Evaluation of hsa_circ_0000018/let-7f-5p/ FAM96A axis in lung adenocarcinoma progression
Abstract
BACKGROUND:
Circular RNAs (circRNAs) are critical regulators of lung adenocarcinoma (LA) progression. Although a molecular marker targeting hsa_circ_0000018 has been developed and used for diagnosing colon cancer, the role of this circRNA in LA progression has not been explored till now.
OBJECTIVES:
This study aimed to elucidate the role and regulatory mechanisms of hsa_circ_0000018 in LA progression.
METHODS:
LA tissues and corresponding adjacent non-tumor tissues were collected from 36 patients to confirm the levels of circRNAs, microRNAs (miRNAs), and messenger RNAs (mRNAs) using quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR). We also cultured two LA cell lines (A549, PC-9), and the human normal lung epithelial cell line BEAS-2B. Cell function experiments were conducted to assess malignancy in LA cells, including proliferation, migration, and invasion, following forced hsa_circ_0000018 expression. The correlation between hsa_circ_0000018, let-7f-5p, and family with sequence similarity 96 member A (FAM96A) was confirmed by using starBase (miRNA-circRNA interaction database), luciferase assay, and western blotting.
RESULTS:
Expression of hsa_circ_0000018 and FAM96A was reduced, whereas that of let-7f-5p was upregulated in LA. Cell function assays revealed that upregulation of hsa_circ_0000018 had a suppressive effect on the proliferation, migration, and invasion of LA cells. Additionally, hsa_circ_0000018 sponge binds let-7f-5p, resulting in upregulation of FAM96A expression.
CONCLUSION:
Our data reveal hsa_circ_0000018 as a tumor suppressor in LA that targets the let-7f-5p/FAM96A axis. Our findings enrich the known regulatory network of circRNAs in LA.
1.Introduction
Lung cancer is a highly prevalent form of cancer worldwide and is further classified into small-cell and non-small cell lung cancer (NSCLC) categories [1]. Lung adenocarcinoma (LA) is the most common subtype of lung cancer, accounting for 45% of all lung cancer cases globally [2]. With improvements in the diagnosis and treatment of LA, patients with LA have longer survival times. However, the prognosis of LA patients remains poor because most cases are diagnosed with LA at late stage when cancer treatments are challenging to deliver [3]. Therefore, it is necessary to identify the critical regulators of LA progression.
Circular RNAs (circRNAs) are a type of non-coding RNA with a covalently bonded closed loop structure that lacks a 5’-cap and a 3’-polyadenylated tail [4]. The circRNA-microRNA (miRNA)- messenger RNA (mRNA) regulatory network is known to play a role in tumor progression, and circRNAs are implicated in this process [5, 6, 7]. For example, hsa_circ_104348 is an oncogenic circRNA in hepatocellular carcinoma that regulates the miR-184-3p/RTKN2 axis [7]. The hsa_circ_101237-miR-490-3p-mitogen-activated protein kinase-1 (MAPK1) network participates in NSCLC tumorigenesis by regulating cell malignancy [8]. CircRNAs circ_0029426 and circ_0007618 show potential as biomarkers for the diagnosis and prognosis of LA [9]. In addition, the knockdown of hsa_circ_0020850 represses LA development by regulating miR-195-5p to enhance insulin receptor substrate 2 (IRS2) expression [10]. Hsa_circ_0020850 suppresses LA progression by targeting the miR-6783-3p/DKK1 axis [11]. Hsa_circ_0000018 (alternative name: hsa_circRNA_00054), located on chr1:15860731-15863309, is downregulated in colon cancer, and a molecular marker targeting hsa_circ_0000018 has been developed for the diagnosis of colon cancer [12]. After detecting the hsa_circ_0000018 expression in lung adenocarcinoma, we found that it was also downregulated in LA. However, a literature search revealed that its function has not been explored in any cancer. Therefore, we hypothesized that the abnormal expression of hsa_circ_0000018 may play a key role in LA and decided to explore its effect and mechanism of action in this cancer.
MiRNAs are short non-coding RNAs that range from 19 to 23 nucleotides in length and can bind to the mRNAs of their target genes, thereby modulating their expression [13]. The miRNA let-7f-5p has been found to act as an oncogenic agent in prostate cancer [14] and a tumor suppressor in osteosarcoma [15]. In lung cancer, let-7f-5p is upregulated in typical and atypical carcinoid tumors; however, its function and mechanism in LA have not been investigated [16]. Using the online tool starBase (miRNA-circRNA interaction database), we predicted that let-7f-5p binds to hsa_circ_0000018. Therefore, we explored the expression of let-7f-5p and its relationship with hsa_circ_0000018 expression in LA.
Family with sequence similarity 96 member A (FAM96A) is a protein belonging to the cytosolic Fe/S protein family and plays a crucial role in biological processes. Previous studies have confirmed that FAM96A suppresses tumor growth in both hepatocellular carcinoma [17] and gastrointestinal stromal tumors [18] via a proapoptotic process. However, the function of FAM96A in LA has not been elucidated.
Based on these previous studies, we speculated that hsa_circ_0000018, let-7f-5p, and FAM96A may be critical regulators of LA. To confirm this hypothesis, we performed a series of cell function experiments to explore the regulatory networks involving hsa_circ_0000018, let-7f-5p, and FAM96A. Our research has the potential to enhance an understanding of the complex regulatory mechanisms underlying LA and could have implications for the development of more effective diagnostic and therapeutic strategies.
2.Material and methods
2.1Clinical tissues
Our study involved the collection of LA tissues and corresponding adjacent non-tumor tissues from 36 patients who had been diagnosed with LA at Puren Hospital in China between January 2021 and January 2022. The Ethics Committee of Puren Hospital approved the study protocol (Approval Number: prll2021019), and all participants provided written informed consent. Patients who were diagnosed with LA and did not undergo any treatment were included, whereas those with a history of LA or other diseases were excluded. Additional information regarding the clinical characteristics of the patients is provided in Supplementary Table 1.
2.2Cell lines
The normal human lung epithelial cell line BEAS-2B (Catalog number: CL-0496) LA cell lines A549 (Catalog number: CL-0016), and PC-9 (Catalog number: CL-0668) were purchased from Procell (China). BEAS-2B cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM, ProCell), whereas A549 and PC-9 cells were maintained in Ham’s F-12K medium (ProCell) and RPMI-1640 medium (ProCell), respectively. All cells were maintained in 10% fetal bovine serum (FBS; Procell) in a cell culture incubator in an atmosphere of 5% CO2 and at 37∘C.
2.3Quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR)
The expression of hsa_circ_0000018, FAM96A, and let-7f-5p in LA was confirmed using qRT-PCR. RNA was extracted using RNAiso Plus (TAKARA, Japan) and cDNA was synthesized using the SuperScript Reverse Transcriptase Kit (Vazyme, Nanjing, China). qRT-PCR was conducted with the help of SYBR Premix Ex Taq kit purchased from TAKARA (Japan) under the following conditions: 95∘C for 30 s, 40 cycles of 95∘C for 5 s, and 60∘C for 30 s. The expression of hsa_circ_0000018, FAM96A, and let-7f-5p was calculated by using the 2-ΔΔCt method as described previously (20) with GAPDH as an internal reference for hsa_circ_0000018 and FAM96A, and U6 for let-7f-5p. Primer sequences are listed in Supplementary Table 2. The experiment was independently repeated thrice.
2.4RNA enzyme digestion assay
RNA enzyme digestion assays are used to confirm the closed-loop structure of RNA molecules because RNA enzymes can digest linear RNA, not circRNAs. Briefly, the RNA from A549 and PC-9 cells was incubated with RNase R (Epicenter Technologies, USA) at 37∘C for 30 min to assess the stability of hsa_circ_0000018 and linear RNA DNAJC16 [19]. After incubation, qRT-PCR was performed to detect the levels of hsa_circ_0000018 and DNAJC16 in LA cells. The experiment was independently repeated thrice.
2.5Cell transfection
The hsa_circ_0000018 overexpression vector(pcDNA 3.1-circ) was constructed by RiboBio (China) using the pcDNA 3.1 circRNA Mini vector. The pcDNA 3.1 circRNA mini vector without the hsa_circ_0000018 overexpression sequences was used as a negative control. Let-7f-5p mimic, mimic-NC, siRNA targeting FAM96A (si-FAM96A), and si-NC (negative control siRNA) were constructed by RiboBio. A549 and PC-9 cells at 60% confluence were transfected with pcDNA 3.1-circ, pcDNA 3.1, let-7f-5p mimic, mimic-NC, si-FAM96A, or si-NC using Lipo6000 (Beyotime, China) and incubated for 48 h. The transfection efficiency was determined by qRT-PCR. The experiment was independently repeated thrice.
2.6Cell proliferation assay
Cell proliferation was assessed using the Cell Counting Kit-8 (CCK8) assay. In a 96-well plate, 2
2.7Transwell assay to assess cell migration and invasion
A transwell assay was performed to evaluate the migratory and invasive capabilities of LA cells as described previously [10]. Transfected LA cells (1
2.8Luciferase assay
The binding sites for hsa_circ_0000018, let-7f-5p, and FAM96A were predicted using StarBase database (https://starbase.sysu.edu.cn/index.php). Based on these predictions, wild-type (WT) vectors of hsa_circ_0000018 and FAM96A carrying binding sites for let-7f-5p were constructed using psiCHECK-2 vectors from RiboBio (China). The mutant (MUT) vectors for hsa_circ_0000018 and FAM96A that lacked the binding sites for let-7f-5p were constructed with psiCHECK-2 vectors by RiboBio. The WT or MUT vectors were transfected into LA cells along with the let-7f-5p mimic or mimic-NC using Lipo6000 (Beyotime, China). Luciferase activity was verified using the Dual-Luciferase Reporter Assay System (Beyotime) after a 48-hour transfection period as described previously [10]. The experiment was independently repeated thrice.
2.9Western blotting
The RIPA buffer (Beyotime, China) was used to extract proteins from LA cells that were quantified using a bicinchoninic acid protein assay kit (Beyotime). Next, 20
2.10Statistical methods
Statistical analysis was conducted by using GraphPad Prism 7.0 software and data are shown as mean
3.Results
3.1Downregulation of hsa_circ_0000018 in LA tissues and cells
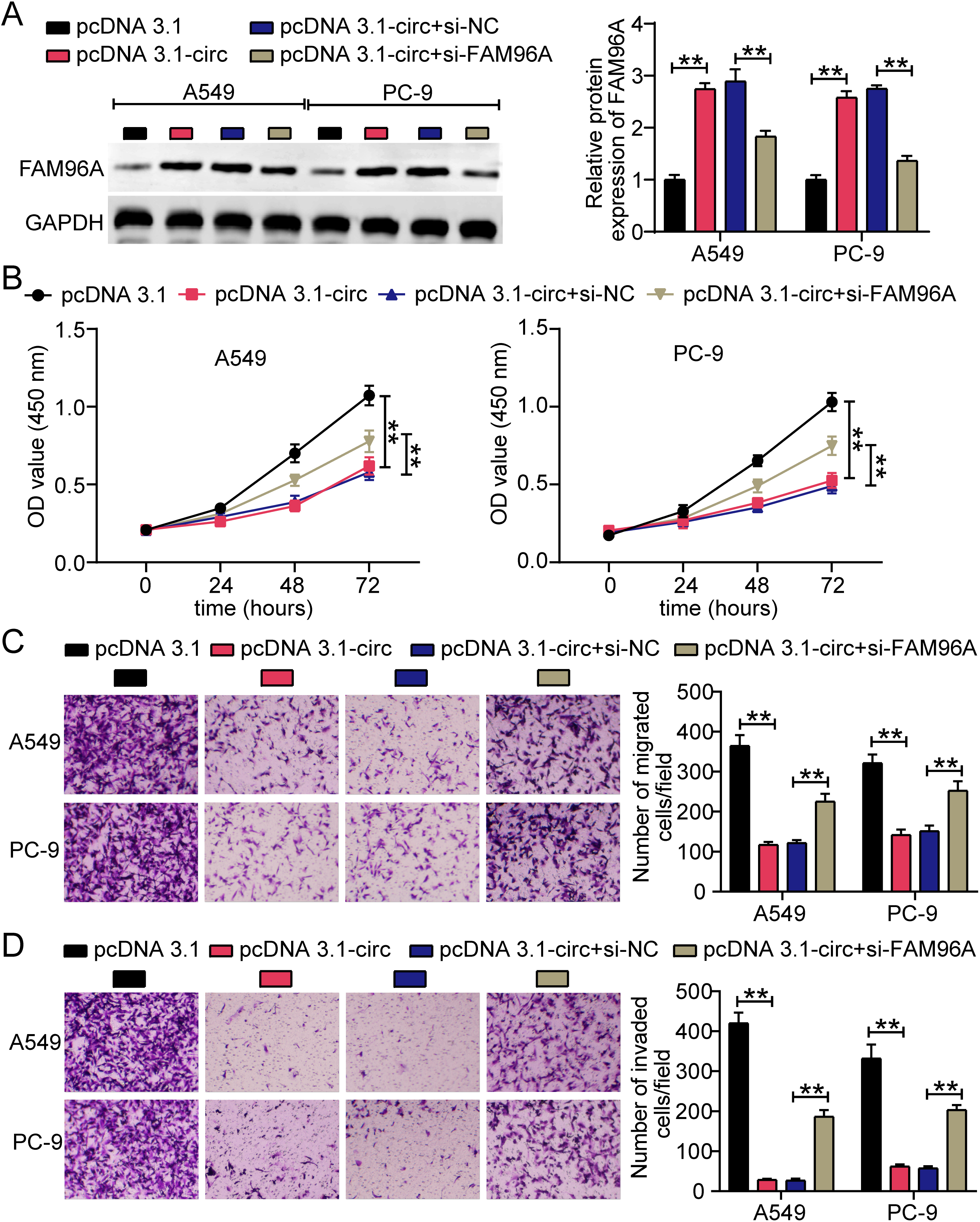
The initial approach used to investigate the role of hsa_circ_0000018 in LA was to assess its expression using qRT-PCR. The results showed that hsa_circ_0000018 expression was reduced by 50% in LA tissues compared to that in adjacent normal tissues (Fig. 1A) and in LA cells (A549 and PC-9) compared to that in BEAS-2B cells (Fig. 1B). A review of the structure of hsa_circ_0000018 (Fig. 1C) showed that its length was 407 nt, and that DNAJC16 was a linear gene of hsa_circ_0000018. In addition, RNase R digestion reduced DNAJC16 levels to a greater extent than hsa_circ_0000018 digestion, proving that hsa_circ_0000018 had a stable closed-loop structure (Fig. 1D). These data proved that hsa_circ_0000018, with a stable closed-loop structure, was downregulated in LA.
Figure 1.
The downregulation of hsa_circ_0000018 in LA. (A) qRT-PCR was used to analyze hsa_circ_0000018 expression in LA tissues and adjacent normal tissues. (B) qRT-PCR was used to analyze hsa_circ_0000018 expression in LA cell lines (A549 and PC-9) and human normal lung epithelial cell line BEAS-2B. (C) The construction of hsa_circ_0000018. (D) The stability of hsa_circ_0000018 was determined by RNA enzyme digestion assay. **P < 0.01.
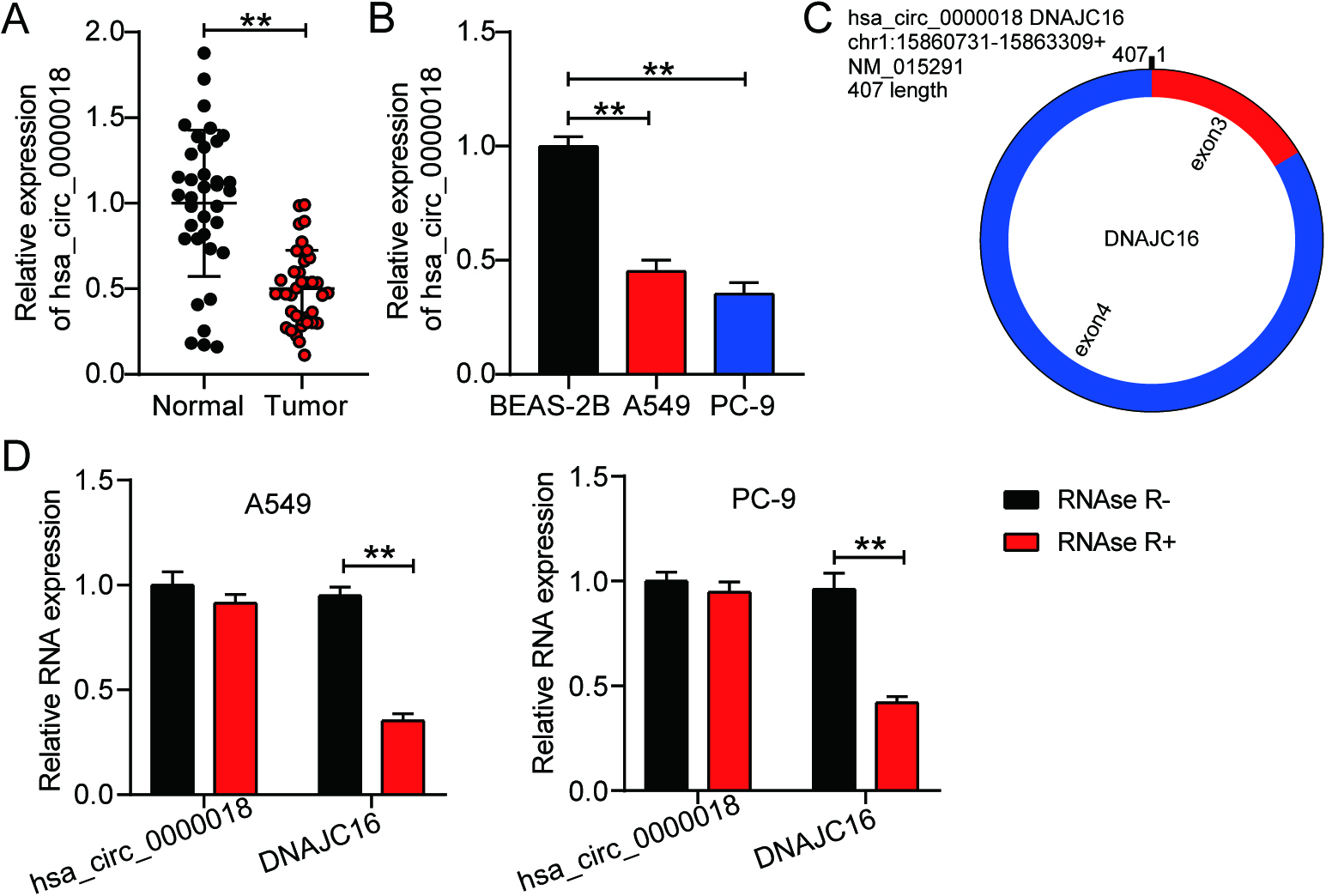
Figure 2.
Hsa_circ_0000018 overexpression inhibits LA progression in vitro. (A) The transfection efficiency of pcDNA 3.1-circ was verified in LA cell lines (A549 and PC-9) using qRT-PCR. (B) The effect of pcDNA 3.1-circ transfection on cell proliferation was assessed using the CCK8 assay in LA cells. (C-D) The migration and invasion abilities of LA cells were evaluated using Transwell assays after transfection with pcDNA 3.1-circ to determine the effect of hsa_circ_0000018 on cell migration (C) and invasion (D). **P < 0.01.
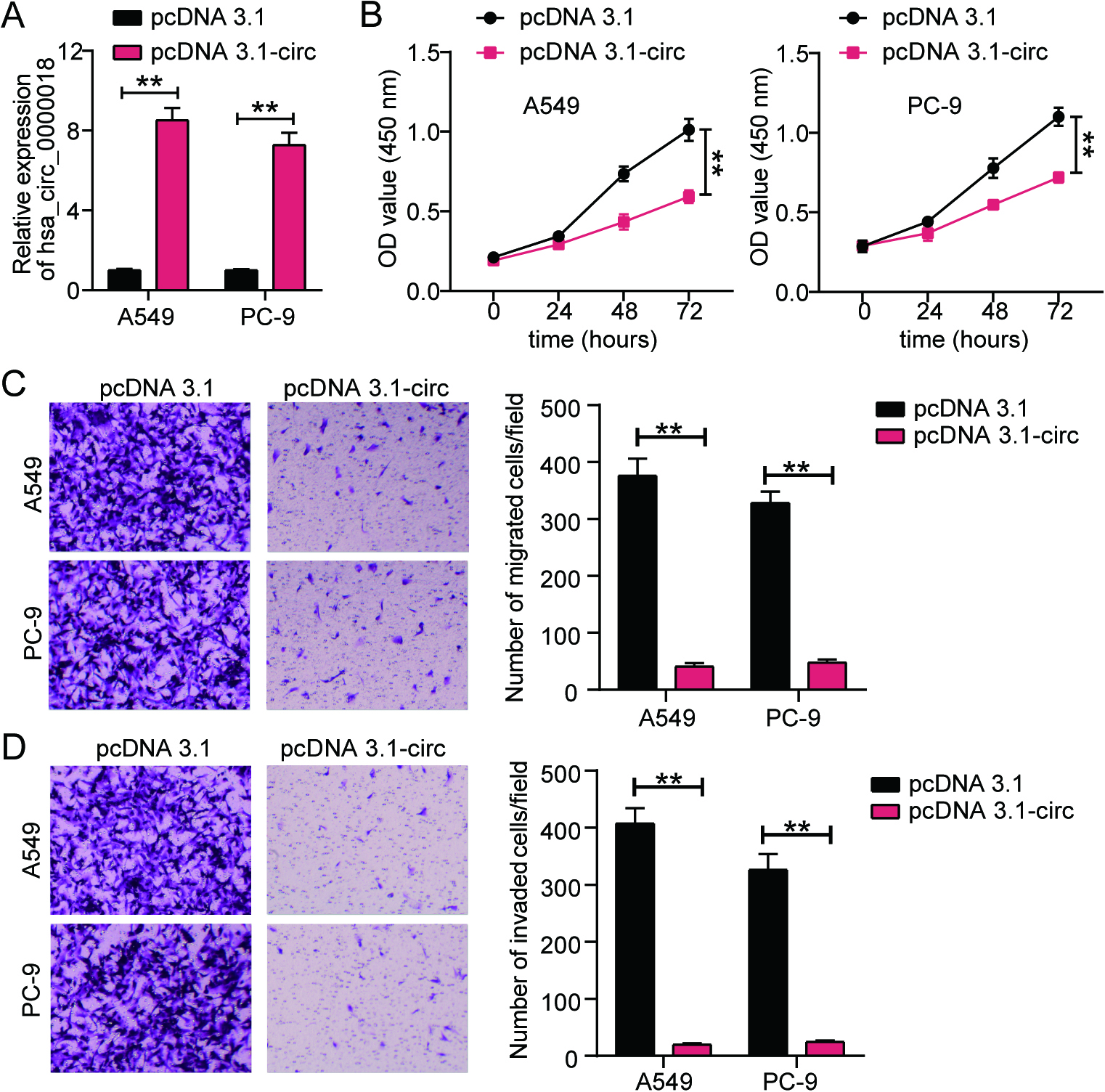
Figure 3.
Hsa_circ_0000018 targets let-7f-5p/FAM96A axis in LA. (A) The prediction of potential binding sites between hsa_circ_0000018 and let-7f-5p using starBase. (B) Luciferase assay further validated the existence of binding sites between hsa_circ_0000018 and let-7f-5p. (C) The prediction of binding sites between FAM96A and let-7f-5p using the starBase database. (D) Luciferase assay further verified the existence of binding sites between FAM96A and let-7f-5p. (E) The expression of let-7f-5p and FAM96A in LA tissues and adjacent normal tissues was detected by using qRT-PCR. (F) Pearson correlation analysis was used to quantify the correlation between hsa_circ_0000018, let-7f-5p, and FAM96A. **P < 0.01.
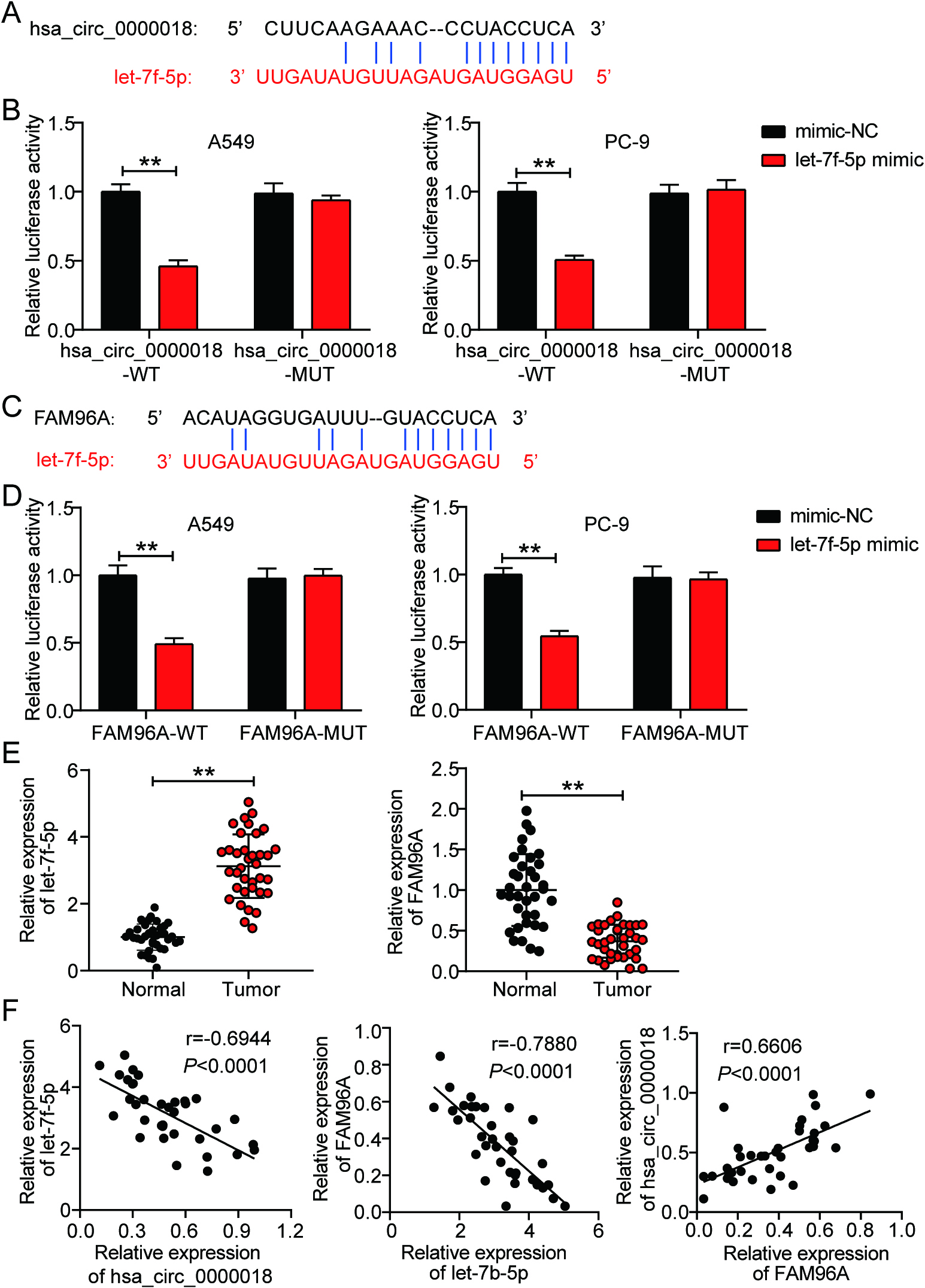
Figure 4.
FAM96A knockdown rescued the inhibitory effect of hsa_circ_0000018 overexpression on LA cells. (A) Western blotting was conducted to verify the expression of FAM96A protein in transfected A549 and PC-9 cells. (B) The cell proliferation ability of transfected A549 and PC-9 cells was evaluated using CCK8 assay. (C-D) The cell migration (C) and invasion (D) abilities of transfected A549 and PC-9 cells were identified using transwell assays. pcDNA 3.1-circ, hsa_circ_0000018 overexpression vector. si-NC, negative control siRNA. si-FAM96A, siRNA targeting FAM96A. **P < 0.01.
3.2hsa_circ_0000018 overexpression inhibits LA progression in vitro
After constructing the hsa_circ_0000018 overexpression vectors using pcDNA 3.1, we found that pcDNA 3.1-circ upregulated hsa_circ_0000018 expression in both A549 and PC-9 cells (Fig. 2A). A series of cell experiments were conducted to demonstrate alterations in LA cell function after overexpression of hsa_circ_0000018. The CCK8 assay showed that hsa_circ_0000018 overexpression attenuated the proliferation of A549 and PC-9 cells (Fig. 2B). In addition, LA cells transfected with pcDNA 3.1-circ showed impaired migration and invasion abilities, as demonstrated by the transwell assay results (Fig. 2C and D). The results of in vitro experiments suggested that hsa_circ_0000018 was an oncogenic circRNA in LA cells.
3.3hsa_circ_0000018 targets let-7f-5p/FAM96A axis in LA
Prediction analysis using starBase revealed the presence of binding sites between let-7f-5p and hsa_circ_0000018 (Fig. 3A). We constructed hsa_circ_0000018-WT and hsa_circ_0000018-MUT vectors with and without binding sites, respectively, to perform a luciferase assay. In the hsa_circ_0000018-WT group, the let-7f-5p mimic reduced luciferase activity, whereas no such effect was observed in the hsa_circ_0000018-MUT group (Fig. 3B). Similarly, starBase predicted the presence of binding sites between FAM96A and let-7f-5p (Fig. 3C), and luciferase activity was reduced only in the FAM96A-WT
3.4FAM96A knockdown reverses the effect of hsa_circ_0000018 overexpression on LA cells
Following transfection of pcDNA 3.1-circ and si-FAM96A into LA cells, western blotting confirmed that pcDNA 3.1-circ enhanced the level of FAM96A protein by
4.Discussion
According to a previous report, LA accounted for approximately 40% of all lung cancer cases diagnosed in 2019 worldwide [22]. Several studies have shown that circRNAs are critical regulators of tumorigenesis and LA progression. Zhou et al. observed that circRNA-ENO1 upregulates enolase 1 (a glycolytic enzyme) and accelerates LA progression by regulating glycolysis, cell proliferation, and epithelial-mesenchymal transition. In contrast to circRNA-ENO1, circDCUN1D4 suppresses tumor metastasis in LA by forming an RNA-protein ternary complex with HuR and TXNIP [23]. Hsa_circ_0000018, also known as hsa_circRNA_000541, is downregulated in colon cancer, and a molecular marker targeting hsa_circ_0000018 has been developed for the diagnosis of colon cancer [12]. Whether hsa_circ_0000018 promotes or inhibits tumorigenicity in LA has not yet been investigated. Here, we show for the first time that hsa_circ_0000018 is downregulated in LA and that its overexpression can effectively reduce the proliferation, migration, and invasion of LA cells. The findings of this study on the function of hsa_circ_0000018 in LA are consistent with those of a previous study on its function in colon cancer, suggesting that a molecular marker targeting hsa_circ_0000018 may also be developed for the diagnosis of LA. Additionally, many studies have shown that circRNAs can sponge the miRNA-mRNA axis, thereby participating in LA progression [10, 24, 25]. For example, hsa_circMMD_007 promotes LA progression by acting as an endogenous sponge for miR-197-3p [26]. In this study, we observed that hsa_circ_0000018 hindered cell malignancy in LA by acting as a sponge for let-7f-5p.
The miRNA let-7f-5p has been implicated in distinct roles in a variety of cancers. For example, let-7f-5p is an oncogenic miRNA in prostate cancer, yet functions as a tumor suppressor in osteosarcoma [15]. Di Fazio et al. analyzed let-7f-5p expression in both typical and atypical carcinoid tumors in lung cancer, confirming that let-7f-5p was upregulated in most tumor samples compared to normal lung tissues. However, the specific functions and mechanisms of action of let-7f-5p in LA have not been explored. The results of this study are consistent with the findings reported by Di Fazio et al., indicating an upregulation of let-7f-5p in LA samples. This study also showed that FAM96A is a target gene of let-7f-5p in LA cells, and that let-7f-5p expression was negatively correlated with both hsa_circ_0000018 and FAM96A in LA samples. Therefore, we hypothesized that let-7f-5p acts as a bridge connecting hsa_circ_0000018 and FAM96A in LA, thereby regulating FAM96A expression. Western blotting confirmed this hypothesis by showing that hsa_circ_0000018 overexpression enhanced FAM96A protein expression.
FAM96A, a cytosolic Fe/S protein, is a pro-apoptotic protein [27]. Evidence supports FAM96A as a tumor suppressor in gastrointestinal stromal tumors, showing that FAM96A re-expressed in gastrointestinal stromal tumors induces cell apoptosis and diminishes tumorigenicity [18]. FAM96A inhibits tumor growth in nude mice, thereby playing an anti-cancer role in hepatocellular carcinoma [17]. Although there have been no studies on the function of FAM96A in LA, we speculate that FAM96A may play an anticancer role in LA, based on a previous study. In our study, we demonstrated that FAM96A is downstream of hsa_circ_0000018 and that its expression can be enhanced by upregulating hsa_circ_0000018. Cell function experiments showed that FAM96A knockdown partially relieved the negative effects of hsa_circ_0000018 overexpression in LA cells.
Although our study elucidated the effect and underlying mechanism of hsa_circ_0000018 on LA cells, further investigation is required to determine whether hsa_circ_0000018 modulates LA progression in vivo. The clinical role of hsa_circ_0000018 in LA, such as in prognosis and drug treatment, warrants further investigation.
5.Conclusion
Our study revealed the role of hsa_circ_0000018 in LA and showed that hsa_circ_0000018 hindered LA progression in vitro by targeting the let-7f-5p/FAM96A axis. Our findings enrich the known regulatory network of circRNAs in LA and may aid in its diagnosis and treatment.
Funding
This work was supported by Wuhan Municipal Health Commission Medical Research Project (grant number: WX21D08).
Availability of data and materials
Not applicable.
Authors contributions
Interpretation or analysis of data: MZ, DDH, and JJQ.
Preparation of the manuscript: QL.
Revision for important intellectual content: WH.
Supervision: all authors.
All authors have read and approved this manuscript.
Ethics approval and consent to participate
All procedures conducted in this study were in compliance with the principles outlined in the Declaration of Helsinki and were approved by the Ethics Committee of Puren Hospital (Approval number: prll2021019). Additionally, this study obtained written informed consent from all patients.
Consent for publication
Consent for publication was obtained from the participants.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230111.
Acknowledgments
Not applicable.
Conflict of interest
The authors have no conflicts of interest related to this research.
References
[1] | Y. Mao, D. Yang, J. He and M.J. Krasna, Epidemiology of lung cancer, Surg Oncol Clin N Am 25: ((2016) ), 439–445. |
[2] | S. Navada, P. Lai, A. Schwartz and G. Kalemkerian, Temporal trends in small cell lung cancer: Analysis of the national Surveillance, Epidemiology, and End-Results (SEER) database, Journal of Clinical Oncology 24: ((2006) ), 7082–7082. |
[3] | T. Tarver, Cancer facts & figures 2012, American cancer society (ACS) Atlanta, GA: American Cancer Society, 2012. 66 p., pdf. Available from, Taylor & Francis, (2012) . |
[4] | L.L. Chen and L. Yang, Regulation of circRNA biogenesis, RNA Biol 12: ((2015) ), 381–388. |
[5] | M. Zhang, X. Bai, X. Zeng, J. Liu, F. Liu and Z. Zhang, circRNA-miRNA-mRNA in breast cancer, Clin Chim Acta 523: ((2021) ), 120–130. |
[6] | Y. Liu, S. Chen, Z.H. Zong, X. Guan and Y. Zhao, CircRNA WHSC1 targets the miR-646/NPM1 pathway to promote the development of endometrial cancer, J Cell Mol Med 24: ((2020) ), 6898–6907. |
[7] | G. Huang, M. Liang, H. Liu, J. Huang, P. Li, C. Wang, Y. Zhang, Y. Lin and X. Jiang, CircRNA hsa_circRNA_104348 promotes hepatocellular carcinoma progression through modulating miR-187-3p/RTKN2 axis and activating Wnt/beta-catenin pathway, Cell Death Dis 11: ((2020) ), 1065. |
[8] | Z.Y. Zhang, X.H. Gao, M.Y. Ma, C.L. Zhao, Y.L. Zhang and S.S. Guo, CircRNA_101237 promotes NSCLC progression via the miRNA-490-3p/MAPK1 axis, Sci Rep 10: ((2020) ), 9024. |
[9] | Q. Zhang, Y. Sun, C. Wang and F. Shao, Circular RNA-microRNA-mRNA network identified circ_0007618 and circ_0029426 as new valuable biomarkers for lung adenocarcinoma, Bioengineered 13: ((2022) ), 6258–6271. |
[10] | T. Xin, S. Li, Y. Zhang, X. Kamali, H. Liu and T. Jia, circRNA Hsa_circ_0020850 Silence Represses the Development of Lung Adenocarcinoma via Regulating miR-195-5p/IRS2 Axis, Cancer Manag Res 12: ((2020) ), 10679–10692. |
[11] | Y. Yao, Q. Hua and Y. Zhou, CircRNA has_circ_0006427 suppresses the progression of lung adenocarcinoma by regulating miR-6783-3p/DKK1 axis and inactivating Wnt/beta-catenin signaling pathway, Biochem Biophys Res Commun 508: ((2019) ), 37–45. |
[12] | C. Zhou, J. Li, T. Bao and T. Gao, Molecular markers and detection reagents for circrnas in colorectal cancer, China Patent CN 201610518307 [P], 2016-10-26. |
[13] | J. Krol, I. Loedige and W. Filipowicz, The widespread regulation of microRNA biogenesis, function and decay, Nat Rev Genet 11: ((2010) ), 597–610. |
[14] | Y. Ge, Q. Wang, W. Shao, Y. Zhao, Q. Shi, Q. Yuan and L. Cui, Circulating let-7f-5p improve risk prediction of prostate cancer in patients with benign prostatic hyperplasia, J Cancer 11: ((2020) ), 4542–4549. |
[15] | G. Chen, H. Gu, T. Fang, K. Zhou, J. Xu and X. Yin, Hypoxia-induced let-7f-5p/TARBP2 feedback loop regulates osteosar-coma cell proliferation and invasion by inhibiting the Wnt signaling pathway, Aging (Albany NY) 12: ((2020) ), 6891–6903. |
[16] | P. Di Fazio, M. Maass, S. Roth, C. Meyer, J. Grups, P. Rexin, D.K. Bartsch and A. Kirschbaum, Expression of hsa-let-7b-5p, hsa-let-7f-5p, and hsa-miR-222-3p and their putative targets HMGA2 and CDKN1B in typical and atypical carcinoid tumors of the lung, Tumour Biol 39: ((2017) ), 1010428317728417. |
[17] | W.P. Wang and H.Y. Gao, Combination therapy of hTERTR and FAM96A for hepatocellular carcinoma through enhancing apoptosis sensitivity, Exp Ther Med 15: ((2018) ), 641–648. |
[18] | B. Schwamb, R. Pick, S.B. Fernandez, K. Volp, J. Heering, V. Dotsch, S. Bosser, J. Jung, R. Beinoraviciute-Kellner, J. Wesely, I. Zornig, M. Hammerschmidt, M. Nowak, R. Penzel, K. Zatloukal, S. Joos, R.J. Rieker, A. Agaimy, S. Soder, K.M. Reid-Lombardo, M.L. Kendrick, M.R. Bardsley, Y. Hayashi, D.T. Asuzu, S.A. Syed, T. Ordog and M. Zornig, FAM96A is a novel pro-apoptotic tumor suppressor in gastrointestinal stromal tumors, Int J Cancer 137: ((2015) ), 1318–1329. |
[19] | J. Wang, X. Zhao, Y. Wang, F. Ren, D. Sun, Y. Yan, X. Kong, J. Bu, M. Liu and S. Xu, circRNA-002178 act as a ceRNA to promote PDL1/PD1 expression in lung adenocarcinoma, Cell Death Dis 11: ((2020) ), 32. |
[20] | Q. Huang, H. Guo, S. Wang, Y. Ma, H. Chen, H. Li, J. Li, X. Li, F. Yang, M. Qiu, S. Zhao and J. Wang, A novel circular RNA, circXPO1, promotes lung adenocarcinoma progression by interacting with IGF2BP1, Cell Death Dis 11: ((2020) ), 1031. |
[21] | X. Zhang, T. Liu, S. Zheng, Q. Liu, T. Shen, X. Han, Q. Zhang, L. Yang and X. Lu, SUMOylation of HSP27 regulates PKM2 to promote esophageal squamous cell carcinoma progression, Oncol Rep 44: ((2020) ), 1355–1364. |
[22] | D. Kerdidani, P. Chouvardas, A.R. Arjo, I. Giopanou, G. Ntaliarda, Y.A. Guo, M. Tsikitis, G. Kazamias, K. Potaris, G.T. Stathopoulos, S. Zakynthinos, I. Kalomenidis, V. Soumelis, G. Kollias and M. Tsoumakidou, Wnt1 silences chemokine genes in dendritic cells and induces adaptive immune resistance in lung adenocarcinoma, Nat Commun 10: ((2019) ), 1405. |
[23] | Y. Liang, H. Wang, B. Chen, Q. Mao, W. Xia, T. Zhang, X. Song, Z. Zhang, L. Xu, G. Dong and F. Jiang, circDCUN1D4 suppresses tumor metastasis and glycolysis in lung adenocarcinoma by stabilizing TXNIP expression, Mol Ther Nucleic Acids 23: ((2021) ), 355–368. |
[24] | S.J. Zhang, J. Ma, J.C. Wu, Z.Z. Hao, Y.N. Zhang and Y.J. Zhang, CircRNA EPB41L2 inhibits tumorigenicity of lung adenocarcinoma through regulating CDH4 by miR-211-5p, Eur Rev Med Pharmacol Sci 25: ((2021) ), 3150. |
[25] | S. Wang, H. Zhang, L. Xia and F. Lan, Circular RNA circ_0061140 accelerates hypoxia-induced glycolysis, migration, and invasion in lung adenocarcinoma through the microRNA-653/hexokinase 2 (HK2) axis, Bioengineered 13: ((2022) ), 7156–7166. |
[26] | L. Zhu, T. Guo, W. Chen, Z. Lin, M. Ye and X. Pan, CircMMD_007 promotes oncogenic effects in the progression of lung adenocarcinoma through microRNA-197-3p/protein tyrosine phosphatase non-receptor type 9 axis, Bioengineered 13: ((2022) ), 4991–5004. |
[27] | O. Stehling, J. Mascarenhas, A.A. Vashisht, A.D. Sheftel, B. Niggemeyer, R. Rosser, A.J. Pierik, J.A. Wohlschlegel and R. Lill, Human CIA2A-FAM96A and CIA2B-FAM96B integrate iron homeostasis and maturation of different subsets of cytosolic-nuclear iron-sulfur proteins, Cell Metab, (2013) , 187–198. |




