Switch Maintenance Therapy for Metastatic Urothelial Carcinoma
Abstract
INTRODUCTION:
While switch maintenance therapy is being increasingly investigated in solid tumors, it is a standard in only a few. We conducted a systematic review on switch maintenance therapy for metastatic urothelial carcinoma.
EVIDENCE ACQUISITION:
In this systematic review, we conducted a literature search in PubMed and Cochrane databases up to 2021, based on PRISMA statement guidelines. One hundred and fifty eight articles were identified and after a three-step selection process and six articles, using different agents were included in evidence synthesis. The primary end points were effect on overall survival, progression free survival, safety and tolerability.
EVIDENCE SYNTHESIS:
In the pre-immunotherapy era, targeted therapies like sunitinib, lapatinib and vinflunine were studied as switch maintenance therapy in metastatic urothelial carcinoma but did not show any overall survival benefit. Use of anti-PD-1/PD-L1 agents have shown promise as switch maintenance therapy; pembrolizumab showed improvement in progression free survival in a phase 2 trial and avelumab showed improvement in overall survival and progression free survival in the phase 3 JAVELIN Bladder 100 trial.
CONCLUSION:
Immunotherapy with anti-PD-1/PD-L1 agents has emerged as an effective switch maintenance strategy in patients with metastatic urothelial carcinoma. Intensification of the immunotherapy backbone in this setting can potentially further enhance outcomes. Emerging evidence shows a potential role of Poly (ADP-ribose) polymerase (PARP) inhibitors in this setting as well. Results from ongoing and planned studies will help us understand which switch maintenance approaches would be most effective for improving outcomes in metastatic urothelial carcinoma.
INTRODUCTION
The standard of care first-line treatment for patients with locally advanced or metastatic urothelial carcinoma (mUC) is platinum-based chemotherapy (DD-MVAC (methotrexate, vinblastine, doxorubicin,cisplatin) or gemcitabine plus cisplatin/carboplatin). The median overall survival (OS) is up to 15 months with cisplatin-based regimens and up to 13 months with carboplatin-based regimens [1–7].
[2–7] However, these agents cannot be continued long-term in those who respond or have stable disease due to cumulative toxicities. Most patients eventually recur and responses with immunotherapy in patients with platinum-refractory mUC are modest. Furthermore, only a minority of patients receive second-line treatment [8–11]. Hence, there is an unmet need to enhance and prolong benefits of first-line chemotherapy and improve survival with effective use of maintenance therapies after first-line therapy in patients with mUC who do not progress on it to offer the best outcomes. The rationale for maintenance therapy is to delay the disease from progressing by either eliminating residual cancer, retarding cell turnover via inhibitory signaling, preventing tumor neo-angiogenesis, or through immunologic control. Major goal of maintenance therapy is to sustain the favorable state achieved with induction chemotherapy and prolonging progression-free survival (PFS) and OS [12]. Maintenance therapy can use two approaches; continuation maintenance, which essentially is continuation of an agent used as part of the combination induction regimen (i.e., treatment de-intensification/de-escalation), and switch maintenance, which adds a different sequential treatment after induction regimen [13]. In this article, we will review the role of switch maintenance treatment strategies in mUC, their role in current clinical practice and future considerations.
EVIDENCE ACQUISITION
Search Strategy
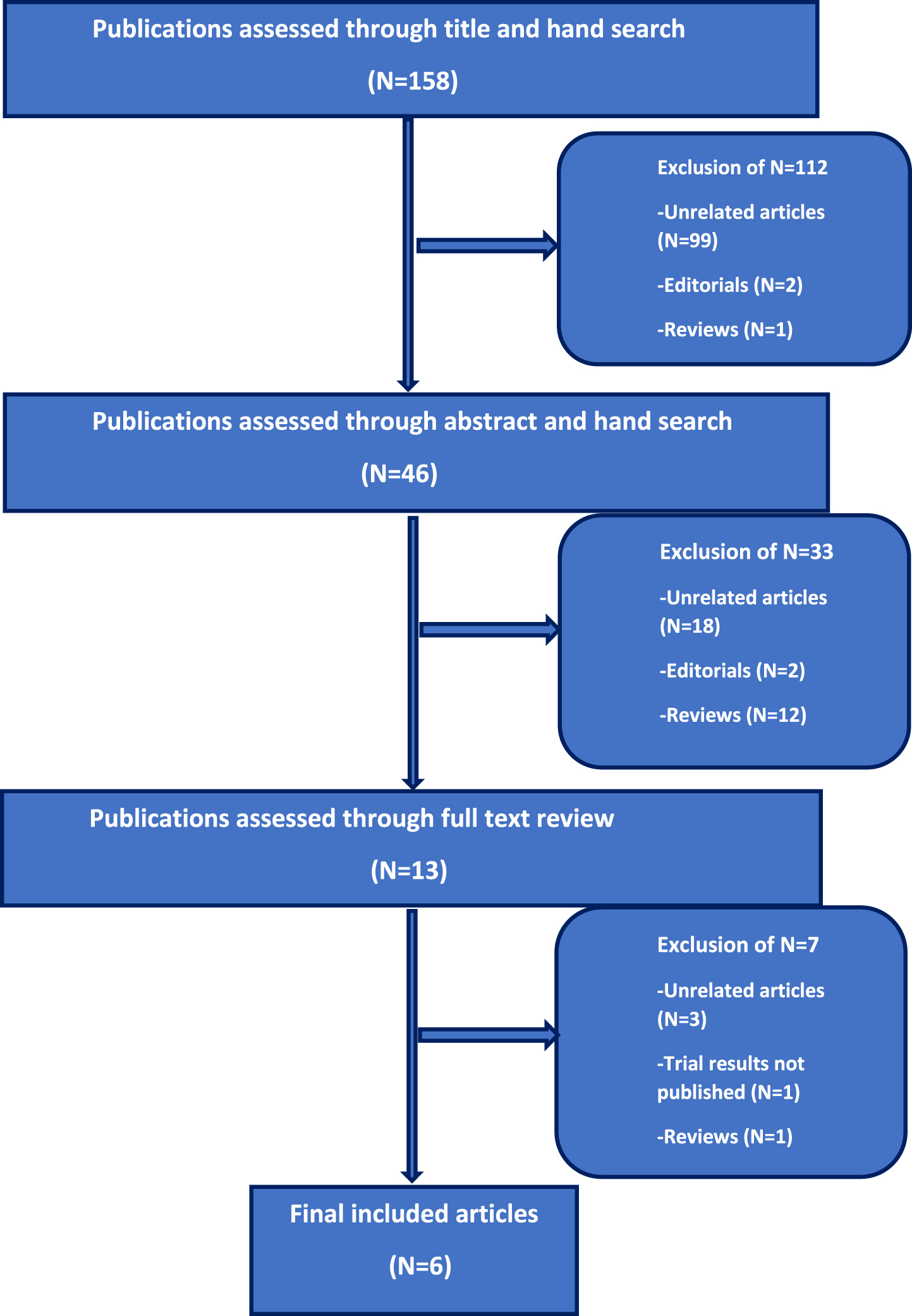
The aim of this systematic review is to analyze clinical trials in patients with mUC who underwent switch maintenance treatment after achieving either treatment response (partial or complete response) or stable disease with first-line chemotherapy and identify differences in outcomes with different agents. We conducted a systematic literature search in the PubMed and Cochrane databases according to the Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) statement for articles published up to February, 2022. We also looked at abstracts from major oncology conferences up to 2022. Articles were searched using one or several combinations of the following terms: (“switch”) AND (“maintenance”) AND (“therapy” OR “treatment”) AND (“metastatic” OR “advanced”) AND (“urothelial” OR “bladder”) AND (“cancer” OR “carcinoma” OR “tumor” OR “malignancy” OR “neoplasm”). One hundred and fifty eight articles were identified on initial screening and we further searched selected articles to identify relevant ones pertaining to our aim. The selection process was done in three steps; 1) initial screening of the title was done to identify eligible publications, 2) screening of selected abstracts and 3) full text reading of respective publications (Fig. 1).
Fig. 1
Flowchart outlining selection process of the includedstudies.
Prespecified questions
Research questions were defined as follows: 1) which agents have been investigated as switch maintenance treatment in patients with mUC? 2) do these switch maintenance therapies improve OS and PFS in patients with mUC? 3) what is the safety and tolerability of these switch maintenance therapies?
Inclusion and exclusion criteria
Only published prospective clinical trials using switch maintenance therapies in patients with locally advanced/mUC who did not have disease progression after first-line chemotherapy were included in this systematic review. Exclusion criteria were review articles, non-English articles, editorial letters, case reports/series, and repeated publications to avoid publication bias.
Six articles met the afore-mentioned criteria and were included for evidence synthesis (Fig. 1).
Evidence Synthesis
Historically, switch maintenance therapy in mUC was attempted with limited success with tyrosine kinase inhibitors and chemotherapy but now, immunotherapy has successfully been established as the only effective approach so far as discussed below.
Tyrosine kinase inhibitors
Sunitinib
Angiogenesis plays a significant role in growth and metastases of UC and vascular endothelial growth factor (VEGF) has been identified as an important regulator of both normal and pathologic angiogenesis and increased VEGF expression has been reported in patients with UC [11, 14, 15]. Sunitinib is an oral tyrosine kinase inhibitor (TKI) with activity against vascular endothelial growth factor receptors (VEGFRs). Based on preclinical activity of sunitinib in bladder cancer models, Grivas et al. conducted a phase 2 randomized controlled trial (RCT) of maintenance sunitinib versus placebo in patients with locally advanced or mUC who did not progress after first-line chemotherapy [16]. Fifty-four patients were randomized to receive either Sunitinib at a dose of 50 mg orally daily (28 days on,14 days off) or placebo for at least 12 weeks until disease progression or toxicity. Primary endpoint was 6-month progression rate. The most common grade 3 or higher adverse events (AEs) in patients who received sunitinib vs placebo were thrombocytopenia (23.1% vs 7.7%), diarrhea and mucositis (15.4% vs 0%), fatigue (15.4 vs 3.8%) and hypertension (11.5% vs 0%) [16]. The study closed prematurely owing to slow accrual and toxicity from sunitinib. Maintenance sunitinib compared to placebo did not improve the 6-month progression rate. The median PFS (2.9 vs 2.7 months) and OS (10.5 vs 10.3 months) did not differ significantly either between treatment groups (p = 0.0002) [16].
Lapatinib
Lapatinib is a TKI against human epidermal growth factor receptor (HER1/HER2). Preclinical data of targeting HER1/HER2 in UC supported its testing in a clinical study in mUC patients with HER1+ or HER2 + tumors [17]. Powles et al. conducted a phase 3 RCT that included 232 mUC patients with HER1/HER2 +tumors who did not have disease progression after first-line chemotherapy [18]. Patients were randomized to receive either maintenance lapatinib at 1500 mg orally daily or placebo. Compared to placebo, lapatinib did not improve PFS which was a primary outcome (HR, 1.07; 95% CI, 0.81 to 1.43; P = 0.63) or OS, a secondary outcome (HR, 0.96; 95% CI, 0.70 to 1.31; P = 0.80). The rate of grade 3 or higher AEs was 8.6% vs 8.1% for lapatinib vs placebo. This trial did not demonstrate any clinical benefit with maintenance lapatinib for patients with HER1/2 + mUC, even in patients with highest level of HER1/2 expression (3+ on immunohistochemistry) [18].
Chemotherapy
Vinflunine
Vinflunine is an antineoplastic agent belonging to the vinca alkaloid class that inhibits microtubule dynamics with a greater efficacy than other microtubule drugs [19]. In a phase 3 RCT comparing vinflunine with best supportive care (BSC) in patients with platinum-refractory mUC, vinflunine showed significant improvement in PFS, response rates and disease control rates compared to BSC, leading to its approval by the European Medicines Agency [20]. Vinflunine is not approved for use outside of Europe.
Vinflunine was subsequently studied as a switch maintenance therapy in patients with mUC in the MAJA; SOGUG 2011/02 trial, a multicenter, open label, phase 2 RCT across Spanish hospitals and included patients with mUC who did not progress after 4 to 6 cycles of cisplatin and gemcitabine (carboplatin was allowed after cycle 4) [21]. Patients were randomized to receive vinflunine or BSC until disease progression. Vinflunine was given every 21 days intravenously at 320 mg/m2 or at 280 mg/m2 in patients with ECOG performance status of 1, age > / = 75 years, prior pelvic radiotherapy or creatinine clearance < 60 mL/min. The primary endpoint was median PFS greater than 5.3 months in the vinflunine group, assessed by modified intention to treat. Between 2012 and 2015, eighty-eight patients with mUC were enrolled, of whom 45 received vinflunine and 43 best supportive care (BSC). PFS was 6.5 months in the vinflunine group and 4.2 months in the BSC group (HR-0.59; 95% CI, 0.37–0.96, p = 0.031). Vinflunine showed a trend towards improved OS; median OS in vinflunine vs BSC group was 16.7 months vs 13.2 months respectively (HR-0.736; 95% CI, 0.44–1.24, p = 0.182); objective responses were achieved in 21% patients in vinflunine group vs 7% in BSC group respectively and vinflunine had an acceptable safety profile. The study showed that vinflunine switch maintenance therapy led to an improvement in PFS that exceeded the acceptable pre-specified threshold compared to BSC alone [21]. However, vinflunine is not approved as a switch maintenance therapy.
Rationale of immunotherapy switch maintenance in mUC
After a long gap of 40 years, anti-PD-1/PD-L1 inhibitors (immune checkpoint inhibitors; ICIs) transformed the treatment paradigm in mUC. Since 2016, multiple ICIs have been approved for patients with platinum-refractory mUC including atezolizumab, durvalumab, avelumab, nivolumab and pembrolizumab [22–27]. Atezolizumab and durvalumab were withdrawn from this indication in the US in 2021 due to lack of benefit in confirmatory trials. Importantly, in mUC, first-line combination of platinum-based chemotherapy and ICI was not significantly better than chemotherapy in the KEYNOTE-361 and IMvigor130 trials [6, 7]. In the Keynote 361 trial, addition of pembrolizumab to chemotherapy did not improve PFS or OS compared to chemotherapy alone [7]. In the IMvigor130 trial, addition of atezolizumab to chemotherapy showed a modest improvement in PFS but no improvement in OS so far [6].
The prior standard of care for mUC patients who did not progress after first-line platinum-based chemotherapy was surveillance and ICI was only indicated for patients who progressed on or after platinum-based chemotherapy. The earlier sequential use of ICI as switch maintenance after first-line chemotherapy in patients with mUC who do not progress is attractive for several reasons. Apart from its cytotoxic effects, conventional chemotherapy can promote tumor immunity by depleting myeloid derived immunosuppressor cells (gemcitabine), or by increasing the expression and presentation of tumor antigens (cisplatin, gemcitabine) thereby making tumor cells more susceptible to T cell mediated lysis [28]. The pro-immunogenic effects of chemotherapy can provide an added advantage while considering switch maintenance therapy with ICIs. The cancer immune-editing conceptual framework provides a rationale for this approach. Tumor growth or disease progression (“escape phase”) happens when rate of tumor proliferation exceeds ability of the immune system to control it. Chemotherapy can help the immune system to regain control (“equilibrium phase”) or even decrease tumor burden (“elimination phase”), but this effect lasts until tumor cells acquire immune escape mechanisms [17]. Use of switch maintenance therapy with ICI after chemotherapy can potentially intensify immune elimination or enable immune system to prolong the equilibrium phase, thus delaying disease progression.
Disease control achieved with chemotherapy provides better patient selection for sequential use of ICI maintenance and incorporation of ICI as a switch maintenance strategy can enable more patients to receive treatment.
Pembrolizumab
Pembrolizumab is an anti-PD-1 agent and showed significant improvement in OS compared to investigator’s choice of second-line chemotherapy in the Phase III Keynote 045 trial [22]. The role of pembrolizumab as switch maintenance therapy was studied in the Hoosier Cancer Research Network GU14-182 trial, a multicenter phase 2 RCT that enrolled 108 patients with mUC achieving at least stable disease on first line chemotherapy [29]. Patients were randomized to receive either Pembrolizumab 200 mg intravenously every 3 weeks or placebo for upto 24 months in the absence of disease progression or severe toxicity. The primary objective was to compare the PFS; it was significantly longer with maintenance pembrolizumab versus placebo (5.4 months vs 3.0 months hazard ratio, 0.65; log-rank P = 0.04; maximum efficiency robust test P = 0.039). This significant improvement in PFS with pembrolizumab was seen despite modification of samples size and analysis plan during study enrollment. The median OS was 22 months with pembrolizumab and 18.7 months with placebo (log-rank P = 0.74). However, OS was a secondary endpoint and the study was not adequately powered to detect an OS improvement. There was no correlation of outcomes with PD-L1 expression in tumors. Safety profile of pembrolizumab was similar to prior experience with pembrolizumab. Treatment related grade 3-4 adverse events occurred in 59% of patients receiving pembrolizumab and 38% of patients receiving placebo. Immune-related adverse events (irAE) from pembrolizumab requiring steroids occurred in 20% of patients and there was 1 death from hepatitis with pembrolizumab. This study showed, for the first time, encouraging activity of switch maintenance with immunotherapy in PFS improvement and deepening of responses achieved with 1st-line chemotherapy in mUC [29].
Avelumab
Avelumab is an anti-PD-L1 agent approved for us in patients with platinum-refractory mUC [27]. The JAVELIN Bladder 100 trial was a phase 3 multicenter, multinational, randomized, open-label, parallel-arm study investigating first-line maintenance treatment with avelumab plus BCS vs BCS alone in patients with locally advanced or mUC who did not have disease progression after 1st-line platinum-containing chemotherapy [30]. Seven hundred patients were randomly assigned to receive either avelumab 10 mg/kg intravenously every 2 weeks plus BSC or BSC alone within 4–10 weeks of first-line platinum-containing chemotherapy. The primary endpoint was OS in overall population and PD-L1 positive population, and secondary endpoints included PFS and safety. Median OS with avelumab and BCS was significantly longer compared to BCS and placebo in all-comers (21.4 vs 14.3 months, respectively; HR 0.69; 95% CI, 0.56 to 0.86) and was not reached with avelumab in patients with PD-L1 + tumors. The landmark OS at 1 year was 79.1% in patients in the avelumab and BSC group and 60.4% in the patients in control group (HR 0.56; 95% CI, 0.40 to 0.79; P < 0.001). The PFS was 3.7 months with avelumab and BCS (95% CI, 3.5 to 5.5) compared to 2 months in the control group (95% CI, 1.9 to 2.7). The safety profile in this trial was similar to previous studies with avelumab. Grade 3 or higher AEs occurred in 47.4% of patients receiving avelumab; and grade 3 irAEs occurred in 7% of patients. Treatment discontinuation from AEs occurred in around 12% patients who received avelumab. The results from this trial led to the Food and Drug Administration (FDA), Health Canada,European Commission and the National Institute for Health and Care Excellence (NICE) approval of avelumab for maintenance treatment of patients with locally advanced or mUC that has not progressed with first-line platinum-containing chemotherapy. This approval is the first and only switch maintenance therapy approval for mUC patients so far [31].
Avelumab benefit was seen despite more frequent use of subsequent treatment in the control group, including ICIs, which highlights the importance of starting maintenance immunotherapy soon after patients receiving induction chemotherapy are deriving benefit (stable disease or better) rather than using when patients experience disease progression. The OS benefit with avelumab maintenance was similar regardless of prior cisplatin or carboplatin-based first-line therapy [34]. The long-term outcomes from the JAVELIN Bladder 100 trial among patients receiving avelumab maintenance therapy were recently reported [31]. The median follow-up in both arms exceeded 38 months and avelumab and BSC continued to show consistent improvement in OS and PFS over BSC (23.8 months vs 15.0 months, respectively; HR 0.76 (0.631–0.915); P = 0.0036) and there were no new safety signals [31]. Importantly, the patient-reported outcomes did not show any detrimental effects of addition of avelumab to BSC compared to BSC alone [32].
RECENT EXPERIENCE WITH OTHER NOVEL AGENTS AS MAINTENANCE THERAPY IN mUC
Poly (ADP-ribose) polymerase (PARP) inhibitors
PARP inhibitors olaparib and niraparib have are approved as first-line maintenance therapy in patients with ovarian cancer who achieve a PR or CR to first-line platinum-based chemotherapy [33, 34]. Exploitation of targeted therapies remains an unmet need in mUC. Recently, results from two trials, ATLANTIS (ISRCTN25859465) and MEET-URO12 (NCT03945084) exploring the role of Poly (ADP-ribose) polymerase (PARP) inhibitors as switch maintenance therapy in patients with mUC were reported [35–37].
The ATLANTIS is an adaptive, multi-comparison, phase II trial platform testing multiple biomarker selected maintenance therapies for patients with mUC in the U.K. who did not progress after 4 to 8 cycles of platinum-based chemotherapy [36]. This study provides a generic framework that will allow new treatments to be introduced into the study in future with prospective stratification based on a molecular target.
A subset of patients with alterations in defined DRD associated genes (ATM, BARD1, BRCA1, BRCA2, BRIP1, CDK12, CHEK2, FANCA, NBN, PALB2, RAD51, RAD51B, RAD51 C, RAD51D, RAD54 L) and/or germline BRCA1 or BRCA2 alteration were randomized to PARP inhibitor rucaparib 600 mg twice daily orally or placebo [36]. The primary endpoint was PFS. Seventy four patients were biomarker positive and 40 patients were randomized to rucaparib arm. Median PFS was 35.3 weeks with rucaparib and 15.1 weeks with placebo (hazard ratio 0.53, 80% CI 0.30–0.92, 1 sided p = 0.07). Median duration of treatment with rucaparib was 10 cycles and 6 cycles with placebo. Rucaparib was well tolerated overall with fatigue, nausea and rash were more common with rucaparib compared to placebo. The promising activity with rucaparib in biomarker-selected mUC patients warrants further investigation as switch maintenance therapy.
The MEET-URO12 trial was a phase II Italian trial of PARP inhibitor, niraparib in non-biomarker selected patients with mUC who did not progress after first-line platinum-based chemotherapy. Patients were randomized 2:1 to niraparib (300 or 200 mg/day) and BSC vs BSC alone [37]. Fifty-eight patients were enrolled with molecular information available for 47 patients; 21 (44.7%) had HRR alterations: 6 (12.8%) had known pathogenic mutations. The study accrual was stopped prematurely due to approval of maintenance avelumab. After a median follow-up of 8.5 months, the median PFS was 2.1 months with niraparib and BSC vs 2.4 months with BSC alone in all comers (HR 0.92; 95% CI 0.49 –1.75, p = 0.81). In patients with HRR alterations, median PFS was 2.0 months with niraparib and BSC vs 1.9 months with BSC. Maintenance niraparib did not improve PFS in this mUC maintenance trial regardless of HRR alterations [37]. Key toxicities with niraparib were myelosuppression, fatigue, constipation, and nausea. The results from this study do not support use of maintenance niraparib in mUC patients.
The ongoing TALASUR trial is a single-arm trial evaluating the combination of another PARP inhibitor talazoparib and avelumab as a switch maintenance therapy (NCT0467836) (Table 2).
Multi-targeted Tyrosine Kinase Inhibitor -Cabozantinib
Cabozantinib is a receptor tyrosine kinase inhibitor (TKI) with activity against a broad range of targets, including MET, RET, AXL, VEGFR2, FLT3, and c-KIT. The ATLANTIS trial platform also explored the role of VEGF TKI, cabozantinib as maintenance therapy in mUC patients who did not progress after 4–8 cycles of platinum-based chemotherapy [36]. The results from the cabozantinib cohort were reported recently [38]. Patients who were not selected for other biomarker-based cohorts were randomized to cabozantinib 40 mg daily or placebo and primary endpoint was PFS. Due to the Covid-19 pandemic, the planned accrual and statistical assumptions were changed and then the study was closed early due to a change in the standard of care with approval of maintenance avelumab. Thirty patients were randomized to cabozantinib and 31 to placebo; the median PFS was 13.7 weeks (80% CI 12.1–23.3) with cabozantinib and 15.8 weeks (80% CI 11.3–23.6) with placebo (adjusted HR 0.89 favoring cabozantinib, 80% CI 0.61–1.3, 1-sided p = 0.35). In this study, cabozantinib did not show significant benefit compared to placebo. However, this could be in part due to selection bias, change in power of the study as well as potential over-estimation of desired PFS benefit. Despite being a negative study, this showed that cabozantinib was tolerable with median duration of treatment of 13 cycles of cabozantinib compared to 10 cycles of placebo. While this study did not show benfit of single-agent cabozanitnib as switch maintenance therapy in mUC, it underscores the importance of considering combination studies with ICI in this setting.
Vaccines
In another phase II trial, a cancer peptide vaccine, S-588410 was studied as a maintenance therapy in patients with mUC after at least 4 cycles of first-line platinum containing chemotherapy without disease progression [39]. S-588410 is a cancer peptide vaccine composed of 5 human leukocyte antigen (HLA)-A*24:02-restricted epitope peptides derived from antigens which are also highly expressed in UC. The aim was to evaluate effect of S-588410 maintenance therapy on peptide-specific cytotoxic T-lymphocyte (CTL) induction and the primary endpoint was CTL induction rate at 12 weeks. Eighty-one patients were enrolled and HLA-A*24:02 positive patients received S-588410 subcutaneously every week for 12 weeks, then every 2 weeks for up to 2 years and HLA-A*24:02 negative patients were on observation arm. The vaccine induced CTLs in 93.3% of patients. Median PFS was 18.1 weeks in the S-588410 group and 12.5 weeks in the observation group; median OS was 71 and 99 weeks respectively. The most frequent treatment-emergent AEs with S-588410 were injection site reaction in 93% patients as well as pyrexia, rash and pruritis. S-588410 showed a potent immune response in maintenance setting and future studies are needed to further establish its potential role.
Intensification of avelumab maintenance in mUC with novel agents
Building upon the maintenance avelumab backbone is the next rational step to further improve outcomes in patients with mUC. The addition of an effective, non-cross resistant therapy with non-overlapping toxicity profile can further intensify and expand the benefits of maintenance avelumab therapy in mUC.
[40, 41] While single-agent cabozantinib is not an effective approach as a switch maintenance therapy after platinum-based chemotherapy in mUC as seen in the ATLANTIS trial, [38] there is preclinical and clinical evidence that owing to it’s immunomodulatory nature, cabozantinib can be more effective in combination with an ICI [41]. Indeed, this provides the rationale for the ongoing MAIN-CAV trial, the first phase III randomized control trial testing the combination of cabozantinib and avelumab versus avelumab after first-line platinum-based chemotherapy in patients with mUC. Six hundred and fifty four adult patients will be randomized 1:1 within 3–10 weeks after last dose of chemotherapy to receive avelumab 800 mg intravenously every 2 weeks or combination of avelumab and cabozantinib 40 mg orally daily for up to 2 years. The primary endpoint is OS (NCT05092958) [42].
A novel, gut microbiome derived, single strain oral live biotherapeutic product, MRx0518 has been shown to have immunomodulatory and anti-tumor effect in multiple cancer models and when combined with ICI, can potentially overcome acquired resistance to ICI [43]. In an ongoing trial, combination of MRx0518 and avelumab is being studied as switch maintenance strategy in patients with mUC (NCT05107427).
Table 2 lists the ongoing switch maintenance trials in mUC.
CONCLUSIONS AND FUTURE DIRECTIONS
Switch maintenance therapy with a variety of agents has been studied extensively in patients with mUC who do not progress after first-line platinum-based chemotherapy [16, 18, 21, 29, 30, 37]. Our systematic review summarizes the six published studies that explored switch maintenance strategy in patients with mUC to date (Table 1). Sunitinib and lapatinib did not show a clinical benefit and vinflunine showed a modest improvement in PFS but is not approved for us for this setting. Niraparib did not show PFS benefit in this patient spopulation, regardless of HRR alterations. Pembrolizumab is the first immunotherapy to show improvement in PFS over placebo in a phase 2 study but there was no improvement in OS. Switch maintenance with avelumab and BSC led to significant improvement in PFS and OS compared to BSC in the pivotal phase 3 Javelin Bladder 100 trial. Based on level 1 evidence from the Javelin Bladder 100 trial, avelumab has been widely approved by regulatory agencies across the US, Europe, Canada and U.K. and is the preferred standard of care for patients with mUC who do not progress after first-line platinum-containing chemotherapy [34, 35].
Table 1
Clinical trials published on switch maintenance therapy in mUC
| STUDY AGENTS | SUNITINIB | LAPATINIB | VINFLUNINE | PEMBROLIZUMAB | AVELUMAB | NIRAPARIB |
| Year study was published | 2013 | 2016 | 2017 | 2020 | 2020 | 2022 |
| Study | NCT00393796 15 | UK NCRI LAMB 17 | MAJA; SOGUG 2011/02 20 | HCRN GU14-182 32 | JAVELIN Bladder 100 34 | Meet-UROL12[37] |
| Phase | 2 | 3 | 2 | 2 | 3 | 2 |
| Patients accrued | 54 | 232 | 88 | 108 | 700 | 58 |
| Median age (yrs) | 69 (48–84) | 70.7 (63.9–77.2) | 63.7 (42.1–83.9) | 68 (41–83) | 68 (37–90) | 69 (44–84) |
| ECOG status | 0:38.5% | 0:45.7% | 0:47% | 0:40% | 0:60.9% | 0:65.5& |
| 1:57.6% | 1:44.8% | 1:53% | 1:60% | 1:38.9% | 1:34.5% | |
| 2:3.9% | >1:9.5% | 2:0.3% | ||||
| Drug dose/route | 50 mg PO daily (28 days on,14 days off) | 1500 mg PO daily (six 250 mg tablets) | 320 mg/m2 IV q3 weeks | 200 mg IV q3 weeks IV | 10 mg/kg IV q2 weeks | 300 mg or 200 mg Po daily |
| Patients requiring dose reduction (%) | 42.3 | 7 | 16 | Not permitted | Not permitted | 47.4 |
| Treatment free interval allowed since first-line chemotherapy | 6 weeks | 4 –10 weeks | 6 weeks | 2 –6 weeks | 4 –10 weeks | 4 weeks |
| Study end points | Primary: 6-month progression rate | Primary: PFS | Primary: Median PFS > 5.3 months | Primary: PFS | Primary: OS | Primary: PFS |
| Secondary: OS, safety, change in serum VEGF/ sVEGFR2 | Secondary: OS, ORR, outcomes of subsets, AEs | Secondary: PFS, OS, ORR, disease control, DOR, TTR, safety, pharmacogenomics | Secondary: OS, treatment outcomes according to PD-L1 status | Secondary: PFS, safety | Secondary: 6-month PFS rate, ORR, DOR,OS, safety and tolerability, PRO | |
| Median follow up (months) | 10.3 | Not known | 15.6 | 12.9 | 19 | 8.5 |
| OS in months (treatment vs control group) | 10.5 vs 10.3 | 12.6 vs 12 (HR-0.96, 95% CI, 0.7 to 1.31; p = 0.8) | 16.7 vs 13.2 (HR-0.736; 95% CI, 0.44–1.24, p = 0.182) | 22 vs 18.7 (HR-0.91, 95% CI, 0.52 to 1.59; p = 0.8) | 21.4 vs 14.3 (HR-0.69, 95% CI, 0.56 to 0.86; p = 0.001) | Not reported |
| PFS in months (treatment vs control group) | 2.9 vs 2.7 | 4.5 vs 5.1 (HR-1.07, 95% CI, 0.81 to 1.43; p = 0.63) | 6.5 vs 4.2 (HR-0.59, 95% CI, 0.37 to 0.96; p = 0.031) | 5.4 vs 3 (HR-0.65; p = 0.04) | 3.7 vs 2 (HR-0.62, 95% CI, 0.52 to 0.75) | 2.1 vs 2.4 (HR 0.92; 95% CI, 0.49 to 1.75, p = 0.81) |
| Objective response rate (ORR) (treatment vs control group) | Not reported | 14% vs 8% (p = 0.14) | 21% vs 7% | 23% vs 10% | 9.7% vs 1.4% (95% CI, 2.82 to 24.45) | Not reported |
| Adverse events (AEs) with study agent | Thrombocytopenia (23.1%), Diarrhea and mucositis (15.4%), fatigue (15.4%), Hypertension (11.5%) | Diarrhea (60.8%), Rash (44.3%), Pain (38.1%), Fatigue (35.1%) | Fatigue (91%), Infection (74%), Constipation (71%), back pain (55%), Neuropathy (52%) | Diarrhea (31%), Cough (25%), Dyspnea (22%), Fatigue (22%), Pruritis (22%) | Fatigue (17.7%), Pruritis (17.2%), UTI (17.2%), Diarrhea (16.6%) | Anemia (50%) Thrombocytopenia (36.8%) Neutropenia (21.1%), Fatigue (31.6%), Constipation (31.6%), Mucositis (13.2%) Nausea (13.2%) |
| Grade 3-4 AEs (%) | >5 | 8.6 | 92 | 59 | 47.4 | 65.8 |
| Drug Discontinuation rate (%) | 96 | 6 | 51 | 87 | 11.9 | Not reported |
| Outcomes | Study closed prematurely. No improvement in 6-month disease progression rate | No PFS improvement | PFS improvement | Prolonged PFS significantly | Prolonged OS and PFS significantly | Study closed prematurely. No PFS improvement. |
Table 2
Ongoing clinical trials exploring switch maintenance therapy in mUC
| Clinical trial | Drug | Phase | Estimated enrollment | Primary outcome |
| NCT05092958 (MAIN-CAV) | Cabozantinib + avelumab vs avelumab | 3 | 654 | OS |
| NCT03193788 (PREMIER) | Pemetrexed | 3 | 74 | PFS |
| NCT05107427 (AVENU) | MRx0518 + Avelumab | 2 | 30 | Safety and PFS at 6 months |
| NCT03945084 (Meet-URO 12) [37] | Niraparib | 2 | 58 | PFS |
| NCT04678362 (TALASUR) | Talazoparib + Avelumab | 2 | 50 | PFS |
| ISRCTN25859465 (ATLANTIS) [38] | Group 1-Cabozantinib | 2 | Group 1-140 | PFS |
| Group 2-Rucaparib | Group 2-48 | |||
| Group 3-Enzalutamide | Group 3-80 |
In summary, first-line switch maintenance therapy with avelumab has revolutionized the treatment paradigm in patients with mUC who do not progress after first-line platinum-based chemotherapy with an improvement in OS and PFS. Ongoing trials of avelumab intensification as well as exploitation of novel targeted therapies will further shape the evolution of switch maintenance therapies in mUC to improve outcomes even more. Opportunities exist to research biomarkers to identify which patients could most benefit from treatment with maintenance therapy approaches as well as understanding of biomarkers of resistance to avoid unnecessary therapy in patients unlikely to respond. Lastly, financial toxicity of treatments needs to be considered and optimal duration of treatment needs to be determined.
ACKNOWLEDGMENTS
The authors have no acknowledgements.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
Both the authors contributed to conception, interpretation of data and writing this article.
CONFLICT OF INTEREST
Dr. Ramsha Ahmed reports no conflict of interest.
Dr. Shilpa Gupta reports consulting fees from Bristol Myers Squibb, Merck, Pfizer, EMD Sorono, Bayer, Gilead, Seattle Genetics, Loxo Oncology; speakers bureau for Bristol Myers Squibb, Janssen, Gilead and Seattle Genetics; stock options for Moderna, BionTek; participation in advisory board for Bristol Myers Squibb, Merck and Seattle Genetics.
REFERENCES
[1] | von der Maase H , Hansen SW , Roberts JT , Dogliotti L , Oliver T , Moore MJ , et al Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. (2000) ;18: (17):3068–77. |
[2] | von der Maase H , Sengelov L , Roberts JT , Ricci S , Dogliotti L , Oliver T , et al Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol. (2005) ;23: (21):4602–8. |
[3] | Sternberg CN , de Mulder P , Schornagel JH , Theodore C , Fossa SD , van Oosterom AT , et al Seven year update of an EORTC phase III trial of high-dose intensity M-VAC chemotherapy and G-CSF versus classic M-VAC in advanced urothelial tract tumours. Eur J Cancer. (2006) ;42: (1):50–4. |
[4] | Dogliotti L , Carteni G , Siena S , Bertetto O , Martoni A , Bono A , et al Gemcitabine plus cisplatin versus gemcitabine plus carboplatin as first-line chemotherapy in advanced transitional cell carcinoma of the urothelium: results of a randomized phase 2 trial. Eur Urol. (2007) ;52: (1):134–41. |
[5] | De Santis M , Bellmunt J , Mead G , Kerst JM , Leahy M , Maroto P , et al Randomized phase II/III trial assessing gemcitabine/carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer who are unfit for cisplatin-based chemotherapy: EORTC study 30986. J Clin Oncol. (2012) ;30: (2):191–9. |
[6] | Galsky MD , Arija JAA , Bamias A , Davis ID , De Santis M , Kikuchi E , et al Atezolizumab with or without chemotherapy inmetastatic urothelial cancer (IMvigor130): amulticentre, randomised, placebo-controlled phase 3 trial. Lancet. (2020) ;395: (10326):1547–57. |
[7] | Powles T , Csoszi T , Ozguroglu M , Matsubara N , Geczi L , Cheng SY , et al Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. Lancet Oncol. (2021) ;22: (7):931–45. |
[8] | Flannery K , Boyd M , Black-Shinn J , Robert N , Kamat AM Outcomes in patients with metastatic bladder cancer in the USA: a retrospective electronic medical record study. Future Oncol. (2019) ;15: (12):1323–34. |
[9] | Herchenhorn D , Freire V , Oliveira T , Tarouquella J Sequential therapies for advanced urothelial cancer: Hope meets new challenges. Crit Rev Oncol Hematol. (2021) ;160: :103248. |
[10] | Swami U , Grivas P , Pal SK , Agarwal N Utilization of systemic therapy for treatment of advanced urothelial carcinoma: Lessons from real world experience. Cancer Treat Res Commun. (2021) ;27: , 100325. |
[11] | Wu W , Shu X , Hovsepyan H , Mosteller RD , Broek D VEGF receptor expression and signaling in human bladder tumors. Oncogene. (2003) ;22: (22):3361–70. |
[12] | Lee JE , Chung CU Update on the evidence regarding maintenance therapy. Tuberc Respir Dis (Seoul). (2014) ;76: (1):1–7. |
[13] | Grivas P , Monk BJ , Petrylak D , Reck M , Foley G , Guenther S , et al Immune Checkpoint Inhibitors as Switch or Continuation Maintenance Therapy in Solid Tumors: Rationale and Current State. Target Oncol. (2019) ;14: (5):505–25. |
[14] | Yang CC , Chu KC , Yeh WM The expression of vascular endothelial growth factor in transitional cell carcinoma of urinary bladder is correlated with cancer progression. Urol Oncol. (2004) ;22: (1):1–6. |
[15] | Bernardini S , Fauconnet S , Chabannes E , Henry PC , Adessi G , Bittard H Serum levels of vascular endothelial growth factor as a prognostic factor in bladder cancer. J Urol. (2001) ;166: (4):1275–9. |
[16] | Grivas PD , Daignault S , Tagawa ST , Nanus DM , Stadler WM , Dreicer R , et al Double-blind, randomized, phase 2 trial of maintenance sunitinib versus placebo after response to chemotherapy in patients with advanced urothelial carcinoma. Cancer. (2014) ;120: (5):692–701. |
[17] | Wulfing C , Machiels JP , Richel DJ , Grimm MO , Treiber U , De Groot MR , et al A single-arm, multicenter, open-label phase 2 study of lapatinib as the second-line treatment of patients with locally advanced or metastatic transitional cell carcinoma. Cancer. (2009) ;115: (13):2881–90. |
[18] | Powles T , Huddart RA , Elliott T , Sarker SJ , Ackerman C , Jones R , et al Phase III, Double-Blind, Randomized Trial That Compared Maintenance Lapatinib Versus Placebo After First-Line Chemotherapy in Patients With Human Epidermal Growth Factor Receptor 1/2-Positive Metastatic Bladder Cancer. J Clin Oncol. (2017) ;35: (1):48–55. |
[19] | Kruczynski A , Hill BT Vinflunine, the latest Vinca alkaloid in clinical development. A review of its preclinical anticancer properties. Crit Rev Oncol Hematol. (2001) ;40: (2):159–73. |
[20] | Bellmunt J , Theodore C , Demkov T , Komyakov B , Sengelov L , Daugaard G , et al Phase III trial of vinflunine plus best supportive care compared with best supportive care alone after a platinum-containing regimen in patients with advanced transitional cell carcinoma of the urothelial tract. J Clin Oncol. (2009) ;27: (27):4454–61. |
[21] | Garcia-Donas J , Font A , Perez-Valderrama B , Virizuela JA , Climent MA , Hernando-Polo S , et al Maintenance therapy with vinflunine plus best supportive care versus best supportive care alone in patients with advanced urothelial carcinoma with a response after first-line chemotherapy (MAJA; SOGUG /02): a multicentre, randomised, controlled, open-label, phase 2 trial. Lancet Oncol.. (2017) ;18: (5):672–81a |
[22] | Bellmunt J , de Wit R , Vaughn DJ , Fradet Y , Lee JL , Fong L , et al Pembrolizumab as Second-Line Therapy for Advanced Urothelial Carcinoma. N Engl J Med. (2017) ;376: (11):1015–26. |
[23] | Bellmunt J , Bajorin DF Pembrolizumab for Advanced Urothelial Carcinoma. N Engl J Med. (2017) ;376: (23):2304. |
[24] | Powles T , O’Donnell PH , Massard C , Arkenau HT , Friedlander TW , Hoimes CJ , et al Efficacy and Safety of Durvalumab in Locally Advanced or Metastatic Urothelial Carcinoma: Updated Results From a Phase 1/2 Open-label Study. JAMA Oncol. (2017) ;3: (9):e172411. |
[25] | Rosenberg JE , Hoffman-Censits J , Powles T , van der Heijden MS , Balar AV , Necchi A , et al Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet. (2016) ;387: (10031):1909–20. |
[26] | Sharma P , Retz M , Siefker-Radtke A , Baron A , Necchi A , Bedke J , et al Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. Lancet Oncol. (2017) ;18: (3):312–22. |
[27] | Apolo AB , Infante JR , Balmanoukian A , Patel MR , Wang D , Kelly K , et al Avelumab, an Anti-Programmed Death-Ligand 1 Antibody, In Patients With Refractory Metastatic Urothelial Carcinoma: Results From a Multicenter, Phase Ib Study. J Clin Oncol. (2017) ;35: (19):2117–24. |
[28] | Zitvogel L , Apetoh L , Ghiringhelli F , Andre F , Tesniere A , Kroemer G The anticancer immune response: indispensable for therapeutic success? .J Clin Invest. (2008) ;118: (6):1991–2001. |
[29] | Galsky MD , Mortazavi A , Milowsky MI , George S , Gupta S , Fleming MT , et al Randomized Double-Blind Phase II Study of Maintenance Pembrolizumab Versus Placebo After First-Line Chemotherapy in Patients With Metastatic Urothelial Cancer. J Clin Oncol. (2020) ;38: (16):1797–806. |
[30] | Powles T , Park SH , Voog E , Caserta C , Valderrama BP , Gurney H , et al Avelumab Maintenance Therapy for Advanced or Metastatic Urothelial Carcinoma. N Engl J Med. (2020) ;383: (13):1218–30. |
[31] | Powles T , Park SH , Voog E , Caserta C , Perez-Valderrama B , Gurney H , et al Avelumab first-line (1L) maintenance for advanced urothelial carcinoma (UC): Long-term follow-up results from the JAVELIN Bladder 100 trial. Journal of Clinical Oncology. (2022) ;40: (6_suppl):487. |
[32] | Grivas P , Kopyltsov E , Su PJ , Parnis FX , Park SH , Yamamoto Y ,et al Patient-reported Outcomes from JAVELIN Bladder 100: Avelumab First-line Maintenance Plus Best Supportive Care Versus Best Supportive Care Alone for Advanced Urothelial Carcinoma. Eur Urol. 2022. |
[33] | Moore K , Colombo N , Scambia G , Kim BG , Oaknin A , Friedlander M , et al Maintenance Olaparib in Patients with Newly Diagnosed AdvancedOvarian Cancer. N Engl J Med. (2018) ;379: (26):2495–505. |
[34] | Gonzalez-Martin A , Pothuri B , Vergote I , DePont Christensen R , Graybill W , Mirza MR , et al Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N Engl J Med. (2019) ;381: (25):2391–402. |
[35] | Crabb SJ , Hussain S , Soulis E , Hinsley S , Dempsey L , Trevethan A , et al A Randomized, Double-Blind, Biomarker-Selected, Phase II Clinical Trial of Maintenance Poly ADP-Ribose Polymerase Inhibition With Rucaparib Following Chemotherapy for Metastatic Urothelial Carcinoma. J Clin Oncol. 2022:JCO2200405. |
[36] | Fulton B , Jones R , Powles T , Crabb S , Paul J , Birtle A , et al ATLANTIS: a randomised multi-arm phase II biomarker-directed umbrella screening trial of maintenance targeted therapy after chemotherapy in patients with advanced or metastatic urothelial cancer. Trials. (2020) ;21: (1):344. |
[37] | Vignani F , Tambaro R , De Giorgi U , Giannatempo P , Bimbatti D , Carella C , et al Addition of Niraparib to Best Supportive Care as Maintenance Treatment in Patients with Advanced Urothelial Carcinoma Whose Disease Did Not Progress After First-line Platinum-based Chemotherapy: The Meet-URO12 Randomized Phase 2 Trial. Eur Urol. 2022. |
[38] | Jones RJ , Hussain SA , Birtle AJ , Song YP , Enting D , Faust G , et al A randomised, double blind, phase II clinical trial of maintenance cabozantinib following chemotherapy for metastatic urothelial carcinoma (mUC): Final analysis of the ATLANTIS cabozantinib comparison. Journal of Clinical Oncology. (2022) ;40: (17_suppl).LBA4505–LBA. |
[39] | Hussain SA , Shimizu N , Obara W , Yamasaki T , Takashima S , Hasegawa T , et al Phase II open-label study of S-10 as maintenance monotherapy after first-line platinum-containing chemotherapy in patients with advanced or metastatic urothelial carcinoma. Journal of Clinical Oncology. (2021) ;39: (6_suppl):440. |
[40] | Apolo AB , Nadal R , Tomita Y , Davarpanah NN , Cordes LM , Steinberg SM , et al Cabozantinib in patients with platinum-refractory metastatic urothelial carcinoma: an open-label, single-centre, phase 2 trial. Lancet Oncol. (2020) ;21: (8):1099–109. |
[41] | Apolo AB , Nadal R , Girardi DM , Niglio SA , Ley L , Cordes LM , et al Phase I Study of Cabozantinib and Nivolumab Alone or With Ipilimumab for Advanced or Metastatic Urothelial Carcinoma and Other Genitourinary Tumors. J Clin Oncol. (2020) ;38: (31):3672–84. |
[42] | Gupta S , Ballman KV , Galsky MD , Morris MJ , Chen RC , Chan TA , et al MAIN-CAV: Phase III randomized trial of maintenance cabozantinib and avelumab versus avelumab after first-line platinum-based chemotherapy in patients with metastatic urothelial cancer (mUC) (Alliance A01). Journal of Clinical Oncology. (2022) ;40: (16_suppl):TPS4607–TPS |
[43] | Parra ERMA , Freitas Pinto Lima C , Li J , Haymaker C , Parikh R , Bernicker E, , et al Baseline biomarkers associated with clinical benefitin patients with solid tumors refractory to immune checkpoint inhibitors (ICIs) treated with live biotherapeutic MRxin combination with pembrolizumab. Annals of Oncology. (2021) ;32: , S858–S9. |



