Technology-Based Counselling for People with Dementia and Their Informal Carers: A Systematic Review and Meta-Analysis
Abstract
Background:
Information technology can enhance timely and individual support for people with Alzheimer’s disease and other dementias and their informal carers.
Objective:
To review the effectiveness of technology-based counselling interventions for people with dementia and informal carers.
Methods:
Randomized controlled trials of remote dementia counselling interventions were included. We searched CINAHL, Cochrane Library, MEDLINE, PsycINFO, and the Web of Science Core Collection (April 2021) in combination with citation tracking and free web searching (October to November 2021). We provide meta-analyses for caregiver depression, burden, and self-efficacy/mastery and structured reporting for other outcomes. The Grading of Recommendations Assessment, Development and Evaluation approach and the Risk of Bias 2 tool were applied.
Results:
We included five randomized controlled trials involving 880 participants. Interventions were provided for carers (four studies) or dyads (one study). Carers were predominantly women and were the spouses or children of people with dementia. Counselling was delivered via telephone or videoconference with two to 23 sessions over 1 to 12 months. Control groups received educational and resource materials only, standard (helpline) services, non-directive support, or home visits. Meta-analysis for our primary outcome, depressive symptoms in carers, revealed no statistically significant effect (SMD –0.15; 95% CI –0.40 to 0.10). There were also no significant effects on burden and self-efficacy/mastery. We rated the certainty of evidence as low to very low and all outcomes at an overall high risk of bias.
Conclusion:
The effectiveness of technology-based counselling interventions for people with dementia and informal carers remains uncertain. Theory-based approaches are needed for the development and evaluation of these interventions.
INTRODUCTION
Frequent consequences of dementia are unmet needs, which can cause changes in behavior and psychological symptoms such as hyperactivity, apathy, depression, or anxiety [1]. The more the disease progresses, the more people with dementia become reliant on carers [2], even if they want to remain independent [3]. The majority of people with dementia are cared for at home, mainly by a spouse [4]. Informal carers of people with dementia face numerous challenges, which have been described frequently in terms of burden [5–7] and stress [8, 9]. Depressive symptoms are a great challenge for both people with dementia [10] and their informal carers [11, 12]. The prevalence of depression in people with dementia ranged from 20% to 37%, compared to 13% in people without dementia [10]. Approximately a third of all informal carers of people with dementia experience depression [13].
Formal interventions aim to support both people with dementia and their carers by addressing diverse support needs. These needs include acquiring relevant knowledge about dementia, obtaining information on accessible services, addressing physical and psychological health conditions, and managing daily living as well as maintaining social connectedness [14, 15]. Because services do not always effectively meet these needs, there is a gap between the support needed and the support received in terms of quality, quantity, and flexibility of services [15].
This gap widened dramatically during the SARS-CoV-2 pandemic when the availability of support services was limited or ceased. This raised great concerns for people with dementia and their carers related to a lack of information, difficulties in safeguarding procedures, or social isolation [16, 17]. Technology-based interventions may be effective options for informal carers to overcome isolation and risk of infection [18]. Beyond the pandemic, the regionally varying availability of support services, the dynamics and complexity of family caregiving with changing support needs make it necessary to find alternative ways for delivering support [19]. There are various types of support, such as information, education, or counselling [20].
Counselling is a conversation therapy that involves a trained therapist listening to individuals and helping them to find ways to cope with an emotional issue. It can be differentiated from information, education, instruction, and case management [21].
Technology-based counselling can be provided through telephone, email, text messaging, or telehealth/videoconferencing modalities [22]. People taking part in telephone counselling sessions believed it to be an attractive option because of convenience, accessibility, control, or inhibition [23]. An earlier mixed-methods systematic review focused on the efficacy of telephone counselling interventions [24]. The review indicated some evidence of moderate quality that telephone counselling can reduce depressive symptoms experienced by carers of people with dementia. Carers found that counselling without having to leave the house brought them more flexibility and less stress. Counsellors discussed the limitations of telephone counselling as they could not see the reactions of the carers [24]. These limitations may be overcome through the use of videoconferencing software [25, 26].
A recent review examined the efficacy of remotely delivered information, training, and support interventions for informal carers of people with dementia [27]. Counselling by health professionals was subsumed under support and the authors found that interventions may have an impact on burden and depression [27]. Saragih et al. investigated the effectiveness of telehealth-delivered psychoeducational and behavioral interventions for persons with dementia and their carers [28]. This comprises a wide range of different interventions, including counselling, which appear to have an effect on depression among persons with dementia and on the perceived competency of carers [28].
Previous reviews therefore focused on counselling provided via telephone or on a wide range of psychosocial interventions using various technologies. In our systematic review, we focused exclusively on counselling interventions delivered through various types of information technology. We aimed at investigating the effectiveness of technology-based counselling interventions for people with dementia and/or their informal carers.
MATERIALS AND METHODS
We followed the Cochrane Handbook for Systematic Reviews of Interventions guidance [29]. The review protocol was prospectively registered in PROSPERO (CRD42021245473) and published [30].
Details about the results of the Qualitative Comparative Analysis will be reported elsewhere.
Eligibility criteria
We included individual and cluster parallel-group randomized controlled trials (RCT), cross-over and stepped-wedge designs assessing counselling interventions for people with dementia and/or their informal carers; published in English or German with no restriction regarding the year of publication. Counselling interventions had to be provided by trained health and social care professionals focusing on the management of daily living, disease and care management as well as functioning and participation by using technologies such as telephone, computer, and web- or mobile-based applications. Both inactive (e.g., labelled as no intervention or no treatment) and active comparisons (e.g., labelled as standard care, usual care, or onsite counselling interventions) were included. We did not make restrictions concerning specific clinical outcomes.
We excluded studies on interventions targeting people with mild cognitive impairment and studies on specific and standardized interventions such as cognitive behavior therapy, or genetic counselling as well as counselling with regard to predictive diagnostics, screening for dementia or diagnostic procedures or educational programs.
Information sources and search strategy
We performed a comprehensive systematic literature search combining an electronic database search with supplementary search methods [31, 32]. The search strategy was developed by one reviewer (JH) and was peer-reviewed by an external consultant using the Peer Review of Electronic Search Strategies (PRESS) [33]. We searched CINAHL, Cochrane Library including Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE via PubMed, PsycINFO via Ovid, and the Web of Science Core Collection (last search: April 22, 2021) without using filters and limiters. Additionally, we conducted forward and backward citation tracking of included studies and eligible reviews using Scopus (last search: October 7, 2021) and web searching via Google and Google Scholar (last search: November 26, 2021). The database-specific search strategies are provided in the Supplementary Material, Chapter A.
Selection process
Two reviewers (out of AB, JH, FW, and DB) independently screened titles, abstracts, and full texts using the Rayyan web app [34]. Disagreements were solved by discussion.
Data collection process
One reviewer (JW or DB) extracted data using a standardized and piloted extraction sheet. Data was cross-checked for accuracy by the other researcher and any discrepancies were resolved by mutual discussion between these two reviewers. We extracted study characteristics, participants’ characteristics (age, gender, ethnic, and educational background), and intervention characteristics (type of intervention and comparison, technology used for delivery, duration and frequency of counselling sessions, outcome(s), measurement(s) and results). In order to generate a detailed description of the reported intervention characteristics and their theoretical underpinning we applied the criteria of the Template for Intervention Description and Replication (TIDieR) checklist [35] and the updated Criteria for Reporting the Development and Evaluation of Complex Interventions (CReDECI-2) guideline [36]. The TIDieR checklist focuses on the description of interventions (e.g., why, what, who provided, where, and how) in order to improve replicability [35]. The CReDECI-2 guideline refers to the development and evaluation stages of complex interventions by applying items such as theoretical underpinning, components of interventions and its interaction, facilitators and barriers potentially influencing the delivery of the intervention [36]. By combining these two guidelines, we were able to comprehensively map the interventions.
Prespecified review outcomes
The predefined primary outcome of our review was depressive symptoms experienced by people with dementia or informal carers. All other outcomes such as burden, self-efficacy, or quality of life were considered as secondary outcomes.
Study risk of bias assessment
We assessed the risk of bias for all outcomes in each study using the revised Risk of Bias tool (RoB 2) [37]. Two reviewers (out of JH, JW, and DB) conducted the RoB 2 assessments independently using the RoB 2 Excel tool for documentation [38]. Conflicting results were discussed between the involved reviewers and, if necessary, with an experienced methodologist (GL). We used robvis to create risk-of-bias assessment figures [39].
Effect measures and synthesis methods
We performed a pooled meta-analysis of the intervention effects with respect to the clinical and statistical heterogeneity. We used the numbers of participants who answered at the different follow-up periods to estimate 95% confidence intervals (CI) irrespective of imputations. The Review Manager Version 5.3 was applied to pool metric data with the random effects model [40]. If studies reported the same outcome with different measurement tools, we computed standardized mean differences (SMD) with their 95% CI using the Comprehensive Meta-Analysis program Version 3.3 and entered the data into RevMan as generic inverse variance data [41]. If a study reported results on different follow-up periods, data from the longest follow-up were used. All meta-analyses base on metric data and used mean differences (MD) or standardized mean differences (SMD) with their 95% CI as intervention effects. The magnitude of an effect was considered as small at an SMD of 0.2, medium at an SMD of 0.5 and large at an SMD of 0.8 [42]. Negative effects are interpreted with the same cut-offs. In cases of more than one measure of the same outcome, we utilized the mean value. Statistical heterogeneity between intervention effects was measured on the basis of I-squared-values (I2) and classified as not important (0% to 40%), moderate (30% to 60%), substantial (50% to 90%), or considerable (75% to 100%) [43]. We conducted a subgroup analysis based on control conditions when substantial heterogeneity across results of included studies was found.
If pooling of data was not possible due to clinical and/or statistical heterogeneity, we performed a narrative synthesis. When no synthesis could be performed due to different effect measures, incompletely reported effect estimates, or single studies reported on outcomes, we tabulated available data for structured reporting of effects [44].
Certainty of evidence assessment
We applied the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach to rate the certainty of evidence as high, moderate, low, or very low [45]. The GRADEpro GDT software was used to create the “Summary of findings” table [46, 47].
In order to determine the importance of the outcomes assessed in the included studies, we involved two persons (entitled “advisory board” in the following). One is a family carer and engaged in a regional support organization for people with dementia and their families. The other is a professional consultant of the Alzheimer’s Society in a German federal state. They were presented with a questionnaire in which they were asked to value the importance of the outcomes on a scale from 1 (not important) to 9 (very important) [48]. According to the GRADE approach, we used three categories to distinguish outcome importance (mean scores of numerical ratings: from 7 to 9 = high importance; from 4 to 6 = moderate importance; 1 to 3 = low importance) [45, 48]. In addition, open-ended questions were asked that addressed outcomes not yet examined in the included studies as well as reasons for the informal carers’ use of counselling services. We used the advisory boards’ assessment to integrate their perspective in the interpretation of results.
Deviations from the intended approach as published in the protocol
Although we planned not to exclude studies because of language [30], we had to exclude reports not written in English or German due to the lack of resources for professional translations.
RESULTS
Study selection
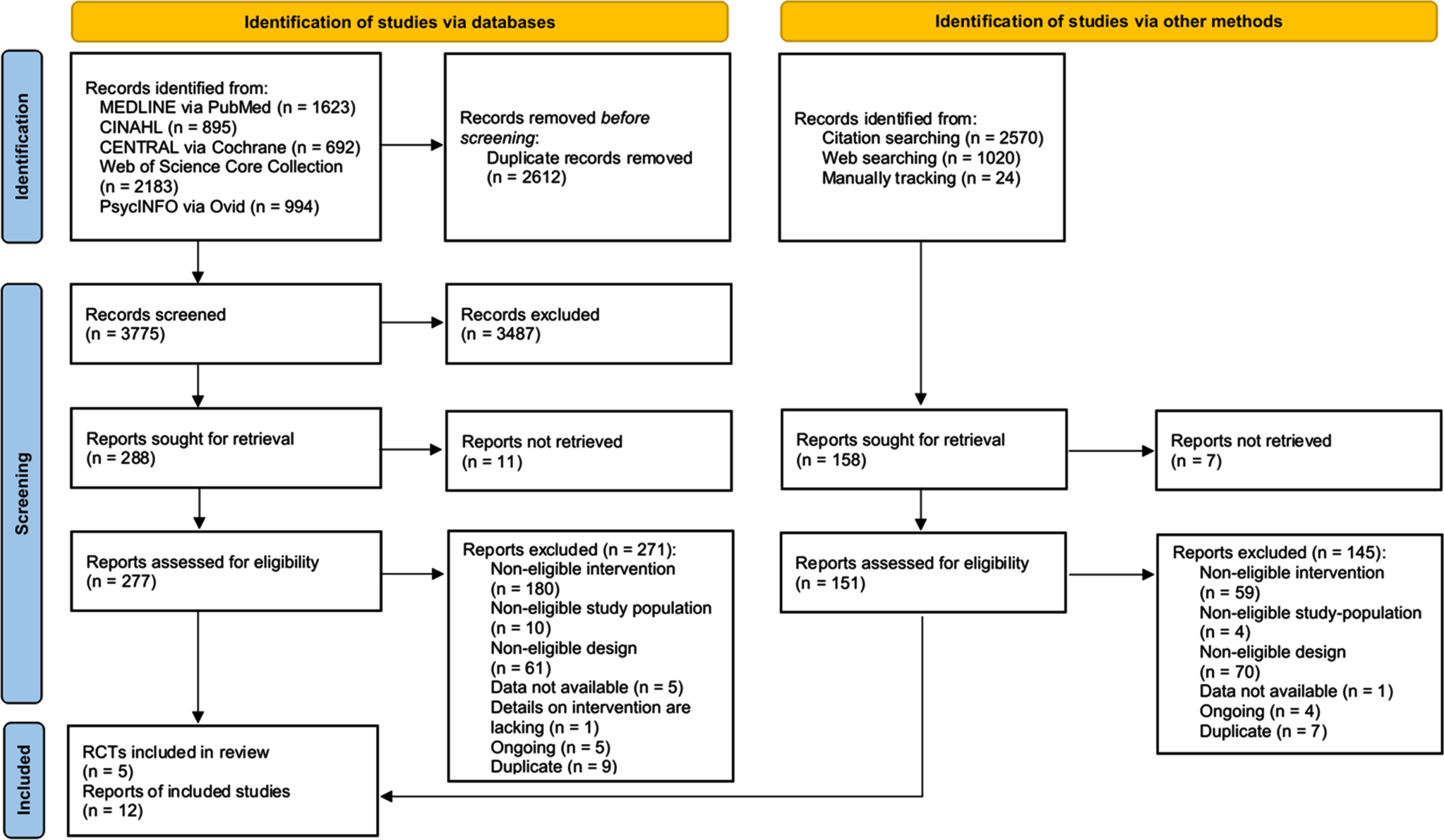
Figure 1 displays the flow diagram of the study selection process. We identified 6,387 records from electronic database search. After removing duplicates, we screened the titles and abstracts of 3,775 records, and the full-text of 277 records for eligibility. Six publications reporting on two studies [49, 50] met the inclusion criteria. We identified 3,614 records from additional sources (citation tracking and web searching). Of these, we assessed 151 full-texts and included six records on three studies [51–54]. We also identified nine records on five ongoing studies [55–59]. We excluded 416 records; the reasons for exclusion are displayed in Fig. 1. Finally, five RCT with a total of 12 publications were included [49–54, 60–65].
Fig. 1
PRISMA flow diagram of study selection.
Study characteristics
Table 1 presents the general characteristics of the included studies. The five RCT involved a total of 880 participants, ranging from 60 to 445 participants per study [49–53]. The studies were published between 1995 and 2021. Four trials were conducted in the USA [49–52] and one in Australia [53]. Counselling interventions were provided for carers by healthcare professionals (four studies, [49–52]) or for carer–care-receiver dyads (one study, [53]). Carers were predominantly spouses (51%) and adult children (42%) of people with dementia (data provided in four studies [49, 51–53]). While women made up the largest proportion of carers (88% [51]; 78% [52]; 76% [53]; 80% [50]), the ethnic and educational backgrounds varied across studies.
Table 1
Characteristics of included studies
| First author Year | Country | Population | Intervention | Comparison | Outcomes (Follow-Up) | ||||
| Target population | N rand. (IG/CG) | Mean age (SD) in years | Women (%) | Type | Duration Frequency Period | ||||
| Coyne 1995 [51] | USA | Carer | 62* (n. a.) | IG: 48.7 (11.3) CG: 51.4 (14,0) | 88 | Telephone counselling | n. a. Helpline call + biweekly follow-up calls 8 weeks | Standard care: Helpline services during a single call | Carer: Depression Carer: Burden Carer/care-receiver: Resource use (8 weeks) |
| Tremont 2008 [49] | USA | Carer | 60 (32 / 28) | IG: 65.75 (13.71) CG: 61.00 (9.60) | – | Telephone counselling with additional materials | Initial 60 min, follow-up 15–30 min 23 telephone contacts 12 months + Educational and resource materials | No counselling + Educational and resource materials | Carer: Depression Carer: Burden Carer: Reaction to care-receiver behavior Carer: Knowledge Carer: General health Carer: Self-efficacy Carer: Family functioning Carer: Perceived social support (12 months) |
| Tremont 2015 [52, 54, 63] | USA | Carer | 250 (133 / 117) | IG: 63.32 (12.30) CG: 62.03 (13.75) | 78 | Telephone counselling with additional materials | Initial 60 min, follow-up 15–30 min 16 telephone contacts 6 months + Educational and resource materials | Telephone Support: Non-directive support through empathic and reflective listening and open-ended questioning; same duration, frequency, and period as IG + Educational and resource materials | Carer: Depression Carer: Burden Carer: Reaction to care-receiver behavior Carer: Family functioning Carer: Self-efficacy Carer: Positive aspects of caregiving Carer: Quality of life (6 months) Carer/care-receiver: Resource use (monthly over 6 months) |
| Laver 2020 [53] | Australia | Dyads | 63 (31 / 32) | Care-receiver: IG: 79.45 (6.52) CG: 80.47 (7.198) Carer: IG: 69.47 (11.849) CG: 70.66 (15.51) | Care-receiver: 40 Carer: 76 | Video- conference-based counselling | 60 min 8 consultations (first 2 home visits) 16 weeks | Home Visits: Same duration, frequency, and period as IG | Carer: Mastery Care-receiver: Functionality Care-receiver: Number of behaviors Carer: Perceived change Carer: Upset (4 months) |
| Hodgson 2021 [50] | USA | Carer | 445 (257 / 188) | IG: 60.42 (13.04) CG: 61.27 (12.23) | 80 | Telephone counselling | n. a. Initial call + one follow-up 1 month** | Standard care: Helpline consultation service | Carer: Global Health Carer: Self-efficacy (1 week, 1 month) |
CG, control group; IG, intervention group; min, minutes; n.a., not available; rand., randomized. *Details on randomization procedures are missing; **Additional booster call was made within a month; the exact time was not specified.
Telephone or videoconferencing software was utilized to deliver counselling. Healthcare professionals assessed the participants’ situation and provided support tailored to the individual needs. Supportive and empathetic approaches were applied, which comprised information as well as educational and problem-solving components. In one trial, technology-based counselling was combined with home visits [53]. Additional material was used for information on local resources and education in two interventions [49, 52]. The theoretical underpinning was described for two interventions [49, 52]. The interventions lasted between 1 and 12 months and comprised two to 23 counselling sessions. Participants in the control group received educational and resource materials only, standard (helpline) services, non-directive support, or home visits. A detailed description of the interventions and comparisons using the TIDieR [35] and CReDECI [36] criteria can be found in the Supplementary Material, Chapter B.
Outcomes were measured using self-reported assessment instruments for carers and proxy-reported for people with dementia. Three studies assessed depressive symptoms and burden experienced by carers of people with dementia [49, 51, 52]. Self-efficacy or mastery in caregiving was assessed in four studies [49, 50, 52, 53] and carers’ reaction to or upset due to dementia-related behavior were reported in three studies [49, 52, 53]. We identified a wide range of outcomes (n = 14) and assessment instruments (n = 21) (Table 1).
Risk of bias in studies
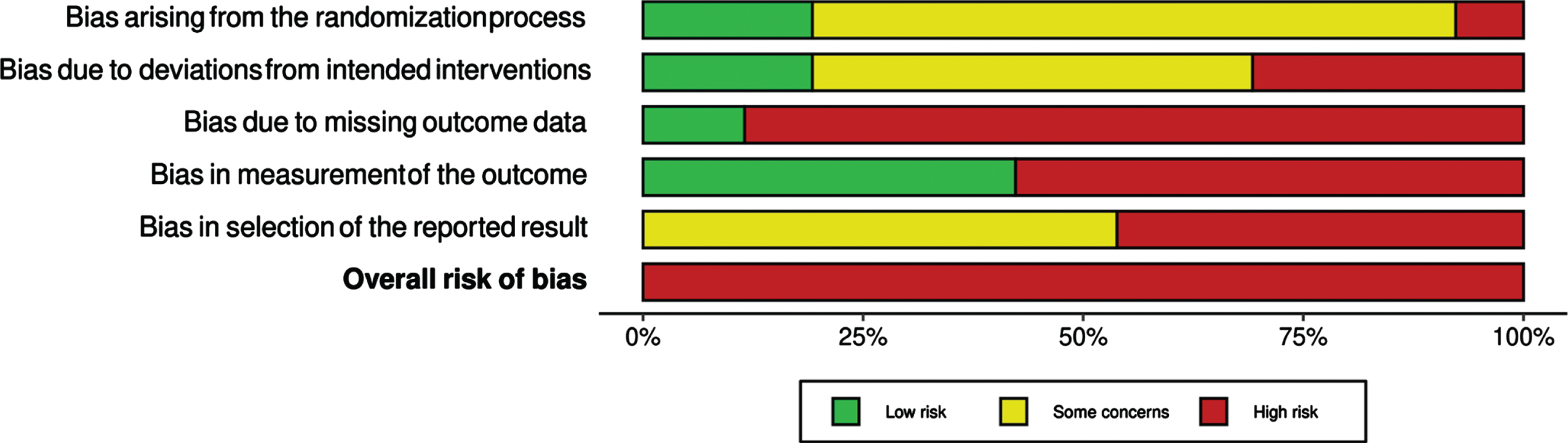
The results of the critical appraisal are displayed in the risk of bias summary plot (Fig. 2). We assessed the risk of bias for 26 outcomes. A high risk of bias due to missing outcome data was found for 23 outcomes. In addition, we found a bias in the selection of the reported results due to deviations in the reported results from the trial registry entry or due to the lack of a pre-specified analysis plan for all outcomes. The risk of bias traffic light plot of assessments for all outcomes as well as justifications of our judgements are provided in the Supplementary Material, Chapter C.
Fig. 2
Risk of bias summary plot of RoB 2 assessments across all outcomes of included studies.
Synthesis
Depressive symptoms
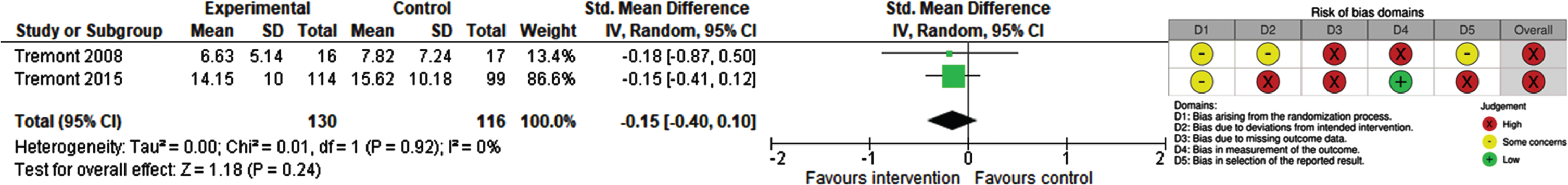
Table 2 shows the summary of findings for our pooled primary and secondary outcomes. Three studies measured depressive symptoms experienced by carers of people with dementia [49, 51, 52]. The outcome was not further defined in the included studies. Coyne 1995 reported no significant effect on depression after 8 weeks (Zung Self-Rating Depression Scale; range 20–80; higher score indicates more depression; 51 participants) [51]. We contacted the first author for numerical data, which are no longer available. Two studies (246 participants) reported data on depression at 6 months or 12 months [49, 52]. Both studies found slightly lower depression scores applying different measurement tools (Tremont 2008: Geriatric Depression Scale (GDS); range 0–30; higher score indicates more depression; Tremont 2015: Center for Epidemiology Studies Depression Scale (CES-D), range 0–60; higher score indicates more depression). Meta-analysis showed no statistically significant difference between intervention and control groups (SMD –0.15; 95% CI –0.40 to 0.10; I2 = 0%) (Fig. 3). The certainty of evidence was rated as low due to a high risk of bias.
Table 2
Summary of findings: Technology-based counselling compared to educational and resource materials only, standard (helpline) services, non-directive support, and home visits for carers of people with dementia
| Certainty assessment | Nunderbaro of patients | Effect | Certainty | Importance | ||||||||
| Nunderbaro of studies | Study design | Risk of bias | Inconsistency | Indirectness | Imprecision | Other considerations | Technology-based counselling | Educational and resource materials only, standard (helpline) services, non-directive support, and home visits | Relative (95% CI) | Absolute (95% CI) | ||
| Depressive symptoms (follow-up: range 6 months to 12 months; assessed with: self-reported scales, higher scores indicate more depressive symptoms) | ||||||||||||
| 2 | randomized trials | very seriousa | not serious | not serious | not serious | none | 130 | 116 | – | SMD 0.15 SD lower (0.4 lower to 0.1 higher) |  Low Low | |
| Burden (follow-up: range 8 weeks to 12 months; assessed with: ZBI) | ||||||||||||
| 3 | randomized trials | very seriousb | seriousc | not serious | seriousd | none | 153 | 144 | – | MD 5.3 lower (11.7 lower to 1.11 higher) |  Very Low Very Low | |
| Self-efficacy/mastery (follow-up: range 1 month to 12 months; assessed with: self-rated scales, higher scores indicate higher self-efficacy/mastery) | ||||||||||||
| 4 | randomized trials | very seriouse | not serious | not serious | not serious | none | 268 | 211 | – | SMD 0.07 SD higher (0.11 lower to 0.25 higher) |  Low Low | |
CI, confidence interval; MD, mean difference; SMD, standardized mean difference; ZBI, Zarit Burden Interview. Explanations:aDowngraded two levels because of high risk of bias due to missing outcome data and in measurement of the outcome in both studies and an additional high risk of bias in selection of the reported result in one study. bDowngraded two levels because of high risk of bias due to missing outcome data and in measurement of the outcome in all three studies and an additional high risk of bias in selection of the reported result in one study. cDowngraded one level due to substantial statistical heterogeneity. dDowngraded one level due to wide confidence intervals. eDowngraded two levels because of high risk of bias due to missing outcome data in all four studies, high risk of bias in measurement of the outcome in three studies, high risk of bias in selection of the reported results in two studies and high risk of bias arising from the randomization process and due to deviations from the intended interventions in one study each.
Fig. 3
Forest Plot and RoB 2 for depressive symptoms experienced by carers of people with dementia.
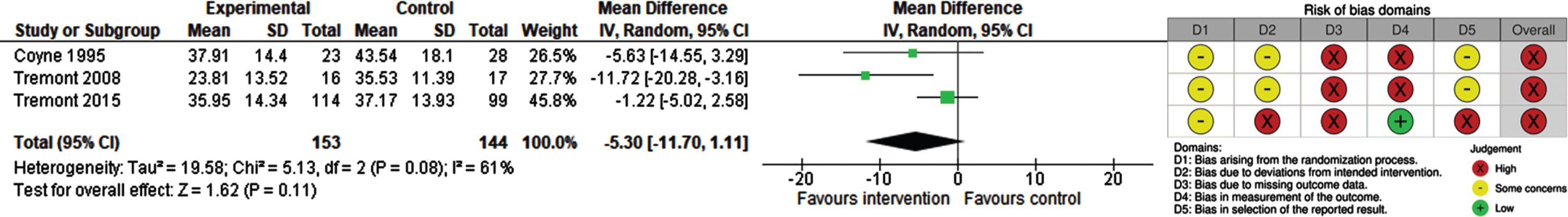
Burden
Three studies (297 participants) reported information on burden at 8 weeks, 6 months and 12 months [49, 51, 52]. All studies used the Zarit Burden Interview (ZBI; range 0–88; higher score indicates higher burden) and reported a lower burden for carers in the intervention group. Pooled data revealed no statistically significant difference between the intervention and control groups (MD –5.30; 95% CI –11.7 to 1.11, I2 = 61%) (Fig. 4). The certainty of evidence was rated as very low because of high risk of bias, substantial heterogeneity, and imprecision.
Fig. 4
Forest Plot and RoB 2 for burden experienced by carers of people with dementia.
Self-efficacy/mastery
Four studies (479 participants) provided low-certainty evidence on self-efficacy/mastery at follow-up periods of 1 week and 1 month, 4 months, 6 months and 12 months [49, 50, 52, 53]. The studies employed different assessment instruments with higher scores describing higher self-efficacy or mastery (Hodgson 2021: PROMIS Self-Efficacy for Managing Emotions; range 8–40; Laver 2020: Caregiving Mastery Index; range 0–24; Tremont 2015: Self Efficacy Questionnaire Symptom Management and Self Efficacy Questionnaire Support Services; range 0–50; Tremont 2008: Self-Efficacy Questionnaire; range 0–100). We used the results from the longest follow-up period of 1 month in Hodgson 2021 and results of measurements with different tools in Tremont 2015 and found no statistically significant difference between groups (SMD 0.07; 95% CI –0.11 to 0.25; I2 = 0%) (Fig. 5).
Fig. 5
Forest Plot and RoB 2 for self-efficacy/mastery experienced by carers of people with dementia.

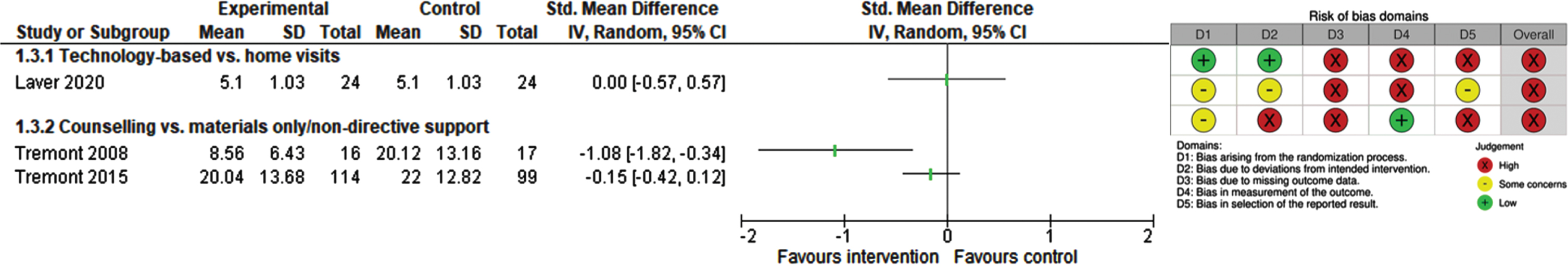
Reaction to/upset due to dementia-related behavior
Three studies (294 participants) reported information on carers’ reactions to or upset due to dementia-related behavior at 4, 6, and 12 months [49, 52, 53]. Due to the substantial heterogeneity across results of all included studies, we added a subgroup analysis based on the different control groups, which did not lead to a reduction of heterogeneity. Figure 6 displays the studies’ results. Comparing technology-based counselling with home visits, Laver 2020 found no differences between groups (SMD 0.00; 95% CI –0.57 to 0.57; Caregiver Behavioral Occurrence and Upset Scale; range not reported; higher score indicates higher upset; 48 participants) [53]. Tremont 2008 reported significantly less severe reactions to dementia-related behaviors after 23 telephone counselling sessions combined with additional material compared to those carers receiving educational and resource materials only (SMD –1.08; 95% CI –1.82 to –0.34; Revised Memory and Behavior Problem Checklist; range 0–96; higher score indicates higher upset; 33 participants) [49]. When compared to non-directive support, telephone counselling had no statistically significant effect on carers’ upset (SMD –0.15; 95% CI –0.42 to 0.12; Revised Memory and Behavior Problem Checklist; range 0–96; higher score indicates higher upset; 213 participants) [52].
Fig. 6
Forest Plot and RoB 2 for reactions/upset experienced by carers of people with dementia.
Structured summary
Table 3 displays the data for the structured summary.
Table 3
Outcomes for structured reporting
| Outcome | Reference | N IG/CG | Intervention Mean (SD) | Control Mean (SD) | MD (95% CI) | p | ||||
| Secondary outcomes: Patient and carer level | Global Health / General Health | Hodgson 2021 [50] | 113 / 68 112 / 68 | GPH 13.70 (2.74) GMH 11.79 (3.38) | GPH 14.07 (2.12) GMH 11.72 (3.02) | n. a. n. a. | 0.26 0.95 | |||
| Tremont 2008 [49] | 16 / 17 | n. a. | n. a. | n. a. | n. a. | |||||
| No statistically significant differences between the groups at the end of treatment | ||||||||||
| Quality of life | Tremont 2015 [52] | 114 / 99 | 79.87 (15.00) | 77.59 (15.69) | n. a. | 0.75 | ||||
| Family functioning | Tremont 2008 [49] | 16 / 17 | n. a. | n. a. | n. a. | n. a. | ||||
| No statistically significant differences between the groups at the end of treatment | ||||||||||
| Tremont 2015 [52] | n. a.*** | 2.09 (0.63) | 2.00 (0.50) | n. a. | 0.80 | |||||
| Perceived social support | Tremont 2008 [49] | 16 / 17 | 2.47 (1.34)** | 2.38 (1.31)** | 0.09* (–0.62 to 0.75)** | n. a. | ||||
| Perceived change | Laver 2020 [53] | 26 / 27 | 37.7 (5.10*) | 40.1 (5.20*) | –2.4 (–5.4 to 0.52) | 0.10 | ||||
| Positive aspects of caregiving | Tremont 2015 [52] | n. a.**** | 34.13 (8.31) | 35.24 (7.55) | n. a. | 0.04 | ||||
| Knowledge | Tremont 2008 [49] | 16 / 17 | n. a. | n. a. | n. a. | n. a. | ||||
| No statistically significant differences between the groups at the end of treatment | ||||||||||
| Behavior | Laver 2020 [53] | 24 / 24 | 6.5 (1.91*) | 8.7 (1.91*) | –2.1 (–3.3 to –1.04) | 0.0003 | ||||
| Functionality | Laver 2020 [53] | 26 / 26 | 59.9 (8.67*) | 63.8 (8.67*) | –3.9 (–8.7 to 1.0) | 0.12 | ||||
| Secondary outcomes System level | Resource use | Average number of services used | Coyne 1995 [51] | 23 / 28 | 6.78 (2.2) | 4.57 (2.5) | n. a. | n. a. | ||
| Number of times per month each community service was used | Tremont 2015 [54] | 105 / 94 | Carer: | 8.47 (5.14) | Carer: | 5.14 (7.05) | n. a. | 0.02 | ||
| Care-receiver: | 6.79 (13.72) | Care-receiver: | 5.11 (10.68) | 0.34 | ||||||
| Number of monthly visits of healthcare resources | 105 / 94 | Carer: | ED 0.02 (0.06) | Carer: | ED 0.05 (0.13) | n. a. | 0.02 | |||
| Hospital 0.01 (0.04) | Hospital 0.04 (0.10) | n. a. | 0.006 | |||||||
| Doctor 0.85 (0.89) | Doctor 0.71 (0.61) | n. a. | 0.88 | |||||||
| Care-receiver: | ED 0.08 (0.16) | Care-receiver: | ED 0.08 (017) | n. a. | 0.83 | |||||
| Doctor 1.32 (1.35) | Doctor 1.30 (1.05) | n. a. | 0.88 | |||||||
CG, control group; CI, confidence interval; ED, emergency department; GMH, global mental health; GPH, global physical health; IG, intervention group; MD, mean difference; n. a., not available; SD, standard deviation. *Data calculated by reviewers; **Data extracted from Lins 2014 [24]; ***N = 171, no specification for IG/CG; ****N = 220, no specification for IG/CG.
Health-related outcomes
Two studies assessed the impact of technology-based counselling on global/general health [50, 49] and one study on health-related quality of life [52]. Telephone counselling (two, 16 or 23 sessions) was combined with additional materials in two studies [49, 52]. The PROMIS Global Health questionnaire (Global Physical Health (GPH) and Global Mental Health (GMH) scores), the EQ-5D VAS and the SF 36 General Health questionnaire were used for assessments after 1, 6, and 12 months. There was no statistically significant effect on health-related outcomes when compared to standard helpline services (GPH: p = 0.26; range 4–20; lower score indicates better health; 181 participants; GMH: p = 0.95; range 4–20; lower score indicates better health; 180 participants) [50], educational and resource materials only (no numerical data reported for SF 36 General Health scores; 33 participants) [49], and non-directive support (p = 0.75; EQ-5D VAS; range 0–100; higher score indicates higher quality of life; 213 participants) [52].
Family functioning and social support
Two studies assessed family functioning using the General Functioning subscale of the Family Assessment Device (FAD GF subscale; range 1–4; higher score indicates poorer functioning) [52, 49]. All participants received educational and resource materials, and telephone counselling was compared to no counselling or non-directive support. There were no statistically significant differences between groups after 6 and 12 months (Tremont 2008: no numerical data reported; 33 participants; Tremont 2015: p = 0.80; 171 participants) [49, 52].
Tremont 2008 found no statistically significant effect of telephone counselling on carers’ perception of social support after 12 months (MD 0.09; 95% CI –0.62 to 0.75; Multidimensional Scale of Perceived Social Support (MSPSS); range 1–12; higher score indicates more perceived support; 33 participants) [49].
Perceived change and benefits of caregiving
The Perceived Change Scale (PCS) was applied in Laver 2020 to assess whether carers perceive the situation as getting better or worse. No statistically significant differences between groups receiving counselling via videoconferencing or during home visits were found (MD –2.4; 95% CI –5.4 to 0.52; PCS; range 13–65; higher score indicates positive change; 53 participants) [53].
Tremont 2015 assessed carers’ perception of benefits within the caregiving context comparing telephone counselling to non-directive support, both combined with educational and resource materials. Participants in the control group showed a statistically significant greater improvement in positive aspects of caregiving (P value = 0.04; Positive Aspects of Caregiving scale (PAC); range 9–45; higher score indicates more positive aspects; 220 participants) [52].
Dementia knowledge
Tremont 2008 measured the carers’ dementia knowledge using the Alzheimer’s Disease Knowledge test. All participants were provided with educational and resource materials and carers in the intervention group received 23 counselling sessions. There were no significant differences between groups after 12 months (no numerical data reported; 33 participants) [49].
Outcomes of people with dementia
Laver 2020 measured the number of dementia-related behavior and the functionality of the care-receiving person using proxy-reported assessment instruments after 4 months of telehealth or home visit delivery of a counselling intervention. While there were no statistically significant differences in functional capacities of people with dementia (MD –3.9; 95% CI –8.7 to 1.0; Caregiver Assessment of Function and Upset (CAFU total); range 15–105; higher score indicates higher functionality; 52 participants), the number of dementia-related behaviors decreased significantly in the telehealth group (MD –2.1; 95% CI –3.3 to –1.04; Caregiver Behavioral Occurrence and Upset Scale; range not reported; higher score indicates higher frequency; 48 participants) [53].
Resource use
Two studies reported data for outcomes on the healthcare system level [51, 54]. Coyne 1995 assessed the number of services summarizing primary care physician visits, prescribed medications, emergency room visits, support group attendance, hospital stays, and over-the-counter medications. Resource use increased in both groups during the follow-up period of 8 weeks. Participants receiving an additional call showed a greater rate of increase than participants receiving standard helpline services [51]. Tremont 2015 differentiated between community and healthcare resources when assessing resource use. Carers receiving 16 counselling sessions over 6 months had a statistically significant higher rate of community service use (p = 0.02) as well as a lower rate of Emergency Department visits (p = 0.02) and hospital stays (p = 0.006) than participants receiving non-directive support. There were no statistically significant differences between groups of care-receiving persons in the use of community resources (p = 0.34) such as respite care, support group attendance, food delivery, and healthcare resources (Emergency Department visits: p = 0.83; Doctor visits: p = 0.88) [54].
Outcome importance
The advisory boards’ ratings for outcome importance resulted in a high importance with a mean score from 7 to 9 for depressive symptoms, burden, family functioning, self-efficacy/mastery, physical and mental health, reaction to dementia-related behavior, frequency of dementia-related behavior, and functionality in activities of daily living of persons with dementia as well as healthcare and community resource use. The following outcomes were considered to be of moderate importance (mean score: 4 to 6): general health, quality of life, perceived social support, perceived change, positive aspects of caregiving, and dementia knowledge. The advisory board suggested further outcomes, i.e., changes in the social support network of carers, the perception of isolation or social participation, and access to support services. In addition, it was indicated that counselling is often sought when the stress levels of the informal carers are high or when they feel overwhelmed. One person reported from her experience that older carers refused the use of technology for counselling.
DISCUSSION
Our systematic review synthesized five RCT evaluating the effectiveness of technology-based counselling interventions for people with dementia and their carers. Meta-analysis was performed with two studies and revealed no evidence that technology-based counselling has an effect on our primary outcome of depressive symptoms experienced by carers of people with dementia. Meta-analyses for burden and self-efficacy/mastery of informal carers also showed no statistically significant differences between groups. Technology-based counselling seems to have some beneficial effects on the frequency of dementia-related behavior [53], carers’ reaction to dementia-related behavior [49], and community and healthcare resource use by informal carers [54] as our narrative synthesis and structured summary demonstrated.
Our findings support earlier research [24, 27, 28]. The Cochrane review by Lins et al. suggested that telephone counselling without additional materials is effective for reducing depressive symptoms in carers of people with dementia [24]. The authors reported inconsistent results of studies on this outcome and findings based on a small number of trials [24]. Remotely delivered interventions comprising training, support or both, with or without information for informal carers of people with dementia may slightly reduce depressive symptoms when compared with information alone, but had no significant effect when compared with usual treatment, waiting list or attention control (moderate certainty) [27]. There was also no effect of telehealth interventions which included counselling interventions as well as psychoeducation and cognitive behavioral therapy on carers’ depression [28]. Effects on burden experienced by carers of people with dementia were described as non-significant [28], uncertain [24], or small, depending on the control intervention [27]. No evidence was found for beneficial effects on further outcomes such as quality of life and self-efficacy [24, 28].
We found a considerable clinical heterogeneity in the included studies. Counselling interventions ranged from extended services of long-established helplines to theory-based, manualized interventions implemented for the first time in the context of the trial setting. There was also a broad range of comparisons, which has to be considered while interpreting the results. While the number of counselling sessions and length of follow-up also varied, individualized counselling covered a similar content and aimed at reducing stress and improving mood and the ability to manage dementia-related challenges.
A variety of outcomes was used to assess the effectiveness in achieving these goals and the use of different outcomes made it difficult to compare the effects of different interventions directly and to conduct meta-analyses [66]. Furthermore, the differences in the importance assigned to the outcomes by the advisory board and the additionally suggested outcomes indicate that further effort is needed to determine adequate outcomes for technology-based counselling interventions. Involvement of people with dementia and their representatives seems to be mandatory in order to define outcomes with better clinical relevance.
The fact that only two studies explicated the theoretical underpinnings [49, 52] suggests that the development of the interventions was not guided by theory. There seems to be a lack of theory-driven approaches in determining objectives of counselling interventions and outcomes, as well as in developing these interventions and their interacting components. To overcome these shortcomings, the framework of the Medical Research Council (MRC) should be applied to the development and evaluation of complex interventions [67].
The information technology used in included studies were telephone [49–52] and videoconferencing software [53]. The telephone as a widespread and undemanding technology was found to enhance accessibility [52]. In contrast, videoconferencing is described as a more direct and immersive mode of communication because it engages verbal and non-verbal communication, and facial expression [25]. Videoconferencing is associated with higher demands on technical skills, and instruction for technical application as well as support for technical equipment may be required [53]. Despite the advantages of using technology for counselling, González-Fraile et al. found that remotely delivered interventions seem to be less acceptable than control interventions such as information alone which is indicated by participant‘s attrition [27]. In addition, technology-assisted counselling was perceived as convenient [23], but it can be more difficult for those seeking advice to maintain privacy. It was not described in included studies whether the person with dementia was home or present during counselling sessions. This illustrates the complexity of using informational technology for counselling interventions in dementia, even though its use was considered beneficial, particularly during the SARS-CoV-2 pandemic [68]. Further research is needed to understand whether the combination of face-to-face and technology-based counselling and the use of multiple technologies such as telephone, email, videoconferencing, and mobile-based services are able to meet the needs of people with dementia and their informal carers.
The quality of evidence of the pooled outcomes was rated as low to very low (Table 2). All results were downgraded two levels due to high risk of bias in several domains. We also downgraded the result for burden because of a substantial statistical heterogeneity and inconsistency between studies.
We found a high risk of bias in all other outcomes. The most common reason for a high risk of bias was due to missing outcome data (Fig. 2). We also rated risk of bias high due to self-reporting of subjective outcomes by participants who were not masked to group assignment. In addition, there was a lack of details in the description of the randomization process in four studies.
Strengths and limitations
We used rigorous methods to perform a comprehensive systematic literature search and to evaluate the effectiveness of technology-based counselling for people with dementia and their informal carers. We focused exclusively on counselling interventions. As these are complex interventions with several components, it was sometimes challenging to differentiate from interventions applying predominantly psychotherapeutic or educational approaches, or standardized procedures. Although counselling is a relatively common service, very few interventions have been evaluated in RCT and only five studies met our inclusion criteria. In order to reflect the complexity and heterogeneity of the interventions included, we applied the criteria of two reporting guidelines [36, 35]. Although we contacted authors for further information on assessment instruments and/or data on results, we received responses from authors but in some cases, information was not provided.
The assessment of the outcome importance is based on the information of only two people. One of them has experience in caring for a person with dementia and both represent regional support organizations for families with dementia. They provided a differentiated view on the outcomes but a broader base is needed to determine adequate outcomes for effectiveness trials.
Conclusions
The available evidence from RCT suggests no effects of technology-based counselling interventions on depressive symptoms, burden, and self-efficacy/mastery experienced by carers of people with dementia. Single studies suggest some beneficial effects on the frequency of dementia-related behavior of people with dementia and on carers’ reaction to dementia-related behavior and resource use. However, these findings need confirmation through future research, which is theory-based and informed by guidance for the development and evaluation of complex interventions.
ACKNOWLEDGMENTS
We thank Thomas Nordhausen from the Institute of Health and Nursing Science at Martin Luther University Halle-Wittenberg for peer reviewing and critically appraising our search strategies. We would like to thank the members of the advisory board, Steffi Bartsch (Landesinitiative Demenz Sachsen e. V.) and Eva-Maria Klecha (Scheibenberger Netz e.V.), for their contribution to our study.
FUNDING
This study is funded by the German Federal Ministry of Education and Research (BMBF), grant number: 01KG2110.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its supplementary material.
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JAD-221194.
REFERENCES
[1] | Hung L , Patience AL , Au-Yeung AT , Kucherova I , Harrigan M ((2016) ) Adopting a clinical assessment framework in older adult mental health. J Psychosoc Nurs Ment Health Serv 54: , 26–31. |
[2] | Prince M , Prina M , Guerchet M ((2013) ) World Alzheimer Report 2013: Journey of Caring: An analysis of long-term care for dementia. Alzheimer’s Disease International, London. |
[3] | Stephan A , Bieber A , Hopper L , Joyce R , Irving K , Zanetti O , Portolani E , Kerpershoek L , Verhey F , Vugt M de , Wolfs C , Eriksen S , Røsvik J , Marques MJ , Gonçalves-Pereira M , Sjölund B-M , Jelley H , Woods B , Meyer G ((2018) ) Barriers and facilitators to the access to and use of formal dementia care: findings of a focus group study with people with dementia, informal carers and health and social care professionals in eight European countries. BMC Geriatr 18: , 131. |
[4] | Wimo A , Gauthier S , Prince M (2018) Global estimates of informal care, https://www.alzint.org/u/global-estimates-of-informal-care.pdf, Accessed August 4, 2022. |
[5] | Springate BA , Tremont G ((2014) ) Dimensions of caregiver burden in dementia: impact of demographic, mood, and care recipient variables. Am J Geriatr Psychiatry 22: , 294–300. |
[6] | Gilsenan J , Gorman C , Shevlin M (2022) Explaining caregiver burden in a large sample of UK dementia caregivers: The role of contextual factors, behavioural problems, psychological resilience, and anticipatory grief. Aging Ment Health, doi: 10.1080/13607863.2022.2102138. |
[7] | Pan Y , Chen R , Yang D ((2022) ) The relationship between filial piety and caregiver burden among adult children: A systematic review and meta-analysis. Geriatr Nurs 43: , 113–123. |
[8] | Peavy G , Mayo AM , Avalos C , Rodriguez A , Shifflett B , Edland SD ((2022) ) Perceived stress in older dementia caregivers: mediation by loneliness and depression. Am J Alzheimers Dis Other Demen 37: , 15333175211064756. |
[9] | Quinn C , Nelis SM , Martyr A , Morris RG , Victor C , Clare L ((2020) ) Caregiver influences on ‘living well’ for people with dementia: Findings from the IDEAL study. Aging Ment Health 24: , 1505–1513. |
[10] | Kuring JK , Mathias JL , Ward L ((2018) ) Prevalence of depression, anxiety and PTSD in people with dementia: a systematic review and meta-analysis. Neuropsychol Rev 28: , 393–416. |
[11] | Wulff J , Fänge AM , Lethin C , Chiatti C ((2020) ) Self-reported symptoms of depression and anxiety among informal caregivers of persons with dementia: a cross-sectional comparative study between Sweden and Italy. BMC Health Serv Res 20: , 1114. |
[12] | Isik AT , Soysal P , Solmi M , Veronese N ((2019) ) Bidirectional relationship between caregiver burden and neuropsychiatric symptoms in patients with Alzheimer’s disease: A narrative review. Int J Geriatr Psychiatry 34: , 1326–1334. |
[13] | Collins RN , Kishita N ((2020) ) Prevalence of depression and burden among informal caregivers of people with dementia: a meta-analysis. Ageing Soc 40: , 2355–2392. |
[14] | McCabe M , You E , Tatangelo G ((2016) ) Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist 56: , e70–88. |
[15] | Morrisby C , Joosten A , Ciccarelli M ((2018) ) Do services meet the needs of people with dementia and carers living in the community? A scoping review of the international literature. Int Psychogeriatr 30: , 5–14. |
[16] | Masoud S , Glassner AA , Mendoza M , Rhodes S , White CL ((2022) ) “A different way to survive": the experiences of family caregivers of persons living with dementia during the COVID-19 pandemic. J Fam Nurs 28: , 243–257. |
[17] | Tam MT , Dosso JA , Robillard JM ((2021) ) The impact of a global pandemic on people living with dementia and their care partners: analysis of 417 lived experience reports. J Alzheimers Dis 80: , 865–875. |
[18] | Aledeh M , Habib Adam P ((2020) ) Caring for dementia caregivers in times of the COVID-19 crisis: a systematic review. Am J Nurs Res 8: , 552–561. |
[19] | Innes A , Morgan D , Kosteniuk J ((2011) ) Dementia care in rural and remote settings: a systematic review of informal/family caregiving. Maturitas 68: , 34–46. |
[20] | Whitlatch CJ , Orsulic-Jeras S ((2018) ) Meeting the informational, educational, and psychosocial support needs of persons living with dementia and their family caregivers. Gerontologist 58: , 58–73. |
[21] | National Health Service (2020) Counselling, https://www.nhs.uk/conditions/counselling/, Last updated December 8, 2020, Accessed November 8, 2022. |
[22] | Waller A , Dilworth S , Mansfield E , Sanson-Fisher R ((2017) ) Computer and telephone delivered interventions to support caregivers of people with dementia: a systematic review of research output and quality. BMC Geriatr 17: , 265. |
[23] | Reese RJ , Conoley , Collie W. , Brossart DF ((2006) ) The attractiveness of telephone counseling: an empirical investigation of client perceptions. J Couns Dev 84: , 54–60. |
[24] | Lins S , Hayder-Beichel D , Rücker G , Motschall E , Antes G , Meyer G , Langer G (2014) Efficacy and experiences of telephone counselling for informal carers of people with dementia. Cochrane Database Syst Rev, CD009126. |
[25] | Lai FH-Y , Yan EW-H , Yu KK-Y , Tsui W-S , Chan DT-H , Yee BK ((2020) ) The protective impact of telemedicine on persons with dementia and their caregivers during the COVID-19 pandemic. Am J Geriatr Psychiatry 28: , 1175–1184. |
[26] | Tarter RR , Tran L , Lindauer A ((2020) ) The state of the science on synchronous video-conferencing interventions for care partners of persons with dementia. Alzheimers Dement 16: , e046173. |
[27] | González-Fraile E , Ballesteros J , Rueda J-R , Santos-Zorrozúa B , Solà I , McCleery J (2021) Remotely delivered information, training and support for informal caregivers of people with dementia. Cochrane Database Syst Rev, CD006440. |
[28] | Saragih ID , Tonapa SI , Porta CM , Lee B-O ((2022) ) Effects of telehealth intervention for people with dementia and their carers: A systematic review and meta-analysis of randomized controlled studies. J Nurs Scholarsh 54: , 704–719. |
[29] | Higgins JPT , Thomas J , Higgins J , Chandler J , Cumpston M , Li T , Page MJ , Welch VA , eds. ((2019) ) Cochrane Handbook for Systematic Reviews of Interventions, 2nd Edition, John Wiley & Sons, Chichester (UK). |
[30] | Hirt J , Langer G , Wilde F , Bauernschmidt D , Meyer G , Bieber A ((2021) ) Technology-based counselling in dementia (TeCoDem): study protocol of a mixed-methods systematic review with qualitative comparative analysis and meta-analysis. BMJ Open 11: , e054157. |
[31] | Cooper C , Booth A , Varley-Campbell J , Britten N , Garside R ((2018) ) Defining the process to literature searching in systematic reviews: a literature review of guidance and supporting studies. BMC Med Res Methodol 18: , 85. |
[32] | Lefebvre C , Glanville J , Briscoe S , Littlewood A , Marshall C , Metzendorf M-I , Noel-Storr A , Rader T , Shokraneh F , Thomas J , Wieland LS ((2019) ) Searching for and selecting studies. In Cochrane Handbook for Systematic Reviews of Interventions, Higgins JPT, Thomas J, Higgins J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, eds., 2nd Edition, John Wiley & Sons: Chichester (UK), pp. 67–108. |
[33] | McGowan J , Sampson M , Salzwedel DM , Cogo E , Foerster V , Lefebvre C ((2016) ) PRESS peer review of electronic search strategies: 2015 guideline statement. J Clin Epidemiol 75: , 40–46. |
[34] | Ouzzani M , Hammady H , Fedorowicz Z , Elmagarmid A ((2016) ) Rayyan-a web and mobile app for systematic reviews. Syst Rev 5: , 210. |
[35] | Hoffmann TC , Glasziou P , Boutron I , Milne R , Perera R , Moher D , Altman DG , Barbour V , MacDonald H , Johnston M , Lamb SE , Dixon-Woods M , McCulloch P , Wyatt JC , Chan A-W , Michie S ((2014) ) Better reporting of interventions: template for intervention description and replication (TIDieR) checklist and guide. BMJ 348: , g1687. |
[36] | Möhler R , Köpke S , Meyer G ((2015) ) Criteria for Reporting the Development and Evaluation of Complex Interventions in healthcare: revised guideline (CReDECI 2). Trials 16: , 204. |
[37] | Sterne JAC , Savović J , Page MJ , Elbers RG , Blencowe NS , Boutron I , Cates CJ , Cheng H-Y , Corbett MS , Eldridge SM , Emberson JR , Hernán MA , Hopewell S , Hróbjartsson A , Junqueira DR , Jüni P , Kirkham JJ , Lasserson T , Li T , McAleenan A , Reeves BC , Shepperd S , Shrier I , Stewart LA , Tilling K , White IR , Whiting PF , Higgins JPT ((2019) ) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366: , l4898. |
[38] | Current Version of RoB 2: Excel tool to implement RoB 2 (2019), https://www.riskofbias.info/welcome/rob-2-0-tool/current-version-of-rob-2, Accessed November 8, 2022. |
[39] | robvis (visualization tool), https://www.riskofbias.info/welcome/robvis-visualization-tool, Accessed November 8, 2022. |
[40] | The Nordic Cochrane Centre, The Cochrane Collaboration (2014) Review Manager (RevMan): Version 5.3, Copenhagen. |
[41] | Comprehensive Meta-Analysis (CMA) Version 3.3, https://www.meta-analysis.com/, Accessed November 19, 2021. |
[42] | Faraone SV ((2008) ) Interpreting estimates of treatment effects: implications for managed care. P T 33: , 700–711. |
[43] | Deeks JJ , Higgins JP , Altman DG ((2019) ) Chapter 10: Analysing data and undertaking meta-analysis. In Cochrane Handbook for Systematic Reviews of Interventions, Higgins JPT, Thomas J, Higgins J, Chandler J, Cumpston M, Li T, Page MJ,Welch VA, eds., 2nd Edition, JohnWiley & Sons: Chichester (UK), pp. 241–284. |
[44] | McKenzie JE , Brennan SE ((2019) ) Chapter 12: Synthesizing and presenting findings using other methods. In Cochrane Handbook for Systematic Reviews of Interventions, Higgins JPT, Thomas J, Higgins J, Chandler J, Cumpston M, Li T, Page MJ,Welch VA, eds., 2nd Edition, JohnWiley & Sons: Chichester (UK), pp. 321–348. |
[45] | Schünemann H , Brozek J , Guyatt G , Oxman A , eds. (2013) GRADE Handbook: Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. |
[46] | Guyatt GH , Thorlund K , Oxman AD , Walter SD , Patrick D , Furukawa TA , Johnston BC , Karanicolas P , Akl EA , Vist G , Kunz R , Brozek J , Kupper LL , Martin SL , Meerpohl JJ , Alonso-Coello P , Christensen R , Schunemann HJ ((2013) ) GRADE guidelines: 13. Preparing summary of findings tables and evidence profiles-continuous outcomes. J Clin Epidemiol 66: , 173–183. |
[47] | GRADEpro Guideline Development Tool (GDT) (2022), https://www.gradepro.org/, Accessed July 4, 2022. |
[48] | Langer G , Meerpohl JJ , Perleth M , Gartlehner G , Kaminski-Hartenthaler A , Schünemann H ((2012) ) GRADE-Leitlinien: 2. Formulierung der Fragestellung und Entscheidung über wichtige Endpunkte. Z Evid Fortbild Qual Gesundhwes 106: , 369–376. |
[49] | Tremont G , Davis JD , Bishop DS , Fortinsky RH ((2008) ) Telephone-delivered psychosocial intervention reduces burden in dementia caregivers. Dementia 7: , 503–520. |
[50] | Hodgson NA , Petrovsky DV , Finegan K , Kallmyer BA , Pike J , Fazio S ((2021) ) One call makes a difference: An evaluation of the Alzheimer’s Association National Helpline on dementia caregiver outcomes. Patient Educ Couns 104: , 896–902. |
[51] | Coyne AC , Potenza M , Broken Nose MA ((1995) ) Caregiving and dementia: The impact of telephone helpline services. Am J Alzheimers Dis Other Demen 10: , 27–32. |
[52] | Tremont G , Davis JD , Papandonatos GD , Ott BR , Fortinsky RH , Gozalo P , Yue MS , Bryant K , Grover C , Bishop DS ((2015) ) Psychosocial telephone intervention for dementia caregivers: A randomized, controlled trial. Alzheimers Dement 11: , 541–548. |
[53] | Laver K , Liu E , Clemson L , Davies O , Gray L , Gitlin LN , Crotty M ((2020) ) Does telehealth delivery of a dyadic dementia care program provide a noninferior alternative to face-to-face delivery of the same program? A randomized, controlled trial. Am J Geriatr Psychiatry 28: , 673–682. |
[54] | Tremont G , Davis JD , Ott BR , Galioto R , Crook C , Papandonatos GD , Fortinsky RH , Gozalo P , Bishop DS ((2017) ) Randomized trial of the family intervention: Telephone Tracking-Caregiver for dementia caregivers: Use of community and healthcare resources. J Am Geriatr Soc 65: , 924–930. |
[55] | Braly T , Muriathiri D , Brown JC , Taylor BM , Boustani MA , Holden RJ ((2021) ) Technology intervention to support caregiving for Alzheimer’s disease (I-CARE): study protocol for a randomized controlled pilot trial. Pilot Feasibility Stud 7: , 23. |
[56] | Ahmed M , Marín M , Bouca-Machado R , How D , Judica E , Tropea P , Bentlage E , Brach M ((2021) ) Investigating users’ and other stakeholders’ needs in the development of a personalized integrated care platform (PROCare4Life) for older people with dementia or parkinson disease: protocol for a mixed methods study. JMIR Res Protoc 10: , e22463. |
[57] | Kabir ZN , Leung AYM , Grundberg Å , Boström A-M , Lämås K , Kallström AP , Moberg C , Cronfalk BS , Meijer S , Konradsen H ((2020) ) Care of family caregivers of persons with dementia (CaFCa) through a tailor-made mobile app: study protocol of a complex intervention study. BMC Geriatr 20: , 305. |
[58] | National Institutes of Health (2015) Tools for distance delivery of an evidence-based AD family caregiver intervention. NCT02347202, https://clinicaltrials.gov/ct2/show/NCT02347202, Accessed November 25, 2021. |
[59] | Gaugler JE , Statz TL , Birkeland RW , Louwagie KW , Peterson CM , Zmora R , Emery A , McCarron HR , Hepburn K , Whitlatch CJ , Mittelman MS , Roth DL ((2020) ) The Residential Care Transition Module: a single-blinded randomized controlled evaluation of a telehealth support intervention for family caregivers of persons with dementia living in residential long-term care. BMC Geriatr 20: , 133. |
[60] | Tremont G , Davis JD , Bryant K , Ott BR , Papandonatos G , Fortinsky R , Gozalo P , Bishop D ((2014) ) Effect of a telephone-based dementia caregiver intervention on use of community support services and health care resources. Alzheimers Dement 10: , 226–227. |
[61] | Tremont G , Davis JD , Grover C , Bryant K , Ott B , Papandonatos G , Fortinsky R , Gozalo P , Bishop D ((2013) ) Randomized controlled trial of a telephone-delivered intervention (FITT-Caregiver) for dementia caregivers. Alzheimers Dement 9: , P324–P325. |
[62] | (2008) Telephone support for dementia caregivers. NCT00735800, https://clinicaltrials.gov/ct2/show/NCT00735800, Accessed November 8, 2022. |
[63] | Tremont G , Davis JD , Papandonatos GD , Grover C , Ott BR , Fortinsky RH , Gozalo P , Bishop DS ((2013) ) A telephone intervention for dementia caregivers: Background, design, and baseline characteristics. Contemp Clin Trials 36: , 338–347. |
[64] | Tremont G , Davis JD , O’Connor K , Grover C , Bishop D , Ott B , Papadonatos G , Fortinsky R ((2011) ) Relationship between expectancy/credibility and early response to telephone-based dementia caregiver interventions. Alzheimers Dement 7: , S435. |
[65] | (2017) Does telehealth delivery of the COPE program provide a non-inferior alternative to face-to-face treatment for community dwelling people with cognitive impairment? ACTRN12617000117314, https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=372140&isReview=true, Accessed November 8, 2022. |
[66] | Williamson PR , Altman DG , Bagley H , Barnes KL , Blazeby JM , Brookes ST , Clarke M , Gargon E , Gorst S , Harman N , Kirkham JJ , McNair A , Prinsen CAC , Schmitt J , Terwee CB , Young B ((2017) ) The COMET Handbook: version 1.0. Trials 18: , 280. |
[67] | Skivington K , Matthews L , Simpson SA , Craig P , Baird J , Blazeby JM , Boyd KA , Craig N , French DP , McIntosh E , Petticrew M , Rycroft-Malone J , White M , Moore L ((2021) ) A new framework for developing and evaluating complex interventions: update of Medical Research Council guidance. BMJ 374: , n2061. |
[68] | Cuffaro L , Di Lorenzo F , Bonavita S , Tedeschi G , Leocani L , Lavorgna L ((2020) ) Dementia care and COVID-19 pandemic: a necessary digital revolution. Neurol Sci 41: , 1977–1979. |




