Hsa_circRNA_001859 regulates pancreatic cancer progression and epithelial-mesenchymal transition through the miR-21-5p/SLC38A2 pathway
Abstract
OBJECTIVE:
This study attempts to investigate whether hsa_circRNA_001859 (circ_001859) could regulate the proliferation and invasion of pancreatic cancer through the miR-21-5p/SLC38A2 pathway.
METHODS:
GSE79634 microarray was analyzed with R package. The expression of circ_001859 in pancreatic cancer tissues and cells was verified by qRT-PCR. After the overexpression of circ_001859, cell proliferation, cell migration and invasion were verified by colony formation and transwell assay. The targeting relationship between miR-21-5p and circ_001859 was predicted by TargetScan and was verified by dual luciferase reporter assay, RNA pull down and qRT-PCR. The effect of miR-21-5p on cell proliferation, migration and invasion were investigated by colony formation and transwell assay respectively. Similarly, the targeting relationship between miR-21-5p and SLC38A2 was predicted by TargetScan and was verified by dual luciferase reporter assay, western blot and qRT-PCR. The effect of SLC38A2 on cell proliferation was investigated by colony formation.
RESULTS:
Circ_001859 was lowly expressed in pancreatic cancer tissues and cells. In vitro assays showed that overexpression of circ_001859 could inhibit the proliferation, migration and invasion of pancreatic cancer. In addition, this effect was also confirmed in xenograft transplantation model. Circ_001859 could be bind to miR-21-5p and sponge its expression in pancreatic cancer cells. Overexpression of miR-21-5p enhanced the proliferation, migration and invasion ability of pancreatic cancer cells, while the inhibition of miR-21-5p expression suppressed these abilities. Moreover, miR-21-5p directly targeted at SLC38A2 and inhibited SLC38A2 expression levels while circ_001859 up-regulated SLC38A2 levels. SLC38A2 expression knockdown enhanced cell proliferation but SLC38A2 overexpression resulted in decreased proliferation, and effects of SLC38A2 could be rescued by miR-21-5p and circ_001859. In addition, both QRT-PCR and immunofluorescence confirmed that circ_001859 could regulate tumor epithelial-mesenchymal transition (EMT) through the miR-21-5p/SLC38A2 pathway.
CONCLUSIONS:
This study suggests that circ_001859 may inhibit the proliferation, invasion and EMT of pancreatic cancer through the miR-21-5p/SLC38A2 pathway.
1.Introduction
Pancreatic cancer is a leading cause of cancer death worldwide and its global burden has more than doubled over the past 25 years. The highest incidence regions for pancreatic cancer include North America, Europe and Australia, and although much of this increase is due to ageing worldwide populations, there are key modifiable risk factors for pancreatic cancer such as cigarette smoking, obesity, diabetes and alcohol intake. The prevalence of these risk factors is increasing in many global regions, resulting in increasing age-adjusted incidence rates for pancreatic cancer [1, 2]. Although great progress in diagnosis and treatment has been made, the 5-year survival rate remains dismal in pancreatic cancer [3]. Pancreatic cancer will probably move to the second primary cause of cancer death around the world by 2020 in the absence of improvements in treatment [2, 4]. Currently, there is no way for the effective diagnosis of early stages of pancreatic cancer [4]. Under this grim situation, it is imperative to understand its molecular mechanisms connected with initiation, progression, and therapy obstacle [5]. In spite of present inspection into some well-known genes, the treatment of pancreatic cancer is still limited. Thus, this study focused on new molecular mechanism to solve treatment obstacle.
Circular RNAs (circRNAs), a novel kind of widespread and diverse endogenous RNAs, have a significant effect on the expression of gene at post-transcriptional level and are extensively expressed in body cells [6]. Currently, circRNAs have been found to be widely expressed in the cytoplasm with the advent of RNA sequencing technology and their complexity of regulation, specificity of expression, and vital function drew concerns in various diseases [7]. CircRNA_100782 promoted pancreatic carcinoma by sponging miR-124 through the IL6-STAT3 pathway [8]. Besides, circ_0000977 silencing suppressed pancreatic cancer cell proliferation and induced cell cycle arrest which was simulated by increased miR-874-3p expression and decreased PLK1 expression [9]. Both results suggested circRNA could be significant in pancreatic cancer diagnosis and treatment.
MicroRNAs (miRNAs), short noncoding RNAs 20–24 nucleotides in length, regulate the gene expression after transcription. Importantly, miRNAs play critical roles in a broad range of biological proceedings, containing proliferation, differentiation, apoptosis, and stress response, linking them with numerous human diseases [10]. In recent years, accumulating evidence has also indicated that the dysregulation of miRNAs in various human cancers may modulate tumor cell proliferation, tumor angiogenesis, invasion and metastasis during tumor initiation and progression [11]. Several researches have suggested that miR-21-5p may play an important part in different diseases, including breast cancer, colorectal cancer and lung cancer [12, 13, 14]. No researches have discussed the intriguing role of miR-21-5p in pancreatic cancer and this study for the first time studied this point.
The transport of glutamine across the plasma membrane is regulated by two systems: the system ASC transporter SLC1A5 and the system A transporter SLC38A1, SLC38A2 and SLC38A4. SLC1A5 is a two-way transporter that transports Ala, Ser, Cys, Asn and Gln in an exchange manner. MYC oncogene can induce the expression of SLC1A5, and it has been found that the high expression of SLC1A5 is related to the poor prognosis of many tumors [15, 16]. It is reported that other glutamine transporters, such as SLC38A1, SLC38A2 and SLC7A5, also play an important role in tumors. Other studies have shown that SLC38A2 plays an important role in pancreatic cancer [17]. These data showed that the tumor cells lacking SLC38A2 fail to concentrate intracellular alanine and undergo a profound metabolic crisis resulting in markedly impaired tumor growth [17]. but the role of miR-21-5p and SLC38A2 in pancreatic cancer is still unclear. However, whether the expression of SLC38A2 is regulated by miRNA or its relationship with miR-21-5p has not been found.
This study intends to make a thorough inquiry about the molecule and biological signaling pathway in pancreatic cancer. Both in vitro and in vivo assays were applied to verify the prediction of function of circ_001859. Besides, the regulation mechanism via circ_001859-miR-21-5p-SLC38A2 axis was analyzed. This study might give a new sight into molecular mechanisms in pancreatic cancer treatment.
2.Experimental methods
2.1Tissue samples
Twenty samples of pancreatic carcinoma tissues were obtained from patients with pancreatic cancer treated at The Second Affiliated Hospital Affiliated to Xinjiang Medical University. A total of 12 males and 8 females were included in this study. The average age was (57.91
2.2Microarray analysis
In the GEO database, GSE79634 was analyzed by chip using the GPL19978 platform (Agilent-069978 Arraystar Human CircRNA microarray) [18]. circRNA expression levels in 20 pancreatic ductal adenocarcinoma samples and 20 normal pancreatic cancer adjacent tissues were analyzed and distinguishable circRNAs were identified. The pathologic validation of all tumor tissues was confirmed by the research group of Wang et al. [18]. Differentially expressed circRNAs were chosen with Fold change values
2.3Cell culture
Pancreatic cancer cell line Panc-1, Capan-1, and normal ductal epithelial cell line HPDE6-C7 were acquired from the American Type Culture Collection (ATCC, Manassas, VA, USA) and maintained according to ATCC’s cell media recommendations in standard conditions (37
2.4Fluorescence in situ hybridization (FISH)
For FISH assay, Fluor 594-labeled probe for detecting hsa_circRNA_001859 and Alexa Fluor 488-labeled probes for detecting miR-21-5p were synthesized by GenePharma. After fixation, cells were incubated with pre-hybridization buffer, then, hybridization was conducted at 55
2.5RT-PCR
Total RNA of samples was extracted using TRIzol
2.6Western blot
Total protein solution was obtained by adding RAPI protein buffer (Beyotime, Shanghai, China). And the protein concentration was measured by Pierce BCA Protein Assay Kit (Pierce, Rockford, IL, USA). After denaturation, the protein received SDS-PAGE electrophoresis and was transferred to a PVDF membrane by using 200mA constant current for about 120 min. The membrane was blocked and incubated with primary antibodies (anti-SLC38A2 ab157492, 1:1000; anti-GAPDH, ab181603, 1:10000, Abcam, Cambridge, MA, USA) at 4
2.7Cell transfection
PcDNA3.1/circ_001859 (pcDNA3.1 as control), pcDNA3.1/SLC38A2 (pcDNA3.1 as control), miR-21-5p-mimics and miR-21-5p-inhibitor were purchased from Shanghai Biotech, Shanghai, China. Interfering nucleotide sequence was designed according to Invitrogen RNA interference sequence design software (BLOCK-iTTM RNAi Designer). At the same time, a negative control was designed according to an unrelated nucleic acid sequence which has the same base number. PANC-1 and Capan-1 cells were seeded in 6-well plates and cultured to 60–70% confluence before transfection, negative controls and overexpression vectors were transiently transfected using Lipofectamine 2000 (Invitrogen) according to instructions and then incubated in a 5% CO
2.8Colony formation
Transfected cells were seeded into a 6-well plate at a density of 1
2.9Transwell migration and invasion experiment
Transwell assay was carried out using 24-well transwell inserts (Corning, Corning, NY, USA) according to the manufacturer’s protocol. Transfected cells were collected, resuspended in serum-free medium and transferred to permeable pores. Cells were seeded at a density of 3
2.10Dual luciferase reporter assay
The circ_001859-wt, circ_001859-mut, SLC38A2-wt, and SLC38A2-mut obtained from Shanghai Biosciences were cloned into the pmirGLO plasmid, the empty plasmid, pmirGLO-NC, pmirGLO-circ001859, miR-21-5p mimics, miR-21-5p inhibitor were cotransfected into HEK293 with Lipofectamie 2000 (Invitrogen, Carlsbad, CA, USA). Luciferase activity was detected on a microplate reader using the dual luciferase reporter assay kit (Promega, Madison, WI, USA) and the experiments were repeated for three times.
2.11RNA pull down experiment
Circ_001859-wt and control group probe were in vitro transcribed and biotin-labeled by using the biotin RNA labeling mix (Roche, Basel, Switzerland), treated with RNase-free DNase I (Roche) and purified using an RNeasy mini kit (Qiagen, Valencia, CA, USA). Then cell protein (1 mg) extract was mixed with biotinylated RNA biotin-labeled RNAs (50 pmol), incubated with streptavidin agarose beads (Invitrogen), and washed three times with NaCl/Pi at room temperature. Dynabeads were used for western blotting. After being washed, mixed with cell lysate, 30
2.12Xenograft transplantation model
BALB/c nude mice were purchased from Charles River (Beijing, China). These mice were given free access to sterile food and water during the whole experiment proceedings. All animal experiments with nude mice were performed strictly in accordance with a protocol approved by The Second Affiliated Hospital of Xinjiang Medical University. Transfected cells were made into suspension in each group (2
2.13Hematoxylin and eosin (HE) staining
HE staining was used to detect the overall condition of tumor tissue. In this study, hematoxylin and eosin was used to stain paraffin sections of mouse tumor tissues after deparaffinization and dehydration. Finally, observe the cell morphology under a microscope.
2.14Immunohistochemistry assay (IHC)
KI67 staining was used to detect cell proliferation in tumor tissues. Paraffin sections of xenograft tumor tissues were cut with a thickness of 4
2.15Immunofluorescence (IF)
TUNEL and EMT-related proteins were stained by using fluorescent staining to verify tumor tissue apoptosis and EMT progression. Paraffin sections of xenograft tumor tissues were cut with a thickness of 4
2.16Statistical analysis
Shapiro-Wilk normality test was used to verify the distribution of data in this study before the analysis began. Data conforming to normal distribution was represented as mean
3.Results
3.1Circ_001859 in was lowly expressed in pancreatic cancer tissues and cells
Figure 1.
Circ_001859 was down-regulated in tumor tissues and cells. (A) GSE79634 revealed circ_001859 as a suppressed circRNA with a criterion of logFC
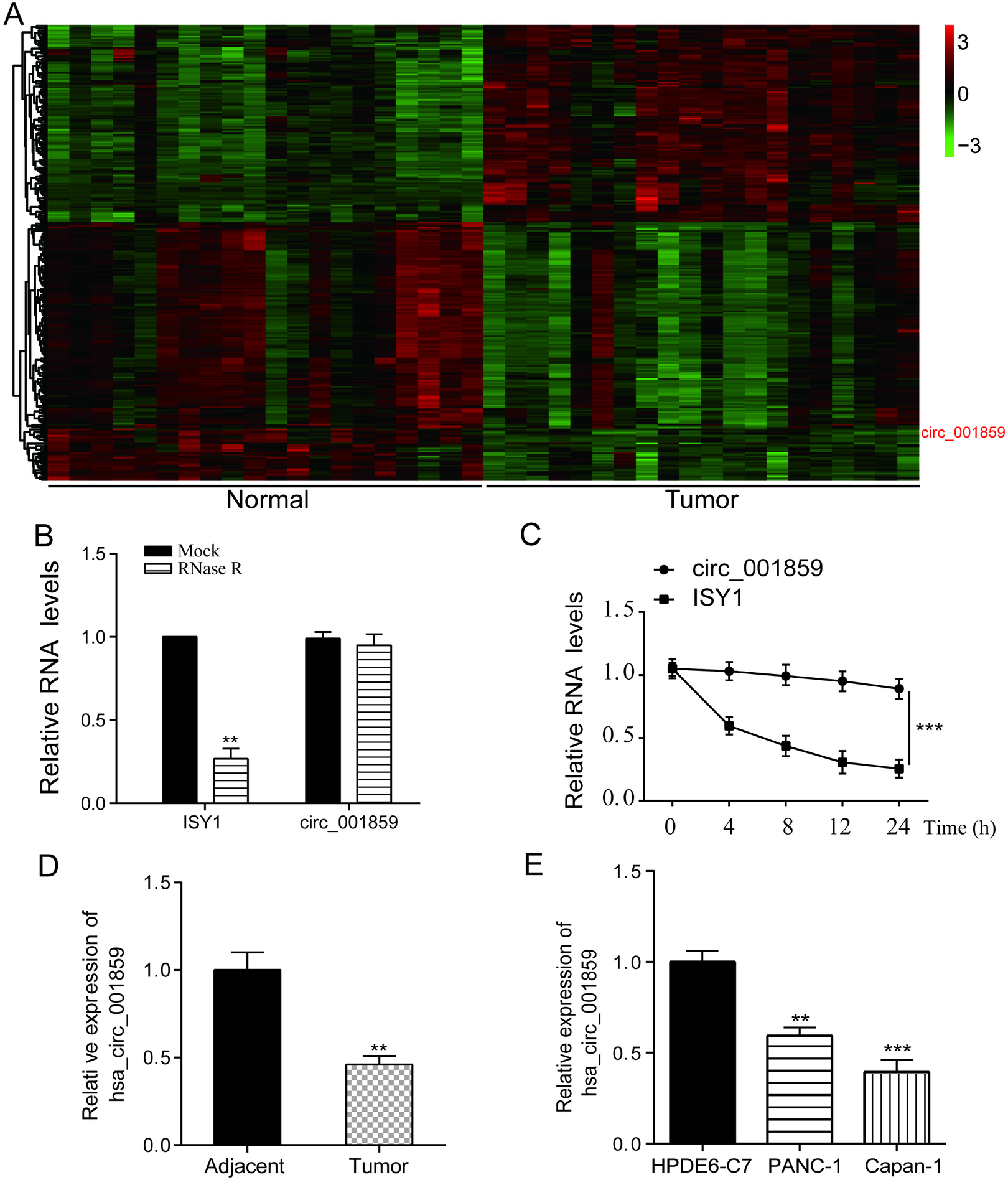
The GSE79634 microarray was selected from the GEO database for analysis. Differentially expressed circRNAs in pancreatic cancer and normal control specimens were analysed by R software, and the heat map was plotted. LogFC
3.2Circ_001859 overexpression suppressed proliferation, migration and invasion of pancreatic cancer cells
Figure 2.
Circ-001859 suppressed cell proliferation, migration and invasion. (A) Sketch of structures of p-circ-001859 and NC vector were shown (B) Circ_001859 expression was enhanced in p-circ_001859 group. (C) Circ_001859 inhibited cell proliferation according to colony formation experiment. (D) Transwell experiment also verified that the overexpression of circ_001859 suppressed invasion and migration (E) The expression levels of EMT markers (E-cadherin, N-cadherin and Vimentin) were detected by QRT-PCR.
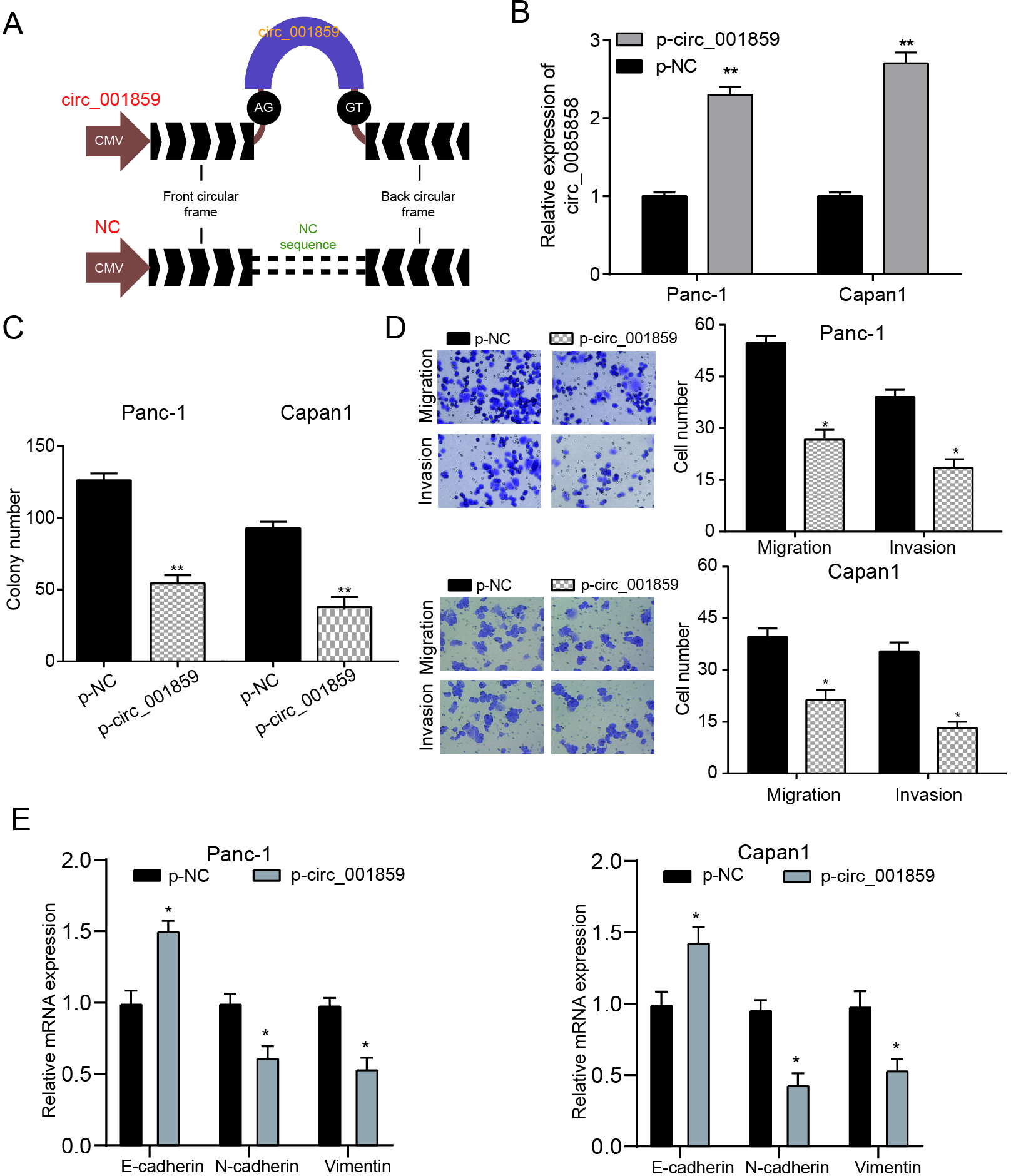
We constructed a circ_001859 overexpression plasmid (p-circ_001859) with circular frame and circ_ 001859 sequence (Fig. 2A) and qPCR verified that circ_001859 was significantly overexpressed in PANC-1 and Capan-1 cells in p-circ_001859 group (Fig. 2B,
3.3Circ_001859 overexpression suppressed tumor growth in pancreatic mice model
Figure 3.
Circ_001859 overexpression suppressed tumor growth. (A–B) The tumor volume of mice in p-circ-001859 group was smaller. (C–D) Images and weight of tumors in nude mice indicated that circ-001859 overexpression inhibited tumor weight growth. (E) HE, KI67 and immunofluorescence detected the regulatory effect of circ_001859 overexpression on tumor proliferation, apoptosis and EMT process (
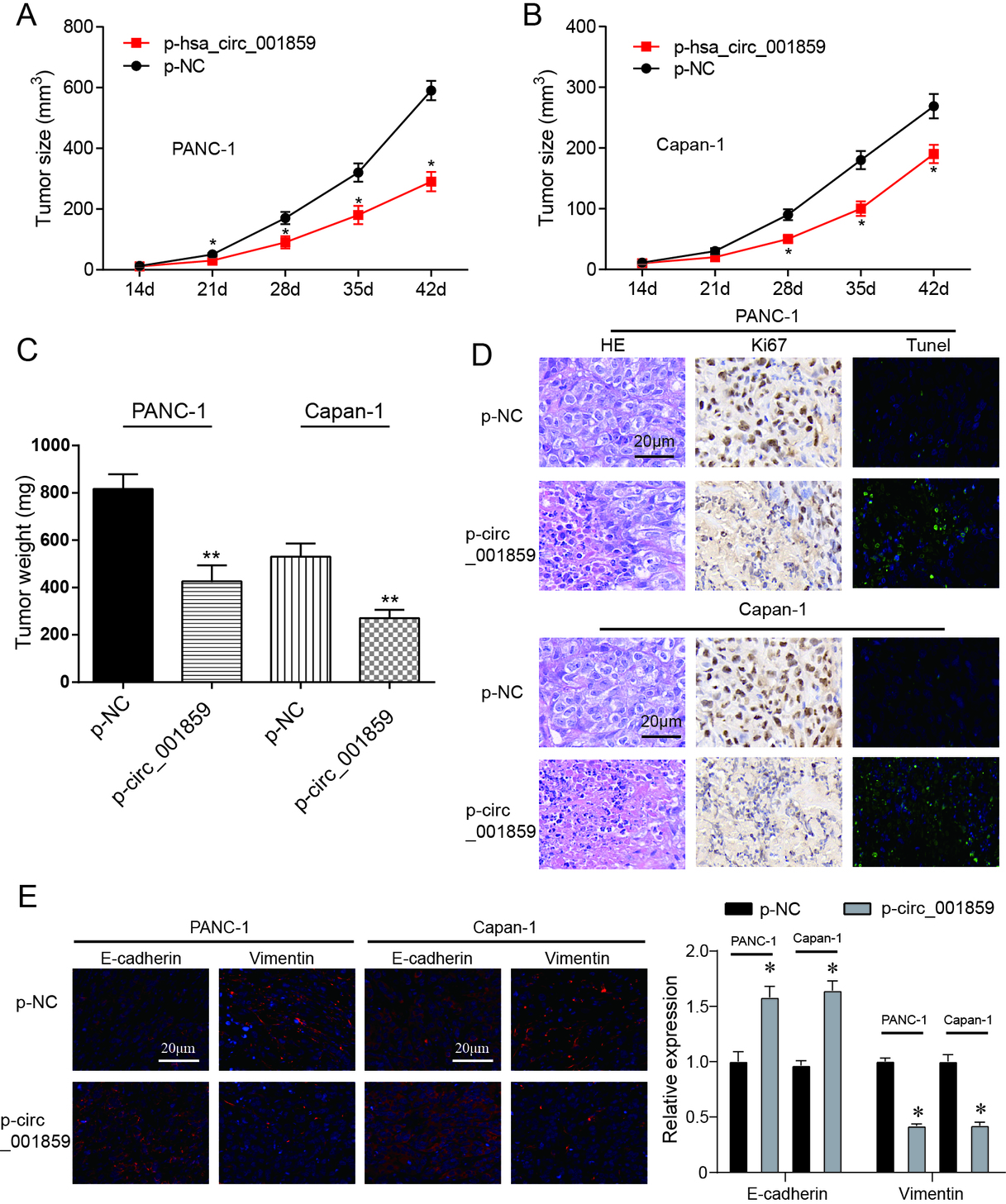
Tumor volume of mice was measured every three days. It was discovered that circ_001859 overexpression significantly suppressed tumor volume growth (Fig. 3A–B,
3.4Direct interaction between circ_001859 and miR-21-5p
Figure 4.
MiR-21-5p was up-regulated in cancer and could be suppressed by circ_001859. (A) Possible sequence of binding sites in the prediction between miR-21-5p and circ_001859. The luciferase reporter assay indicated that miR-21-5p-mimics directly targeted at circ_001859. (B) Circ_001859 pulled down miR-21-5p expression by RNA pull down assay. (C) The FISH assay verified the expression and localization of circ_001859 and miR-21-5p in pancreatic cancer cells (x 200). (D) MiR-21-5p expression was enhanced in tumor tissues and cells. (E) TCGA data validated the overall survival of miR-21-5p in pancreatic cancer. (F) MiR-21-5p expression was enhanced in cells. (G) p-circ_001859 suppressed miR-21-5p expression.
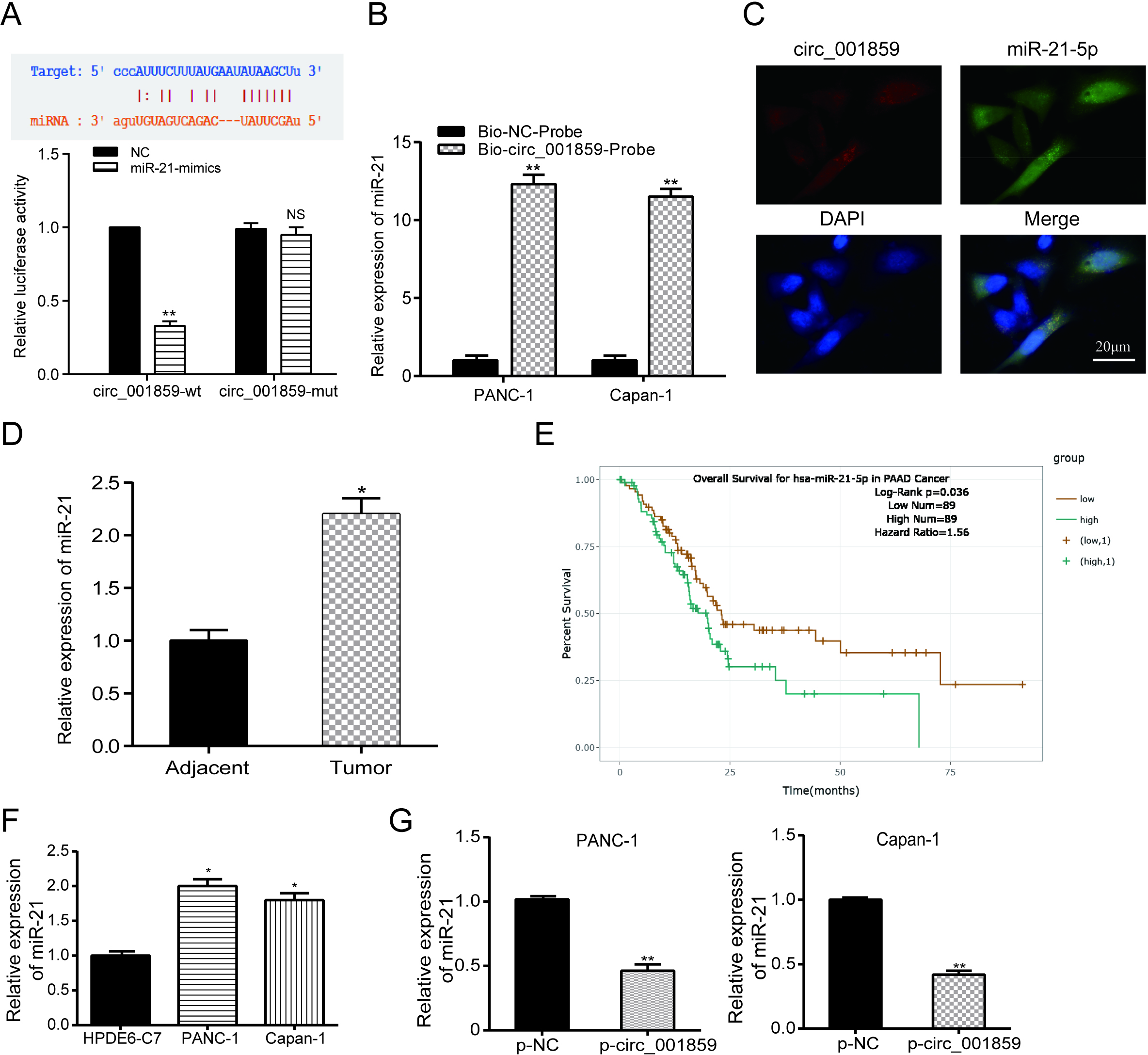
The Starbase database was used to predicted the direct targeting relationship between miR-21-5p and circ_001859. In dual luciferase reporter assay, it can be founded that the fluorescence intensity in circ_001859-wt
3.5MiR-21-5p mimics promoted proliferation, migration and invasion
Figure 5.
MiR-21-5p mimics promoted cell proferation, migration and invasion. (A–B) The expression of miR-21-5p in miR-21-5p-mimics, miR-21-5p-inhibitor, p-circ_001859
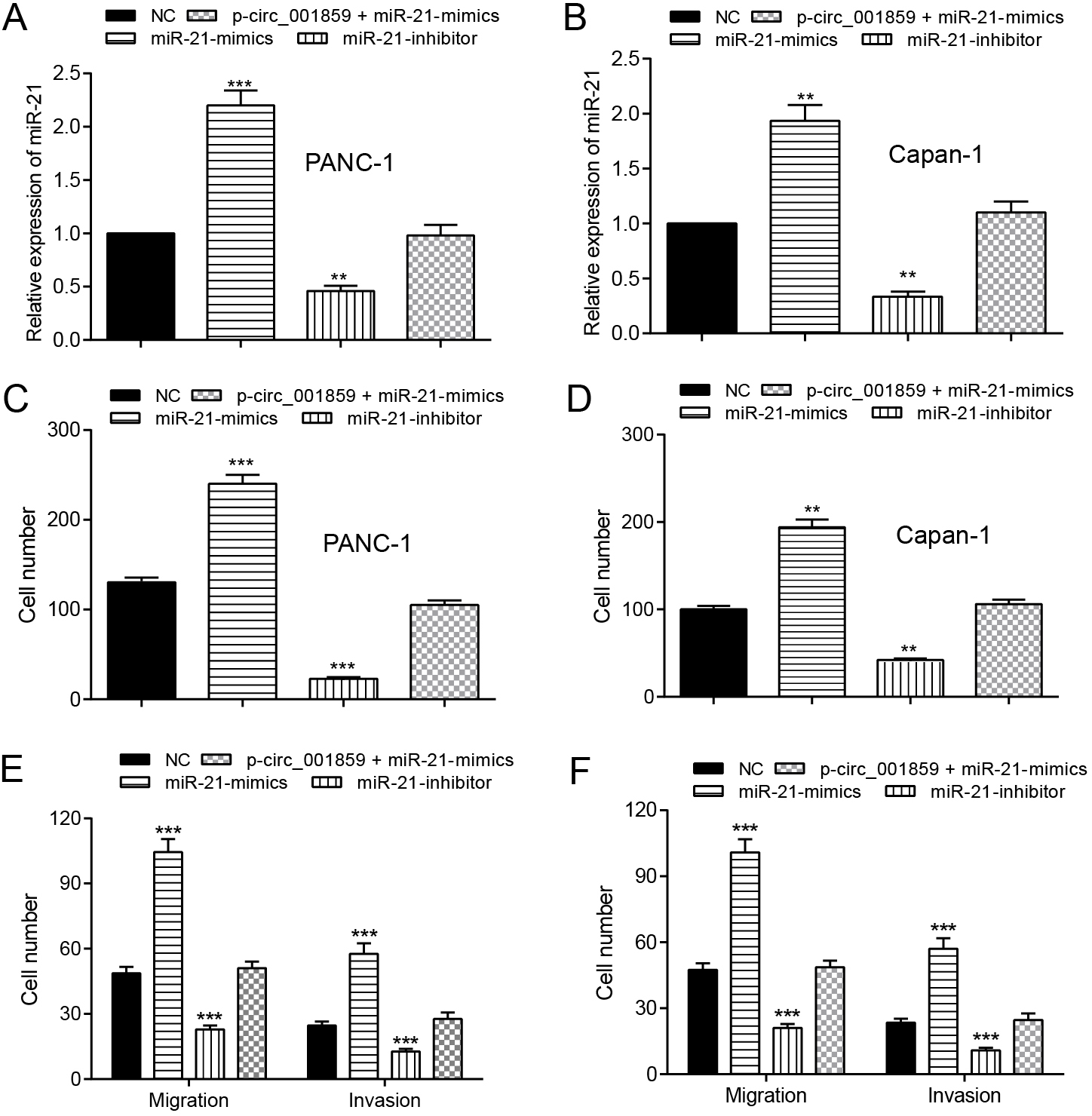
In order to study the regulation of miR-21-5p on the ability of invasion, proliferation and migration, four groups were divided as empty vector NC, miR-21-5p-mimics, miR-21-5p-inhibitor and p-circ_001859
3.6SLC38A2 was regulated by miR-21-5p and circ_001859
Figure 6.
MiR-21-5p suppressed SLC38A2 expression and SLC38A2 suppression promoted cell proliferation. (A) MiR-21-5p directly targeted at SLC38A2. (B) TCGA data validated the overall survival of SLC38A2 in pancreatic cancer. (C–D) MiR-21-5p mimics suppressed SLC38A2 mRNA or protein expression but p-circ_001859 enhanced SLC38A2 expression. (E) Si-SLC38A2 suppressed SLC38A2 protein and mRNA expression and p-circ_001859 could restore the suppression. p-SLC38A2 enhanced SLC38A2 protein and mRNA expression and miR-21-5p mimics rescued the enhancement. (F) Si-SLC38A2 increased cell proliferation and p-SLC38A2 suppressed the proliferation.
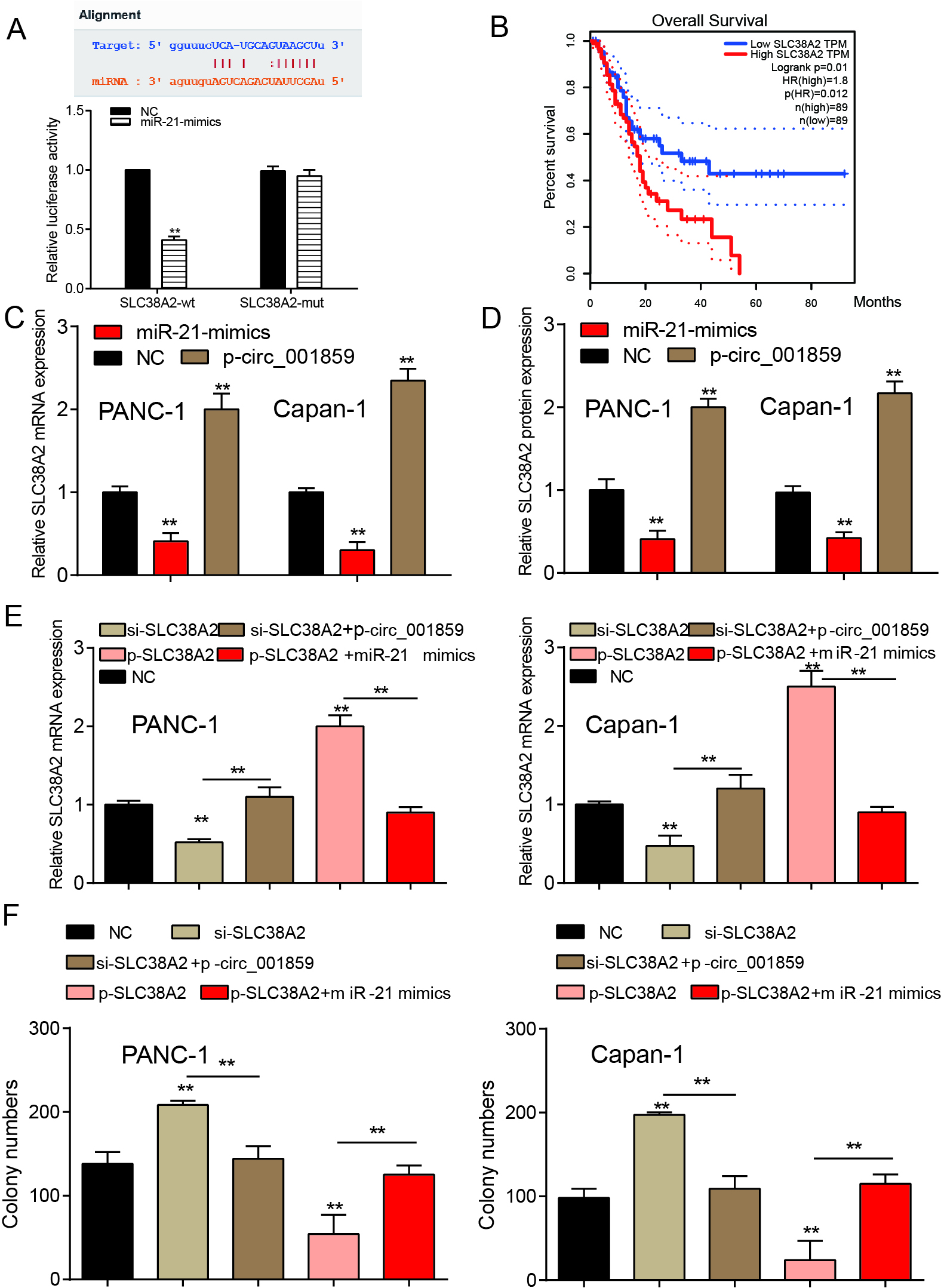
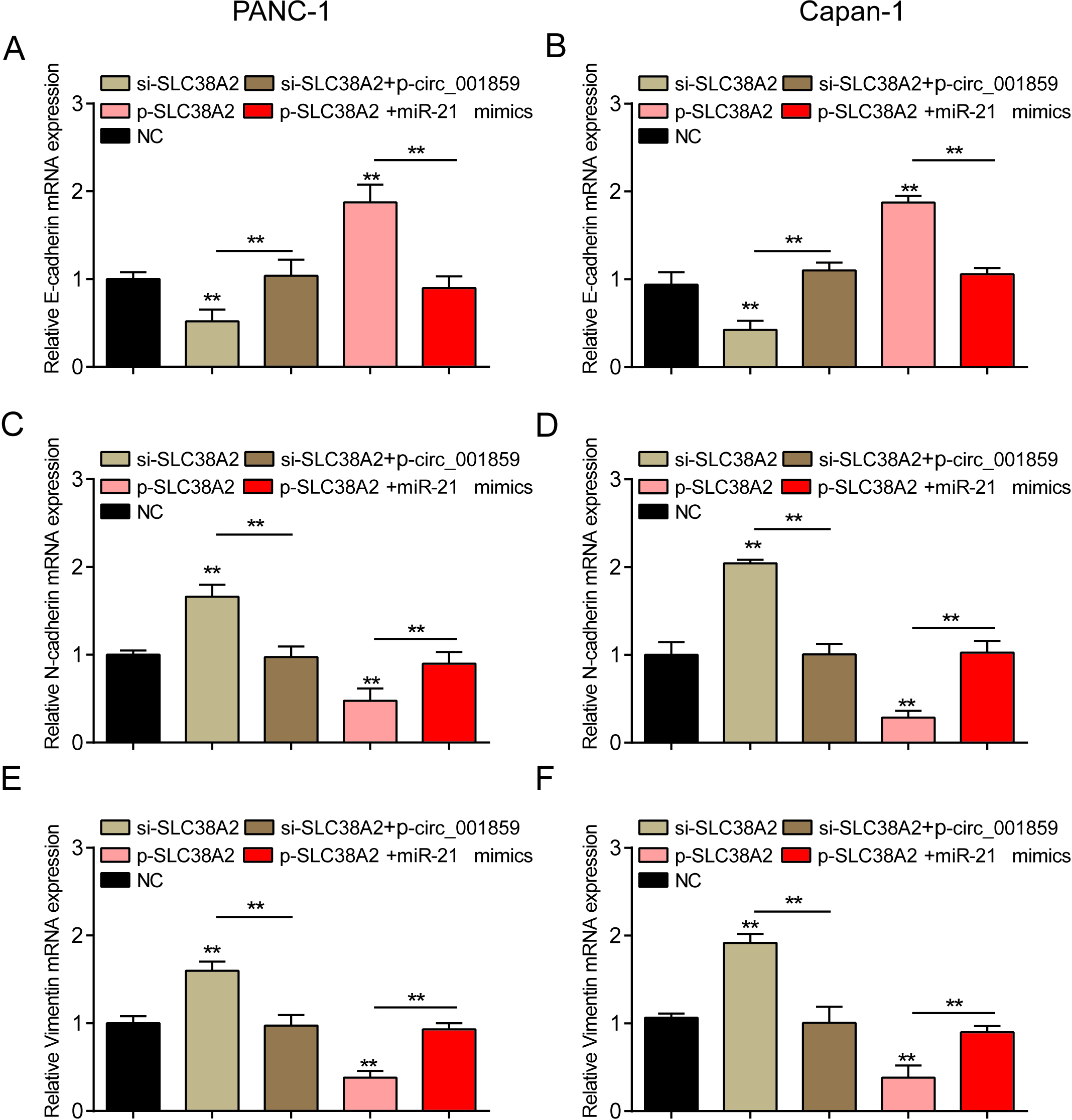
Figure 7.
Circ_001859 affects EMT process through miR-21-5p/SLC38A2. (A–B) The expression level of E-cadherin mRNA was detected by QRT-PCR in each treatment group. (C–D) The expression level of N-cadherin mRNA was detected by QRT-PCR in each treatment group. (E–F) The expression level of Vimentin mRNA was detected by QRT-PCR in each treatment group.
By bioinformatics analysis, we predicted the protein SLC38A2 that may be regulated by miR-21-5p. The dual luciferase assay showed that the SLC38A2-wt group’s fluorescence intensity decreased remarkably, however, the fluorescence intensity of miR-21-5p-mimics or NC group remained the same (Fig. 6A,
Finally, in order to verify the regulation of the three factors on the process of tumor cells, we used cloning and EMT marker detection respectively and found that overexpression of SLC38A2 or inhibition of SLC38A2 could reverse the regulatory effects of miR-21-5p or circ_001859 overexpression on the process of pancreatic cancer (Figs 6F and 7,
4.Discussion
Pancreatic cancer is a lethal malignancy that responsible for about 227,000 deaths every year on a global scale [19]. There’s a grim five-year low survival rate of this disease, which is only 6%. Therefore, better treatment strategies are sorely needed and early detection is of critical importance [20]. In this study, the low expression of hsa_circ_001859 in tumor tissues was found after screening GEO data. qRT-PCR, RNA half-life assay and other methods to verify its expression and ring-like characteristics. In vitro and in vivo evidence suggests that overexpression of circ_001859 could inhibit the progression of pancreatic cancer. This is the first report of this molecule as a tumor suppressor gene in tumors. Further, through bioinformatics and molecular biological verification, we found that circ_001859 might regulate the proliferation, metastasis and EMT proceedings of pancreatic cancer through the miR-21-5p/SLC38A2 pathway. In addition, through our study of prognostic data, we believe that our data will increase the feasibility of this molecule in the selection of diagnostic markers for pancreatic cancer and the development of targets for targeted therapy.
This study indicated that circRNA circ_001859 was down-regulated in pancreatic cancer patients. Furthermore, the proliferation, migration and invasion ability of pancreatic cancer cells were inhibited by the overexpression of circ_001859. Nair et al. observed that a lot of circRNAs in normal-adjacent samples of ER
Besides, this study firstly demonstrated that miR-21-5p was overexpressed in pancreatic cancer tissues. Overexpression of miR-21-5p stimulated the proliferation, invasion and migration ability of pancreatic cancer cells. Recent literatures were referred to that Pai et al. have discovered that the numerous miRNAs were aberrantly expressed in pancreatic cancer. A number of them could prove to be effective drug targets; however, effective targeting of the affected organ and minimizing off-target effects of the miRNA-based therapeutic agent represent formidable challenges [20]. Meanwhile, it is now becoming more and more evident that miRNAs have the potential to be utilized as bio-markers for cancer diagnosis. For example, certain miRNAs such as miR-376a, miR-221, miR-155, miR-21-5p, miR-301, and miR-222 are overexpressed in pancreatic cancer [5]. This study then properly proved the oncogenic role of miR-21-5p via a regulation loop.
Finally, we investigated the relationship between miR-21-5p and SLC38A2, results demonstrated that SLC38A2 was the target protein of miR-21-5p, and inhibited by miR-21-5p, circ_001859 can affect SLC38A2 by regulating miR-21-5p. Several downstream proteins have been discussed to be tumor regulator in pancreatic cancer, such as FBXO11 [24], IL-6 [25] and RBM3 [26] except SLC38A2. The blank intersection was discussed in this report and SLC38A2 was supported to be a tumor suppressor in pancreatic cancer as well.
The role of circ_0001859 in cancer progression has not been fully figured out. The interactions between circRNAs and miRNAs on the process of pancreatic cancer remain largely confused [5]. MiR-21-5p, as one of miRNA, has been studied in pancreatic cancer rarely. However, in our study, we found the effect of SLC38A2 as a target protein of miR-21-5p. Nevertheless, in this research, there were still some defects which should be taken into account. For instance, the SLC38A2 was not the only one gene of miR-21-5p, but this study merely investigated SLC38A2. Thus, we set out to make some improvements in the subsequent work.
5.Conclusion
Taken all together, circ_001859 which was lowly expressed in pancreatic carcinoma could suppress the ability of proliferation, migration and invasion of pancreatic cancer cells by down-regulating miR-21-5pand up-regulating SLC38A2.
Authors’ contributions
Conception: Li Jiangang.
Interpretation or analysis of data: Li Liang, Wang Nan, Wang Jun.
Preparation of the manuscript: Li Liang.
Revision for important intellectual content: Li Liang, Li Jiangang.
Supervision: Li Jiangang.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220229.
Acknowledgments
This research was supported by the Natural Science Foundation of Xinjiang Uygur Autonomous Region (2021D01C375).
References
[1] | M. Ilic and I. Ilic, Epidemiology of pancreatic cancer, World J Gastroenterol 22: ((2016) ), 9694–9705. |
[2] | A.P. Klein, Pancreatic cancer epidemiology: understanding the role oflifestyle and inherited risk factors, Nat Rev Gastroenterol Hepatol 18: ((2021) ), 493–502. |
[3] | H. He, S.J. Hao, L. Yao, F. Yang, Y. Di, J. Li, Y.J. Jiang, C. Jin and D.L. Fu, MicroRNA-218 inhibits cell invasion and migration of pancreaticcancer via regulating ROBO1, Cancer Biol Ther 15: ((2014) ), 1333–9. |
[4] | M. Humeau, A. Vignolle-Vidoni, F. Sicard, F. Martins, B. Bournet, L. Buscail, J. Torrisani and P. Cordelier, Salivary MicroRNA in PancreaticCancer Patients, PLoS One 10: ((2015) ), e0130996. |
[5] | M.A. Khan, H. Zubair, S.K. Srivastava, S. Singh and A.P. Singh, Insightsinto the Role of microRNAs in Pancreatic Cancer Pathogenesis: Potential forDiagnosis, Prognosis, and Therapy, Adv Exp Med Biol 889: ((2015) ), 71–87. |
[6] | H. Xie, X. Ren, S. Xin, X. Lan, G. Lu, Y. Lin, S. Yang, Z. Zeng, W. Liao, Y.Q. Ding and L. Liang, Emerging roles of circRNA_001569 targetingmiR-145 in the proliferation and invasion of colorectal cancer, Oncotarget 7: ((2016) ), 26680–91. |
[7] | M. Taylor-Teeples, L. Lin, M. de Lucas, G. Turco, T.W. Toal, A. Gaudinier, N.F. Young, G.M. Trabucco, M.T. Veling, R. Lamothe, P.P. Handakumbura, G. Xiong, C. Wang, J. Corwin, A. Tsoukalas, L. Zhang, D. Ware, M. Pauly, D.J. Kliebenstein, K. Dehesh, I. Tagkopoulos, G. Breton, J.L. Pruneda-Paz, S.E. Ahnert, S.A. Kay, S.P. Hazen and S.M. Brady, AnArabidopsis gene regulatory network for secondary cell wall synthesis, Nature 517: ((2015) ), 571–5. |
[8] | G. Chen, Y. Shi, Y. Zhang and J. Sun, CircRNA_100782 regulatespancreatic carcinoma proliferation through the IL6-STAT3 pathway, Onco Targets Ther 10: ((2017) ), 5783–5794. |
[9] | W.J. Huang, Y. Wang, S. Liu, J. Yang, S.X. Guo, L. Wang, H. Wang and Y.F. Fan, Silencing circular RNA hsa_circ_0000977 suppressespancreatic ductal adenocarcinoma progression by stimulating miR-874-3p andinhibiting PLK1 expression, Cancer Lett 422: ((2018) ), 70–80. |
[10] | M. Shi, D. Xie, Y. Gaod and K. Xie, Targeting miRNAs for pancreaticcancer therapy, Curr Pharm Des 20: ((2014) ), 5279–86. |
[11] | Y. Zhang, X. Guo, L. Xiong, L. Yu, Z. Li, Q. Guo, Z. Li, B. Li and N. Lin, Comprehensive analysis of microRNA-regulated protein interactionnetwork reveals the tumor suppressive role of microRNA-149 in humanhepatocellular carcinoma via targeting AKT-mTOR pathway, Mol Cancer 13: ((2014) ), 253. |
[12] | Y. An and Q. Yang, MiR-21 modulates the polarization of macrophages andincreases the effects of M2 macrophages on promoting the chemoresistance ofovarian cancer, Life Sci 242: ((2020) ), 117162. |
[13] | W. Ren, J. Hou, C. Yang, H. Wang, S. Wu, Y. Wu, X. Zhao and C. Lu, Extracellular vesicles secreted by hypoxia pre-challenged mesenchymal stemcells promote non-small cell lung cancer cell growth and mobility as well asmacrophage M2 polarization via miR-21-5p delivery, J Exp Clin Cancer Res 38: ((2019) ), 62. |
[14] | S.R. Pfeffer, C.H. Yang and L.M. Pfeffer, The Role of miR-21 in Cancer, Drug Dev Res 76: ((2015) ), 270–7. |
[15] | M.S. Huang, J.H. Chang, W.C. Lin, Y.H. Cheng, F.A. Li, C.S. Suen, M.J. Hwang, C.K. Chang and K.Y. Mou, SLC38A2 Overexpression Induces a Cancer-likeMetabolic Profile and Cooperates with SLC1A5 in Pan-cancer Prognosis, Chem Asian J 15: ((2020) ), 3861–3872. |
[16] | M. Morotti, C.E. Zois, R. El-Ansari, M.L. Craze, E.A. Rakha, S.J. Fan, A. Valli, S. Haider, D.C.I. Goberdhan, A.R. Green and A.L. Harris, Increasedexpression of glutamine transporter SNAT2/SLC38A2 promotes glutaminedependence and oxidative stress resistance, and is associated with worseprognosis in triple-negative breast cancer, Br J Cancer 124: ((2021) ), 494–505. |
[17] | S.J. Parker, C.R. Amendola, K.E.R. Hollinshead, Q. Yu, K. Yamamoto, J. Encarnacion-Rosado, R.E. Rose, M.M. LaRue, A.S.W. Sohn, D.E. Biancur, J.A. Paulo, S.P. Gygi, D.R. Jones, H. Wang, M.R. Philips, D. Bar-Sagi, J.D. Mancias and A.C. Kimmelman, Selective Alanine Transporter UtilizationCreates a Targetable Metabolic Niche in Pancreatic Cancer, Cancer Discov 10: ((2020) ), 1018–1037. |
[18] | S. Guo, X. Xu, Y. Ouyang, Y. Wang, J. Yang, L. Yin, J. Ge and H. Wang, Microarray expression profile analysis of circular RNAs in pancreaticcancer, Mol Med Rep 17: ((2018) ), 7661–7671. |
[19] | B.S. Li, H. Liu and W.L. Yang, Reduced miRNA-218 expression inpancreatic cancer patients as a predictor of poor prognosis, Genet Mol Res 14: ((2015) ), 16372–8. |
[20] | P. Pai, S. Rachagani, C. Are and S.K. Batra, Prospects of miRNA-basedtherapy for pancreatic cancer, Curr Drug Targets 14: ((2013) ), 1101–9. |
[21] | A. Singh, V.K. Patel, D.K. Jain, P. Patel and H. Rajak, Panobinostat as Pan-deacetylase Inhibitor for the Treatment of Pancreatic Cancer: Recent Progress and Future Prospects, Oncol Ther 4: ((2016) ), 73–89. |
[22] | A.A. Nair, N. Niu, X. Tang, K.J. Thompson, L. Wang, J.P. Kocher, S. Subramanian and K.R. Kalari, Circular RNAs and their associations withbreast cancer subtypes, Oncotarget 7: ((2016) ), 80967–80979. |
[23] | F. Yang, D.Y. Liu, J.T. Guo, N. Ge, P. Zhu, X. Liu, S. Wang, G.X. Wang and S.Y. Sun, Circular RNA circ-LDLRAD3 as a biomarker in diagnosis ofpancreatic cancer, World J Gastroenterol 23: ((2017) ), 8345–8354. |
[24] | Y. Ma, F. Deng, P. Li, G. Chen, Y. Tao and H. Wang, The tumorsuppressive miR-26a regulation of FBXO11 inhibits proliferation, migrationand invasion of hepatocellular carcinoma cells, Biomed Pharmacother 101: ((2018) ), 648–655. |
[25] | K. Otani, D. Inoue, K. Fujikura, T. Komori, S. Abe-Suzuki, T. Tajiri, T. Itoh and Y. Zen, Idiopathic multicentric Castleman’s disease: aclinicopathologic study in comparison with IgG4-related disease, Oncotarget 9: ((2018) ), 6691–6706. |
[26] | E. Karnevi, L.B. Dror, A. Mardinoglu, J. Elebro, M. Heby, S.E. Olofsson, B. Nodin, J. Eberhard, W. Gallagher, M. Uhlen and K. Jirstrom, Translational study reveals a two-faced role of RBM3 in pancreatic cancerand suggests its potential value as a biomarker for improved patientstratification, Oncotarget 9: ((2018) ), 6188–6200. |




