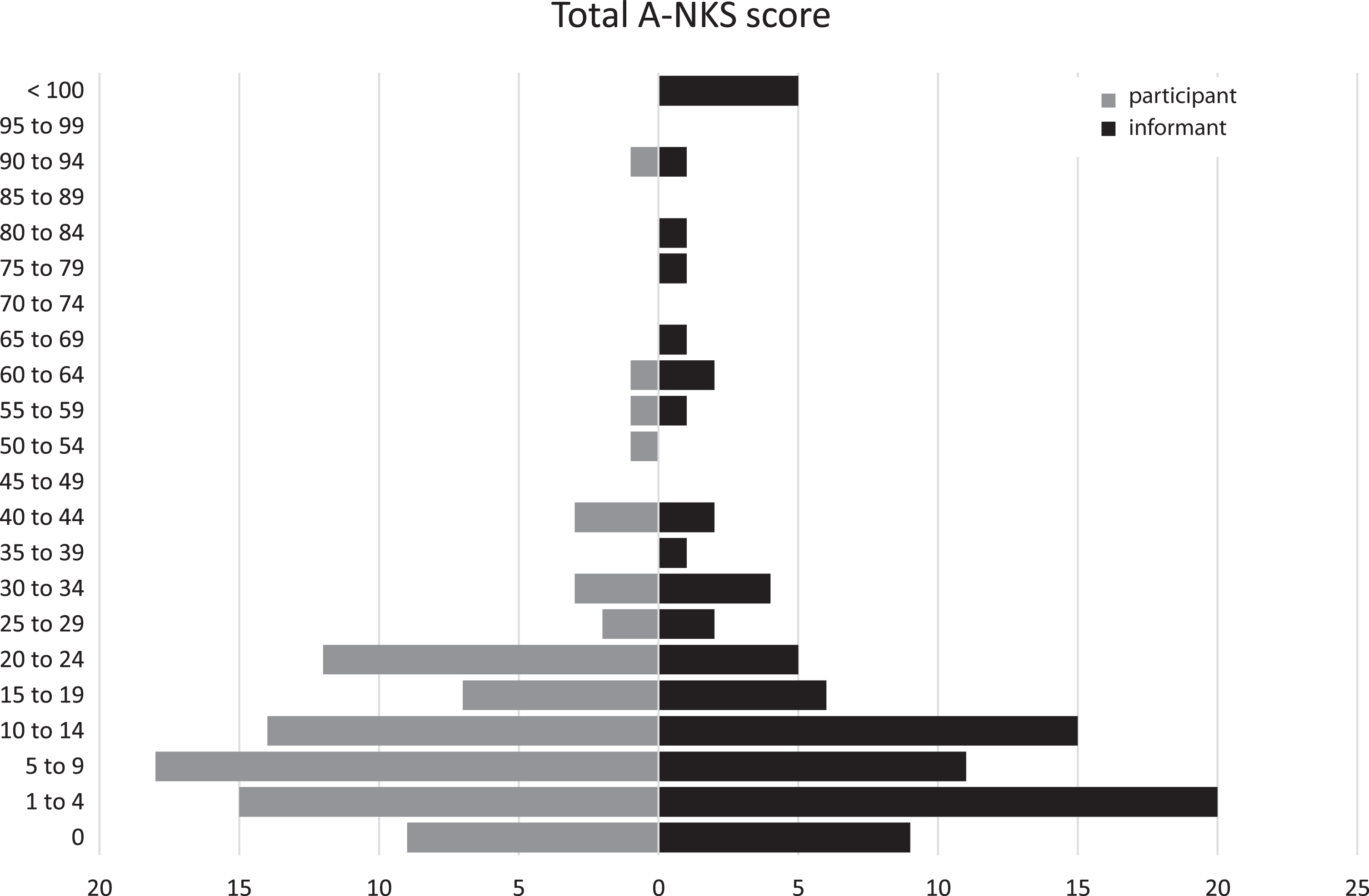
The highest total A-NKS score reported in the informant-rated A-NKS was slightly higher than the participant-rated A-NKS (M = 14.1; SD = 15.7) versus M = 21.8; SD = 30.7). Overall, few ADL impairments as recorded by the A-NKS were reported (see Fig. 3 and Table 5).
Fig. 3
Distribution of A-NKS total score (absolute frequencies) (N = 87).
Agreement between participant- and informant-rated A-NKS
Agreement between the participant- and informant-rated A-NKS is at a moderate level (ICC = 0.62; 95% -KI [0.41 – 0.75]). For the majority of items, there is low agreement between both versions of the A-NKS, and for questions 6, 8, 15, 21, 22, 24, and 26, there is no agreement.
Acceptability
Both versions of the A-NKS were rated as acceptable or very acceptable. The majority of them rated the questions as clearly formulated (participants: n = 89 (96.7%); informants: n = 83 (90.2%)). All informants rated the length of the A-NKS, as well as the length of each question, as appropriate for use with individuals aged 60 years and older. Two participants (2.2%) disagreed for the length of the total instrument and one for the length of the single questions (1.1%). The response possibilities were also considered acceptable to most (participants: n = 90 (97.8%); informants: n = 87 (94.6%)).
Overall, there are very few missing values, which underscores the acceptance of the A-NKS. participant-rated A-NKS had no missing values in 18 questions and informant-rated A-NKS in 9 questions. More than 5% missing values were found in participant-rated A-NKS for items 8, 35, and 37, as well as items 8, 9, and 18 in the informant-rated A-NKS. Table 6 provides an overview.
Internal consistency
Consistency of both versions of the A-NKS was on excellent level (participants: α= 0.95; informants: α= 0.99) [31]. A total of two items in the participant-rated A-NKS (15 and 18) had low item-total correlation. Alpha did not increase when these items were removed. Table 8 provides an overview.
Table 8
Summary of the psychometric properties of the individual questions of the A-NKS
| Psychometric characteristics | Item-total correlation | Convergent validity: participant-agreement | |
| Statistics | Spearman‘s r | Cohen‘s k | |
| A-NKS-item | Participant | Informant | |
| 1. ... concentrating on the content when reading, watching TV or listening to the radio. | 0.69 | 0.83 | 0.14 |
| 2. ... remembering new information when I get distracted. | 0.67 | 0.85 | 0.21 |
| 3. ... concentrating for a longer period of time. | 0.70 | 0.86 | 0.20 |
| 4. ... following a thought to the end. | 0.76 | 0.85 | 0.23 |
| 5. ... continuing an activity after I was briefly interrupted. | 0.76 | 0.87 | 0.17 |
| 6. ... doing two things at the same time. | 0.74 | 0.83 | 0.08 |
| 7. ... remembering new things (e.g., a new PIN code, the names of new people). | 0.67 | 0.83 | 0.19 |
| 8. ... taking medication correctly (at the right time and the right quantity). | 0.55 | 0.89 | 0.07 |
| 9. ... remembering engagements or appointments without any aids (e.g., using a calendar or taking notes). | 0.69 | 0.82 | 0.15 |
| 10. ... remembering recent events (e.g., the activities of the previous day). | 0.77 | 0.89 | 0.24 |
| 11. ... remembering the names of familiar people (e.g., relatives, friends, acquaintances). | 0.60 | 0.78 | 0.12 |
| 12. ... remembering where I put things (e.g., my apartment keys, glasses, wallet). | 0.71 | 0.84 | 0.22 |
| 13. ... remembering what I wanted to say or do. | 0.70 | 0.89 | 0.23 |
| 14. ... remembering the content of recent conversations. | 0.73 | 0.91 | 0.11 |
| 15. ... finding my way around in my familiar surroundings (e.g., house, close neighborhood). | 0.28 | 0.68 | 0.03 |
| 16. ... finding my way around outside of my familiar surroundings. | 0.54 | 0.85 | 0.15 |
| 17. ... doing previously familiar activities (e.g., driving, cycling, using public transport). | 0.49 | 0.82 | 0.23 |
| 18. ... doing manual activities or hobbies (e.g., gardening, crafting, repairing things, doing needlework or handicraft). | 0.31 | 0.85 | 0.22 |
| 19. ... recognizing faces. | 0.62 | 0.83 | 0.23 |
| 20. ... estimating gaps or distances correctly (e.g., in traffic, when climbing stairs). | 0.31 | 0.75 | 0.12 |
| 21. ... not faltering while speaking. | 0.77 | 0.84 | -0.04 |
| 22. ... formulating longer sentences in conversations. | 0.75 | 0.82 | 0.05 |
| 23. ... finding the right words in conversations. | 0.73 | 0.81 | 0.24 |
| 24. ... understanding the content of questions directed to me. | 0.71 | 0.87 | 0.09 |
| 25. ... following longer sentences in conversations. | 0.77 | 0.88 | 0.15 |
| 26. ... understanding the content of what is said on television or radio. | 0.78 | 0.90 | 0.07 |
| 27. ... maintaining social contacts. | 0.52 | 0.86 | 0.18 |
| 28. ... understanding feelings of other people (e.g., interpreting a facial expression). | 0.65 | 0.85 | 0.10 |
| 29. ... responding to the opinions of other people in conversations. | 0.46 | 0.79 | 0.12 |
| 30. ... having my feelings under control. | 0.55 | 0.88 | 0.12 |
| 31. ... grasping the intentions of other people. | 0.45 | 0.81 | 0.16 |
| 32. ... keeping patience. | 0.64 | 0.71 | 0.14 |
| 33. ... making decisions. | 0.40 | 0.87 | 0.18 |
| 34. ... preparing meals or hot drinks (e.g., coffee) independently. | 0.73 | 0.88 | 0.33 |
| 35. ... planning my grocery shopping. | 0.77 | 0.89 | 0.29 |
| 36. ... operating technical devices (e.g., (mobile) telephone or television). | 0.61 | 0.84 | 0.09 |
| 37. ... paying in cash or electronically in shops (e.g., with a debit or credit card). | 0.65 | 0.89 | 0.22 |
| 38. ... managing my own financial affairs independently (e.g., making bank transfers). | 0.71 | 0.94 | 0.33 |
| 39. ... planning/ structuring my day. | 0.70 | 0.91 | 0.25 |
| Total | 0.96 | 0.99 | 0.62 |
| 95% -CI | 0.41; 0.75 | ||
| Statistics for A-NKS score | Cronbach’s α | Intraclass Correlation Coefficient | |
A-NKS, pilot version of the instrument for the assessment of everyday impairments due to neurodegenerative decline [15]; CI, confidence interval; k, Cohen’s kappa; r, rank correlation coefficient; Italics indicate areas of low reliability or agreement: item-total correlation: Cronbach’s α increases if item is deleted. Value cannot be calculated since no variance in the data.
Validity
When comparing the subscales, significant differences were found between the results of each group for each subscale in both A-NKS versions, with participants with major NCD reporting more difficulties than those with mild NCD. Cognitively healthy participants reported the least ADL difficulties (Table 5). Additionally, the means and standard deviations of the subscales differed from each other (Table 5).
Convergent validity
The total score of the participant-rated A-NKS correlated strongly with the Amsterdam IADL, moderately with the SIDAM IADL, the subscale autonomy of the WHOQOL-OLD, the sum score of items 2 and 3 of the EQ-5D, and physical and cognitive activities. In contrast, only a weak correlation was found with the BI. The total score of the informant-rated version correlates moderately with the following instruments: Amsterdam IADL, SIDAM IADL, BI, the subscale autonomy of the WHOQOL-OLD, and the sum score of items 2 and 3 of the EQ-5D. A strong relationship is shown between the total A-NKS score of the informant-rated A-NKS and physical and cognitive activities. See Table 9.
Table 9
Spearman correlations between external criteria of the A-NKS subscales and the A-NKS as a total score
| Participant | Informant | |||||||||||||
| Total | 1 | 2 | 3 | 4 | 5 | 6 | Total | 1 | 2 | 3 | 4 | 5 | 6 | |
| T-MoCA | –0.40*** | –0.40*** | –0.40*** | –0.26* | –0.35*** | –0.24* | –3.60*** | –0.52*** | –0.49*** | –0.55*** | –0.44*** | –0.55*** | –0.37*** | –0.47*** |
| CERAD TMT A | 0.27** | 0.17 | 0.30** | 0.33** | 0.20 | 0.17 | 0.40** | 0.41*** | 0.40*** | 0.47*** | 0.46*** | 0.40*** | 0.16 | 0.31** |
| CERAD TMT B | 0.34** | 0.33** | 0.35*** | 0.33** | 0.31** | 0.22* | 0.43*** | 0.40*** | 0.38*** | 0.45*** | 0.38*** | 0.46*** | 0.19 | 0.37*** |
| CERAD wordlist recall | –0.35*** | –0.37*** | –0.33*** | –0.39*** | –0.23* | –0.14 | –0.38*** | –0.48*** | –0.43*** | –0.53*** | –0.56*** | –0.47*** | –0.32** | –0.48*** |
| CERAD constructive practice | –0.02 | –0.00 | –0.03 | –0.19 | –0.01 | 0.04 | –0.20 | –0.29** | –0.28** | –0.26** | –0.33*** | –0.30** | –0.18 | –0.21* |
| CERAD verbal fluency | –0.29** | –0.30** | –0.24* | –0.40*** | –0.31** | –0.14 | –0.34*** | –0.49*** | –0.39*** | –0.50*** | –0.57*** | –0.48*** | –0.33** | –0.45*** |
| RMET | –0.09 | –0.10 | –0.08 | –0.25* | –0.03 | –0.08 | –0.17 | –0.20* | –0.17 | –0.30** | –0.30* | –0.28** | –0.15 | –0.24* |
| Barthel Index | –0.26* | –0.16 | –0.19 | –0.35*** | –0.19 | –0.12 | –0.28** | –0.30** | –0.34*** | –0.33** | –0.28** | –0.34*** | –0.16 | –0.18 |
| Amsterdam IADL | 0.59** | 0.56*** | 0.53*** | 0.57*** | 0.32** | 0.41*** | 0.54*** | 0.48** | 0.47*** | 0.50*** | 0.38*** | 0.50*** | 0.31** | 0.36*** |
| SIDAM ADL | 0.46*** | 0.41*** | 0.47*** | 0.55*** | 0.31** | 0.30** | 0.41*** | 0.47*** | 0.45*** | 0.51*** | 0.40*** | 0.41*** | 0.34*** | 0.37*** |
| WHOQOL-OLD Autonomy | –0.50*** | –0.48*** | –0.37*** | –0.36*** | –0.36*** | –0.51*** | –0.46*** | –0.37*** | –0.35*** | –0.44*** | –0.33*** | –0.22* | –0.32** | –0.24* |
| GDS | 0.61*** | 0.60*** | 0.50*** | 0.48*** | 0.45*** | 0.55*** | 0.60*** | 0.46*** | 0.45*** | 0.48*** | 0.43*** | 0.41*** | 0.38*** | 0.30** |
| EQ-02 | 0.47*** | 0.50*** | 0.42*** | 0.54*** | 0.27** | 0.27** | 0.54*** | 0.39** | 0.36*** | 0.37*** | 0.39*** | 0.43*** | 0.22* | 0.31** |
| EQ-03 | 0.13 | 0.14 | 0.17 | 0.28** | 0.09 | 0.09 | 0.30** | 0.37*** | 0.36*** | 0.33** | 0.38*** | 0.42*** | 0.34** | 0.37*** |
| Activities (EQ-02 + EQ-03) | 0.48*** | 0.51*** | 0.43*** | 0.55*** | 0.27** | 0.29** | 0.55*** | 0.41*** | 0.38*** | 0.39*** | 0.41*** | 0.45*** | 0.25* | 0.34** |
| Physical and cognitive activity | –0.39*** | –0.35*** | –0.31** | –0.45*** | –0.35*** | –0.27** | –0.36*** | –0.50*** | –0.46*** | –0.47*** | –0.50*** | –0.48*** | –0.36*** | –0.46*** |
1 = Complex attention; 2 = Learning and memory; 3 = Perceptual-motor function; 4 = Language; 5 = Social cognition; 6 = Executive functions; Underlined values = expected correlations. * p≤0.05; ** p≤0.01; *** p≤0.001. A-NKS, pilot version of the Instrument for the Assessment of Daily Living Impairments due to Neurodegenerative Decline [15]; Amsterdam IADL, Amsterdam Instrumental Activities of Daily living [21]; Barthel Index [20]; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease [26]; EQ2 and EQ3, Item 2 and Item 3 from the Quality of Life Questionnaire EQ-5D [24]; GDS, Geriatric Depression Scale [19]; RMET, Reading the Mind in the Eyes – Short Form [28]; SIDAM-IADL, Structured Interview for the Diagnosis of Dementia of the Alzheimer’s Type of Multiinfarct Dementia and Dementias of Other Etiologies according to DSM-III-R and ICD-10. Manual [22]; T-MoCA, Telephone Montreal Cognitive Assessment [16]; TMT A, Trail Making Test A from CERAD [26]; TMT B, Trail Making Test B from CERAD [26]; WHOQOL-OLD Autonomy, Quality of Life in Aging – subscale autonomy [23].
Translation into English
We performed a multi-step and team-based translation process following the TRAPD model (Translation, Review, Adjudication, Pretesting, and Documentation) (see Fig. 4) [32, 33]. Several individuals were involved over several stages for optimization of the translation process. First, the original German A-NKS was translated into English by two independent professional translators certificated according to the norms DIN EN 15038 and ISO 1700. Second, these two versions were reviewed, discussed, and agreed upon by several scientists, including members of the research team and a bilingual external reviewer. This resulted in the English version of the A-NKS.
DISCUSSION
The aim of our study was to investigate psychometric characteristics of the A-NKS—an instrument to assess ADL impairments, specifically based on the diagnostic specifications of the DSM-5 [4]. The assessment of ADL impairment is important for the diagnosis of mild and major NCD, especially to discriminate both forms from each other. This is because ADL functioning is a decisive criterion: in major NCD, impairment of ADL caused by cognitive difficulties is required, while in mild NCD, cognitive deficits do not or only minimally interfere with the capacity for independence.
We found a very good acceptance of the A-NKS by the target population, as shown in a high agreement regarding the length of the individual items, the length of the instrument, as well as the applicability of the answer categories. In addition, the low number of missing values in our study also indicates a high acceptability of the A-NKS. This further supports the notion that the A-NKS is a well-received instrument for assessing ADL impairments in individuals with mild and major NCD according to DSM-5 criteria.
The internal consistency of the A-NKS is at an excellent level for both, participant- and informant-rated version. Regarding convergent validity, the hypothesized associations were mostly confirmed for both A-NKS versions. For informant-rated A-NKS, all expected associations were confirmed except for the correlation between the RMET and the A-NKS subscale “social cognition”, which was also not confirmed for participant-rated version. Additionally, in participant-rated A-NKS, no significant correlation was observed between TMT A and the subscale “complex attention”, as well as between the subscale “perceptual-motor function” and the CERAD subscale “constructive practice”. Nonetheless, it can be concluded that the A-NKS is suitable to assess impairment of activities of daily living and, as we found significant associations between ADL impairment assessed by the A-NKS and mild and major cognitive deficits in the DSM-5 specified neurocognitive domains, that it appears particularly suitable to assess impairment of activities of daily living in DSM-5 mild and major NCD. The weak correlation between the A-NKS subscale “social cognition” and RMET performance may reflect that RMET assesses only one specific social cognitive domain (facial recognition) and does not test empathy or theory of mind, therefore, this association should be re-tested with other social cognition instruments, particularly in larger populations including more individuals with mild and major NCD.
In addition, we observed significant correlations between the participant-rated and informant-rated A-NKS score and various established instruments assessing (I)ADL, physical and cognitive activities. For instance, the strong correlation between the self-reported A-NKS and the Amsterdam IADL suggests that individuals were reliable in reporting their own ADL impairments.
However, it is worth noting the weaker correlation with the Barthel Index (BI) in participant-reported A-NKS scores. This may be attributed to differences in the constructs measured by the two instruments. While the A-NKS offers a more comprehensive assessment of ADL impairments across specific neurocognitive domains, the BI focuses on basic ADLs, such as bathing and dressing. This discrepancy highlights the importance of considering the scope and depth of ADL assessments in research and clinical practice.
Furthermore, the moderate correlations observed with the SIDAM IADL, the WHOQOL-OLD subscale ‘autonomy,’ and the EQ-5D indicate convergent validity and suggest that the A-NKS captures relevant aspects of daily functioning. These findings are promising and support the utility of the A-NKS in assessing ADL impairments.
The results of this study on the mean values and standard deviations of the A-NKS, including the subscales (see Table 5), allow for a preliminary differentiation by neurocognitive domain and severity of cognitive deficits. However, for more reliable statements on corresponding cut-off values, as well as for the final factor structure of the A-NKS, a subsequent study should be conducted with more comprehensive samples and a larger sample size, including participants with mild and severe NCD as well as those without such disorders.
Strengths and limitations
The present study provides the first validated instrument for the assessment of ADL impairments in regard to mild and major NCD according to the DSM-5 [4]. Therefore, the study contributes decisively to the diagnostics of NCDs as it may allow differentiating between ADL impairment (I) due to mild and major deficits (differentiation according to the severity of cognitive deficits) and (II) due to deficits in specific neurocognitive domains as specified by the DSM-5 (differentiation according to the domain of cognitive deficits).
Our study has several noteworthy limitations. First, we did not inquire about the medication intake of self-rated participants, which can potentially influence brain health. This decision was made to streamline the survey and reduce participant burden, given our primary focus on cognitive health and its correlation with activities of daily living. As a result, we may not have accounted for the impact of medications on cognitive function. Future research could delve into this aspect to provide a more comprehensive understanding of the factors affecting cognitive health.
Secondly, due to the small sample size comprising only a few cognitively impaired individuals, the level of functional impairment due to neurocognitive deficits was rather low which led to a low variance and to limitations in the calculation of statistical parameters, especially Cohen’s kappa. As this is only a pilot study aimed at gaining initial insights, we will forgo conducting a factor analysis. Further research should address a larger sample of individuals with mild and major NCDs to confirm our findings and to test interrater- and test-retest reliability. In addition, it is important to investigate the factor structure of the A-NKS to enable subscale-specific analysis.
Conclusion
To the best of our knowledge, this is the first instrument to assess ADL impairments based on the six neurocognitive domains of the DSM-5 diagnoses of mild and major NCD. Our study provides evidence that the A-NKS can be used as a participant-informant report to assess ADL impairments in cognitively healthy individuals, individuals with mild NCD, and those with major NCD according to the DSM-5 criteria. The A-NKS should be further tested in different settings, such as nursing homes or among community-dwelling individuals, to assess its applicability to a broad range of users in research and practice (e.g., general practitioners, specialists, neuropsychologists, etc.). Further studies should apply the instrument to a larger samples of individuals with mild and major NCD allowing to investigate the A-NKS’s factor and internal structure. The new developed and psychometrically tested A-NKS may help to improve diagnosis of mild and major NCD according to DSM-5 criteria by providing differentiation in terms of severity and cognitive domain. In this way, the A-NKS may support the early detection and (differential) diagnosis of major versus mild NCD in research and clinical practice and helps to improve the care situation of individuals with dementia themselves as well as to relieve the burden on their relatives.
ACKNOWLEDGMENTS
We would like to thank all research participants, without whom this work would not have been feasible. We would also like to thank Andrea Bräsecke, Joana Raub, Elisabeth Schultheiß, Antonia Buß, and Stephan Gemp for their great support.
FUNDING
This study was funded by the Deutsche Alzheimer Gesellschaft e. V. – Selbsthilfe Demenz (Forschungsförderung 2019).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
Data supporting this study are available upon reasonable request. Please direct all data inquiries to the corresponding author.
REFERENCES
[1] | Alzheimer’s Disease International ((2019) ) World Alzheimer Report 2019. Attitudes to dementia. Alzheimer’s Disease International. London. |
[2] | Thyrian JR , Stentzel U ((2021) ) Die Entwicklung der geschätzten Anzahl an Demenz erkrankter Menschen auf Kreisebene in Deutschland für 2030. Psychiatr Prax 48: , 79–84. |
[3] | ((2009) ) Diagnostic and statistical manual of mental disorders: DSM-IV-TR, 4th ed., text revision. American Psychiatric Association, Arlington, VA. |
[4] | American Psychiatric Association ((2013) ) Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5) . American Psychiatric Association, Arlington, VA. |
[5] | Petersen RC ((2004) ) Mild cognitive impairment as a diagnostic entity. J Intern Med 256: , 183–194. |
[6] | Winblad B , Palmer K , Kivipelto M , Jelic V , Fratiglioni L , Wahlund L-O , Nordberg A , Bäckman L , Albert M , Almkvist O , Arai H , Basun H , Blennow K , Leon M de , DeCarli C , Erkinjuntti T , Giacobini E , Graff C , Hardy J , Jack C , Jorm A , Ritchie K , van Duijn C , Visser P , Petersen RC ((2004) ) Mild cognitive impairment–beyond controversies, towards a consensus: Report of the International Working Group on Mild Cognitive Impairment. J Intern Med 256: , 240–246. |
[7] | World Health Organization ((1992) ) The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines . World Health Organization, Geneva. |
[8] | Reppermund S , Brodaty H , Crawford JD , Kochan NA , Draper B , Slavin MJ , Trollor JN , Sachdev PS ((2013) ) Impairment in instrumental activities of daily living with high cognitive demand is an early marker of mild cognitive impairment: The Sydney memory and ageing study. Psychol Med 43: , 2437–2445. |
[9] | Njegovan V , Hing MM , Mitchell SL , Molnar FJ ((2001) ) The hierarchy of functional loss associated with cognitive decline in older persons. J Gerontol A Biol Sci Med Sci 56: , M638–M643. |
[10] | Knopman DS , Petersen RC ((2014) ) Mild cognitive impairment and mild dementia: A clinical perspective. Mayo Clin Proc 89: , 1452–1459. |
[11] | Deuschl G , Maier W ((2016) ) Diagnose- und Behandlungsleitlinie Demenz - Interdisziplinäre S3 Praxisleitlinien 2016, Deutsche Gesellschaft für Psychiatrie, Psychotherapie und Nervenheilkunde (DGPPN), Deutsche Gesellschaft für Neurologie (DGN), https://www.dgppn.de/_Resources/Persistent/ade50e44afc7eb8024e7f65ed3f44e995583c3a0/S3-LL-Demenzen-240116.pdf. Accessed May 15, 2020. |
[12] | Falkai P ed , ((2018) ) Diagnostisches und statistisches Manual psychischer Störungen DSM-5 . Korrigierte Auflage, Hogrefe, Göttingen. |
[13] | Gélinas I , Gauthier L , McIntyre M , Gauthier S ((1999) ) Development of a functional measure for persons with Alzheimer’s disease: The disability assessment for dementia. Am J Occup Ther 53: , 471–481. |
[14] | Lawton MP , Brody EM ((1969) ) Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 9: , 179–186. |
[15] | Funke K , Bernard M , Luppa M , Riedel-Heller SG , Luck T ((2022) ) Alltagsbeeinträchtigungen bei neurokognitiven Störungen Entwicklung eines differenzierten Erfassungsinstrumentes für Forschung und Praxis. Nervenarzt 93: , 840–843. |
[16] | Katz MJ , Wang C , Nester CO , Derby CA , Zimmerman ME , Lipton RB , Sliwinski MJ , Rabin LA ((2021) ) T-MoCA: A valid phone screen for cognitive impairment in diverse community samples. Alzheimers Dement (Amst) 13: , e12144. |
[17] | Beesdo-Baum K , Zaudig M , Wittchen H-U ((2019) ) Strukturiertes Klinisches Interview fu¨r DSM-5®-Sto¨rungen – Klinische Version: Deutsche Bearbeitung des Structured Clinical Interview for DSM-5® Disorders - Clinician Version von Michael B. First, Janet B. W. Williams, Rhonda S. Karg, Robert L. Spitzer , Hogrefe, Go¨ttingen. |
[18] | Löwe B , Kroenke K , Gräfe K ((2005) ) Detecting and monitoring depression with a two-item questionnaire (PHQ-2). J Psychosom Res 58: , 163–171. |
[19] | Gauggel S , Birkner B ((1999) ) Validität und Reliabilität einer deutschen Version der Geriatrischen Depressionsskala (GDS). Z Klin Psychol Psychother 28: , 18–27. |
[20] | Mahoney FI , Barthel D ((1965) ) Functional evaluation: The Barthel Index. Md State Med J 14: , 61–65. |
[21] | Jutten RJ , Peeters CFW , Leijdesdorff SMJ , Visser PJ , Maier AB , Terwee CB , Scheltens P , Sikkes SAM ((2017) ) Detecting functional decline from normal aging to dementia: Development and validation of a short version of the Amsterdam IADL Questionnaire. Alzheimers Dement (Amst) 8: , 26–35. |
[22] | Zaudig M , Mittelhammer J , Hiller W , Pauls A , Thora C , Morinigo A , Mombour W ((1991) ) SIDAM–A structured interview for the diagnosis of dementia of the Alzheimer type, multi-infarct dementia and dementias of other aetiology according to ICD-10 and DSM-III-R. Psychol Med 21: , 225–236. |
[23] | Winkler I , Matschinger H , Angermeyer MC ((2006) ) Der WHOQOL-OLD – Ein Fragebogen zur interkulturellen Erfassung der Lebensqualität im Alter. Psychother Psychosom Med Psychol 56: , 63–69. |
[24] | Graf J-M , Claes C , Greiner W , Uber A ((1998) ) Die deutsche Version des EuroQol-Fragebogens. J Public Health 6: , 3–20. |
[25] | Verghese J , Lipton RB , Katz MJ , Hall CB , Derby CA , Kuslansky G , Ambrose AF , Sliwinski M , Buschke H ((2003) ) Leisure activities and the risk of dementia in the elderly. N Engl J Med 348: , 2508–2516. |
[26] | Morris JC , Heyman A , Mohs RC , Hughes JP , van Belle G , Fillenbaum G , Mellits ED , Clark C ((1989) ) The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part I. Clinical and neuropsychological assessment of Alzheimer’s disease. Neurology 39: , 1159–1165. |
[27] | Rosen WG , Mohs RC , Davis KL ((1984) ) A new rating scale for Alzheimer’s disease. Am J Psychiatry 141: , 1356–1364. |
[28] | Burke T , Pinto-Grau M , Costello E , Peelo C , Lonergan K , Heverin M , Hardiman O , Pender N ((2020) ) The reading the mind in the eyes test short form (A & B): Validation and outcomes in an amyotrophic lateral sclerosis cohort. Amyotroph Lateral Scler Frontotemporal Degener 21: , 380–388. |
[29] | Cohen J ((1968) ) Weighted kappa: Nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 70: , 213–220. |
[30] | StataCorp ((2019) ) StataCorp LLC, College Station, TX. Stata Statistical Software: Release 16. |
[31] | Tavakol M , Dennick R ((2011) ) Making sense of Cronbach’s alpha. Int J Med Educ 2: , 53–55. |
[32] | Harkness J ((2008) ) Round 4 ESS Translation Strategies and Procedures. |
[33] | Harkness J , van de Vijver F , Mohler P ((2003) ) Questionnaire translation. In Cross-cultural survey methods, Wiley J, ed., pp. 35–56. |

![Stages of the development of the A-NKS [15].](https://ip.ios.semcs.net:443/media/jad/2024/97-1/jad-97-1-jad230627/jad-97-jad230627-g001.jpg)
![Translation of the A-NKS following the TRAPD-model (according [32, 33]).](https://ip.ios.semcs.net:443/media/jad/2024/97-1/jad-97-1-jad230627/jad-97-jad230627-g004.jpg)



