The Clinical Use of Alzheimer’s Disease Biomarkers in Patients with Mild Cognitive Impairment: A European Alzheimer’s Disease Consortium Survey
Abstract
Background:
Recent advances occurred in the field of Alzheimer’s disease (AD) biomarkers and the introduction of a research framework grounded on a biomarker-based definition of AD might have fostered an increased clinical use of AD biomarkers. For this reason, an up-to-date depiction of the clinical use of AD biomarkers is needed.
Objective:
To investigate the clinical use of the main AD biomarkers in patients with mild cognitive impairment (MCI) by examining the beliefs and preferences of professionals (clinicians and biomarker experts) of the European Alzheimer’s Disease Consortium (EADC).
Methods:
150 professionals filled in an online survey from May to September 2020. The investigated biomarkers were medial temporal lobe atrophy score (MTA) on structural MRI, typical AD (i.e., temporoparietal and posterior cingulate) hypometabolism on FDG-PET, CSF (Aβ42, p-tau, t-tau), amyloid-PET and tau-PET.
Results:
The frequency of responders reporting a frequent-to-constant use of MTA (77%) is higher than that of those reporting a frequent-to-constant use of the other AD biomarkers (i.e., CSF: 45%, p = 0.014; FDG-PET: 32%, p < 0.001; amyloid-PET: 8%, p < 0.001; and tau-PET: 2%, p < 0.001). CSF is considered the most valuable biomarker in terms of additional diagnostic value, followed by amyloid-PET, tau-PET, and typical AD hypometabolism on FDG-PET.
Conclusion:
AD biomarkers are widely used across European memory clinics with a clinical research background for the diagnosis of MCI. Overall, we observed that CSF is currently considered as the most useful biomarker, followed by amyloid-PET.
INTRODUCTION
The clinical understanding of Alzheimer’s disease (AD) and its diagnostic implications have radically changed over time. Currently, AD is defined as a biological disease characterized by the abnormal accumulation of amyloid plaques and neurofibrillary tangles in the brain [1, 2], and is clinically considered a continuum ranging from a long preclinical phase (abnormal biomarkers in asymptomatic individuals) to mild cognitive impairment (MCI) and finally dementia. Even if this mainly represents a research framework, it points to the clinical relevance of AD biomarkers, and supports their increasing use in clinical practice. Consistently, several studies demonstrated that the use of biomarkers has different clinical implications [3–6] with MCI patients being the ones who might benefit the most from biomarker testing [7]. Indeed, an early etiological diagnosis of AD in MCI patients should initiate specific counseling [8] and allow interventions to potentially delay the progression to dementia [9]. Moreover, in light of the recent conditional approval of aducanumab by the FDA [10], an early and biomarker-supported etiological diagnosis of AD might drastically influence patient management.
For an etiological diagnosis of early symptomatic patients, this concept has already been picked up by most academic memory clinics and is implemented in clinical practice. However, to date, no clinical guidelines recommend a “biomarker-based diagnosis of AD” as the state-of-the-art for the etiological diagnosis of MCI (or even dementia) patients.
The main biomarkers able to capture AD-related changes can be classified into two categories: 1) diagnostic/pathophysiological biomarkers are measures of amyloid (i.e., positive amyloid-PET, low cerebrospinal fluid (CSF) Aβ42) and tau (i.e., positive tau-PET, high CSF p-tau) deposition, and have the necessary specificity for a diagnosis of AD [11]; 2) progression/topographical biomarkers include measures of neurodegeneration (e.g., medio-temporal lobe atrophy (MTA) on structural MRI, hypometabolism on FDG-PET, high CSF t-tau), are not specific to AD but can be used to track the disease progression [11]. Among all these biomarkers, a major progress in recent years is the advent of amyloid-PET and tau-PET: these novel molecular imaging techniques make possible to visualize the topography of amyloid and tau deposits in the brain, previously measurable only indirectly via CSF. Specifically, given the strong association between tau pathology and symptoms, already known from neuropathological studies, and given the relative specificity of tau-PET tracers for tau deposits in AD, tau-PET could represent the most efficient imaging modality in AD: it has been shown that a positive tau-PET with Flortaucipir (a first-generation tau-PET tracer) might be an indicator of both amyloid and tau pathology and thus provide a definite diagnostic answer with only one test [12, 13], even though this might not be the case of second-generation tau-PET tracers (for which pathological confirmation has not been provided yet). However, this interpretation might depend on the relative role attributed by the physician to amyloid and tau pathology in the physiopathology of AD [14].
Recent evidence on the use of the main AD biomarkers is needed to 1) understand if and to what extent they are used in academic clinical settings, 2) evaluate the quality of the diagnostic procedure in European countries, 3) assess if their use is consistent with the latest AD diagnostic criteria, and 4) evaluate the memory clinics preparedness for disease-modifying therapies. A previous European survey conducted in 2012 investigated the use of biomarkers in MCI patients and showed that physicians felt comfortable delivering a diagnosis of MCI due to AD when both amyloid and neurodegeneration biomarkers were abnormal [15]. The most frequently used biomarkers were found to be MTA (75% of responders reported to use it at least frequently), Aβ42, p-tau, t-tau levels in CSF (22%), FDG-PET (16%), and amyloid-PET (3%). In light of the notable advances recently occurring in the field of AD biomarkers, namely clinical studies on real-word patients and introduction of newly developed techniques, an up-to-date depiction of their clinical use is needed.
Thus, the aim of this study is to investigate the clinical use of the main AD biomarkers in MCI patients by examining the beliefs and preferences of clinicians (i.e., neurologists, geriatricians, psychiatrists) and biomarker experts (i.e., nuclear medicine, radiology, and laboratory physicians) within the framework of the European Alzheimer’s Disease Consortium (EADC), a functional network comprising European memory clinics of excellence with a clinical research background working in the field of AD.
METHODS
The survey
A survey consisting of an online questionnaire assessing the clinical use of biomarkers in MCI patients (see the Supplementary Material for the full questionnaire) was made accessible from May 14 to September 30, 2020. The number of items/questions depended on the respondent specialty and answers: 26 for clinicians (i.e. neurologists, geriatricians, psychiatrists) and 14 for biomarker experts (i.e. nuclear medicine, radiology, and laboratory physicians); the difference being due to 12 questions specific to the treating clinicians.
The questionnaire was subdivided in ten sections assessing respectively: (i) respondent’s details, i.e., specialty, center, city, country, role in memory clinic, years of experience in the field of neurodegenerative disorders, competence to answer to questions regarding neurodegenerative disorders; (ii) for clinicians only, the request to provide names and contacts of nuclear medicine, radiology, and laboratory physicians associated with the respondent’s memory clinic; (iii) beliefs about the pathogenic role of amyloid and tau in AD; (iv) respondent’s clinical work, i.e., consultation with MCI, number of new MCI patients per month; (v) frequency of use of biomarkers (MRI, FDG-PET, CSF, amyloid-PET, tau-PET); (vi) use of imaging biomarkers in clinical reports (vii); use of biochemical biomarkers in clinical reports; (viii) diagnostic additional value of biomarkers; (ix) clinical vignette where respondents were asked to rate the diagnostic confidence on the basis of abnormality of the biomarkers (alone or combined); and (x) perceived clinical utility of amyloid-PET and tau-PET.
Concerning tau-PET, the following disclaimer was added to the survey: “Please note that some of the following questions concern tau-PET. We are aware that while amyloid-PET tracers are well established and show similar performances, tau-PET tracers have been more recently developed and are less established, e.g., Flortaucipir is the most used tracer and has been validated against neuropathology and second-generation tracers are promising for an increased diagnostic accuracy. For this reason, we chose not to specify further which tau-PET tracer, and we would ask you to consider in answering a “theoretical” tau-PET tracer with a diagnostic accuracy deemed adequate for clinical use in AD.”
The survey took place in two steps: first, we contacted the clinicians working in the EADC memory clinics asking them to fill in the online questionnaire and to provide names and contacts of nuclear medicine, radiology, and laboratory physicians associated with their memory clinics; second, we contacted the nuclear medicine, radiology, and laboratory physicians recommended by the clinicians during the first step.
Outcomes of interest
In the present study, we assessed the 1) responders’ belief about the pathogenic role of amyloid and tau in AD pathology and symptoms, 2) frequency of use of AD biomarkers in MCI patients, 3) use of AD biomarkers to support etiological diagnosis in MCI, 4) additional value of FDG-PET, CSF, amyloid-PET and tau-PET over neuropsychological testing and structural MRI in an MCI patient, 5) responders’ confidence in making a diagnosis of MCI due to AD in a typical MCI patient based on different AD biomarkers, and 6) responders’ opinion on the perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia patients.
Belief on the pathogenic role of amyloid and tau in AD
The following question was posed to all responders: “What is your belief/opinion about the pathogenic role of amyloid and tau in Alzheimer’s disease pathology and symptoms?”. Possible answers went from 0 to 10 (11-point Likert scale): 0, the abnormal accumulation of amyloid is the initial cause of AD; 5, amyloid and tau have the same relevance in causing AD, or neither amyloid nor tau are the initial cause of AD; 10, the abnormal accumulation of tau is the initial cause of AD. Answers were grouped into three categories: 0–4, favoring amyloid; 5, neutral; 6–10, favoring tau.
Frequency of use of AD biomarkers in MCI patients
The following question was posed: “In MCI, in your clinical practice, please state frequency of use for medial temporal lobe atrophy (MRI), FDG-PET, CSF (e.g., A β42, p-tau, t-tau), amyloid-PET, tau-PET”. Possible answers were: not used (0%), rarely (<10%), regularly (20–60%), frequently (60–80%), always (>80%). Answers were grouped into three categories: no use, rare-to-regular use (rarely or regularly), frequent-to-constant use (frequently or always). This item was applicable only to clinicians. Since the frequency of use of biomarkers is strongly dependent on the center expertise, policies, guidelines, and facilities, we included only the answers of the head of the memory clinic or, if not possible, the answers of the most experienced (years of expertise in the field of neurodegenerative disorders) clinician of that memory clinic.
Use of AD biomarkers to support etiological diagnosis in MCI
The following question was posed: “Do you use imaging biomarkers / CSF collection (e.g., A β42, p-tau, t-tau) / APOE genotyping to support your etiological diagnosis in MCI?”. Possible answers were yes or no. Concerning imaging biomarkers, an additional question was posed: “Do you use any quantitative reading tool (e.g., SPM) or scale (e.g., MTA scale; Scheltens et al., 1992) for your clinical reports?”. Possible answers were yes or no. In case of affirmative answer, a further question was posed: “In MCI please state what kind of quantitative reading tool (e.g., SPM) or scale (e.g., MTA scale; Scheltens et al., 1992) you use for your clinical reports for the answers indicated in previous question”. These items were applicable only to clinicians.
Additional value over neuropsychological testing and structural MRI in MCI
The following question was posed to all responders: “Assuming that clinical examination with neuropsychological testing and brain structural MRI are the most feasible procedures in most memory clinics, please rate the additional diagnostic value (i.e., the ability to provide diagnostic information in excess of that already provided by neuropsychological testing and brain structural MRI) in an MCI patient of FDG-PET, CSF markers (e.g., A β42, p-tau, t-tau), amyloid-PET, tau-PET”. Possible answers were: none, little, moderately significant, greatly significant, decisive. Answers were grouped into three categories: none-to-little (none or little), moderate (moderately significant), great-to-decisive (greatly significant or decisive).
Confidence in an etiological diagnosis of AD in MCI
The following case vignette was proposed to all responders: “A 75 years old person comes into your office complaining of memory deterioration in the past 6–12 months, he/she is in good physical health, has no problems in his/her daily chores, but is clearly worried. Routine labs are normal, but he/she performs 1.5 SD below the age-and education-adjusted mean on a test of verbal or non-verbal recall. How confident would you be with a diagnosis of MCI due to AD (or prodromal AD) on the basis of i) evidence of clear-cut medial temporal lobe atrophy alone, ii) clear-cut temporoparietal and posterior cingulate hypometabolism on FDG-PET alone, iii) clearly abnormal CSF levels of A β and tau alone, iv) clearly positive amyloid-PET, v) clearly positive tau-PET, vi) at least one clearly positive amyloid marker and at least one clearly positive neuronal injury marker.”. Possible answers were: not at all comfortable, moderately comfortable, comfortable, very comfortable, extremely comfortable. Answers were grouped into three categories: not comfortable, sufficiently comfortable (moderately comfortable or comfortable), very-to-extremely comfortable (very comfortable or extremely comfortable).
Perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia
The following question was posed to all responders: “Independent of any specific patient’s feature and based on your clinical experience with patients usually seen in your memory clinic, what is, in your opinion, the most clinically useful exam for etiological diagnosis of MCI and mild dementia?”. Possible answers went from 0 to 10 (11-point Likert scale): 0, amyloid-PET is in general the most useful exam; 5, amyloid-PET and tau-PET are equally useful, or neither amyloid-PET nor tau-PET are the most useful exam; 10, tau-PET is in general the most useful exam. Answers were grouped into three categories: 0–4, favoring amyloid-PET; 5, neutral; 6–10, favoring tau-PET.
Statistical methods
The outcomes of interest were assessed using proportion test (χ2). Significance was set at p < 0.05 and post-hoc pairwise comparisons were adjusted using Bonferroni correction.
All statistical analyses were performed with R, version 3.4.2 (R Foundation for statistical computing, https://www.r-project.org/).
RESULTS
Responders’ features
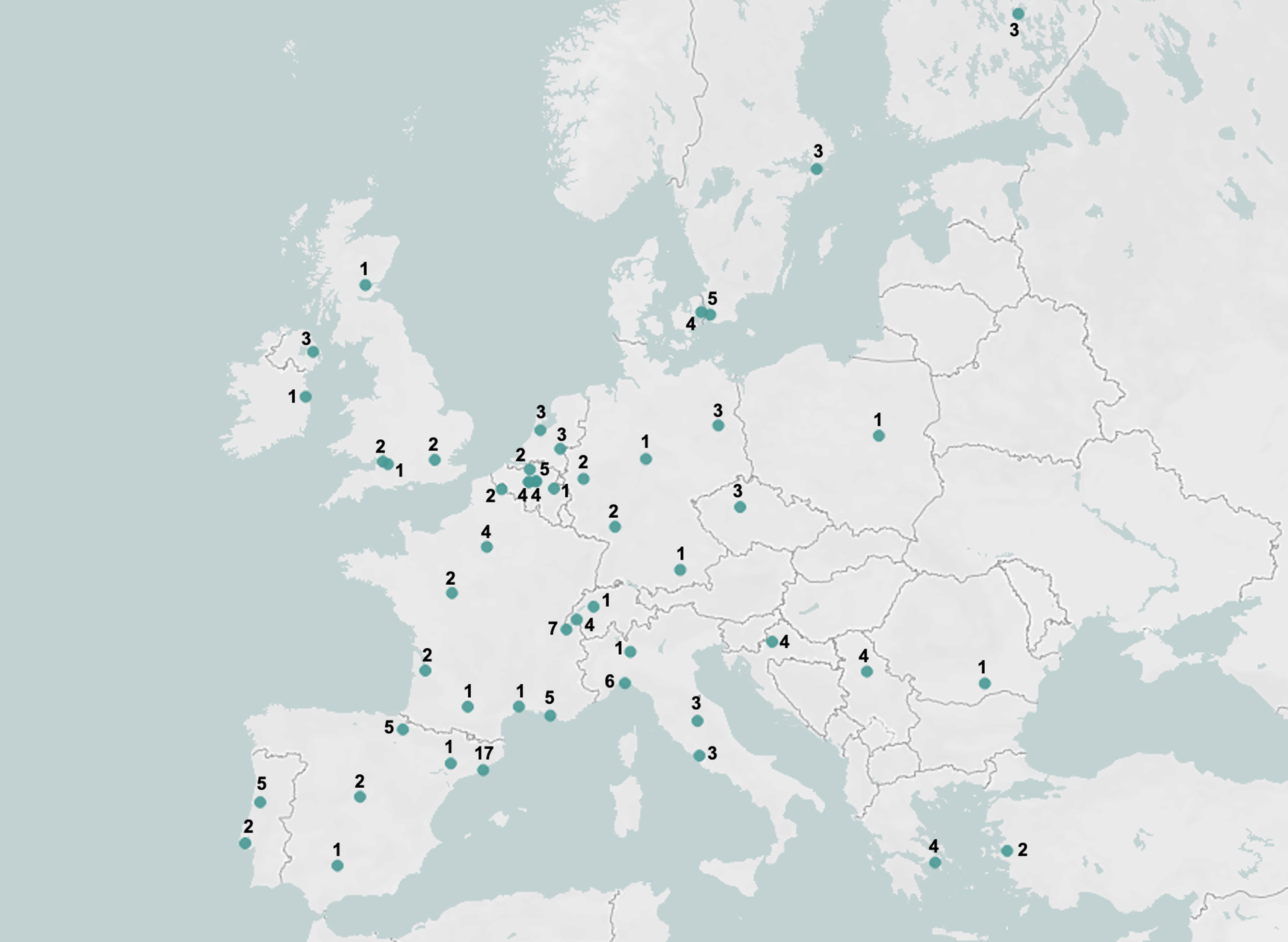
First, 168 clinicians working in the EADC memory clinics were contacted, and 51% of them (86/168) filled in the survey questionnaire recommending 138 additional professionals (20 clinicians, 42 biomarker experts, 1 neuroscientist, and 1 psychologist) related to their memory clinics, 46% (64/138) of whom filled in the survey. In total, 150 professionals filled in the survey questionnaire, with an overall answer rate of 49% (150/306). Among them, 6 did not fall into the pre-defined categories of interest (i.e., clinicians and biomarker experts), being research staff (n = 4) or general practitioner (n = 2), and were therefore excluded from the analyses. Among the remaining responders, 5% (7/144) declared that they were not competent enough in the field of neurodegenerative disorders to fulfill the questionnaire, and therefore did not proceed with the questionnaire. The final sample consisted of 100 clinicians and 37 biomarker experts (9 radiologists, 18 nuclear medicine physicians, and 10 laboratory physicians). Among clinicians, 45% (45/100) were head of their memory clinics, while the remaining ones were memory clinic staff or collaborators. Clinicians had more years of experience in the field of neurodegenerative disorder than biomarker experts (clinicians: 19±9, biomarker experts: 13±8; p < 0.001). On average, clinicians consulted 17±13 new patients with MCI in a typical month. Detailed respondent’s characteristics are reported in Table 1, while Fig. 1 shows the geographic distribution of responders. The list of responding centers and number of responders per center is reported in the Supplementary Material.
Table 1
Respondent’s characteristics
| Respondent’s characteristics | Clinicians n = 100 | Biomarker experts n = 37 |
| Specialty | Neurologist 72% | Radiologist 24% |
| Geriatrician 14% | Nuclear medicine physicians 49% | |
| Psychiatrist 18% | Laboratory physicians 27% | |
| Role | Head of memory clinic 45% | Head of memory clinic 0% |
| Staff 39% | Staff 0% | |
| Collaborator 7% | Collaborator 92% | |
| Other 9% | Other 8% | |
| Years of experience in the field of neurodegenerative disorders | 19±9 | 13±8 |
| Providing clinical consultation for MCI | 92% | NA |
| New MCI patients consulted in a typical month | 17±13 | NA |
Staff: professionals which are actually hired and employed by the memory clinics involved in the survey. Collaborator: professionals employed elsewhere (e.g., other units or facilities) which support and collaborate with the memory clinic involved in the survey. Other: professional connected to the memory clinic not falling in the other categories (e.g., scientific director, head of clinical research, research Principal Investigator).
Fig. 1
Geographical distribution of the responders and number of responders per center.
Pathogenic role of amyloid and tau in AD
Figure 2A shows the beliefs on the pathogenic role of amyloid and tau in AD reported by clinicians and biomarker experts, while Supplementary Figure 1A shows the beliefs on the pathogenic role of amyloid and tau in AD reported by different biomarker experts groups, i.e., radiologists, nuclear medicine physicians, and laboratory physicians. Clinicians deemed the abnormal accumulation of amyloid (answers from 0 to 4, 42% of cases) rather than tau (answers from 6 to 10, 20% of cases, p = 0.001; Fig. 2A) as the initial cause of AD. A similar, although not significant possibly due to the smaller sample size, pattern was observed among biomarker experts (amyloid: 43%, tau: 27%, p = 0.223; Fig. 2A). When comparing the proportion of clinicians and biomarker experts favoring amyloid, no significant difference emerges. The same result was observed for tau.
Fig. 2
Beliefs on the pathogenic role of amyloid and tau in AD [A] and perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia [B]. (A) The question asked to responders was: “What is your belief/opinion about the pathogenic role of amyloid and tau in Alzheimer’s disease pathology and symptoms?”. Answers were grouped into three categories: 0–4, favoring the abnormal accumulation of amyloid as the initial cause of AD; 5, amyloid and tau have the same relevance in causing AD, or neither amyloid nor tau are the initial cause of AD; 6–10, favoring the abnormal accumulation of tau as the initial cause of AD. (B) The question posed to responders was: “Independent of any specific patient’s feature and based on your clinical experience with patients usually seen in your memory clinic, what is, in your opinion, the most clinically useful exam for etiological diagnosis of MCI and mild dementia?”. Answers were grouped into three categories: 0–4, favoring amyloid-PET; 5, neutral; 6–10, favoring tau-PET.
![Beliefs on the pathogenic role of amyloid and tau in AD [A] and perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia [B]. (A) The question asked to responders was: “What is your belief/opinion about the pathogenic role of amyloid and tau in Alzheimer’s disease pathology and symptoms?”. Answers were grouped into three categories: 0–4, favoring the abnormal accumulation of amyloid as the initial cause of AD; 5, amyloid and tau have the same relevance in causing AD, or neither amyloid nor tau are the initial cause of AD; 6–10, favoring the abnormal accumulation of tau as the initial cause of AD. (B) The question posed to responders was: “Independent of any specific patient’s feature and based on your clinical experience with patients usually seen in your memory clinic, what is, in your opinion, the most clinically useful exam for etiological diagnosis of MCI and mild dementia?”. Answers were grouped into three categories: 0–4, favoring amyloid-PET; 5, neutral; 6–10, favoring tau-PET.](https://ip.ios.semcs.net:443/media/jad/2022/89-2/jad-89-2-jad220333/jad-89-jad220333-g002.jpg)
Frequency of use of AD biomarkers in MCI patients
Figure 3 shows the detailed frequency of use of each biomarker. Among clinicians, 8% (8/100) did not provide clinical consultation for patients with MCI and were therefore excluded from the analyses on the frequency of use of AD biomarkers in MCI. Moreover, due to the above-explained reasons, we included only the answers of the head of the memory clinic or, if not possible, the answers of the most experienced (as measured by years of expertise in the field of neurodegenerative disorders) clinician of that memory clinic.
Fig. 3
Frequency of use of AD biomarkers in MCI patients. The question asked to clinicians was: “In MCI, in your clinical practice, please state frequency of use for medial temporal lobe atrophy (MRI), FDG-PET, CSF (e.g., A β42, p-tau, t-tau), amyloid-PET, tau-PET”. Possible answers were: not used (0%), rarely (<10%), regularly (20–60%), frequently (60-80%), always (>80%). Answers were grouped into three categories: no use, rare-to-regular use (rarely or regularly), frequent-to-constant use (frequently or always).
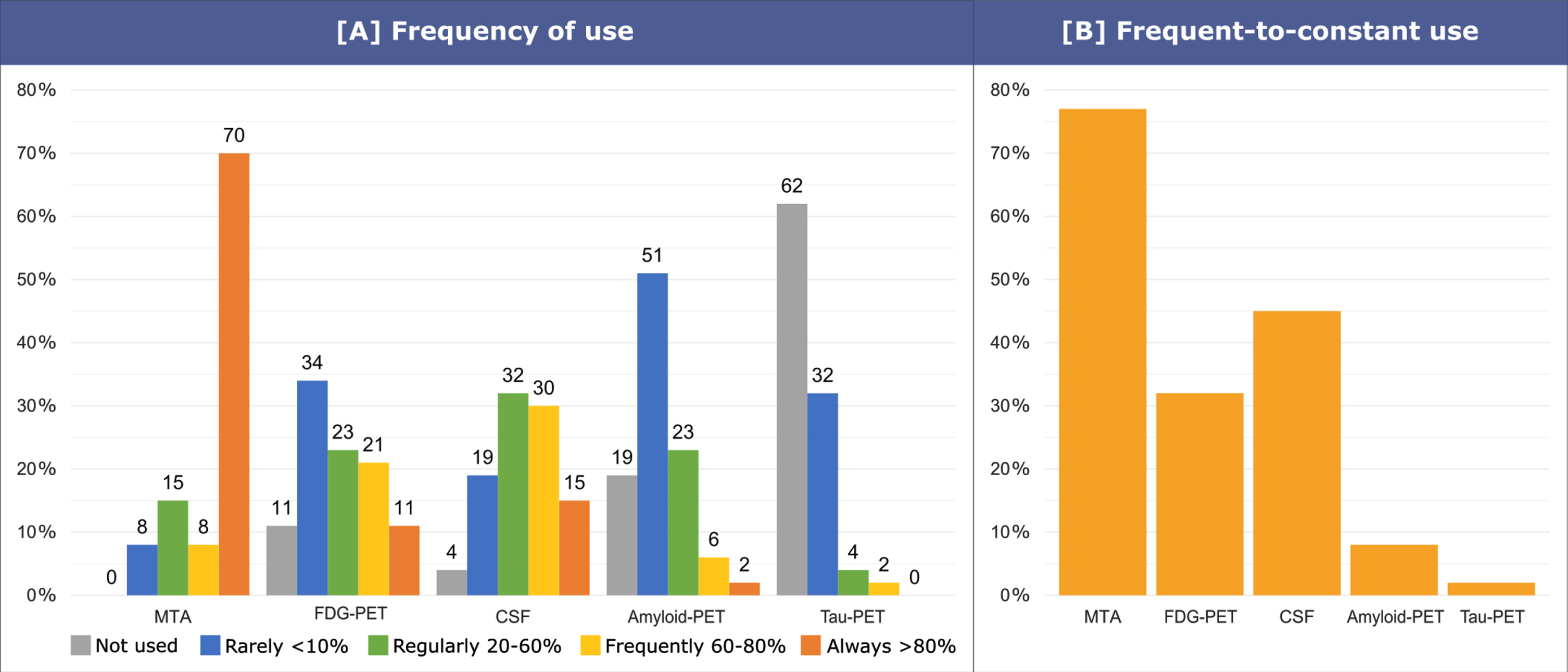
The frequency of clinicians reporting a frequent-to-constant use of MTA (77%, 41/53) is higher than that of those reporting a frequent-to-constant use of CSF (45%, 24/53; p = 0.014), FDG-PET (32%, 17/53, p < 0.001), amyloid-PET (8%, 4/53; p < 0.001), and tau-PET (2%, 1/53; p < 0.001). Moreover, the frequency of clinicians reporting a frequent-to-constant use of CSF and FDG-PET is higher than that of those reporting a frequent-to-constant use of amyloid-PET (p < 0.001 and p = 0.035) and tau-PET (p < 0.001 and p = 0.001).
Use of AD biomarkers to support etiological diagnosis in MCI
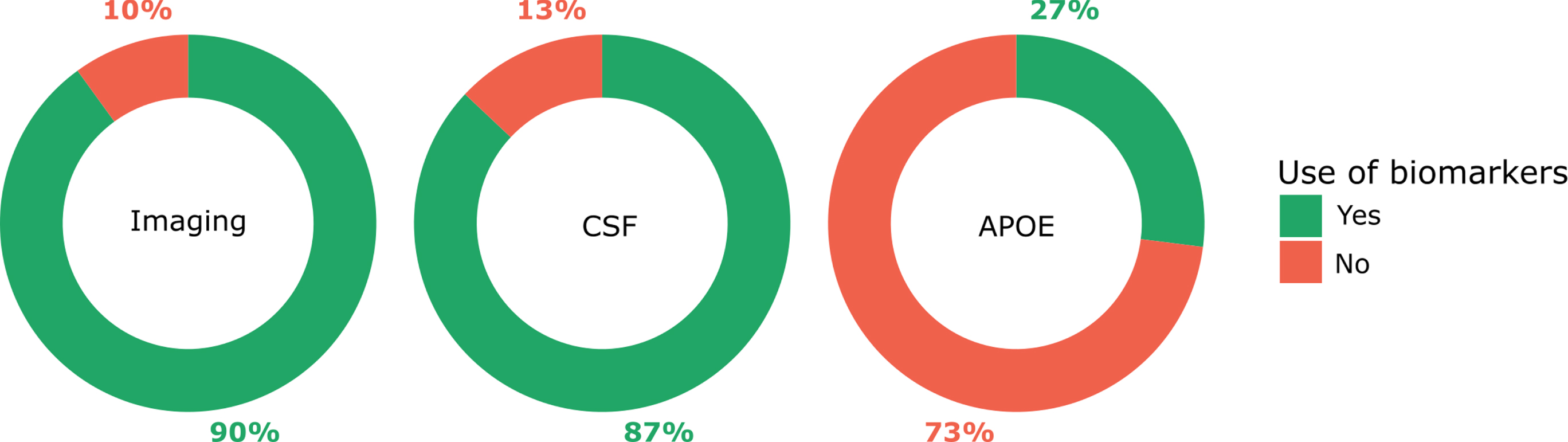
Figure 4 shows the use of AD biomarkers to support etiological diagnosis in MCI. Among clinicians, 8% (8/100) did not provide clinical consultation for patients with MCI and were therefore excluded from the analyses on use of AD biomarkers to support etiological diagnosis in MCI. Clinicians providing clinical consultation for patients with MCI used imaging (90%, 83/92) and CSF (87%, 80/92) biomarkers to support etiological diagnosis in MCI more frequently than APOE genotyping (27%, 25/92; p < 0.001).
Fig. 4
Use of AD biomarkers to support etiological diagnosis in MCI. The question asked to clinicians was: “Do you use imaging biomarkers / CSF collection (e.g., A β42, p-tau, t-tau) / APOE genotyping to support your etiological diagnosis in MCI?”. Possible answers were yes or no. Clinicians reported to use imaging to support their etiological diagnosis in MCI in 90% of cases, CSF in 87% of cases and APOE in 27% of cases.
As for imaging biomarker, 67% of clinicians (62/92) used quantitative reading tools and scales for reporting imaging biomarkers data in clinical reports. Specific results on quantitative tools or scales are presented in Supplementary Table 1.
Additional value over neuropsychological testing and structural MRI in MCI
Figure 5 shows the detailed reported additional value over neuropsychological testing and structural MRI in MCI, while Supplementary Figure 2 shows the detailed reported additional value over neuropsychological testing and structural MRI in MCI by different biomarker experts groups, i.e., radiologists, nuclear medicine physicians, and laboratory physicians. Clinicians report more frequently a great-to-decisive additional value of Aβ42, p-tau, and t-tau levels in CSF (85%, 85/100) with respect to amyloid-PET (72%, 72/100; p < 0.05), tau-PET (54%, 54/100; p < 0.001) and FDG-PET (35%, 35/100; p < 0.001). Amyloid-PET, instead, has been reported as having a great-to-decisive additional value more frequently than FDG-PET only (p < 0.001). No significant differences emerged between amyloid-PET and tau-PET (p = 0.077), and between FDG-PET and tau-PET (p = 0.063).
Fig. 5
Additional value over neuropsychological testing and structural MRI in MCI. The question asked to responders was: “Assuming that clinical examination with neuropsychological testing and brain structural MRI are the most feasible procedures in most memory clinics, please rate the additional diagnostic value (i.e., the ability to provide diagnostic information in excess of that already provided by neuropsychological testing and brain structural MRI) in an MCI patient of FDG-PET, CSF markers (e.g., Aβ42, p-tau, t-tau), amyloid-PET, tau-PET”. Possible answers were: none, little, moderately significant, greatly significant, decisive. Answers were grouped into three categories: none-to-little (none or little), moderate (moderately significant), great-to-decisive (greatly significant or decisive).
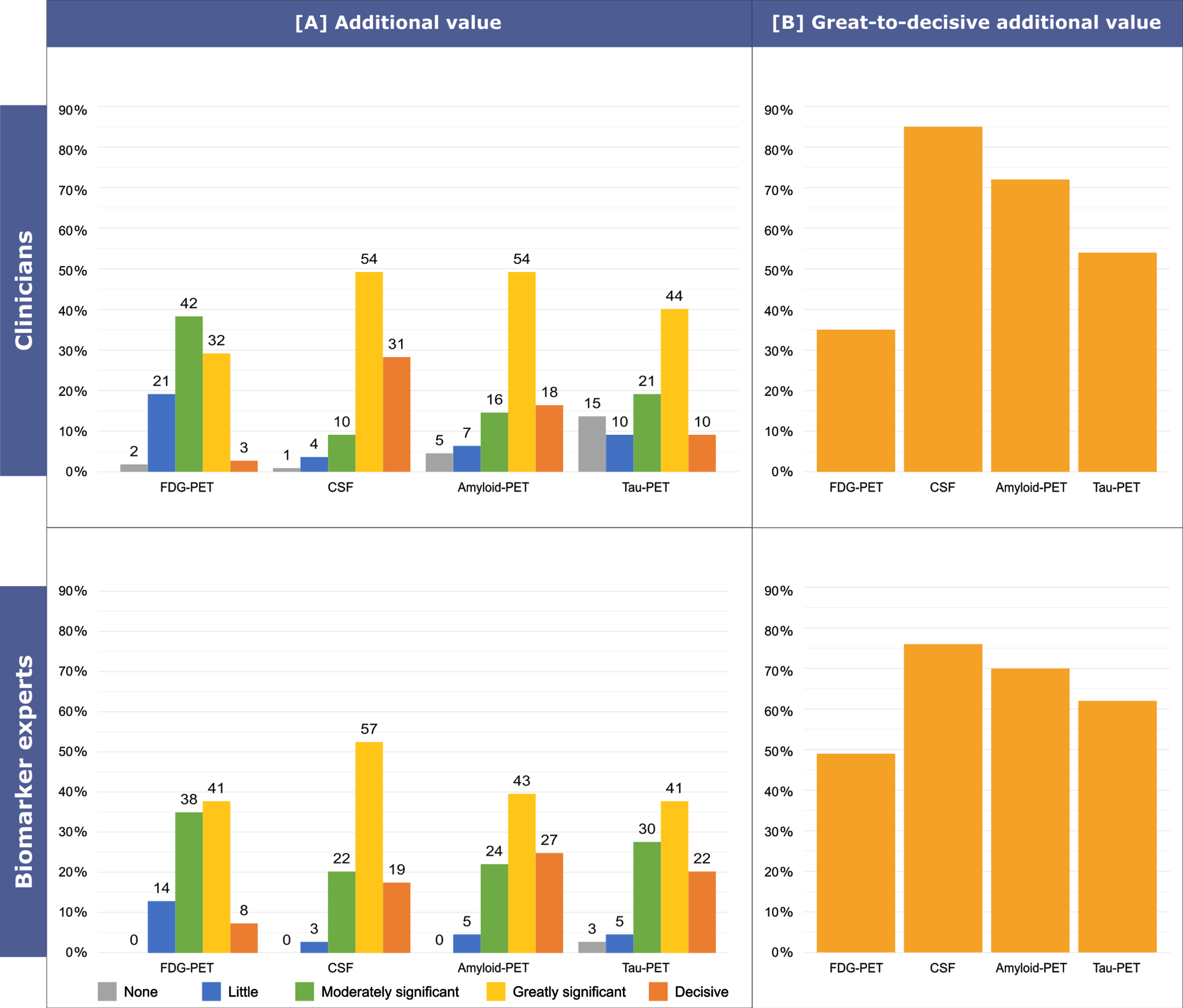
Among biomarker experts, we observed no significant differences (p = 0.083), with the frequency of a great-to-decisive additional value being 76% (28/37) for Aβ42, p-tau, and t-tau levels in CSF, 70% (26/37) for amyloid-PET, 62% (23/37) for tau-PET, and 49% (18/37) for FDG-PET.
No significant differences were observed when comparing the frequency of clinicians and biomarker experts reporting a great-to-decisive additional value for each technique.
Confidence in an etiological diagnosis of AD in MCI
Figure 6 shows the detailed level of confidence in an etiological diagnosis of AD in MCI, while Supplementary Figure 3 shows the detailed level of confidence in an etiological diagnosis of AD in MCI by different biomarker experts groups, i.e., radiologists, nuclear medicine physicians, and laboratory physicians. Clinicians reported to be more frequently very-to-extremely comfortable with an etiological diagnosis of AD on the basis of at least one clearly positive amyloid marker and at least one clearly positive neuronal injury marker (86%, 86/100) with respect to abnormal CSF levels of Aβ42, p-tau, and t-tau (64%, 64/100; p = 0.009), positive amyloid-PET (52%, 52/100; p < 0.001), positive tau-PET (41%, 41/100; p < 0.001), typical AD (i.e., temporoparietal and posterior cingulate) hypometabolism on FDG-PET (22% 22/100; p < 0.001), and evidence of MTA on structural MRI (14%, 14/100; p < 0.001). Moreover, clinicians reported to be more frequently very-to-extremely comfortable with an etiological diagnosis of AD on the basis of abnormal CSF levels of Aβ and tau or of a positive amyloid-PET than typical AD hypometabolism on FDG-PET (p < 0.001 and p < 0.001, respectively) and evidence of MTA (p < 0.001 and p < 0.001, respectively). Additionally, they reported to be more frequently very-to-extremely comfortable with an etiological diagnosis of AD on the basis of abnormal CSF levels of Aβ and tau rather than of positive tau-PET (p = 0.028). Finally, clinicians reported to be more frequently very-to-extremely comfortable with an etiological diagnosis of AD on the basis of positive tau-PET than evidence of MTA (p < 0.001).
Fig. 6
Confidence in an etiological diagnosis of AD in MCI. The following case vignette was proposed to responders: “A 75 years old person comes into your office complaining of memory deterioration in the past 6–12 months, he/she is in good physical health, has no problems in his/her daily chores, but is clearly worried. Routine labs are normal, but he/she performs 1.5 SD below the age-and education-adjusted mean on a test of verbal or non-verbal recall. How confident would you be with a diagnosis of MCI due to AD (or prodromal AD) on the basis of i) evidence of clear-cut medial temporal lobe atrophy alone, ii) clear-cut temporoparietal and posterior cingulate hypometabolism on FDG-PET alone, iii) clearly abnormal CSF levels of Aβ and tau alone, iv) clearly positive amyloid-PET, v) clearly positive tau-PET, vi) at least one clearly positive amyloid marker and at least one clearly positive neuronal injury marker.”. Possible answers were: not at all comfortable, moderately comfortable, comfortable, very comfortable, extremely comfortable. Answers were grouped into three categories: not comfortable, sufficiently comfortable (moderately comfortable or comfortable), very-to-extremely comfortable (very comfortable or extremely comfortable).
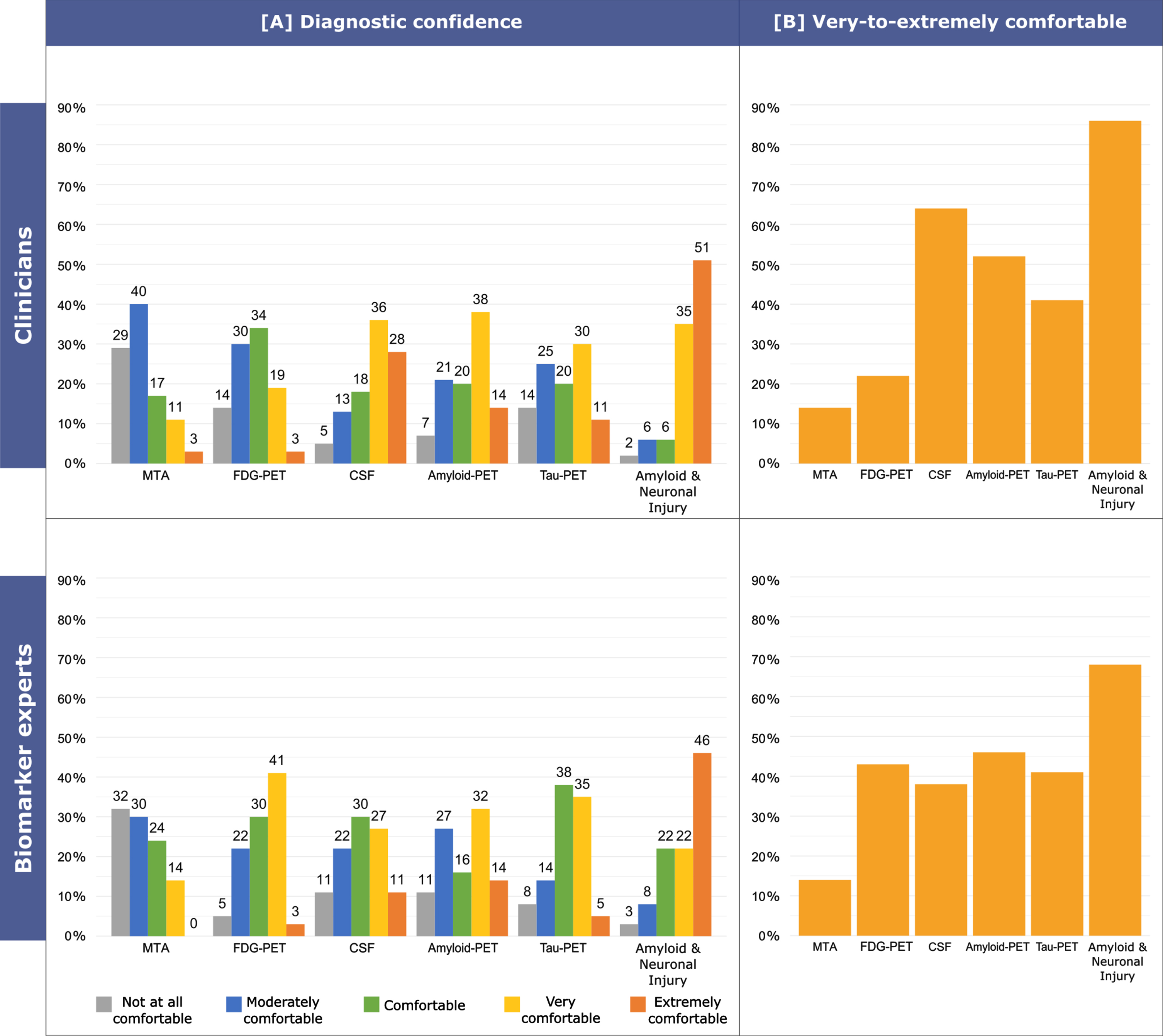
Biomarker experts stated to be more frequently very-to-extremely comfortable with an etiological diagnosis of AD on the basis of at least one clearly positive amyloid marker and at least one clearly positive neuronal injury marker (68%, 25/37) rather than on the evidence of MTA (14%, 5/37; p < 0.001). Among biomarker experts we observed no other significant differences, possibly due to the smaller sample size, with the frequency of those stating to be very-to-extremely comfortable with an etiological diagnosis of AD being 46% (17/37) for positive amyloid-PET, 43% (16/37) for typical AD hypometabolism on FDG-PET, 41% (15/37) for positive tau-PET and 38% (14/37) for abnormal CSF levels of Aβ and tau.
No significant differences were observed when comparing the frequency of clinicians and biomarker experts reporting to be very-to-extremely comfortable with an etiological diagnosis of AD on the basis of MTA, amyloid-PET, and tau-PET. Biomarker experts reported to be very-to-extremely comfortable with an etiological diagnosis of AD on the basis of typical AD hypometabolism on FDG-PET more frequently than clinicians (p = 0.024). Conversely, clinicians reported to be very-to-extremely comfortable with an etiological diagnosis of AD on the basis abnormal CSF levels (p = 0.010), or of at least one clearly positive amyloid marker and at least one clearly positive neuronal injury marker (p = 0.028) more frequently than biomarker experts.
Perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia
Figure 2B shows the perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia reported by clinicians and biomarker experts, while Supplementary Figure 1B shows the perceived clinical utility of amyloid-PET versus tau-PET in MCI and mild dementia reported by different biomarker experts groups, i.e., radiologists, nuclear medicine physicians, and laboratory physicians. Clinicians considered amyloid-PET and tau-PET equally useful to support an etiological diagnosis in MCI and mild dementia patients (amyloid-PET: 35% versus tau-PET: 24%, p = 0.121; Fig. 2B). A non-significant trend towards an amyloid-PET preference was observed among biomarker experts (amyloid-PET: 46% versus tau-PET: 24%, p = 0.088). When comparing the proportion of clinicians and biomarker experts favoring amyloid-PET or tau-PET, no significant difference emerged.
DISCUSSION
This study reports the results of the largest multidisciplinary survey on the use and the perceived utility of AD biomarkers in clinical practice in MCI patients. The investigation on the clinicians and biomarker experts’ beliefs on the initial cause of AD revealed that a prevalent pathogenic role was attributed to amyloid rather than tau. However, when the focus switched from the theoretical belief to the perceived clinical utility of PET scans assessing amyloid and tau, responders did not show a clear preference for amyloid-PET. In the clinical assessment of MCI patients, the most widely used biomarker is MTA on structural MRI, followed by CSF (i.e., levels of Aβ42, p-tau, t-tau), typical AD hypometabolism on FDG-PET, amyloid-PET, and lastly tau-PET. Both clinicians and biomarker experts deemed CSF (i.e., levels of Aβ42, p-tau, t-tau) as the most valuable biomarker in terms of additional diagnostic value over neuropsychological testing and structural MRI, followed by amyloid-PET, tau-PET, and lastly typical AD hypometabolism on FDG-PET. Finally, concerning diagnostic confidence in an etiological diagnosis of AD in MCI, both clinician and biomarker experts quite predictably deemed the combination of amyloidosis and neuronal injury biomarkers as the most convincing in vivo signature of AD, while MTA alone was perceived as the less reliable biomarker.
In MCI patients, only 19% have no neurodegenerative pathology, while 51% of MCI cases are in the AD continuum (i.e., evidence of amyloid pathology), and 30% have non-AD pathology [16]. Positive AD biomarkers support an early diagnosis of AD (diagnostic value) [17], and allow to predict cognitive decline (prognostic value) in MCI patients [18]. An early and biomarker-supported diagnosis of AD might have several implications: for example, it can be the gateway to disease-modifying therapies (also playing a pivotal role in their efficacy).
Consistent with the publication of the latest diagnostic criteria of AD where biomarkers play a key role, our results showed an overall increase in the use of all biomarkers, as compared with a previous survey launched in 2012 [15], and with another one launched in 2014 among members of European Academy of Neurology and EADC [19]. In a period of less than 10 years, amyloid-PET went from being frequently or constantly used in 3% of cases to 8% (and from being used at least regularly in 16% of cases to 31%), thus highlighting a remarkable spread of this biomarker in the clinical practice. Moreover, tau-PET, which has been recently introduced [20] and was not even included in the previous survey, now appears to be used frequently or constantly in 2% of cases and at least regularly in 6% by the responders in their clinical practice. The use of these advanced techniques might be limited by the absence of disease modifying therapies, thus their use will probably increase as new effective therapies will become widely available. It must be remarked that the frequency of use of each biomarker is clearly influenced by its local availability, which varies significantly from center to center with MRI being already widely available and tau-PET being available only in research contexts, despite the promising preliminary results exhibited since its recent introduction [4, 21]. Of note, the limited frequency of use detected for amyloid-PET and tau-PET in clinical practice might be influenced by the lack of reimbursement for these examinations. As for CSF markers, their frequency of use might be further influenced not only by their availability but also by the attitude toward lumbar puncture among clinicians and patients. Performing lumbar puncture, indeed, requires specific training and evaluation of potential contraindications; however, the overall risk of complications is relatively low [22].
Our results showed a slight decrease in the use of APOE genotyping to support etiological diagnosis (27% in our study versus 38% in 2012) which seems at odds with its non-invasivity and affordability and with the crucial information that this exam might reveal [23]. Indeed, even though the estimated risk conferred by an APOE ɛ4 allele varies between studies, odd ratios ranging between 1.8 and 9.9 have been reported [24]. Moreover, it has been recently shown that APOE ɛ4 carriers have an increased rate of progression from preclinical AD to MCI, and from MCI to mild AD dementia [25]. APOE ɛ4 allele, however, is neither necessary nor sufficient to cause AD. Thus, APOE genotyping should not be used alone for a diagnosis of MCI due to AD in a single case [26]. Indeed, current guidelines do not do not recommend APOE genotyping in the workup of dementia and MCI either in isolation or as part of the diagnostic process [27, 28].
Notably, we observed that the perceived diagnostic value of CSF and amyloid-PET dramatically increased from previous survey (85% and 72% in this study versus 60% and 43% in 2012), while that of FDG-PET decreased (35% in this study vs 46% in 2012). The remarkable level of perceived utility attributed to CSF (i.e., levels of Aβ42, p-tau, t-tau) can be easily explained in light of the different pathophysiological process which is able to capture (i.e., amyloid and tau deposition, and neurodegeneration). Consistently, Falgas and colleagues have shown that both CSF and amyloid-PET increase diagnostic confidence in AD and non-neurodegenerative conditions, leading to diagnostic and treatment changes [29]. A recent survey further confirmed the value of amyloid pathology markers, highlighting that these biomarkers are perceived by clinicians as the most valuable to predict progression and rate of progression in MCI patients [8]. However, Ramusino and colleagues recently compared the relative incremental diagnostic value of amyloid-PET and CSF (i.e., levels of Aβ42, p-tau, t-tau), showing that amyloid-PET induces greater changes in the diagnosis of AD patients as compared to CSF [3].
Consistently with our results indicating a similar preference for amyloid-PET and tau-PET, a recent work assessing the diagnostic value of amyloid-PET and tau-PET in a memory clinic population showed that both exams significantly impacted diagnosis and diagnostic confidence in a similar way [4]. The relatively low additional value attributed to FDG-PET might be due its nature of nonspecific measure of neurodegeneration, detecting damage that may derive not only from AD but from a variety of etiologies, for example cerebrovascular injury [2].
Limitations
The here-reported results allow an up-to-date depiction of the clinical use of AD biomarkers in patients with MCI in EADC memory clinics and of their perceived utility among a multidisciplinary group of experts in the field of neurodegenerative disorders. Nevertheless, this study has some limitations. Firstly, the participation of EADC centers was only partial (51% response rate). Secondly, the nature of the responding centers, i.e., memory clinics with a clinical research background, might prevent to generalize our results to ordinary memory clinics. Thirdly, since tau-PET tracers have been developed only recently and are less established, we had to ask responders to consider a “theoretical” tau-PET tracer with a diagnostic accuracy deemed adequate for clinical use in AD, maybe affecting their answers concerning this technique. Fourthly, plasma biomarkers were not included in the present survey. However, since they are emerging as potentially scalable and valuable biomarkers [30], their inclusion in future survey is needed. Lastly, the results of the survey might be influenced by the local indications for prescription, reimbursement policies, and costs of biomarkers, which are not always consistent across Europe [7]. Indeed, the same biomarker might be indicated without restrictions and reimbursed in some countries (e.g., CSF in France), and indicated only in specific cases (e.g., CSF in UK) or not reimbursed (e.g., CSF in Spain) in others [7]. As a consequence, in some cases, wealthy patients might pay out of pocket to have access to advanced diagnostic exams that would otherwise be inaccessible. In other words, the European heterogeneity in indications for prescription and reimbursement policies and other financial aspects might have influenced the results of the survey.
Conclusions
Altogether, the results of this ample and multidisciplinary survey suggest that AD biomarkers are widely adopted in clinical practice in MCI patients across European memory clinics with a clinical research background. Overall, we observed that CSF is currently considered as the most useful biomarker by clinicians and biomarker experts, followed by amyloid-PET.
ACKNOWLEDGMENTS
The Geneva Memory Center is funded by the following private donors under the supervision of the Private Foundation of Geneva University Hospitals: A.P.R.A. – Association Suisse pour la Recherche sur la Maladie d’Alzheimer, Genève; Fondation Segré, Genève; Ivan Pictet, Genève; Fondazione Agusta, Lugano; Fondation Chmielewski, Genève. Competitive research projects have been funded by: H2020, Human Brain Project, Innovative Medicines Initiative (IMI), IMI2, Swiss National Science Foundation, VELUX Foundation.
Valentina Garibotto received funding by the Swiss National Science Foundation (grant numbers: 320030_169876, 320030_185028 and IZSEZ0_188355) and the VELUX Foundation (grant number 1123).
Giovanni B. Frisoni received funding by: the EU-EFPIA Innovative Medicines Initiatives 2 Joint Undertaking (IMI 2 JU) European Prevention of Alzheimer’s Dementia consortium (EPAD, grant agreement number: 115736) and Amyloid Imaging to Prevent Alzheimer’s Disease (AMYPAD, grant agreement number: 115952); the Swiss National Science Foundation: Brain connectivity and metacognition in persons with subjective cognitive decline (COSCODE): correlation with clinical features and in vivo neuropathology (grant number: 320030_182772); A.P.R.A. – Association Suisse pour la Recherche sur la Maladie d’Alzheimer, Genève; Fondation Segré, Genève; Ivan Pictet, Genève; Fondazione Agusta, Lugano; Fondation Chmielewski, Genève; VELUX Foundation.
Daniele Altomare received funding by the Fondation Recherche Alzheimer and the Swiss National Science Foundation (project CRSK-3_196354 / 1).
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/22-0333r2).
The authors would like to thank the responders for taking part to this survey: Carla Abdelnour (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Emel Ada (Dokuz Eylul University, Izmir, Turkey), Eduardo Agüera Morales (Hospital Universitario Reina Sofía, Cordoba, Spain), Daniel Alcolea (Hôpital Sant Pau, Barcelona, Spain), Gilles Allali (Geneva University Hospital, Geneva, Switzerland), Gert-Jan Allemeersch (Radiology Department, Universitair Ziekenhuis Brussel, Brussel, Belgium), Javier Arbizu (Clinica Universidad de Navarra, Pamplona, Spain), Frederic Assal (Geneva University Hospital, Geneva, Switzerland), Josep Augé (Hospital Clinic IDIBAPS, Barcelona, Spain), Sophie Auriacombe (Centre Hospitalier Universitaire (CHU) Pellegrin, Bordeaux, France), Mircea Balasa (Hospital Clinic IDIBAPS, Barcelona, Spain), Inês Baldeiras (University Hospital of Coimbra, Coimbra, Portugal), Nuria Bargallo (Hospital Clinic IDIBAPS, Barcelona, Spain), Frederik Barkhof (Vrije University Medical Centre, Amsterdam, The Netherlands), Matteo Bauckneht (University of Genoa, Genoa, Italy), Jean-Louis Bayart (Université catholique de Louvain & Cliniques Universitaires Saint-Luc, Louvain, Belgium), Aurélie Bedel (CHU Pellegrin, Bordeaux, France), Lisa Bjartmar (Skåne University Hospital & Lund University, Malmö, Sweden), Marina Boban (University Hospital Center Zagreb & University of Zagreb School of Medicine, Zagreb, Croatia), Anne Börjesson Hanson (Karolinska Institutet, Stockholm, Sweden), Fran Borovecki (University Hospital Center Zagreb & University of Zagreb School of Medicine, Zagreb, Croatia), José Boucraut (CHU Timone, Marseille, France), Lela Brajkovic (Institute of Neurobiology, Belgrade, Serbia), Valle Camacho (Hôpital Sant Pau, Barcelona, Spain), Vincent Camus (Clinique Universitaire CHRU & Université François Rabelais, Tours, France), Mathieu Ceccaldi (CHU Timone, Marseille, France), Joaquim Cerejeira (University Hospital of Coimbra, Coimbra, Portugal), Jiri Cerman (Charles University & Motol University Hospital, Prague, Czech Republic), Pietro Chiarini (Geriatric Department, University of Perugia, Perugia, Italy), Jurgen Claassen (Radboud University Medical Center, Nijmegen, The Netherlands), Peter Connelly (National Health Service, Perth, UK), Liz Coulthard (Bristol Medical School, University of Bristol, Bristol, UK), Peter De Deyn (University of Antwerp, Antwerp, Belgium), Alexandre de Mendonça (Faculdade de Medicina de Lisboa, Lisbon, Portugal), Philippe Demaerel (University Hospital Gasthuisberg, Leuven, Belgium), Jean-François Démonet (Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland), Thomas Desmidt (Clinique Universitaire CHRU & Université François Rabelais, Tours, France), Daniela Di Giuda (Università Cattolica del Sacro Cuore, Roma, Italy), Mira Didic (CHU Timone, Marseille, France), Massimo Eugenio Dottorini (Geriatric Department, University of Perugia, Perugia, Italy), Alexander Drzezga (Uniklinik, Cologne, Germany), Sebastiaan Engelborghs (Vrije Universiteit Brussel, Brussel, Belgium), Stephane Epelbaum (Hôpital Salpétrière, Paris, France), Mohamed Eshmawey (Geneva University Hospital, Geneva, Switzerland), Olivier Félician (CHU Timone, Marseille, France), Marta Fernandez-Mattarubia (Clinica Universidad de Navarra, Pamplona, Spain), Kristian Steen Frederiksen (Rigshospitalet, University of Copenhagen, Copenhagen, Denmark), Giovanni Battista Frisoni (Geneva University Hospital, Geneva, Switzerland), Lutz Frölich (Zentralinstitut für Seelische Gesundheit, Mannheim, Germany), Domenico Fusco (Università Cattolica del Sacro Cuore, Roma, Italy), Tomasz Gabryelewicz (Polish Academy of Sciences, Medical Research Center, Warsaw, Poland), Guillermo García-Ribas (Hospital Universitario Ramón y Cajal, Madrid, Spain), Valentina Garibotto (Geneva University Hospital, Geneva, Switzerland), Silvia Gil Navarro (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Timo Grimmer (Technische Universität, Munich, Germany), Eric Guedj (CHU Timone, Marseille, France), Marie-Odile Habert (Hôpital Salpétrière, Paris, France), Douglas Hägerström (Skåne University Hospital & Lund University, Malmö, Sweden), Sara Hall (Skåne University Hospital & Lund University, Malmö, Sweden), Merja Hallikainen (Kuopio University Hospital, Kuopio, Finland), Bernard Hanseeuw (Université catholique de Louvain (UCL) & Cliniques Universitaires Saint-Luc, Louvain, Belgium), Oskar Hansson (Skåne University Hospital & Lund University, Malmö, Sweden), Steen Hasselbalch (Rigshospitalet, University of Copenhagen, Copenhagen, Denmark), Lucrezia Hausner (Zentralinstitut für Seelische Gesundheit, Mannheim, Germany), Isabel Hernández-Ruiz (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Isabella Heuser (Charitá – Universitätsmedizin, Berlin, Germany), Jakub Hort (Charles University & Motol University Hospital, Prague, Czech Republic), Jacques Hugon (University of Paris Diderot, Paris, France), Adrian Ivanoiu (Université catholique de Louvain (UCL) & Cliniques Universitaires Saint-Luc, Louvain, Belgium), Alice Jaillard (CHU Inserm, Lille, France), Frank Jessen (Uniklinik, Cologne, Germany), Roy Jones (RICE (The Research Institute for the Care of Older People), Bath, UK), Marleen Keyaerts (Vrije Universiteit Brussel, Brussel, Belgium), Anne Maria Koivisto (Kuopio University Hospital, Kuopio, Finland), Asunción Lafuente (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Catharina Lange (Charité – Universitätsmedizin, Berlin, Germany), Vibeke Andree Larsen (Rigshospitalet, University of Copenhagen, Copenhagen, Denmark), Ian Law (Rigshospitalet, University of Copenhagen, Copenhagen, Denmark), Brian Lawlor (Mercer’s Institute for Research on Ageing, St. James’ Hospital, Dublin, Ireland), Thibaud Lebouvier (CHU Inserm, Lille, France), Ondrej Lerch (Charles University & Motol University Hospital, Prague, Czech Republic), Marcus Likeman (Bristol Medical School, University of Bristol, Bristol, UK), Rosa Liperoti (Università Cattolica del Sacro Cuore, Roma, Italy), Albert Lladó (Hospital Clinic IDIBAPS, Barcelona, Spain), Alberto Lleó (Hôpital Sant Pau, Barcelona, Spain), Rogelio López-Cuevas (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Finn Lornsen (University of Bern, Bern, Switzerland), Ivanka Markovic (Institute of Neurobiology, Belgrade, Serbia), Marta Marquié (Fundació ACE Institut Català de Neurociències Aplicades – Universitat Internacional de Catalunya (UIC), Barcelona, Spain), Federico Massa (University of Genoa, Genoa, Italy), Bernadette McGuinness (Centre for Public Health, Belfast, UK), Patrizia Mecocci (Geriatric Department, University of Perugia, Perugia, Italy), Aline Mendes (Geneva University Hospital, Geneva, Switzerland), Marie Meyer (Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland), Lefkos Middleton (Imperial College, London, UK), Angela Milan (Clinica Universidad de Navarra, Pamplona, Spain), José Luis Molinuevo (Hospital Clinic IDIBAPS, Barcelona, Spain), Ricardo Morais (University Hospital of Coimbra, Coimbra, Portugal), Silvia Morbelli (University of Genoa, Genoa, Italy), Karen Mullin (Centre for Public Health, Belfast, UK), Sophie Navucet (Montpellier University Hospital, Montpellier, France), Ivan Nikolic (Institute of Neurobiology, Belgrade, Serbia), Flavio Nobili (University of Genoa, Genoa, Italy), Marcel Olde-Rikkert (Radboud University Medical Center, Nijmegen, The Netherlands), Pierre-Jean Ousset (CHU La Grave-Casselardit, Toulouse, France), David Ozretic (University Hospital Center Zagreb & University of Zagreb School of Medicine, Zagreb, Croatia), George P. Paraskevas (2nd Department of Neurology, National and Kapodistrian University of Athens, “Attikon” General University Hospital, Athens, Greece), Matteo Pardini (University of Genoa, Genoa, Italy), Patricia Paredes (Hospital Universitario Ramón y Cajal, Madrid, Spain), Peter Passmore (Centre for Public Health, Belfast, UK), Andres Perissinotti (Hospital Clinic IDIBAPS, Barcelona, Spain), Samuel Perivier (Geneva University Hospital, Geneva, Switzerland), Oliver Peters (Charité – Universitätsmedizin, Berlin, Germany), Fabrizio Piazza (School of Medicine, University of Milano-Bicocca, Milan, Italy), Gerard Piñol Ripoll (Hospital Universitario Santa Maria, Lleida, Spain), Koen Poesen (University Hospital Gasthuisberg, Leuven, Belgium), John Prior (Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland), Mario Riverol (Clinica Universidad de Navarra, Pamplona, Spain), Luca Roccatagliata (University of Genoa, Genoa, Italy), Olivier Rouaud (Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland), Minna Rusanen (Kuopio University Hospital, Kuopio, Finland), Eric Salmon (University of Liège, Liège, Belgium), Raquel Sanchez-Valle (Hospital Clinic IDIBAPS, Barcelona, Spain), Isabel Santana (University Hospital of Coimbra, Coimbra, Portugal), Nikos Scarmeas (National and Kapodistrian University of Athens, Medical School, Aiginition Hospital, Athens, Greece), Philip Scheltens (Vrije University Medical Centre, Amsterdam, The Netherlands), Jonathan Schott (University College London, London, UK), Luiza Spiru (Elias University Hospital, Bucharest, Romania), Elka Stefanova (Institute of Neurobiology, Belgrade, Serbia), Erik Stomrud (Skåne University Hospital & Lund University, Malmö, Sweden), Víctor Suárez (Clinica Universidad de Navarra, Pamplona, Spain), Miguel Tábuas-Pereira (University Hospital of Coimbra, Coimbra, Portugal), Charlotte Teunissen (Vrije University Medical Centre, Amsterdam, The Netherlands), Mireia Tondo Colomer (Hôpital Sant Pau, Barcelona, Spain), Koen Van Laere (University Hospital Gasthuisberg, Leuven, Belgium), Vincent Van Pesch (Université catholique de Louvain (UCL) & Cliniques Universitaires Saint-Luc, Louvain, Belgium), Rik Vandenberghe (University Hospital Gasthuisberg, Leuven, Belgium), Mathieu Vandenbulcke (University Hospital Gasthuisberg, Leuven, Belgium), Tim Vanderhasselt (Vrije Universiteit Brussel, Brussel, Belgium), Georgios Velonakis (National and Kapodistrian University of Athens, Medical School, Aiginition Hospital, Athens, Greece), Marcel Verbeek (Radboud University Medical Center, Nijmegen, The Netherlands), Ana Verdelho (Faculdade de Medicina de Lisboa, Lisbon, Portugal), Yannick Vermeiren (University of Antwerp, Antwerp, Belgium), Nicolas Villain (Hôpital Salpétrière, Paris, France), Giorgos Vlachos (National and Kapodistrian University of Athens, Medical School, Aiginition Hospital, Athens, Greece), Zeljka Vogrinc (University Hospital Center Zagreb & University of Zagreb School of Medicine, Zagreb, Croatia), Anders Wimo (Karolinska Institutet, Stockholm, Sweden), Bengt Winblad (Karolinska Institutet, Stockholm, Sweden), Görsev Yener (Dokuz Eylul University, Izmir, Turkey), Inga Zerr (Clinical Dementia Center, Department of Neurology, University Medical Center, Georg August University, Göttingen, Germany).
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JAD-220333.
REFERENCES
[1] | Dubois B , Hampel H , Feldman HH , Scheltens P , Aisen P , Andrieu S , Bakardjian H , Benali H , Bertram L , Blennow K , Broich K , Cavedo E , Crutch S , Dartigues JF , Duyckaerts C , Epelbaum S , Frisoni GB , Gauthier S , Genthon R , Gouw AA , Habert MO , Holtzman DM , Kivipelto M , Lista S , Molinuevo JL , O’Bryant SE , Rabinovici GD , Rowe C , Salloway S , Schneider LS , Sperling R , Teichmann M , Carrillo MC , Cummings J , Jack CR ((2016) ) Preclinical Alzheimer’s disease: Definition, natural history, and diagnostic criteria. Alzheimers Dement 12: , 292–323. |
[2] | Jack CR , Bennett DA , Blennow K , Carrillo MC , Dunn B , Haeberlein SB , Holtzman DM , Jagust W , Jessen F , Karlawish J , Liu E , Molinuevo JL , Montine T , Phelps C , Rankin KP , Rowe CC , Scheltens P , Siemers E , Snyder HM , Sperling R , Elliott C , Masliah E , Ryan L , Silverberg N ((2018) ) NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement 14: , 535–562. |
[3] | Ramusino MC , Garibotto V , Bacchin R , Altomare D , Dodich A , Assal F , Mendes A , Costa A , Tinazzi M , Morbelli SD , Bauckneht M , Picco A , Dottorini ME , Tranfaglia C , Farotti L , Salvadori N , Moretti D , Savelli G , Tarallo A , Nobili F , Parapini M , Cavaliere C , Salvatore E , Salvatore M , Boccardi M , Frisoni GB ((2020) ) Incremental value of amyloid-PET versus CSF in the diagnosis of Alzheimer’s disease. Eur J Nucl Med Mol Imaging 47: , 270–280. |
[4] | Altomare D , Caprioglio C , Assal F , Allali G , Mendes A , Ribaldi F , Ceyzeriat K , Martins M , Tomczyk S , Stampacchia S , Dodich A ((2021) ) Diagnostic value of amyloid-PET and tau-PET: A head-to-head comparison. Eur J Nucl Med Mol Imaging 48: , 2200–2211. |
[5] | Barthel H , Sabri O ((2017) ) Clinical use and utility of amyloid imaging. J Nucl Med 58: , 1711–1717. |
[6] | Blennow K , Dubois B , Fagan AM , Lewczuk P , De Leon MJ , Hampel H ((2015) ) Clinical utility of cerebrospinal fluid biomarkers in the diagnosis of early Alzheimer’s disease. Alzheimers Dement 11: , 58–69. |
[7] | Frisoni GB , Boccardi M , Barkhof F , Blennow K , Cappa S , Chiotis K , Démonet JF , Garibotto V , Giannakopoulos P , Gietl A , Hansson O , Herholz K , Jack CR , Nobili F , Nordberg A , Snyder HM , Ten Kate M , Varrone A , Albanese E , Becker S , Bossuyt P , Carrillo MC , Cerami C , Dubois B , Gallo V , Giacobini E , Gold G , Hurst S , Lönneborg A , Lovblad KO , Mattsson N , Molinuevo JL , Monsch AU , Mosimann U , Padovani A , Picco A , Porteri C , Ratib O , Saint-Aubert L , Scerri C , Scheltens P , Schott JM , Sonni I , Teipel S , Vineis P , Visser PJ , Yasui Y , Winblad B ((2017) ) Strategic roadmap for an early diagnosis of Alzheimer’s disease based on biomarkers. Lancet Neurol 16: , 661–676. |
[8] | Frederiksen KS , Nielsen TR , Appollonio I , Andersen BB , Riverol M , Boada M , Ceccaldi M , Dubois B , Engelborghs S , Frölich L , Hausner L , Gabelle A , Gabryelewicz T , Grimmer T , Hanseeuw B , Hort J , Hugon J , Jelic V , Koivisto A , Kramberger MG , Lebouvier T , Lleó A , de Mendonça A , Nobili F , Ousset PJ , Perneczky R , Olde Rikkert M , Robinson D , Rouaud O , Sánchez E , Santana I , Scarmeas N , Sheardova K , Sloan S , Spiru L , Stefanova E , Traykov L , Yener G , Waldemar G ((2021) ) Biomarker counseling, disclosure of diagnosis and follow-up in patients with mild cognitive impairment: A European Alzheimer’s disease consortium survey. Int J Geriatr Psychiatry 36: , 324–333. |
[9] | Petersen RC , Lopez O , Armstrong MJ , Getchius TSD , Ganguli M , Gloss D , Gronseth GS , Marson D , Pringsheim T , Day GS , Sager M , Stevens J , Rae-Grant A ((2018) ) Practice guideline update summary: Mild cognitive impairment report of theguideline development, dissemination, and implementation. Neurology 90: , 126–135. |
[10] | Aducanumab (marketed as Aduhelm) Information, Last updated 2021, Accessed on 2022. |
[11] | Dubois B , Feldman HH , Jacova C , Hampel H , Molinuevo JL , Blennow K , Dekosky ST , Gauthier S , Selkoe D , Bateman R , Cappa S , Crutch S , Engelborghs S , Frisoni GB , Fox NC , Galasko D , Habert MO , Jicha GA , Nordberg A , Pasquier F , Rabinovici G , Robert P , Rowe C , Salloway S , Sarazin M , Epelbaum S , de Souza LC , Vellas B , Visser PJ , Schneider L , Stern Y , Scheltens P , Cummings JL ((2014) ) Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol 13: , 614–629. |
[12] | Hammes J , Bischof GN , Bohn KP , Onur Ö , Schneider A , Fliessbach K , Hönig MC , Jessen F , Neumaier B , Drzezga A , van Eimeren T ((2021) ) One-stop shop: 18 F-Flortaucipir PET differentiates amyloid-positive and -negative forms of neurodegenerative diseases. J Nucl Med 62: , 240–246. |
[13] | Fleisher AS , Pontecorvo MJ , Devous MD , Lu M , Arora AK , Truocchio SP , Aldea P , Flitter M , Locascio T , Devine M , Siderowf A , Beach TG , Montine TJ , Serrano GE , Curtis C , Perrin A , Salloway S , Daniel M , Wellman C , Joshi AD , Irwin DJ , Lowe VJ , Seeley WW , Ikonomovic MD , Masdeu JC , Kennedy I , Harris T , Navitsky M , Southekal S , Mintun MA ((2020) ) Positron emission tomography imaging with [18F]flortaucipir and postmortem assessment of Alzheimer disease neuropathologic changes. JAMA Neurol 77: , 829–839. |
[14] | Boccardi M , Altomare D , Ferrari C , Festari C , Antelmi L , Pievani M , Tarallo A , Muscio C , Guerra UP , Paghera B , Padovani A , Frisoni GB , Zanetti O , Anzola GP , Bertocchi M , Chitò E , Galluzzi S , Geroldi C , Lussignoli G , Mattioli F , Moretti D , Pizzocaro C , Borroni B , Rozzini L , Prelle A , Gennuso M , Villani D , Raimondi MC , Gentile S , Bellelli G , Morandi A , Turco R , Bellandi D , Carbone P , Abruzzi L , Bettoni L , Bianchetti A , Facchi E , Di Fazio I , Turla M , Cotelli MS , Volta GD , Bigni B , Bilotti G , Vollaro S , Rozzini R , Boffelli S , Cappuccio M , Conti MZ , Guizzetti G , Defanti C , Mirabile D , Fascendini S , Manzoni L , Salvi G Pietro , Belotti G , Cavaliere S , Fiacco F , Valente L , Ciccone A , Lanari A , Selletti L , Palmerini F , Avanzi S , Vezzadini G ((2016) ) Do beliefs about the pathogenetic role of amyloid affect the interpretation of amyloid PET in the clinic? Neurodegener Dis 16: , 1–7. |
[15] | Bocchetta M , Galluzzi S , Kehoe PG , Aguera E , Bernabei R , Bullock R , Ceccaldi M , Dartigues JF , De Mendonça A , Didic M , Eriksdotter M , Félician O , Frölich L , Gertz HJ , Hallikainen M , Hasselbalch SG , Hausner L , Heuser I , Jessen F , Jones RW , Kurz A , Lawlor B , Lleo A , Martinez-Lage P , Mecocci P , Mehrabian S , Monsch A , Nobili F , Nordberg A , Rikkert MO , Orgogozo JM , Pasquier F , Peters O , Salmon E , Sänchez-Castellano C , Santana I , Sarazin M , Traykov L , Tsolaki M , Visser PJ , Wallin AK , Wilcock G , Wilkinson D , Wolf H , Yener G , Zekry D , Frisoni GB ((2015) ) The use of biomarkers for the etiologic diagnosis of MCI in Europe: An EADC survey. Alzheimers Dement 11: , 195–206. |
[16] | Altomare D , De Wilde A , Ossenkoppele R , Pelkmans W , Bouwman F , Groot C , Van Maurik I , Zwan M , Yaqub M , Barkhof F , Van Berckel BN , Teunissen CE , Frisoni GB , Scheltens P , Van Der Flier WM ((2019) ) Applying the ATN scheme in a memory clinic population: The ABIDE project. Neurology 93: , E1635–E1646. |
[17] | Albert MS , DeKosky ST , Dickson D , Dubois B , Feldman HH , Fox NC , Gamst A , Holtzman DM , Jagust WJ , Petersen RC , Snyder PJ , Carrillo MC , Thies B , Phelps CH ((2011) ) The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7: , 270–279. |
[18] | Altomare D , Ferrari C , Caroli A , Galluzzi S , Prestia A , van der Flier WM , Ossenkoppele R , Van Berckel B , Barkhof F , Teunissen CE , Wall A , Carter SF , Schöll M , Choo IH , Grimmer T , Redolfi A , Nordberg A , Scheltens P , Drzezga A , Frisoni GB ((2019) ) Prognostic value of Alzheimer’s biomarkers in mild cognitive impairment: The effect of age at onset. J Neurol 266: , 2535–2545. |
[19] | Bertens D , Vos S , Kehoe P , Wolf H , Nobili F , Mendonça A , Van Rossum I , Hort J , Molinuevo JL , Heneka M , Petersen R , Scheltens P , Visser PJ ((2019) ) Use of mild cognitive impairment and prodromal AD/MCI due to AD in clinical care: A European survey. Alzheimers Res Ther 11: , 1–12. |
[20] | Chien DT , Szardenings AK , Bahri S , Walsh JC , Mu F , Xia C , Shankle WR , Lerner AJ , Su MY , Elizarov A , Kolb HC ((2014) ) Early clinical PET imaging results with the novel PHF-tau radioligand [F18]-T808. J Alzheimers Dis 38: , 171–184. |
[21] | Ossenkoppele R , Rabinovici GD , Smith R , Cho H , Schöll M , Strandberg O , Palmqvist S , Mattsson N , Janelidze S , Santillo A , Ohlsson T , Jogi J , Tsai R , La Joie R , Kramer J , Boxer AL , Gorno-tempini ML , Miller BL , Choi JY , Ryu YH , Lyoo CH , Hansson O ((2018) ) Discriminative accuracy of [18F]flortaucipir positron emission tomography for Alzheimer disease vs other neurodegenerative disorders. JAMA 320: , 1151–1162. |
[22] | Duits FH , Martinez-Lage P , Paquet C , Engelborghs S , Lleó A , Hausner L , Molinuevo JL , Stomrud E , Farotti L , Ramakers IHGB, Tsolaki M , Skarsgård C , åstrand R , Wallin A , Vyhnalek M , Holmber-Clausen M , Forlenza O V. , Ghezzi L , Ingelsson M , Hoff EI , Roks G , De Mendonça A , Papma JM , Izagirre A , Taga M , Struyfs H , Alcolea DA , Frölich L , Balasa M , Minthon L , Twisk JWR , Persson S , Zetterberg H , Van Der Flier WM , Teunissen CE , Scheltens P , Blennow K ((2016) ) Performance and complications of lumbar puncture in memory clinics: Results of the multicenter lumbar puncture feasibility study. Alzheimers Dement 12: , 154–163. |
[23] | Frisoni GB , Altomare D , Thal DR , Ribaldi F , van der Kant R , Ossenkoppele R , Blennow K , Cummings J , van Duijn C , Nilsson PM , Dietrich PY , Scheltens P , Dubois B ((2022) ) The probabilistic model of Alzheimer disease: The amyloid hypothesis revised. Nat Rev Neurosci 23: , 53–66. |
[24] | Koriath CAM , Kenny J , Ryan NS , Rohrer JD , Schott JM , Houlden H , Fox NC , Tabrizi SJ , Mead S ((2021) ) Genetic testing in dementia — utility and clinical strategies. Nat Rev Neurol 17: , 23–36. |
[25] | Vermunt L , Sikkes SAM , van den Hout A , Handels R , Bos I , van der Flier WM , Kern S , Ousset PJ , Maruff P , Skoog I , Verhey FRJ , Freund-Levi Y , Tsolaki M , Wallin ÅK , Olde Rikkert M , Soininen H , Spiru L , Zetterberg H , Blennow K , Scheltens P , Muniz-Terrera G , Visser PJ , Vellas B , Reynish E , Ousset PJ , Andrieu S , Burns A , Pasquier F , Frisoni G , Salmon E , Michel JP , Zekry DS , Boada M , Dartigues JF , Olde-Rikkert MGM , Rigaud AS , Winblad B , Malick A , Sinclair A , Frölich L , Scheltens P , Ribera C , Touchon J , Robert P , Salva A , Waldemar G , Bullock R , Tsolaki M , Rodriguez G , Spiru L , Jones RW , Stiens G , Stoppe G , Eriksdotter Jönhagen M , Cherubini A , Lage PM , Gomez-Isla T , Camus V , Agüera-Morales E , Lopez F , Savy S , Cantet C , Coley N ((2019) ) Duration of preclinical, prodromal, and dementia stages of Alzheimer’s disease in relation to age, sex, and APOE genotype. Alzheimers Dement 15: , 888–898. |
[26] | Reitz C , Rogaeva E , Beecham GW ((2020) ) Late-onset vs nonmendelian early-onset Alzheimer disease. Neurol Genet 6: , e512. |
[27] | Goldman JS , Hahn SE , Catania JW , Larusse-Eckert S , Butson MB , Rumbaugh M , Strecker MN , Roberts JS , Burke W , Mayeux R , Bird T ((2011) ) Genetic counseling and testing for Alzheimer disease: Joint practice guidelines of the American College of Medical Genetics and the National Society of Genetic Counselors. Genet Med 13: , 597–605. |
[28] | Waldemar G , Dubois B , Emre M , Georges J , McKeith IG , Rossor M , Scheltens P , Tariska P , Winblad B ((2007) ) Recommendations for the diagnosis and management of Alzheimer’s disease and other disorders associated with dementia: EFNS guideline. Eur J Neurol 14: , 1–26. |
[29] | Falgàs N , Tort-Merino A , Balasa M , Borrego-Écija S , Castellví M , Olives J , Bosch B , Férnandez-Villullas G , Antonell A , Augé JM , Lomeña F , Perissinotti A , Bargalló N , Sánchez-Valle R , Lladó A ((2019) ) Clinical applicability of diagnostic biomarkers in early-onset cognitive impairment. Eur J Neurol 26: , 1098–1104. |
[30] | Ribaldi F , Altomare D , Frisoni GB ((2019) ) Is a large-scale screening for Alzheimer’s disease possible? Yes, in a few years. J Prev Alzheimers Dis 6: , 221–222. |




