Peripheral blood biomarkers associated with combination of immune checkpoint blockade plus chemotherapy in NSCLC
Abstract
BACKGROUND:
Biomarkers predicting clinical outcomes of treating non-small cell lung cancer (NSCLC) with combination of immune checkpoint inhibitors (ICIs) and chemotherapy would be valuable.
OBJECTIVE:
This study aims to seek predictors of combination of ICI/chemotherapy response in NSCLC patients using peripheral blood samples.
METHODS:
Patients diagnosed with advanced NSCLC between July 2019 and May 2021 receiving combination of ICI/chemotherapy were included and assessed for partial responses (PR), stable disease (SD) or progressive disease (PD). We measured circulating immune cells, plasma cytokines and chemokines.
RESULTS:
Nineteen patients were enrolled. The proportions of circulating natural killer (NK) cells within CD45 + cells, programmed death 1 (PD-1) + Tim-3 + T cells within CD4 + cells, and the amount of chemokine C-X-C ligand (CXCL10) in the plasma were significantly elevated in PR relative to SD/PD patients (median 8.1%-vs-2.1%,
CONCLUSIONS:
We conclude that NK cells, CD4 + PD-1 + Tim-3 + T cells, and CXCL10 levels in pre-treatment peripheral blood may predict the efficacy of combination of ICI/chemotherapy in NSCLC.
1.Introduction
Lung cancer is a common cancer causing 1.9 million deaths per year globally [1]. The introduction of immune checkpoint inhibitors (ICIs) has led to breakthroughs in the treatment of advanced non-small cell lung carcinoma (NSCLC). Based on the results of phase III trials, combined ICI/chemotherapy has become the current standard of care for the initial treatment of advanced NSCLC without driver mutations that can be targeted by small molecule inhibitors [2, 3]. Patients with excellent responses to such ICI/chemotherapy combinations are expected to have long-term survival, but predicting in advance which patients will fall into this category is challenging. The availability of reliable biomarkers would be valuable in this context.
Several clinical trials that have indicated that the efficacy of ICI monotherapy for advanced NSCLC is associated with the proportion of cells expressing programmed death ligand 1 (PD-L1) in the tumor tissue [4, 5]. ICIs are most effective in patients with high PD-L1 expression, with a reported 5-year survival rate of 31% [6]. This relationship between PD-L1 expression and clinical response has also been observed in combination of ICI/chemotherapy [2, 3]. Consequently, the proportion of PD-L1 is used to predict the efficacy of ICIs in clinical practice. However, treatment strategies based on PD-L1 expression alone remain unsatisfactory; some patients with a PD-L1 high tumor proportion score (TPS) nonetheless fail to respond to ICIs, and some patients with low or negative TPS respond well to ICIs. In addition, there is a practical constraint in that the PD-L1 TPS cannot be evaluated in some patients due to the small amounts of tumor tissue available, or to having received only a cytological diagnosis. Therefore, in addition to PD-L1 expression analysis, non-invasive, reliable, and simple methods are warranted to predict the efficacy of ICI therapy.
Peripheral blood is easily accessible in a minimally invasive manner. Several previous studies have reported that CD8 + T cells, CD4 + T cells, Natural Killer (NK) cells, and the neutrophil to lymphocyte ratio (NLR) in blood samples are useful for predicting the efficacy of ICIs in patients with lung cancer.
ICIs mediate their anti-tumor effects inter alia by blocking immune suppressive signals and preventing CD8 + T cell exhaustion [7]. Hence, CD8 + T cells have been well studied, not only in terms of quantity but also whether they are exhausted or activated and which subpopulations are present, all of which have been reported as predictors of response [8, 9, 10, 11]. CD4 + T cells and NK cells in the tumor microenvironment (TME) were also reported as predictors [9, 12, 13, 14]. Several other predictors that have been reported are immune suppressive cells such as regulatory T cells (Tregs) [13] and myeloid-derived suppressor cells (MDSCs) [15, 16, 17], cytokines and chemokines such as interleukin (IL)-6 [18], IL-8 [19, 20] and chemokine C-X-C ligand 10 (CXCL10) [21]. The NLR and absolute counts of lymphocytes and monocytes can easily be measured in routine clinical practice [22, 23, 24].
However, the conclusions in these reports were based on data from patients who received ICI monotherapy, and some predictors have conflicting functions between ICI monotherapy and chemotherapy. Therefore, whether the same applies to combination of ICI/chemotherapy is unclear. Cytotoxic anticancer drugs affect the TME in multiple ways, including by reducing immune cell numbers due to bone marrow suppression or by activating immune cells via the release of damage-associated molecular patterns (DAMPs) [25]. It is therefore likely that predictors of responses to combination of ICI/chemotherapy and ICI monotherapy will differ. Despite advances in combination immunotherapy regimens, it is also unclear whether peripheral blood biomarkers can function as useful predictors for the success of such therapies. To this end, here we conducted a retrospective study to identify predictors of combination of ICI/chemotherapy response using pre-treatment peripheral blood samples.
2.Materials and methods
2.1Patients
Patients diagnosed with unresectable locally advanced, postoperatively recurrent, or metastatic NSCLC at the Tohoku University Hospital between July 2019 and May 2021 were reviewed. Inclusion criteria for this study were age
This study was approved by the institutional review boards of Tohoku University Hospital.
The medical records of patients were reviewed to extract clinical information, as follows: age, sex, ECOG PS, histology, clinical stage, PD-L1 expression by the tumor, complete blood count, first-line chemotherapy regimen, tumor response, and progression-free survival (PFS). A total of 19 patients was enrolled based on the above criteria.
All subjects were evaluated for tumor responses according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1). Patients were dichotomized into 1) Partial Response (PR) or 2) Stable Disease (SD)/Progressive Disease (PD) groups. PFS was defined as the time elapsed from the initiation of combination of ICI/chemotherapy until the first diagnosis of progressive disease, or death from any cause. Patients without documented clinical or radiographic disease progression or who were still alive were censored on the date of the last follow-up. The cutoff date of the study was March 31st, 2022.
2.2Peripheral blood collection
Peripheral blood samples were collected the day before initiation and just before the third dose of the first-line treatment in all patients. Mononuclear cell preparation tubes (BD Vacutainer® CPT
2.3Flow cytometry
Cryopreserved PBMCs were thawed in a 37∘C water bath and resuspended in PBS with 3% fetal bovine serum. Total cell numbers were counted, and 1
2.4Cytometric bead array
Concentrations of plasma Concentrations of plasma IL-4, IL-2, CXCL10, IL-1
2.5ELISA
Concentrations of plasma HMGB1 and calreticulin were measured using ELISA kits (Cloud-Clone Corp., Houston, USA), following the manufacturer’s protocol. The concentration of these DAMPs was quantified by SpectraMax ABS (MOLECULAR DEVICES, Tokyo, Japan).
2.6Statistical analysis
Statistical analyses were performed using the commercial software JMP (SAS Institute Japan, Tokyo, Japan) or GraphPad prism (GraphPad Software Inc. California, USA). Medians with 95% confidence intervals (CI) are given for most data. Differences between continuous variables were assessed using the Mann–Whitney U test. The Chi-square test was used to analyze categorical variables. Correlations were measured as Pearson correlation coefficients. PFS was evaluated with the Kaplan–Meier method, and differences between survival curves were evaluated using the log-rank test. Considering the exploratory approaches used,
3.Results
3.1Patients’ characteristics
Table 1
Patients’ characteristics at baseline
| Characteristics | Number (%) or Median (Range) | ||||||
|---|---|---|---|---|---|---|---|
| ALL | ( | PR | ( | SD/PD | ( | ||
| Age | 69 | (52–78) | 71 | (52–78) | 67 | (63–77) | 0.5346 |
| Gender | |||||||
| Male | 14 | (73.7) | 9 | (69.2) | 5 | (83.3) | 0.5164 |
| Female | 5 | (26.3) | 4 | (30.8) | 1 | (16.7) | |
| Performance status | 0.1112 | ||||||
| 0 | 14 | (73.7) | 11 | (84.6) | 3 | (50.0) | |
| 1 | 5 | (26.3) | 2 | (15.4) | 3 | (50.0) | |
| Histology | 0.3421 | ||||||
| Adenocarcinoma | 13 | (68.4) | 8 | (61.5) | 5 | (83.3) | |
| Squamous cell carcinoma | 6 | (31.6) | 5 | (38.5) | 1 | (16.7) | |
| Disease stage | 0.5986 | ||||||
| IVB | 10 | (52.6) | 6 | (46.2) | 4 | (66.7) | |
| IVA | 5 | (26.3) | 4 | (30.8) | 1 | (16.7) | |
| IIIB | 2 | (10.5) | 2 | (15.4) | 0 | ||
| Postoperative recurrence | 2 | (10.5) | 1 | (7.7) | 1 | (16.7) | |
| PD-L1 TPS | 0.2522 | ||||||
| | 3 | (15.8) | 3 | (23.1) | 0 | ||
| 1–49% | 4 | (21.1) | 2 | (15.4) | 2 | (33.3) | |
| | 9 | (47.4) | 7 | (53.8) | 2 | (33.3) | |
| Unknown | 3 | (15.8) | 1 | (7.7) | 2 | (33.3) | |
| Regimen of ICI/chemotherapy | 0.1927 | ||||||
| CDDP | 4 | (21.1) | 1 | (7.7) | 3 | (50.0) | |
| CBDCA | 8 | (42.1) | 6 | (46.2) | 2 | (33.3) | |
| CBDCA | 6 | (31.6) | 5 | (38.4) | 1 | (16.7) | |
| CBDCA | 1 | (5.3) | 1 | (7.7) | 0 | ||
| Leukocyte count (/ | |||||||
| Neutrophils | 5320 | (2420–10410) | 5000 | (2420–10410) | 5770 | (4810–7750) | 0.1791 |
| Lymphocytes | 1380 | (730–2570) | 1360 | (1120–2420) | 1550 | (730–2570) | 0.9488 |
| Monocytes | 480 | (250–1670) | 480 | (250–1670) | 505 | (440–920) | 0.9487 |
TPS, tumor proportion score; CDDP, Cisplatin; PEM, Pemetrexed; CBDCA, Carboplatin; PTX, Paclitaxel; Pembro, Pembrolizumab; Atezo, Atezolizumab; BEV, Bevacizumab.
The background characteristics of the patients are summarized in Table 1. Of the 19 patients 13 exhibited a PR and 6 SD/PD. The median age was 69 years (range, 52–78 years), and 14 (73.7%) were male. There were 13 (68.4%) adenocarcinomas and 6 (31.6%) squamous cell carcinomas. All patients received platinum-based chemotherapy together with ICIs, as follows: cisplatin
The median follow-up time was 14.8 months and median PFS of all patients was 7.4 months. The median PFS of patients with PR was 12.4 months, significantly longer those with SD/PD (4.6 months,
3.2NK cells and CD4 + PD-1 + Tim-3 + T cells are associated with clinical efficacy
Figure 1.
Peripheral NK cells and CD4 + PD-1 + Tim-3 + cells predict clinical efficacy. (A, B) Percentages of NK cells within the CD45 + population, and %CD4 + PD-1 + Tim-3 + cells within the CD4 + population in peripheral blood of patients with PR or SD/PD. Data are medians
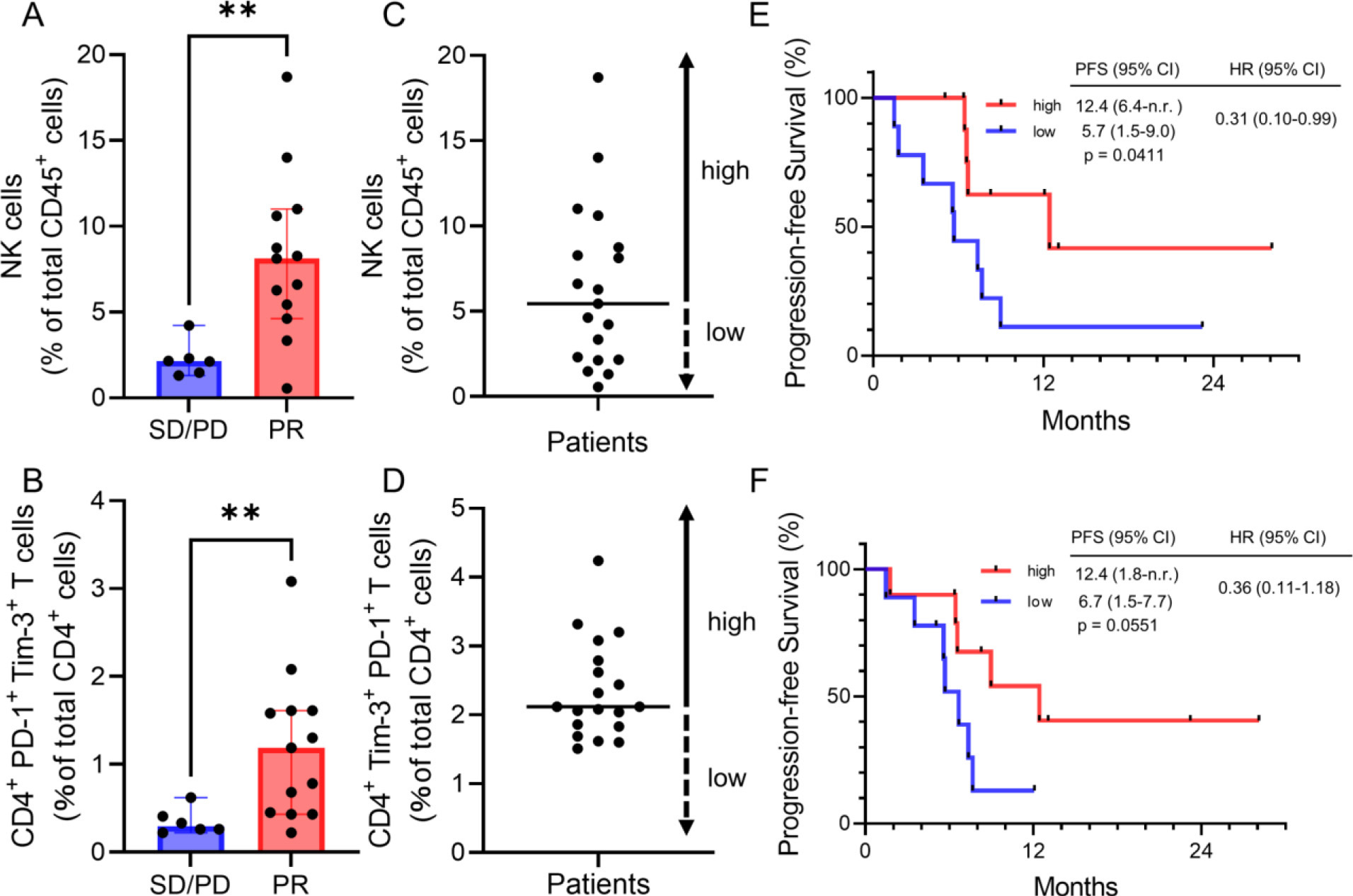
In order to determine which if any blood immune cells correlated with clinical response, we compared the distribution of immune cell populations in PBMCs between the PR and the SD/PD groups. We found that the proportions of circulating NK cells and CD4 + PD-1 + Tim-3 + T cells within the CD45 + population were significantly higher in the PR group (
3.3CXCL10 is associated with clinical efficacy
Figure 2.
Plasma CXCL10 predicts clinical efficacy. (A) Plasma CXCL10 levels in PR and SD/PD patients. Data are medians
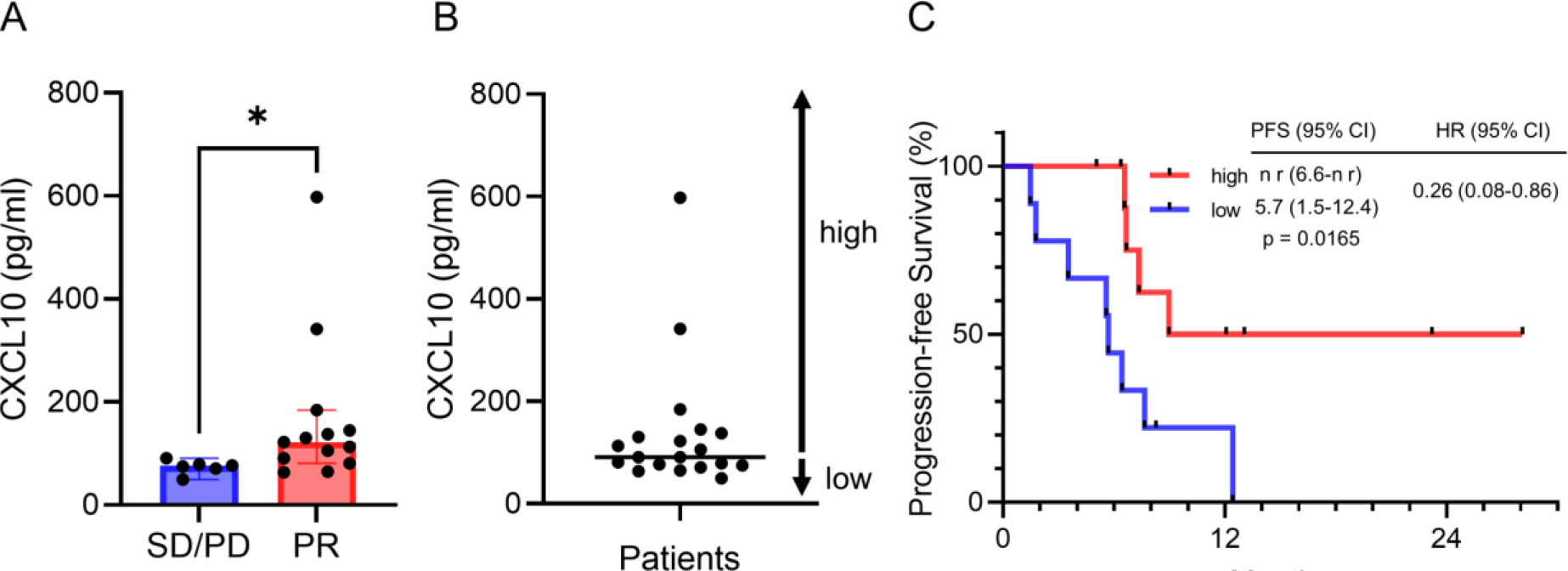
Figure 3.
Plasma cytokine concentrations. Plasma cytokines in PR and SD/PD patients. Dot plots are each patient’s data and horizontal bars are medians.
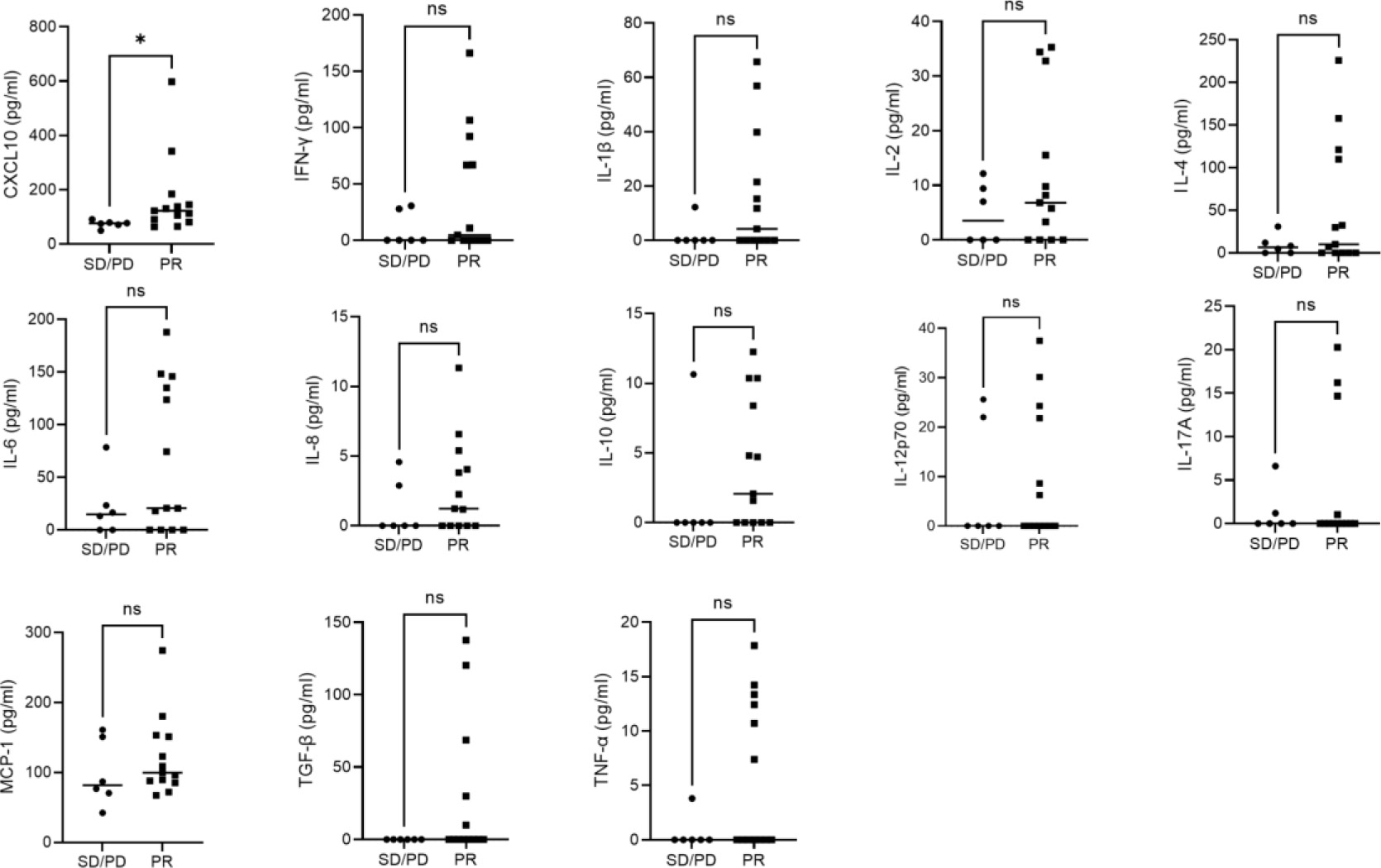
In the TME, CD4 + T cells help CD8 + cytotoxic T cells via IL-2, as well as producing cytokines with direct anti-tumor activity, such as IFN-
3.4Plasma DAMPs are not correlated with clinical outcome
DAMPs are released from dying cells or cells damaged by chemotherapy, and can activate several types of immune cells [28, 29]. We hypothesized that DAMPs affect the efficacy of the combination of ICI/chemotherapy. We quantified HMGB1 and calreticulin in patients’ plasma because they are DAMPs reported to be released by chemotherapy [30, 31]. However, there were no differences between PR and SD/PD patients for either of these DAMPs (Supplementary Fig. 4); neither were there any significant differences when comparing pre- and post-treatment. Therefore, we concluded that measuring these DAMPs is not useful for predicting treatment outcome.
3.5Prediction of the efficacy of combination immunotherapy
Figure 4.
Combinations of NK cells, CD4 + PD-1 + Tim-3 + T cells and CXCL10 are better predictors of ICI/chemotherapy outcome than any single factor. (A) Each patient is shown as a column, indicating the PD-L1 TPS, NK cells, CD4 + PD-1 + Tim-3 + T cells, CXCL10, best overall response, and PFS. (B) Kaplan-Meier curves of PFS for patients with two or three elevated factors among NK cells, CD4 + PD-1 + Tim-3 + , and CXCL10 versus those with zero or one factor. ICIs, immune checkpoint inhibitors; PD-L1, programmed death ligand 1; TPS, tumor proportion score; NK cells, natural killer cells; CD4 + Tex, exhausted T cells (CD4 + PD-1 + Tim-3 + T cells); CXCL10, chemokine C-X-C ligand 10; PFS, progression-free survival; PR, partial response; SD/PD, stable disease/progressive disease; HR, hazard ratio; CI, confidence interval; n r, not reached, ND; no data.
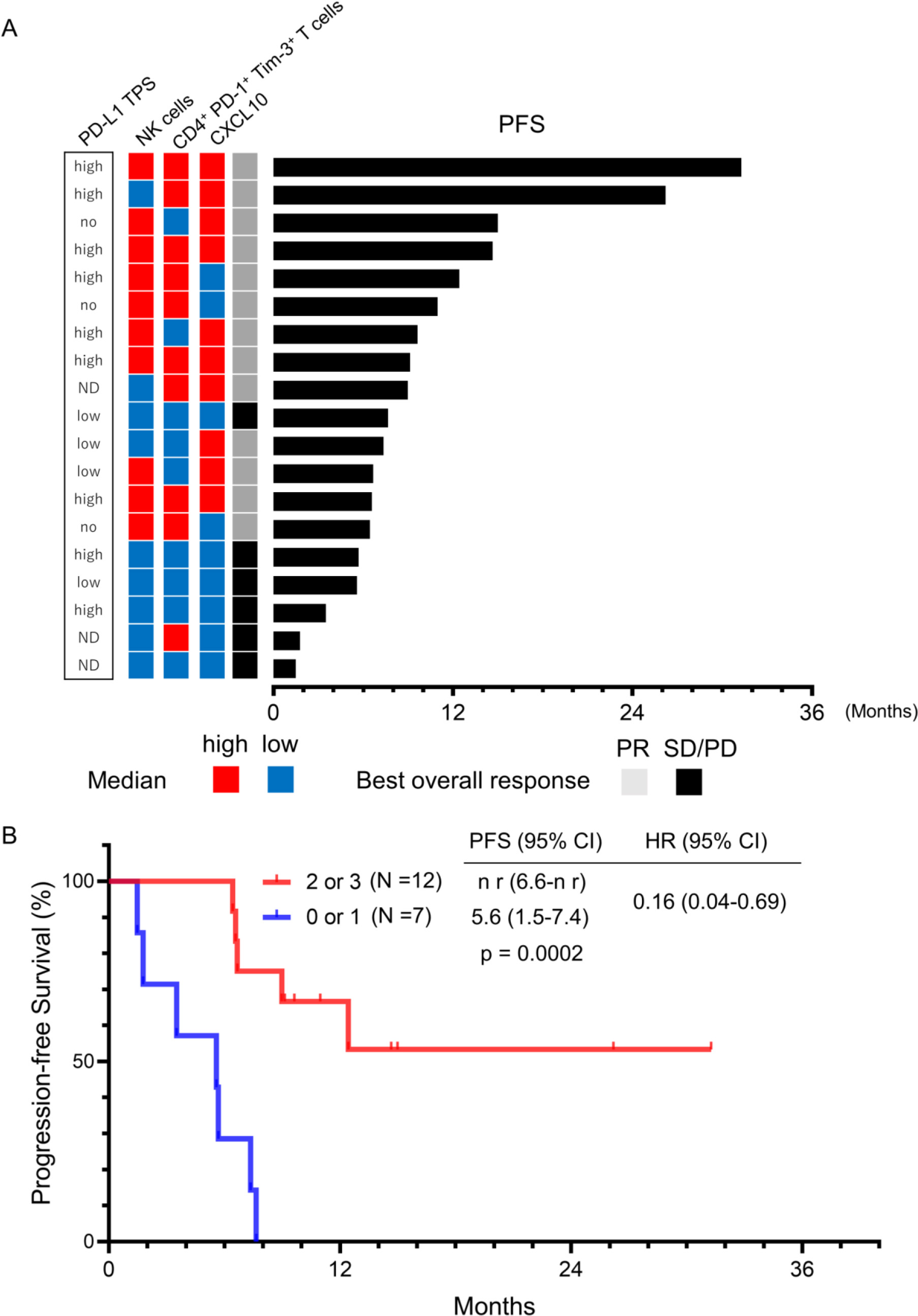
Based on the above results, we concluded that 3 factors (NK cells, CD4 + PD-1 + Tim-3 + T cells and CXCL10) could each predict the efficacy of combination of ICI/chemotherapy. We then investigated whether these factors were correlated with each other by means of Pearson correlation coefficient estimates
(Supplementary Fig. 5) no significant correlation was found among them. Figure 4 shows the impact of PD-L1 TPS, the proportions of NK cells and CD4 + PD-1 + Tim-3 + T cells, and the level of CXCL10 on response to treatment and on PFS. Patients with higher levels of two or all three of these 3 factors exhibited a higher PR rate (
4.Discussion
In this study, we sought peripheral blood biomarkers informative for the outcome of combination of ICI/chemotherapy in patients with NSCLC. The main finding was that patients experiencing a partial response had higher percentages of NK cells and CD4 + PD-1 + Tim-3 + T cells and a higher CXCL10 level in the peripheral blood than patients with stable or progressive disease. In addition, higher levels of NK cells, CD4 + PD-1 + Tim-3 + T cells and CXCL10 predicted a better PFS, and a combination of these biomarkers further improved the accuracy of prediction. In contrast, PD-L1 TPS failed to predict treatment efficacy in this study.
To the best of our knowledge, only two studies have previously reported predictors for combination of ICI/chemotherapy in NSCLC patients [32, 33]. Miriam et al reported that elevation of NK cells in peripheral blood was a favorable predictive factor, consistent with our study. In the TME, NK cells play important roles such as preventing metastasis, attracting different immune cells into the tumor and directly lysing cancer cells that are resistant to ICI-facilitated CD8 + T cell cytotoxicity due to lack of expression MHC class I molecules [34, 35, 36, 37].
Patients with higher proportion of CD4 + PD-1 + Tim-3 + T cells showed better clinical responses in our study. We hypothesized that PD-1 and Tim-3 are markers of exhaustion of CD4 + as well as CD8 + T cells and thus are targets for ICIs, such that patients with higher levels of these cells have a better clinical response to blockade of these checkpoints. In a previous study of patients not receiving immunotherapy, those with high CD4 + PD-1 + Tim3high T cells were reported to have a worse prognosis [38]. This is consistent with exhaustion of these cells and their potential susceptibility to checkpoint blockade. However, exhaustion of CD4 + T cells have not been fully uncovered. We used PD-1 and Tim-3 as markers of exhaustion because they are the most commonly-used markers of exhaustion of CD8 + T cells [39], and it was reported that CD4 + T cells with reduced effector functions upregulate Tim-3 and PD-1, paralleling phenotypes observed in exhausted CD8 + T cells [40]. However, after completing our analysis, a study was published suggesting that CD39 rather than Tim-3 should be used to assess exhaustion of CD4 + T cells [41]. Therefore, the function of PD-1 + Tim-3 + CD4 + T cells requires further clarification in order to determine the role of these cells in the TME and to assess which markers are most appropriate for assessing exhaustion status.
Of the 13 cytokines we analyzed, only CXCL10 was significantly elevated in the PR group relative to the SD/PD group, and patients with higher CXCL10 had longer PFS. Several studies reported that CXCL10 induced migration of immune cells into the tumor and that this correlated with clinical efficacy [42, 43]. Thus, high levels of CXCL10 in the peripheral blood may indicate active immune cell infiltration into tumor tissue also in combination therapy.
As mentioned, we hypothesized that changes in TME by chemotherapy might alter predictors between ICI monotherapy and combination of ICI/chemotherapy. For the ICI monotherapy, NK cells, CD4 + PD-1 + T cells, and CXCL10 were reported as good predictors of response [14, 21, 44]. On the other hand, for the chemotherapy, NK cells and CXCL10 did not have function as predictors [45, 46], and CD4 + PD-1 + T cells were reported as a poor predictor [38, 47]. Although further studies are needed, these results may suggest that predictors of ICI monotherapy are also useful to predict combination of ICI/chemotherapy.
Our study has several limitations. First, it was retrospective with only a small number of patients. To validate our results, we searched for publicly-available data on PBMC from patients who received ICIs plus chemotherapy. However, there appear to be no such cohorts with information on treatment and response. In addition considering that our biomarker analysis was exploratory, we did not correct
In conclusion, this study revealed that NK cells, CD4 PD-1 + Tim-3 + T cells, and CXCL10 in pre-treatment peripheral blood may predict the efficacy of combination of ICI/chemotherapy in NSCLC. Because the TME is very complex due to the variety of immune cells and cytokines involved, the elevation of two or more of these three markers may be a more useful predictive biomarker in clinical practice than any single factor. A further prospective large-scale multi-center study is warranted to validate these results.
Author contributions
For every author, his or her contribution to the manuscript needs to be provided using the following categories:
Conception: Nozomu Kimura, Yoko Tsukita, Risa Ebina-Shibuya, Eisaku Miyauchi.
Interpretation or analysis of data: Nozomu Kimura, Yoko Tsukita, Risa Ebina-Shibuya, Eisaku Miyauchi, Mitsuhiro Yamada, Chihiro Inoue Daisuke Narita.
Preparation of the manuscript: Nozomu Kimura, Yoko Tsukita.
Revision for important intellectual content: Ryota Saito, Naoya Fujino, Tomohiro Ichikawa, Tsutomu Tamada, Hisatoshi Sugiura.
Supervision: Eisaku Miyauchi, Hisatoshi Sugiura.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230301.
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 20K17207.
NAI Inc. assisted with editing the English language.
References
[1] | C. Fitzmaurice, D. Abate, N. Abbasi et al., Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: A systematic analysis for the global burden of disease study, JAMA Oncol 5: ((2019) ), 1749–1768. |
[2] | L. Gandhi, D. Rodríguez-Abreu, S. Gadgeel, E. Esteban, E. Felip, F. De Angelis, M. Domine, P. Clingan, M.J. Hochmair, S.F. Powell, S.Y. Cheng, H.G. Bischoff, N. Peled, F. Grossi, R.R. Jennens, M. Reck, R. Hui, E.B. Garon, M. Boyer, B. Rubio-Viqueira, S. Novello, T. Kurata, J.E. Gray, J. Vida, Z. Wei, J. Yang, H. Raftopoulos, M.C. Pietanza and M.C. Garassino, Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer, N Engl J Med 378: ((2018) ), 2078–2092. |
[3] | L. Paz-Ares, A. Luft, D. Vicente, A. Tafreshi, M. Gümüş, J. Mazières, B. Hermes, F. Çay Şenler, T. Csöszi, A. Fülöp, J. Rodríguez-Cid, J. Wilson, S. Sugawara, T. Kato, K.H. Lee, Y. Cheng, S. Novello, B. Halmos, X. Li, G.M. Lubiniecki, B. Piperdi and D.M. Kowalski, Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer, N Engl J Med 379: ((2018) ), 2040–2051. |
[4] | R.S. Herbst, G. Giaccone, F. de Marinis, N. Reinmuth, A. Vergnenegre, C.H. Barrios, M. Morise, E. Felip, Z. Andric, S. Geater, M. Özgüroğlu, W. Zou, A. Sandler, I. Enquist, K. Komatsubara, Y. Deng, H. Kuriki, X. Wen, M. McCleland, S. Mocci, J. Jassem and D.R. Spigel, Atezolizumab for first-line treatment of PD-L1-selected patients with NSCLC, N Engl J Med 383: ((2020) ), 1328–1339. |
[5] | E.B. Garon, N.A. Rizvi, R. Hui, N. Leighl, A.S. Balmanoukian, J.P. Eder, A. Patnaik, C. Aggarwal, M. Gubens, L. Horn, E. Carcereny, M.J. Ahn, E. Felip, J.S. Lee, M.D. Hellmann, O. Hamid, J.W. Goldman, J.C. Soria, M. Dolled-Filhart, R.Z. Rutledge, J. Zhang, J.K. Lunceford, R. Rangwala, G.M. Lubiniecki, C. Roach, K. Emancipator and L. Gandhi, Pembrolizumab for the treatment of non-small-cell lung cancer, N Engl J Med 372: ((2015) ), 2018–28. |
[6] | M. Reck, D. Rodríguez-Abreu, A.G. Robinson, R. Hui, T. Csöszi, A. Fülöp, M. Gottfried, N. Peled, A. Tafreshi, S. Cuffe, M. O’Brien, S. Rao, K. Hotta, T.A. Leal, J.W. Riess, E. Jensen, B. Zhao, M.C. Pietanza and J.R. Brahmer, five-year outcomes with Pembrolizumab versus chemotherapy for metastatic non-small-cell lung cancer with PD-L1 tumor proportion score ⩾ 50, J Clin Oncol 39: ((2021) ), 2339–2349. |
[7] | A. Ribas and J.D. Wolchok, Cancer immunotherapy using checkpoint blockade, Science 359: ((2018) ), 1350–1355. |
[8] | B.Y. Nabet, M.S. Esfahani, E.J. Moding, E.G. Hamilton, J.J. Chabon, H. Rizvi, C.B. Steen, A.A. Chaudhuri, C.L. Liu, A.B. Hui, D. Almanza, H. Stehr, L. Gojenola, R.F. Bonilla, M.C. Jin, Y.J. Jeon, D. Tseng, C. Liu, T. Merghoub, J.W. Neal, H.A. Wakelee, S.K. Padda, K.J. Ramchandran, M. Das, A.J. Plodkowski, C. Yoo, E.L. Chen, R.B. Ko, A.M. Newman, M.D. Hellmann, A.A. Alizadeh and M. Diehn, Noninvasive early identification of therapeutic benefit from immune checkpoint inhibition, Cell 183: ((2020) ), 363–376e13. |
[9] | G. Mazzaschi, F. Facchinetti, G. Missale, D. Canetti, D. Madeddu, A. Zecca, M. Veneziani, F. Gelsomino, M. Goldoni, S. Buti, P. Bordi, F. Aversa, A. Ardizzoni, F. Quaini and M. Tiseo, The circulating pool of functionally competent NK and CD8+ cells predicts the outcome of anti-PD1 treatment in advanced NSCLC, Lung Cancer 127: ((2019) ), 153–163. |
[10] | C.G. Kim, K.H. Kim, K.H. Pyo, C.F. Xin, M.H. Hong, B.C. Ahn, Y. Kim, S.J. Choi, H.I. Yoon, J.G. Lee, C.Y. Lee, S.Y. Park, S.H. Park, B.C. Cho, H.S. Shim, E.C. Shin and H.R. Kim, Hyperprogressive disease during PD-1/PD-L1 blockade in patients with non-small-cell lung cancer, Ann Oncol 30: ((2019) ), 1104–1113. |
[11] | A.O. Kamphorst, R.N. Pillai, S. Yang, T.H. Nasti, R.S. Akondy, A. Wieland, G.L. Sica, K. Yu, L. Koenig, N.T. Patel, M. Behera, H. Wu, M. McCausland, Z. Chen, C. Zhang, F.R. Khuri, T.K. Owonikoko, R. Ahmed and S.S. Ramalingam, Proliferation of PD-1+ CD8 T cells in peripheral blood after PD-1-targeted therapy in lung cancer patients, Proc Natl Acad Sci USA 114: ((2017) ), 4993–4998. |
[12] | M. Zuazo, H. Arasanz, G. Fernández-Hinojal, M.J. García-Granda, M. Gato, A. Bocanegra, M. Martínez, B. Hernández, L. Teijeira, I. Morilla, M.J. Lecumberri, A. Fernández de Lascoiti, R. Vera, G. Kochan and D. Escors, Functional systemic CD4 immunity is required for clinical responses to PD-L1/PD-1 blockade therapy, EMBO Mol Med 11: ((2019) ), e10293. |
[13] | H. Kagamu, S. Kitano, O. Yamaguchi, K. Yoshimura, K. Horimoto, M. Kitazawa, K. Fukui, A. Shiono, A. Mouri, F. Nishihara, Y. Miura, K. Hashimoto, Y. Murayama, K. Kaira and K. Kobayashi, CD4(+) T-cell immunity in the peripheral blood correlates with response to Anti-PD-1 therapy, Cancer Immunol Res 8: ((2020) ), 334–344. |
[14] | Y.H. Cho, M.G. Choi, D.H. Kim, Y.J. Choi, S.Y. Kim, K.J. Sung, J.C. Lee, S.Y. Kim, J.K. Rho and C.M. Choi, Natural killer cells as a potential biomarker for predicting immunotherapy efficacy in patients with non-small cell lung cancer, Target Oncol 15: ((2020) ), 241–247. |
[15] | H.R. Kim, S.M. Park, S.U. Seo, I. Jung, H.I. Yoon, D.I. Gabrilovich, B.C. Cho, S.Y. Seong, S.J. Ha and J.I. Youn, The ratio of peripheral regulatory T cells to Lox-1(+) polymorphonuclear myeloid-derived suppressor cells predicts the early response to Anti-PD-1 therapy in patients with non-small cell lung cancer, Am J Respir Crit Care Med 199: ((2019) ), 243–246. |
[16] | E. Limagne, C. Richard, M. Thibaudin, J.D. Fumet, C. Truntzer, A. Lagrange, L. Favier, B. Coudert and F. Ghiringhelli, Tim-3/galectin-9 pathway and mMDSC control primary and secondary resistances to PD-1 blockade in lung cancer patients, Oncoimmunology 8: ((2019) ), e1564505. |
[17] | A. Passaro, P. Mancuso, S. Gandini, G. Spitaleri, V. Labanca, E. Guerini-Rocco, M. Barberis, C. Catania, E. Del Signore, F. de Marinis and F. Bertolini, Gr-MDSC-linked asset as a potential immune biomarker in pretreated NSCLC receiving nivolumab as second-line therapy, Clin Transl Oncol 22: ((2020) ), 603–611. |
[18] | A. Keegan, B. Ricciuti, P. Garden, L. Cohen, R. Nishihara, A. Adeni, C. Paweletz, J. Supplee, P.A. Jänne, M. Severgnini, M.M. Awad and D.R. Walt, Plasma IL-6 changes correlate to PD-1 inhibitor responses in NSCLC, J Immunother Cancer 8: ((2020) ). |
[19] | M.F. Sanmamed, J.L. Perez-Gracia, K.A. Schalper, J.P. Fusco, A. Gonzalez, M.E. Rodriguez-Ruiz, C. Oñate, G. Perez, C. Alfaro, S. Martín-Algarra, M.P. Andueza, A. Gurpide, M. Morgado, J. Wang, A. Bacchiocchi, R. Halaban, H. Kluger, L. Chen, M. Sznol and I. Melero, Changes in serum interleukin-8 (IL-8) levels reflect and predict response to anti-PD-1 treatment in melanoma and non-small-cell lung cancer patients, Ann Oncol 28: ((2017) ), 1988–1995. |
[20] | K.C. Yuen, L.F. Liu, V. Gupta, S. Madireddi, S. Keerthivasan, C. Li, D. Rishipathak, P. Williams, E.E. Kadel 3rd, H. Koeppen, Y.J. Chen, Z. Modrusan, J.L. Grogan, R. Banchereau, N. Leng, A. Thastrom, X. Shen, K. Hashimoto, D. Tayama, M.S. van der Heijden, J.E. Rosenberg, D.F. McDermott, T. Powles, P.S. Hegde, M.A. Huseni and S. Mariathasan, High systemic and tumor-associated IL-8 correlates with reduced clinical benefit of PD-L1 blockade, Nat Med 26: ((2020) ), 693–698. |
[21] | Y. Wang, H. Chen, T. Zhang, X. Yang, J. Zhong, Y. Wang, Y. Chi, M. Wu, T. An, J. Li, X. Zhao, Z. Dong, Z. Wang, J. Zhao, M. Zhuo and J. Huang, Plasma cytokines interleukin-18 and C-X-C motif chemokine ligand 10 are indicative of the anti-programmed cell death protein-1 treatment response in lung cancer patients, Ann Transl Med 9: ((2021) ), 33. |
[22] | D. Cao, H. Xu, X. Xu, T. Guo and W. Ge, A reliable and feasible way to predict the benefits of Nivolumab in patients with non-small cell lung cancer: A pooled analysis of 14 retrospective studies, Oncoimmunology 7: ((2018) ), e1507262. |
[23] | L. Zhang, L. Bai, X. Liu, Y. Liu, S. Li, J. Liu, S. Zhang, C. Yang, X. Ren and Y. Cheng, Factors related to rapid progression of non-small cell lung cancer in Chinese patients treated using single-agent immune checkpoint inhibitor treatment, Thorac Cancer 11: ((2020) ), 1170–1179. |
[24] | J. Tanizaki, K. Haratani, H. Hayashi, Y. Chiba, Y. Nakamura, K. Yonesaka, K. Kudo, H. Kaneda, Y. Hasegawa, K. Tanaka, M. Takeda, A. Ito and K. Nakagawa, Peripheral blood biomarkers associated with clinical outcome in non-small cell lung cancer patients treated with nivolumab, J Thorac Oncol 13: ((2018) ), 97–105. |
[25] | J.I.G. Solari, E. Filippi-Chiela, E.S. Pilar, V. Nunes, E.A. Gonzalez, F. Figueiro, C.F. Andrade and F. Klamt, Damage-associated molecular patterns (DAMPs) related to immunogenic cell death are differentially triggered by clinically relevant chemotherapeutics in lung adenocarcinoma cells, BMC Cancer 20: ((2020) ), 474. |
[26] | R.E. Tay, E.K. Richardson and H.C. Toh, Revisiting the role of CD4(+) T cells in cancer immunotherapy-new insights into old paradigms, Cancer Gene Ther 28: ((2021) ), 5–17. |
[27] | N.D. Huntington, J. Cursons and J. Rautela, The cancer-natural killer cell immunity cycle, Nat Rev Cancer 20: ((2020) ), 437–454. |
[28] | X.J. Wu, Y.Y. Chen, C.C. Gong and D.S. Pei, The role of high-mobility group protein box 1 in lung cancer, J Cell Biochem 119: ((2018) ), 6354–6365. |
[29] | J. Fucikova, R. Spisek, G. Kroemer and L. Galluzzi, Calreticulin and cancer, Cell Res 31: ((2021) ), 5–16. |
[30] | Q. Gao, S. Wang, X. Chen, S. Cheng, Z. Zhang, F. Li, L. Huang, Y. Yang, B. Zhou, D. Yue, D. Wang, L. Cao, N.R. Maimela, B. Zhang, J. Yu, L. Wang and Y. Zhang, Cancer-cell-secreted CXCL11 promoted CD8(+) T cells infiltration through docetaxel-induced-release of HMGB1 in NSCLC, J Immunother Cancer 7: ((2019) ), 42. |
[31] | P. Giglio, M. Gagliardi, N. Tumino, F. Antunes, S. Smaili, D. Cotella, C. Santoro, R. Bernardini, M. Mattei, M. Piacentini and M. Corazzari, PKR and GCN2 stress kinases promote an ER stress-independent eIF2α phosphorylation responsible for calreticulin exposure in melanoma cells, Oncoimmunology 7: ((2018) ), e1466765. |
[32] | M. Moller, S. Turzer, G. Ganchev, A. Wienke, W. Schutte, B. Seliger and D. Riemann, Blood immune cell biomarkers in lung cancer patients undergoing treatment with a combination of chemotherapy and immune checkpoint blockade, Cancers (Basel) 14: ((2022) ). |
[33] | E. Abdelfatah, M.D. Long, R. Kajihara, T. Oba, T. Yamauchi, H. Chen, J. Sarkar, K. Attwood, J. Matsuzaki, B.H. Segal, G.K. Dy and F. Ito, Predictive and prognostic implications of circulating CX3CR1(+) CD8(+) T cells in non-small cell lung cancer patients treated with chemo-immunotherapy, Cancer Res Commun 3: ((2023) ), 510–520. |
[34] | A. Lopez-Soto, S. Gonzalez, M.J. Smyth and L. Galluzzi, Control of metastasis by NK cells, Cancer Cell 32: ((2017) ), 135–154. |
[35] | P. Sathe, R.B. Delconte, F. Souza-Fonseca-Guimaraes, C. Seillet, M. Chopin, C.J. Vandenberg, L.C. Rankin, L.A. Mielke, I. Vikstrom, T.B. Kolesnik, S.E. Nicholson, E. Vivier, M.J. Smyth, S.L. Nutt, S.P. Glaser, A. Strasser, G.T. Belz, S. Carotta and N.D. Huntington, Innate immunodeficiency following genetic ablation of Mcl1 in natural killer cells, Nat Commun 5: ((2014) ), 4539. |
[36] | J.P. Böttcher, E. Bonavita, P. Chakravarty, H. Blees, M. Cabeza-Cabrerizo, S. Sammicheli, N.C. Rogers, E. Sahai, S. Zelenay and C. Reis e Sousa, NK cells stimulate recruitment of cDC1 into the tumor microenvironment promoting cancer immune control, Cell 172: ((2018) ), 1022–1037e14.. |
[37] | A.J. Freeman, S.J. Vervoort, K.M. Ramsbottom, M.J. Kelly, J. Michie, L. Pijpers, R.W. Johnstone, C.J. Kearney and J. Oliaro, Natural killer cells suppress T cell-associated tumor immune evasion, Cell Rep 28: ((2019) ), 2784–2794e5.. |
[38] | C. Laheurte, M. Dosset, D. Vernerey, L. Boullerot, B. Gaugler, E. Gravelin, V. Kaulek, M. Jacquin, L. Cuche, G. Eberst, P. Jacoulet, E. Fabre, F. Le Pimpec-Barthes, E. Tartour, M. De Carvalho Bittencourt, V. Westeel and O. Adotevi, Distinct prognostic value of circulating anti-telomerase CD4(+) Th1 immunity and exhausted PD-1(+)/TIM-3(+) T cells in lung cancer, Br J Cancer 121: ((2019) ), 405–416. |
[39] | E.J. Wherry, T cell exhaustion, Nat Immunol 12: ((2011) ), 492-9. |
[40] | A.M. Miggelbrink, J.D. Jackson, S.J. Lorrey, E.S. Srinivasan, J. Waibl-Polania, D.S. Wilkinson and P.E. Fecci, CD4 T-Cell Exhaustion: Does It Exist and What Are Its Roles in Cancer? Clin Cancer Res 27: ((2021) ), 5742-5752. |
[41] | C.C. Balança, A. Salvioni, C.M. Scarlata, M. Michelas, C. Martinez-Gomez, C. Gomez-Roca, V. Sarradin, M. Tosolini, C. Valle, F. Pont, G. Ferron, L. Gladieff, S. Vergez, A. Dupret-Bories, E. Mery, P. Rochaix, J.J. Fournié, J.P. Delord, C. Devaud, A. Martinez and M. Ayyoub, PD-1 blockade restores helper activity of tumor-infiltrating, exhausted PD-1hiCD39+ CD4 T cells, JCI Insight 6: ((2021) ). |
[42] | E. Wennerberg, V. Kremer, R. Childs and A. Lundqvist, CXCL10-induced migration of adoptively transferred human natural killer cells toward solid tumors causes regression of tumor growth in vivo, Cancer Immunol Immunother 64: ((2015) ), 225–35. |
[43] | I.G. House, P. Savas, J. Lai, A.X.Y. Chen, A.J. Oliver, Z.L. Teo, K.L. Todd, M.A. Henderson, L. Giuffrida, E.V. Petley, K. Sek, S. Mardiana, T.N. Gide, C. Quek, R.A. Scolyer, G.V. Long, J.S. Wilmott, S. Loi, P.K. Darcy and P.A. Beavis, Macrophage-derived CXCL9 and CXCL10 are required for antitumor immune responses following immune checkpoint blockade, Clin Cancer Res 26: ((2020) ), 487-504. |
[44] | M. Inomata, T. Kado, S. Okazawa, S. Imanishi, C. Taka, K. Kambara, T. Hirai, H. Tanaka, K. Tokui, K. Hayashi, T. Miwa, R. Hayashi, S. Matsui and K. Tobe, Peripheral PD1-positive CD4 T-Lymphocyte count can predict progression-free survival in patients with non-small cell lung cancer receiving immune checkpoint inhibitor, Anticancer Res 39: ((2019) ), 6887–6893. |
[45] | M. Aldarouish, X. Su, J. Qiao, C. Gao, Y. Chen, A. Dai, T. Zhang, Y. Shu and C. Wang, Immunomodulatory effects of chemotherapy on blood lymphocytes and survival of patients with advanced non-small cell lung cancer, Int J Immunopathol Pharmacol 33: ((2019) ), 2058738419839592. |
[46] | S.H. Kim, J.W. Kim, I.G. Hwang, J.S. Jang, S. Hong, T.Y. Kim, J.Y. Baek, S.H. Shin, S. Sun, D.S. Hong, H.J. Kim, Y.S. Hong, I.S. Woo, J.H. Lee and J.H. Kim, Serum biomarkers for predicting overall survival and early mortality in older patients with metastatic solid tumors, J Geriatr Oncol 10: ((2019) ), 749–756. |
[47] | H. Zheng, X. Liu, J. Zhang, S.J. Rice, M. Wagman, Y. Kong, L. Zhu, J. Zhu, M. Joshi and C.P. Belani, Expression of PD-1 on CD4+ T cells in peripheral blood associates with poor clinical outcome in non-small cell lung cancer, Oncotarget 7: ((2016) ), 56233–56240. |




