The Association between Diabetes Medication Use and Tumour Characteristics at Diagnosis in Patients with Urothelial Carcinoma: A Retrospective Registry-Based Study1
Abstract
BACKGROUND:
Observational studies indicate a potential association between diabetes medication use and aggressiveness of bladder cancer.
OBJECTIVE:
The objective is to exploratively study the association between diabetes medication use, as proxy for diabetes, and cancer characteristics of urothelial carcinoma at diagnosis. Furthermore, differences in associations between specific types of diabetes medication are studied.
METHODS:
The association between use of diabetes medication and urothelial carcinoma (UC) characteristics at diagnosis is studied. A retrospective registry-based study among UC patients in the Netherlands was performed for which two large linked registries from PHARMO and IKNL were used. Patients diagnosed with UC between 2000 and 2016 and no previous cancer were included in this study. In this study, 1,168 UC patients who were diabetes medication users were included as well as 3,609 non-users. Conditional logistic regression analysis was performed to determine odds ratios comparing cancer characteristics between different types of diabetes medication users to non-users.
RESULTS:
Noninsulin antidiabetic drugs (NIAD) use was associated with a muscle-invasive type of UC compared to non-users (OR = 1.31, 95% CI: 1.10–1.55 for T2+ versus Ta) as well as a poorly differentiated tumour (OR = 1.31, 95% CI: 1.07–1.59 for poorly versus well differentiated tumours).
CONCLUSION:
Users of diabetes medication are potentially more likely to be diagnosed with a more aggressive tumour than non-users; however, lifestyle factors could not be adjusted for.
INTRODUCTION
Bladder cancer is the eleventh most prevalent cancer type worldwide [1], with about 75% of cases being men [2]. GLOBOCAN estimated globally 573,000 new bladder cancer cases and 213,000 bladder cancer deaths in 2020 [3]. The most important risk factors for bladder cancer are smoking and historically occupational exposure to several chemicals [4]. Furthermore, lifestyle factors such as physical activity, diet and BMI can potentially influence bladder cancer risk [5] and perhaps subsequently, people diagnosed with diabetes mellitus are at higher risk of bladder cancer (RR = 1.35, 95% CI 1.17–1.56, I2 = 94.7) in a meta-analysis performed by Zhu and colleagues [6]. Moreover, a higher bladder cancer risk for patients diagnosed with diabetes mellitus less than 5 years ago has been estimated (RR = 1.52, 95% CI:1.05–2.21) compared to people having diabetes mellitus for 5 years or longer [6]. However, pooling results is perhaps not warranted because of heterogeneity on e.g. diabetes measurements, between studies on diabetes medication and bladder cancer risk and therefore results should not be taken at face value [7].
Potential mechanisms to explain the possible association between diabetes mellitus and bladder cancer development and progression can be found in either shared risk factors or in metabolic explanations. The most frequently proposed metabolic mechanisms associated with cancer development in literature are related to insulin and insulin-like growth factors (IGF) [8, 9]. Zhao and colleagues [10] indicated higher levels of IGF-1 in plasma and lower levels of IGF binding protein-3 in patients with bladder cancer compared to controls. However, Lin and colleagues [11] studied this association prospectively and found no association between IGF-1 plasma concentrations and bladder cancer risk as well as Eich and colleagues who did not observe an association between IGF-1 receptor expression and tumor progression (HR = 1.6, CI = 0.8–3.1) [12]. In a study on urothelial carcinoma (UC) specimens, the IGF-1 receptor was upregulated in 74% of the samples [13]. Moreover, IGF-1 receptor expression in muscle-invasive tumours compared to non-muscle-invasive tumours appeared to be higher in one study [14], while another study indicated opposite results [15]. Therefore, a potential indirect influence of diabetes on IGF-1 levels could result in development of invasive tumours, but studies are still conflicting.
In addition to potentially increasing bladder cancer risk, an observational study by Hwang and colleagues [16] among 251 patients showed that patients with poor glycaemic control (HbA1c> = 7) had high-grade tumours more often and a higher tumour multiplicity in a sample of non-muscle invasive UC. Also, diabetes mellitus was associated with an increased risk of recurrence (OR = 2.11, 95% CI:1.4–3.2) and progression (OR = 9.35, 95% CI:3.1–28.6). Nevertheless, a Dutch cohort study showed no associations between BMI and diabetes mellitus and bladder cancer recurrence and progression [17].
The aim of this study is to exploratively determine in a large registry study whether there is an association between diabetes medication use, as an indicator for diabetes, and aggressiveness of UC tumours in the bladder at diagnosis. Additionally, using different types of diabetes medication, as a proxy for distributed glucose metabolism, and the association with UC aggressiveness will be determined. It is hypothesized that in patients using any diabetes medication a higher tumour stage is observed. Also, diabetes medication use is possibly associated with multifocality and poor differentiation grade at diagnosis of UC in the bladder.
METHODS
Data sources
Data was obtained from registries of the Out-patient Pharmacy Database of the PHARMO Database Network on medication usage. The PHARMO Database Network contains data from primary and secondary health care institutes in the Netherlands [18]. The Out-patient Pharmacy Database contains drug dispensings from both the general practitioner as well as the medical specialist. Drug dispensing was recorded with dates, name of product, quantities and with corresponding Anatomical Therapeutic Chemical (ATC) codes according to the World Health Organization.
The Netherlands Comprehensive Cancer Organization (IKNL) provided data on cancer diagnosis and treatment which were recorded in the Netherlands Cancer Registry (NCR). The NCR contains information on cancer type, stage, site, and morphology. T stage was determined according to the TNM classification (4th edition from 1999–2002, 5th edition from 2003–2009 and 6th edition from 2010–2016) [19]. Differentiation grade was determined as either 1 (well differentiated), 2 (moderately differentiated) or 3 (poorly differentiated) based on the pathologist report as recorded in different hospitals, according to the 1973 classification. A primary tumour was categorised as multifocal when pathology reports indicated evidence of multiple tumour locations within the bladder. This study has been approved by the PHARMO Institute for Drug Outcomes Research and the privacy review board of the NCR (project number K19.210).
Study population
The source population included patients living in the Netherlands diagnosed with UC in the bladder between 2000 and 2016 who were registered in the NCR and present in the Out-patient Pharmacy Database from the PHARMO Database Network (n = 5,666), which covers predominantly the southern part of the Netherlands. Patients who used diabetes medication, had to be continuously enrolled in the PHARMO Database Network for at least 2 years prior to date of UC diagnosis to be included. When data was not available, it could not be assessed whether diabetes medication use started recently or whether medication was used for a longer period before being enrolled in the PHARMO Database Network. Participants who already had cancer (excluding basal cell carcinoma) before diagnosis of UC were excluded from analyses (n = 560). Also, remaining patients with unkown TNM stage were excluded (n = 28). Finally, patients with only past insulin use were not further analysed because of too low numbers (n = 14).
Study variables
The main independent variable was the category of diabetes medication use. Firstly, patients were defined as insulin users when they received insulin from their pharmacy at least two times in six months before UC diagnosis. Similar rules applied for non-insulin anti-diabetes medication (NIAD). Furthermore, a group was defined as using a combination of insulin and NIAD, for which it is likely that they started with NIAD medication after which they required insulin.
Clinical characteristics such as age, sex and comorbidities of UC patients were collected at diagnosis. In addition, the chronic disease score (CDS) was provided through the PHARMO Database Network. This score represents the burden of chronic diseases based on medication use. The CDS was categorized into either having a low score (lower than 7) or high (equal or higher than 7). Patients with a high CDS have higher risk on hospitalization and mortality compared to patients with no chronic diseases [20].
Statistical analysis
Patient characteristics were expressed as mean±standard deviation for continuous variables or frequencies and percentages for categorical variables. To compare differences in baseline characteristics between users of diabetes medication and controls, who never used diabetes medication, Chi square tests were performed for categorical variables and ANOVA/t-tests were performed for continuous variables. Comparisons were made between NIAD users, the combination of insulin and NIAD users, and non-users. Conditional logistic regression analysis was performed to estimate odds ratios comparing UC characteristics between diabetes medication users and non-users using dummy variables for endpoints stage (Tis, T1 and T2 compared to Ta) and differentiation grade (poor, moderate versus well) which are not dichotomous variables. In the analyses, adjustments were made for age, sex and CDS (continous). CDS was included as confounder, because multimorbidity [21, 22] can also influence tumour growth.
Moreover, conditional logistic regression was performed to study specific types of NIADs. UC characteristics were compared between metformin only, sulfonylurea (SU) only, mixed NIAD users and non-medication users, with adjustment for age, sex and CDS. Analyses were performed in SAS version 14.
RESULTS
Among the total population of 5,078 patients with UC, 23% (n = 1,182) used any type of diabetes medication regularly with dispensing patterns of at least two in six months before cancer diagnosis. The mean age was 72±9 years and 80% was male. 932 UC patients used NIADs only, while the combination of insulin and NIAD was used by 236 patients, for a period of at least 2 years. Compared to non-users, the proportion of patients with a CDS score≥7, a T2+ stage and a poorly differentiated tumour was significantly higher in NIAD users (Table 1).
Table 1
Urothelial carcinoma (UC) patient and cancer characteristics by use of diabetes medication
| Total | No medication | Medication | ||||
| NIAD > 2 years | Insulin and NIAD > 2 years | P-value any diabetes medication versus no medication | P-value insulin and NIAD versus NIAD | |||
| n | 5078 | 3609 | 932 | 236 | ||
| Patient characteristics | ||||||
| Sex (n, % men) | 4079, 80% | 2887, 80% | 754, 81% | 187, 81% | p = 0.671 | p = 0.564 |
| Age (μ±sd) | 72.3 +–9.2 | 72.0 +9.2 | 73.7 +9.1 | 73.0 +–8.7 | p = <0.001 | p = 0.274 |
| Chronic disease score (n, %) | ||||||
| <7 | 2224, 44% | 1630, 45% | 380, 41% | 75, 32% | ||
| ≥7 | 2854, 56% | 1979, 55% | 552, 59% | 161, 68% | p = <0.001 | p = 0.011 |
| Cancer characteristics | ||||||
| TNM stage at diagnosis | 0.005 | 0.462 | ||||
| T0A | 2705, 53% | 1981, 55% | 459, 49% | 117, 50% | ||
| T0IS | 185, 4.0% | 134, 4% | 29, 3% | 13, 6% | ||
| T1 | 1088, 21% | 759, 21% | 214, 23% | 50, 21% | ||
| T2+ | 1100, 22% | 735, 21% | 230, 25% | 56, 24% | ||
| Differentiation grade (n, %) | ||||||
| well differentiated | 1111, 23% | 812, 24% | 178, 20% | 62, 28% | ||
| moderately differentiated | 1547, 33% | 1128, 33% | 268, 31% | 62, 28% | ||
| poorly differentiated | 2100, 44% | 1434, 43% | 432, 49% | 100, 45% | ||
| Frequency unkown = 320 | P = 0.004 | p = 0.057 | ||||
| Multifocality (n, % yes) | 1164, 27% | 807, 27% | 228, 28% | 60, 29% | ||
| Frequency unkown = 830 | p = 0.383 | p = 0.827 | ||||
NIAD users were more likely to be diagnosed with a muscle-invasive tumour at diagnosis (stage T2+ versus Ta) and a poorly differentiated tumour compared to non-users (OR = 1.31, 95% CI: 1.10–1.55 and 1.31, 95% CI: 1.07–1.59, respectively; Fig. 1). No associations between Tis or T1 versus Ta, moderately versus well differentiated or regarding multifocality were observed. The data give little indication of an association between the combined use of insulin and NIAD and risk on T2+ stage, a poorly differentiated tumour and multifocality (OR = 1.09 (95% CI:0.68–1.73), OR = 0.90, 95% CI:0.65–1.25, OR = 1.11, 95% CI:0.81–1.52).
Fig. 1
Odds ratios displayed separately for NIAD users and for users of both insulin and NIADs, compared to UC patients not using diabetes medication (reference group) after adjustment for age, sex and CDS category.
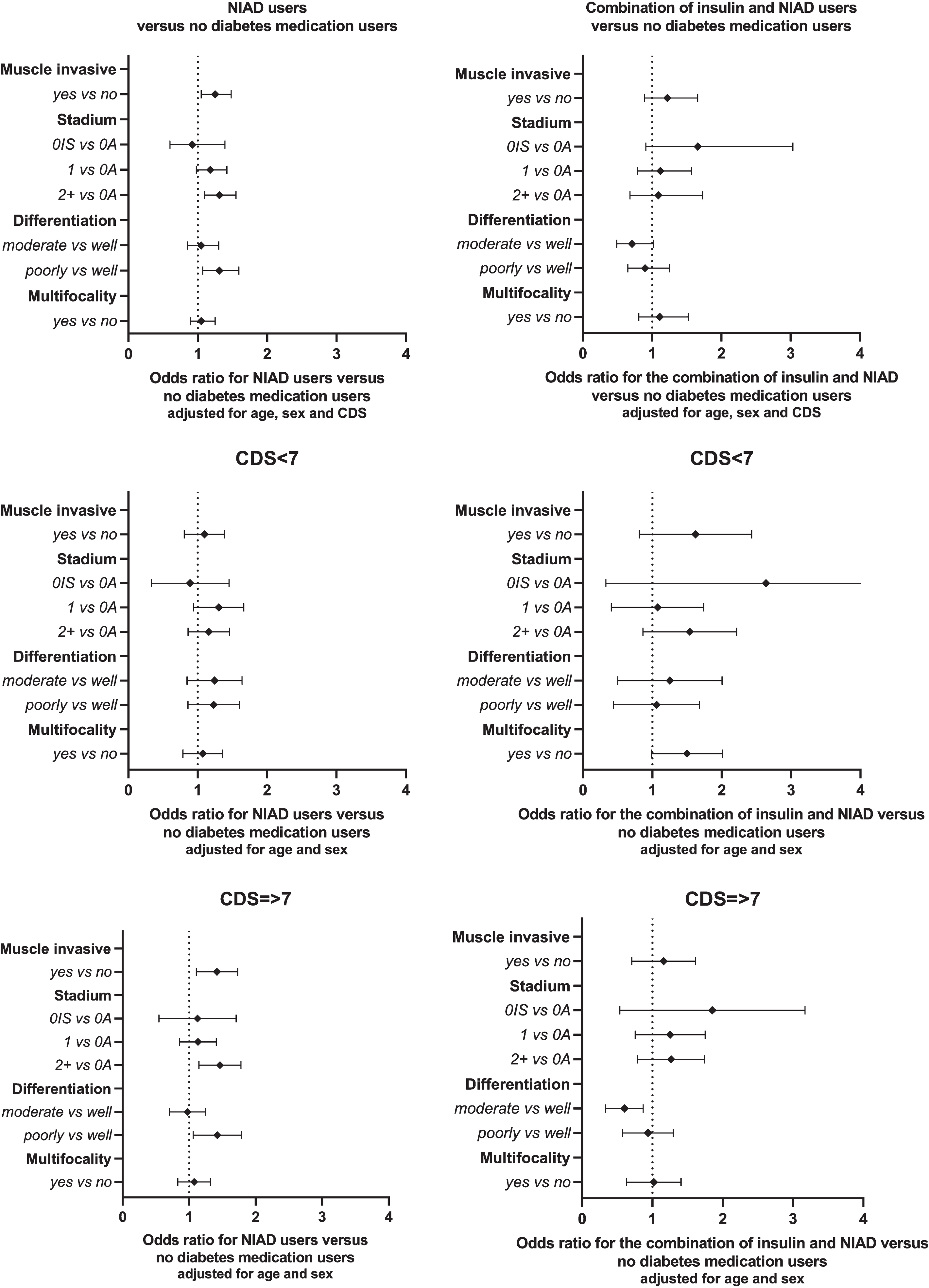
After stratification for CDS, within the analysis on the combined use of NIAD and insulin, no differences in probability of the cancer characteristics could be observed between patients with a high and low CDS. When comparing cancer characteristics between NIAD users and non-users, results for the high CDS group are similar to the overall analysis while the low CDS group show similar ORs with wider 95% confidence intervals (Fig. 1). Furthermore, those with a relatively high number of comorbidities are at a higher risk of a T2+ tumour compared to a Ta if they used NIADs compared to non-users (OR = 1.44 95% CI:1.16–1.79). Also, a higher odds ratio on multifocality of the UC tumour was observed for people with a relatively low number of comorbities when using both NIAD and insulin anti-diabetes medication, albeit not statistically significant (OR = 1.49, 95% CI:0.99–2.02).
Separate analyses for specific NIAD groups showed that mainly metformin (n = 263), sulfonylureas (SU) (n = 145), or a combination of the two (n = 520) were used (Table 2). Patients who used the combination of metformin and SU were more likely to be diagnosed with a T2+ tumour (OR = 1.52, 95% CI: 1.13–2.04, p for trend < 0.001) and with a poorly differentiated tumour (OR = 1.37, 95% CI: 1.07–1.76) compared to non-users. No significant association could be determined for odds ratios on multifocality. For metformin users only and sulfonylureas users only no significant associations could be observed for the different cancer characteristics.
Table 2
Odds ratios (and 95% CI) for specific NIAD medication groups (according to ATC) compared to UC patients not using diabetes medication (reference group), adjusted for age, sex, and chronic disease score
| No medication use | Total NIAD | Metformin only | Sulfonylureas only | Metformin and sulfonylureas | |
| n | 3609 | 929* | 263 | 145 | 520 |
| T stage (OR 95% CI) | |||||
| Ta | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Tis | 1.00 | 0.92 (0.60–1.39) | 0.51 (0.20–1.26) | 1.59 (0.75–3.39) | 0.96 (0.56–1.64) |
| T1 | 1.00 | 1.18 (0.98–1.42) | 1.05 (0.76–1.43) | 1.14 (0.75–1.74) | 1.28 (1.01–1.62) |
| T2+ | 1.00 | 1.31 (1.10–1.55) | 1.01 (0.76–1.37) | 1.07 (0.73–1.56) | 1.52 (1.13–2.04) |
| P for trend | 0.001 | 0.876 | 0.534 | <0.001 | |
| Differentiation grade (OR 95% CI) | |||||
| Well-differentiated | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Moderately differentiated | 1.00 | 1.05 (0.85–1.30) | 1.04 (0.73–1.48) | 1.61 (0.97–2.68) | 0.95 (0.72–1.76) |
| Poorly differentiated | 1.00 | 1.31 (1.07–1.59) | 1.14 (0.82–1.59) | 1.41 (0.86–2.33) | 1.37 (1.07–1.76) |
| P for trend | 0.006 | 0.429 | 0.088 | 0.015 | |
| Multifocality (OR 95% CI) | |||||
| No | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Yes | 1.00 | 1.05 (0.89–1.25) | 0.94 (0.70–1.26) | 0.99 (0.64–1.53) | 1.14 (0.91–1.41) |
*Only 4 patients used other types of NIADs and could not be placed in either of the three stratified analyses.
DISCUSSION
Analyses of this historic cohort of UC patients diagnosed between 2000–2016 show that UC patients having diabetes, based on diabetes medication use, had a higher risk on a muscle-invasive UC compared to UC patients not having diabetes. More specifically, we observed that UC patients using a combination of NIADs (both metformin and SU) had a worse differentiation grade and were more likely to be diagnosed with a muscle invasive UC compared to patients who did not use diabetes medication. In patients with a high CDS, a stronger association between NIAD use and cancer aggressiveness was observed compared to patients with a low CDS.
The results of this study are in line with conclusions from studies in other types of cancer. In a study with colorectal cancer patients, T2DM was associated with a more advanced stage at diagnosis [23]. [24]. The study by Overbeek and colleagues [25] indicated that women with type 2 diabetes, as defined by medication use, generally have a higher tumour stage (OR = 1.28, 95% CI:1.13–1.44) at breast cancer diagnosis which is in line with this study. Nevertheless, direct comparison of results can be difficult because of different ways of defining diabetes and the inclusion of different covariates.
23% of UC patients in this study used any type of diabetes medication which is relatively high compared to the general Dutch population in which the prevalence of diabetes is about 15% for the age group 65 and older (Statline, 2021). This indicates that diabetes might occur more often when other comorbidities, such as cancer, are also present.
A potential explanation of the observed results is through poor glycaemic control in patients before they used diabetes medication. The study by Chang et al. indicated a worse breast cancer survival for women with a bad glycaemic control (HR = 3.55, 95% CI:1.37–9.30). In further specific analyses on types of non-insulin medication, the associations observed for T-stage and tumour grade remained in the group of patients using both metformin and SU medication, while no significant associations were observed in the metformin and SU only users. The type and number of medication used could be an indication for aggressivenes of diabetes phenotype and duration of having diabetes [26], as beta cell functioning decreases over time. People using multiple types of diabetes medication could potentially have worse glycaemic control compared to people only requiring one type of medication. However, the reason for the use of specific types of medication is unknown and could not be retrieved due to the use of registry data in this study. Also, it is not possible to define someone’s glycaemic control based on medication use and therefore studies including data on glycaemic control are required to be able to explain the results found in this study on UC patients. It should also be noted that people receiving diabetes medication may have relatively good glycaemic control compared to people with diabetes not receiving medication. Moreover, diabetes medication could potentially have an effect on cancer development as previous studies indicate [27–31]. Nonetheless, it is difficult to determine this effect independently from correlated health status in observational studies. Furthermore, the group of people who used diabetes medication might be different to the group of people who did not use any diabetes medication because of multiple other factors, such as lifestyle factors.
To our knowledge, very few studies have been performed in such a large sample size on the association between diabetes medication and bladder cancer characteristics. This study covers a large proportion of the population of bladder cancer patients within the Netherlands. The combination of using two national registries and thereby being able to link detailed information on medication use to cancer diagnosis data is a strength of this study.
A limitation of this registry study is that important potential confounding variables could not be included as this information was not available; mainly BMI, smoking and other lifestyle factors are potential causal factors in determining tumour aggressiveness that are also associated with T2DM [32]. It is expected that if smoking or BMI could have been included as a confounders, results would be attenuated, as smoking and BMI are both potentially associated with higher prevalence of T2DM and higher aggressiveness of bladder cancer [32–34]. Patients with a higher CDS score are expected to have a less healthy lifestyle overall and perhaps are more likely to be smokers and have a higher BMI. Notwithstanding, no relevant effect modification by CDS score was observed. Therefore, we cannot exclude residual confounding due to BMI, smoking or other lifestyle factors. Furthermore, the risk estimates for grade 2 (moderately differentiated) tumours are likely to be heterogenous as different classification systems were used by different pathologists throughout the country. Low grade and high grade tumours were always classified as such, however within the grade 2 category, potentially some poorly and well-differentiated tumours were also included.
Reversed causality could not have been excluded as no dose-response relationship could be determined. No information on medication duration was available retrospectively from the accessed registry from before 1998. Therefore, no information on age of diabetes diagnosis and medication initation and duration was available. This could potentially have influenced the results as a higher bladder cancer incidence was associated with a longer duration of diabetes, with a peak at 8 years [35, 36]. As a proxy for diabetes status, we included patients based on the prescribed T2DM medication but this has its limitations. Patients with uncontrolled diabetes are grouped as people not using any diabetes medication and therefore it is not an optimal proxy for diabetes status. It is expected that this may have attenuated the results as people with diabetes could be in the control group if they do not use any diabetes medication and therefore the observed association is lower than the expected true association. Also, no information on glycaemic control and medication compliance was available, which would have been better indicators for diabetes.
CONCLUSION
In this large explorative registry study, diabetes medication users appeared more likely to be diagnosed with a T2+ stage compared to patients who did not use any diabetes medication. NIAD users could potentially have a more aggressive/invasive type of UC. Nevertheless, no conclusions can be drawn as residual confounding due to smoking or other lifestyle related factors cannot be disregarded. Therefore, future studies require information on glycaemic control and lifestyle factors such as smoking to be able to substantiate the observed associations.
ACKNOWLEDGMENTS
The authors have no acknowledgements.
FUNDING
This research was supported by the VieCuri Medical Centre’s local Science and Innovation Fund.
AUTHOR CONTRIBUTIONS
JF: performance of work, interpretation of data & writing the article. DA: interpretation of data & writing the article. MdJ: interpretation of data & writing the article. JO: performance of work, interpretation of data & writing the article. PV: interpretation of data & writing the article. KA: interpretation of data & writing the article. MJH: interpretation of data & writing the article. JvdB: conception, interpretation of data & writing the article. FvO: conception, performance of work, interpretation of data & writing the article.
ETHICAL CONSIDERATIONS
Data is obtained from Dutch national registries that adhere to European privacy and ethics regulations and are ISO and NEN certified.
CONFLICTS OF INTEREST
J.P.M. Faessen, D.J.A.J. Oerlemans, M.R.P.A. de Jong, J.A. Overbeek, P.A.J. Vissers, K.K.H. Aben, M.L.G.Janssen-Heijnen, J.P.W. van den Bergh and F. H.M. van Osch have no conflicts of interests to report.
REFERENCES
[1] | Babjuk M , Burger M , Compérat ME , Gontero P , Mostafid AH , Palou J , et al. EA Guidelines: non-muscle invasive bladder cancer [Internet]. Available from: https://uroweb.org/guideline/non-muscle-invasive-bladder-cancer/#4 |
[2] | Antoni S , Ferlay J , Soerjomataram I , Znaor A , Jemal A , Bray F Bladder Cancer Incidence and Mortality: A Global Overview and Recent Trends, Eur Urol (2017) ;71: (1):96–108. |
[3] | Sung H , Ferlay J , Siegel RL , Laversanne M , Soerjomataram I , Jemal A , et al. Global Cancer Statistics GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries, CA Cancer J Clin (2021) ;71: (3):209–49. |
[4] | Babjuk M , Burger M , Compérat ME , Gontero P , Mostafid AH , Palou J , et al. EA Guidelines: non-muscle invasive bladder cancer [Internet]. Available from: https://uroweb.org/guideline/non-muscle-invasive-bladder-cancer/#4 |
[5] | Al-Zalabani AH , Stewart KFJ , Wesselius A , Schols AMWJ , Zeegers MP Modifiable risk factors for the prevention of bladder cancer: a systematic review of meta-analyses, Eur J Epidemiol (2016) ;31: (9):811–51. |
[6] | Zhu Z , Wang X , Shen Z , Lu Y , Zhong S , Xu C Risk of bladder cancer in patients with diabetes mellitus: An updated meta-analysis of 36 observational studies. BMC Cancer. 2013;13. |
[7] | Ripamonti E , Azoulay L , Abrahamowicz M , Platt RW , Suissa S A systematic review of observational studies of the association between pioglitazone use and bladder cancer. Diabetic Medicine. 2019. |
[8] | Feng YH , Velazquez-Torres G , Gully C , Chen J , Lee MH , Yeung SCJ The impact of type 2 diabetes and antidiabetic drugs on cancer cell growth, J Cell Mol Med (2011) ;15: (4):825–36. |
[9] | Zanders M Diabetes and cancer - a dangerous liaison? Zanders, M. M. J. Publisher’ s PDF, also known as Version of record Publication date: 2015. |
[10] | Zhao H , Grossman HB , Spitz MR , Lerner SP , Zhang K , Wu X Plasma levels of insulin-like growth factor-1 and binding protein-3, and their association with bladder cancer risk, Journal of Urology (2003) ;169: (2):714–7. |
[11] | Lin C , Travis RC , Appleby PN , Tipper S , Weiderpass E , Chang-Claude J , et al. Pre-diagnostic circulating insulin-like growth factor-I and bladder cancer risk in the European Prospective Investigation into Cancer and Nutrition, Int J Cancer (2018) ;143: (10):2351–8. |
[12] | Eich ML , Tregnago AC , Faraj SF , Palsgrove DN , Fujita K , Bezerra SM , et al. Insulin-like growth factor-1 receptor expression in upper tract urothelial carcinoma, Virchows Archiv (2019) ;474: (1):21–7. |
[13] | Rochester MA , Patel N , Turney BW , Davies DR , Roberts IS , Crew J , et al. The type 1 insulin-like growth factor receptor is over-expressed in bladder cancer, BJU Int (2007) ;100: (6):1396–401. |
[14] | Rochester MA , Patel N , Turney BW , Davies DR , Roberts IS , Crew J , et al. The type 1 insulin-like growth factor receptor is over-expressed in bladder cancer, BJU Int (2007) ;100: (6):1396–401. |
[15] | Neuzillet Y , Chapeaublanc E , Krucker C , de Koning L , Lebret T , Radvanyi F , et al. IGF1R activation and the in vitro antiproliferative efficacy of IGF1R inhibitor are inversely correlated with IGFBP5 expression in bladder cancer, BMC Cancer (2017) ;17: (1). |
[16] | Hwang EC , Kim YJ , Hwang IS , Hwang JE , Jungil S , Kwon DD , et al. Impact of diabetes mellitus on recurrence and progression in patients with non-muscle invasive bladder carcinoma: A retrospective cohort study, International Journal of Urology (2011) ;18: (11):769–76. |
[17] | Evers J , Grotenhuis AJ , Aben KKH , Kiemeney LALM , Vrieling A No clear associations of adult BMI and diabetes mellitus with non-muscle invasive bladder cancer recurrence and progression. PLoS One. 2020; |
[18] | Kuiper JG , Bakker M , Penning-Van Beest FJA , Herings RMC Existing data sources for clinical epidemiology: The pharmo database network, Clin Epidemiol (2020) ;12: , 412–22. |
[19] | Brierley JD , Gospodarowicz MK , Wittekind C TNM classification of malignant tumours - 8th edition. Union for International Cancer Control. 2017; |
[20] | Von Korff M , Wagner EH , Saunders K A chronic disease score from automated pharmacy data, J Clin Epidemiol (1992) ;45: (2):197–203. |
[21] | Licher S , Heshmatollah A , van der Willik KD , Stricker BHC , Ruiter R , de Roos EW , et al. Lifetime risk and multimorbidity of non-communicable diseases and disease-free life expectancy in the general population: A population-based cohort study, PLoS Med (2019) ;16: (2):1–17. |
[22] | Katikireddi SV , Skivington K , Leyland AH , Hunt K , Mercer SW The contribution of risk factors to socioeconomic inequalities in multimorbidity across the lifecourse: A longitudinal analysis of the twenty-07 cohort, BMC Med (2017) ;15: (1):1–10. |
[23] | Siddiqui AA , Spechler SJ , Huerta S , Dredar S , Little BB , Cryer B Elevated HbA1c is an independent predictor of aggressive clinical behavior in patients with colorectal cancer: A case-control study, Dig Dis Sci (2008) ;53: (9):2486–94. |
[24] | Faiena I , Dombrovskiy VY , Sultan RC , Salmasi AH , Singer EA , Weiss RE Effect of Uncontrolled Diabetes on Outcomes After Cystectomy in Patients With Bladder Cancer: A Population-Based Study, Clin Genitourin Cancer [Internet] (2016) ;14: (5):e509–14. http://dx.doi.org/10.1016/j.clgc.2016.02.004 |
[25] | Overbeek JA , van Herk-Sukel MPP , Vissers PAJ , van der Heijden AAWA , Bronsveld HK , Herings RMC , et al. Type 2 diabetes, but not insulin (analog) treatment, is associated with more advanced stages of breast cancer: A national linkage of cancer and pharmacy registries, Diabetes Care (2019) ;42: (3):434–42. |
[26] | José GJ , Jorge E , Lujan F , Ivanna Q , Jean-Marc C Type 2 diabetes: Prescription patterns and treatment outcomes of IDMPS survey in Argentina, Diabetes Res Clin Pract (2019) ;153: :86–92. |
[27] | Gallagher EJ , Leroith D Diabetes, antihyperglycemic medications and cancer risk: Smoke or fire? Curr Opin Endocrinol Diabetes Obes (2013) ;20: (5):485–94. |
[28] | Dankner R , Roth J More recent, better designed studies have weakened links between antidiabetes medications and cancer risk, Diabetic Medicine (2020) ;37: (2):194–202. |
[29] | Liu CQ , Sun JX , Xu JZ , Qian XY , Hong SY , Xu MY , et al. Metformin Use on Incidence and Oncologic Outcomes of Bladder Cancer Patients With T2DM: An Updated Meta-Analysis, Front Pharmacol (2022) ;13: (April):1–9. |
[30] | Mamtani R , Pfanzelter N , Haynes K , Finkelman BS , Wang X , Keefe SM , et al. Incidence of bladder cancer in patients with type 2 diabetes treated with metformin or sulfonylureas, Diabetes Care (2014) ;37: (7):1910–7. |
[31] | Tseng CH Pioglitazone and bladder cancer in human studies: Is it diabetes itself, diabetes drugs, flawed analyses or different ethnicities? Journal of the Formosan Medical Association (2012) ;111: (3):123–31. |
[32] | Maddatu J , Anderson-Baucum E , Evans-Molina C Smoking and the Risk of Type 2 Diabetes HHS Public Access, Physiol Behav (2017) ;176: (3):139–48. |
[33] | Ferro M , Vartolomei MD , Russo GI , Cantiello F , Farhan ARA , Terracciano D , et al. An increased body mass index is associated with a worse prognosis in patients administered BCG immunotherapy for T1 bladder cancer, World J Urol (2019) ;37: (3):507–14. |
[34] | Van Roekel EH , Cheng KK , James ND , Wallace DMA , Billingham LJ , Murray PG , et al. Smoking is associated with lower age, higher grade, higher stage, and larger size of malignant bladder tumors at diagnosis, Int J Cancer (2013) ;133: (2):446–54. |
[35] | Hu Y , Zhang X , Ma Y , Yuan C , Wang M , Wu K , et al. Incident Type 2 Diabetes Duration and Cancer Risk: A Prospective Study in Two US Cohorts, J Natl Cancer Inst (2021) ;113: (4):381–9. |
[36] | MacKenzie T , Zens MS , Ferrara A , Schned A , Karagas MR Diabetes and risk of bladder cancer: evidence from a case control study in New England. Cancer. 2011; |




