A Systematic Review of the Diagnostic and Prognostic Value of Urinary Protein Biomarkers in Urothelial Bladder Cancer
Abstract
For over 80 years, cystoscopy has remained the gold-standard for detecting tumours of the urinary bladder. Since bladder tumours have a tendency to recur and progress, many patients are subjected to repeated cystoscopies during long-term surveillance, with the procedure being both unpleasant for the patient and expensive for healthcare providers. The identification and validation of bladder tumour specific molecular markers in urine could enable tumour detection and reduce reliance on cystoscopy, and numerous classes of biomarkers have been studied. Proteins represent the most intensively studied class of biomolecule in this setting.
As an aid to researchers searching for better urinary biomarkers, we report a comprehensive systematic review of the literature and a searchable database of proteins that have been investigated to date. Our objective was to classify these proteins as: 1) those with robustly characterised sensitivity and specificity for bladder cancer detection; 2) those that show potential but further investigation is required; 3) those unlikely to warrant further investigation; and 4) those investigated as prognostic markers. This work should help to prioritise certain biomarkers for rigorous validation, whilst preventing wasted effort on proteins that have shown no association whatsoever with the disease, or only modest biomarker performance despite large-scale efforts at validation.
INTRODUCTION
Non-muscle-invasive bladder cancer (NMIBC: stages pTa/pT1/pTis) is characterised by a high incidence of recurrence and a risk of progression to muscle-invasive disease (MIBC: stages pT2+) [1]. Long-term surveillance thus remains the cornerstone of long-term management, and cystoscopy has represented the gold standard modality for over 80 years. However, the cystoscopic approach is both burdensome for patients and expensive for healthcare providers, and so there has been a decades-long search for non-invasive urinary biomarkers that can match or even improve upon the sensitivity and specificity of cystoscopy. However, current guidelines do not recommend the use of urinary biomarkers in the management of bladder cancer patients [2]. Despite this, urine cytology is often used as an adjunct to cystoscopy and whilst visual detection of cancer cells in urine is a very specific test with high sensitivity for high-grade bladder cancer, sensitivity for low-grade bladder cancer is only 4–31% [3]. Thus any new biomarkers might be considered useful if they outperform cytology i.e show very high specificity and high sensitivity for both low and high-grade bladder cancer.
Many classes of biomolecule have been investigated as urinary biomarkers but the majority of studies have analysed proteins with hundreds of published reports where specific proteins have been measured in urine as potential indicators of bladder cancer. In this review we focus entirely on soluble proteins (those that can be measured in the supernatant rather than the pellet following centrifugation). Most proteins can be measured reliably using inexpensive immunoassays. These may be rapid and qualitative, enabling point-of-care testing, or laboratory based quantitative immunoassays but in either case, multiplex testing for a panel of protein biomarkers (if such a panel was defined) should be relatively straightforward to implement.
The proteins measured in biomarker studies will have been selected based on prior knowledge of the biology of bladder cancer or the biology of the particular protein or based on data from hypothesis generating approaches such as gene expression profiling and proteomic analyses of bladder cancer tissue, cell lines or indeed urine itself. In most cases, the measurements will have been made using a validated antibody-based assay such as an ELISA. The reliability of such assays is dependent on the specificity of the antibodies used; certain types of assay which rely on the specificity of a single antibody may be more susceptible to interference than ‘sandwich assays’ (those that rely on the specificity of a pair of antibodies). However, in many bladder cancer biomarker studies, patient selection is of more concern than the assay used. That is to say, “do the patients (and the non-cancer subjects) being used in the study represent the patient population where the biomarker test would be applied in the real world?”. Ideally in studies of biomarkers for first presentation diagnosis both “cases” and “controls” should be patients undergoing investigation for suspected bladder cancer e.g. patients from haematuria clinics. For surveillance markers both cases and controls should be patients undergoing surveillance for disease recurrence. However, in many biomarker studies we see bias introduced by enrichment for high-grade and advanced disease which is likely to increase apparent sensitivity (% cases correctly identified) and inclusion of healthy volunteers which is likely to increase apparent specificity (% controls correctly identified), or the use of patients with large-primary tumours when the goal is to detect small recurrent tumours [4].
Another major pitfall in the measurement of urinary biomarkers for bladder cancer is haematuria [5]: haematuria is a symptom and sign of bladder cancer but is not the biological cause of bladder cancer. Thus, any protein present in blood may appear to act as a biomarker in case-control studies where haematuria is not matched, but will not be bladder cancer-specific.
Reviews of urinary biomarkers for bladder cancer (e.g. [6, 7]) tend to focus on the biomarkers that have been most extensively validated for detecting disease, especially those with FDA approval (NMP22, BTA, UroVysion, ImmuoCyt), but also others such as MMP9 which have been extensively measured but fall short of clinical utility, and perhaps a handful of ‘promising candidates’. In the current review we attempt to comprehensively review all proteins which have been investigated as urinary biomarkers for bladder cancer. Our main rationale for doing so is to generate a useful resource for researchers that may indicate the potential (or otherwise) of a particular urinary protein under investigation. A secondary aim of is to collate and assess the literature on prognostic urinary biomarkers, an area which is often neglected but which could be incorporated into risk stratification algorithms and so aid patient management.
The biomarker studies reviewed are heterogeneous in terms of the populations studied. The non-bladder cancer control cohorts vary from healthy controls to non-malignant urological disorders to non-bladder urological malignancies and patients undergoing surveillance for bladder cancer recurrence with no detectable disease (or a mix of all four). The bladder cancer cases vary in stage and grade (which we have partially controlled for with our selection criteria, see below) but are also either primary or recurrent tumours or a mix of both (or unspecified) in different studies. Thus, to be inclusive, we have used the term “detection biomarker” and present sensitivity for bladder cancer versus non-bladder cancer throughout this review rather than attempting to distinguish between proposed diagnostic and surveillance roles for biomarkers in individual studies.
MATERIALS AND METHODS
Systematic review methods were employed to find primary studies that reported test results on measured soluble protein biomarkers in urine. The search was conducted in Medline via the Pubmed search platform on the 25th August 2015 using the following search terms: ((“urinary bladder neoplasms”[MeSH Terms] OR (“urinary”[All Fields] AND “bladder”[All Fields] AND “neoplasms”[All Fields]) OR “urinary bladder neoplasms”[All Fields] OR (“bladder”[All Fields] AND “cancer”[All Fields]) OR “bladder cancer”[All Fields]) AND (“urine”[Subheading] OR “urine”[All Fields] OR “urine”[MeSH Terms])) AND (“biological markers”[MeSH Terms] OR (“biological”[All Fields] AND “markers”[All Fields]) OR “biological markers”[All Fields] OR “biomarker”[All Fields]). Studies retrieved from the PubMed search were assessed for eligibility by two people (JD & JG) using the title and abstract or where necessary the full text. Disagreements regarding inclusion were resolved by discussion and moderation by the rest of the team. Papers were included if they reported on tests that measured soluble protein biomarkers in the urine of bladder cancer patients (any stage). We excluded papers that did not measure protein biomarkers in urine, measured only enzyme activities, or that analysed urinary cell pellets. Prognostic biomarker studies were manually identified from the full set of included publications.
The included papers underwent a quality filter step to separate the better designed and reported studies from those with poor reporting or design. Studies were categorized into “unequivocal” or “equivocal” categories based on whether they met the following selection criteria:
• ≥20 cancer patients
• ≥20 controls or more
• Specificity and sensitivity presented
• ≥25% pTa bladder cancer
• ≥15% grade 1 bladder cancer
The thresholds for the percentage of pTa and grade 1 tumours are well below the percentages for incident bladder cancer in the UK [8] but enable identification of studies with bias towards reporting protein biomarkers associated with high-grade/stage bladder cancer. Studies which were not available to view or purchase online were also excluded. The proteins reported in unequivocal studies were further categorised into those in 3 or more unequivocal studies (validated detection biomarkers), those with a combined sensitivity and specificity (sensitivity+specificity/2) ≥80% (possible biomarkers), and those with a combined sensitivity and specificity ≤80% (unlikely biomarkers). The value of 80% for combined specificity and sensitivity was selected on the basis that it is close to the performance of cystoscopy, the gold standard to which biomarkers are usually referenced (which is the only method of detection of bladder cancer recommended by NICE [2]). The reported sensitivity and specificity of white light cystoscopy vary greatly but a recent meta-analysis arrived at values of 85 and 87% [9]. The full search and categorisation strategy is shown in Fig. 1.
RESULTS
Search result overview
Our initial literature search identified 1310 publications. From these, we identified 350 reports in which one or more proteins were measured in the urine of bladder cancer patients. Of the 350 reports, only 49 met our criteria to be considered as “unequivocal”. The remaining 301 studies either had too few cases or controls, did not report a high enough proportion of pTa and/or grade 1 tumours, did not provide values for sensitivity and specificity, or were not readily accessible. In total, we found evidence that 161 proteins have been investigated as urinary biomarkers for bladder cancer (Table 1 and Supplemental Information Table S1).
Validated detection biomarkers
Of the 161 proteins investigated, 27 were investigated in one or more “unequivocal studies” (Table 1). Only 4 proteins appear in 3 or more of the unequivocal studies and thus met our criteria as “validated detection biomarkers”. These are the well-known biomarkers: nuclear matrix protein 22 (NMP22), bladder tumour antigen (BTA), and the cytokeratin-based tests urinary bladder carcinoma antigen (UBC) and Cyfra 21-1. NMP22, BTA and UBC are commercially available as both quantitative and point-of-care assays. NMP22 (gene symbol: NUMA1) is a nuclear matrix protein overexpressed in bladder cancer cells and has been measured in 25 unequivocal studies with a weighted mean sensitivity and specificity of 61.8% and 80.3% (4528 cancer patients vs. 7728 non-cancer). The antigen recognised in the BTA assay is reported as complement factor H related protein which is released from bladder cancer cells [10]: the weighted mean sensitivity and specificity for using BTA to detect bladder cancer across 23 unequivocal studies are 64.0% and 76.6%, respectively (2258 cancer patients v 2994 non-cancer). The UBC test measures soluble fragments of cytokeratins 8 and 18 and has been reported in 11 unequivocal studies with a mean sensitivity and specificity of 64.4% and 80.3% (753 cancer patients v 1072 non-cancer). The Cyfra 21-1 test measures soluble fragments of cytokeratin 19 and has been reported in 3 unequivocal studies with a mean sensitivity and specificity of 64.4% and 85.5%, respectively (293 cancer patients v 331 non-cancer).
The mean sensitivities and specificities for the 4 well validated biomarkers across multiple studies are all very similar, in accordance with studies performing side-by-side comparisons: the results of such studies vary as to which biomarker performs best but seldom find substantial differences between them [11–13]. All 4 validated biomarkers also show a similar dependence on stage and grade with high sensitivity for high-grade and muscle-invasive disease but lower sensitivity for low-grade disease. Based on data from those studies listed in Table 1 which present sensitivity for different grades of disease, the mean sensitivities for grade 1/grade 3 are 53.4% /77.4% for NMP22, 51.4% /87.5% for BTA, 48.5% /76.0% for UBC and 55.7% /91.9% for Cyfra 21-1. Thus, these markers do have higher sensitivity for low-grade disease than cytology (albeit with lower specificity) but fall short of cystoscopy in terms of both sensitivity and specificity [3, 9]. Whilst there may be some utility for these commercially-available biomarkers in the surveillance of HR-NMIBC (when index tumour grade and stage are known), they cannot be solely relied upon for the diagnosis of incident disease.
Possible biomarkers
There are 11 proteins that have been evaluated in an unequivocal study and have a combined sensitivity and specificity (sensitivity+specificity)/2) of greater than 80%. We have designated these as “possible biomarkers”. They are apolipoprotein A4, calprotectin, CD147, CEACAM1, clusterin, coronin-1A, DJ-1, fibronectin, reg-1, stathmin-1, and γ-synuclein. Several of these possible biomarkers are also supported by evidence from “equivocal studies”. Each protein is discussed briefly below. These are proteins that may merit further investigation: all require independent validation in appropriate patient cohorts.
The utility of urinary fibronectin for detecting bladder cancer reported in 2 unequivocal studies [14, 15] is supported by 9 “equivocal” studies: 5 of these studies present moderately high sensitivities and specificities with weighted means across the studies of 82.5% and 80.2%, respectively (390 cases and 520 controls) [16–20]. Although there is substantial evidence that increased urinary fibronectin is indicative of bladder cancer, Alias-Melgar [21] found that urinary fibronectin is increased in urolithiasis, and Eissa [17] reported that in side-by–side comparison NMP22 slightly outperforms fibronectin.
The utility of urinary clusterin is also reported by 2 unequivocal studies [22, 23] and 1 equivocal study [24] with the latter reporting a sensitivity of 73% but only 55% specificity. Clusterin is a multifunctional chaperone protein with alternatively spliced forms exhibiting different cellular locations and functions [25]. Hazzaa et al. [22] found increased clusterin gene expression in bladder cancer, particularly in invasive disease, and that high clusterin expression was associated with poor prognosis. Although clusterin is widely expressed and found in all body fluids, which may limit it’s specificity [26], measurement of individual splice variants rather than total clusterin levels might merit further research.
CEACAM1 (Carcinoembryonic antigen-related cell adhesion molecule 1, also known as CD66a) was reported as a novel urinary marker for bladder cancer by Tilki et al. [27]. In this study comparing 93 cases (72 NMIBC, 17 low-grade) with 82 controls (30 healthy subjects, 10 with benign disease and 42 with a history of bladder cancer but no evidence of disease) urinary CEACAM1 generated a sensitivity of 74% at 95% specificity. As with most urinary biomarkers sensitivity was higher for MIBC than NMIBC. It is not stated whether the tumours were incident or recurrent and subjects with diabetes were excluded from the study but CEACAM1 appears to merit further investigation. Tilki et al. [27] also reported that CEACAM1 immunostaining was detected on endothelial cells rather than cancer cells in bladder tumours. It has not been reported whether the urinary CEACAM1 is expressed as a soluble isoform (lacking the transmembrane domain) or if the ectodomain is shed by proteolytic cleavage.
Ebbing et al. [28] reported that urinary calprotectin (a heterodimer of S100A8 and S100A9 proteins with antimicrobial properties) can be used to detect bladder cancer with 80% sensitivity at 92% specificity in a study with 46 cases (38 NMIBC, 25 low-grade) and 40 healthy controls. The median calprotectin level was 10-fold higher in bladder cancer patients than healthy controls and less than 2-fold increased in patients with prostate and renal cancers (although their inclusion in the data analysis slightly decreased specificity). Calprotectin has been reported as a prognostic indicator in bladder cancer and to be upregulated both in tumours and sera [29, 30]. However, calprotectin is released by neutrophils during inflammation which may compromise its role as a tumour marker.
Two urinary proteins, stathmin-1 and CD147 make the “possible biomarkers” category on the basis of a study by Bhagirath et al. [31] analysing the urine of 30 bladder cancer cases (21 NMIBC, 13 low-grade) and 30 controls (15 healthy, 15 benign prostatic hyperplasia). Sensitivities and specificities were 90% and 87% for stathmin-1, respectively (also known as oncoprotein-18) and 97% and 100% for CD147, respectively (also known as basigin or EMMPRIN). Although a small single study, both stathmin-1 and CD147 are known cancer-related proteins and there are reports that overexpression of stathmin-1 [32] and CD147 [33] are associated with aggressive bladder cancer and a poor prognosis.
Another 4 possible biomarkers were reported in a study by Kumar et al. [34]: γ-synuclein (87.5% sensitivity at 90.0% specificity), DJ-1 (83.3% sensitivity at 100% specificity), apolipoprotein A4 (79.2% sensitivity at 100% specificity) and Coronin-1A (66.7% sensitivity at 100% specificity) based on 173 cases (110 NMIBC, 89 low-grade) and 212 controls (66 healthy, 91 other malignancy, 121 assorted chronic conditions). Various apolipoproteins have been reported as increased in the urine of bladder cancer patients [35–37]; however, being moderately abundant in plasma, their specificity is not assured and, in the case of apolipoprotein A4, Chen et al. [38] found no evidence of elevation in bladder cancer patients. γ-synuclein was also investigated by one equivocal study which reported only 40.2% sensitivity albeit at 96.5% specificity [39] (112 cancer and 230 controls). Although DJ-1 has been reported to be overexpressed in aggressive high-grade bladder cancer [40], no other urine studies have been published. We have performed a pilot study and found urinary DJ-1 to be significantly increased only in MIBC, disputing a high sensitivity for all stages and grades of bladder cancer (Ward, unpublished data). Coronin-1A is a cytoskeletal protein that has not been otherwise reported in bladder cancer. It seems likely that Kumar et al’s study [34] may have overestimated the performance of these 4 possible biomarkers.
Using proteomics, Orenes-Piñero et al. [41] identified a protein at increased levels in the urine of bladder cancer patients as Reg-1 (lithostathine-1-alpha). Immunohistochemistry showed Reg-1 overexpression in bladder tumours and measurement in 32 cases (16 NMIBC, 5 low-grade) and 48 controls (cystoscopy negative) gave 81% sensitivity at 81% specificity.
Unlikely biomarkers
Sixteen of the 27 proteins evaluated in unequivocal studies have a combined sensitivity and specificity of less than 80% (including the 4 validated detection biomarkers). As the diagnostic performance of these proteins is well below that achieved by flexible cystoscopy it is unlikely that any of these would find widespread clinical use as standalone biomarkers for detecting bladder cancer. Nonetheless, the evidence suggests that some cases of bladder cancer result in an increase in the urinary concentration of these proteins and consequently they may be diagnostically useful when used in combination with one another or other markers.
Equivocal biomarkers
We found that the majority of the biomarker studies we reviewed could not be classified as unequivocal. This is due to missing information (stage/grade/sensitivity/specificity) or due to a non-representative patient population which is likely to inflate the estimated sensitivity and specificity. Fifteen proteins that have not been investigated in an unequivocal study have been evaluated in at least 5 equivocal studies (Fig. 2), and some of the equivocal studies report high sensitivities and specificities. With the caveat that these “equivocal biomarkers” may not be robust in the clinical setting, we briefly discuss those reported in 5 or more studies below.
Urinary carcinoembryonic antigen (CEA) has been investigated in 28 equivocal studies. The weighted mean values for sensitivity and specificity are 54.0% and 90.5% respectively (814 cases and 578 controls) based on accessible studies reporting sensitivity and specificity [42–47]. Most of the CEA studies were published in the 1970s and 1980s (Fig. 3) and interest presumably waned due to the sensitivity being too low for clinical utility. However, as expected for an oncofetal antigen, specificity appears to be high and therefore urinary CEA could prove useful in the context of a multimarker panel.
The role of matrix metallopeptidase 9 (MMP9) as a urinary biomarker has been investigated in 16 equivocal studies with 8 presenting values for sensitivity and specificity [48–55]. The weighted mean values for sensitivity and specificity are 72.1% and 77.2%, respectively (based on 707 cases and 917 controls). MMP9 is biologically plausible as a biomarker for invasive bladder cancer and urinary levels are clearly elevated in many cases of bladder cancer; however, modest sensitivity and specificity (especially for low-grade disease) limit its usefulness.
Urinary vascular endothelial growth factor (VEGF) has been investigated in 13 equivocal studies with 6 presenting sensitivity and specificity data [48, 54, 56–59]. The weighted mean values for sensitivity and specificity are 71.4% and 78.1%, respectively (based on 509 cases and 389 controls).
Tissue polypeptide antigen (TPA, a complex of cytokeratins 8, 18 & 19) has been investigated in 10 equivocal studies with 3 presenting sensitivity and specificity data [46, 60, 61]. The weighted mean values for sensitivity and specificity are 84.1% and 96.6%, respectively (based on 277 cases and 311 controls). However, Stefanovic et al reported that TPA lacks diagnostic accuracy [62], and Carbin et al reported that TPA is only effective if 24 hour urine samples are analysed [63]. Additionally, although the averaged sensitivity/specificity appear higher than the averaged sensitivity/specificity for other cytokeratin based tests (UBC and Cyfra 21-1), in a direct comparison of TPA and Cyfra 21-1 Sanchez-Carbayo found TPA to be slightly inferior to Cyfra 21-1, indicating that the equivocal studies have overestimated the performance of TPA [61, 62].
Survivin has been investigated in 9 equivocal studies with 6 reporting sensitivity and specificity data [58, 64–68]. The weighted mean values for sensitivity and specificity are 69.4% and 88.3%, respectively (based on 437 cases and 313 controls).
Matrix metallopeptidase 2 (MMP2) has been investigated in 8 equivocal studies with 4 presenting sensitivity and specificity data [49, 52, 53, 69]. The weighted mean values for sensitivity and specificity are 68.2% and 88.8% (based on 345 cases and 681 controls).
A number of interleukins have been repeatedly investigated in the urine of bladder cancer patients. Interleukin-8 (IL-8) has been investigated in 10 equivocal studies with 4 presenting sensitivity and specificity data [54, 56, 70, 71]. The weighted mean values for sensitivity and specificity are 66.4% and 83.1%, respectively (based on 225 cases and 273 controls). Interleukins 2 and 6 (IL-2 and IL-6) have been measured in 5 and 7 equivocal studies respectively. However most of these studies focus on response to BCG treatment rather than bladder cancer detection [72]. With only one study [56] reporting sensitivity and specificity data for IL-6 (67% and 63%), and none for IL-2, there is no evidence that either is likely to be useful for detecting bladder cancer.
CA19-9 has been investigated as a urinary biomarker in 8 equivocal studies, but with only two reporting sensitivity and specificity (83.3% and 50.8% [73], and 71.6% and 91.6% [74], respectively). Strictly speaking, this is a glycan biomarker rather than a glycoprotein biomarker [75]. CA19-9 may be a useful biomarker for bladder cancer when interpreted with reference to secretor phenotype [76, 77].
BCLA-4 has been investigated as a urinary biomarker in 6 equivocal studies. BCLA-4 was first reported in 1996 as a spot in 2D-electrophoresis analyses of nuclear matrix extracts that was more intense in bladder cancer than normal urothelium [78]. The authors then partially sequenced the protein in the gel spot and generated antibodies to a synthetic peptide (EISQLNAG), despite the sequence not matching any known human protein sequences. The antibodies were used to generate immunoassays and BCLA-4 measured in the urine of 54 bladder cancer patients (predominantly high-grade) and 51 control subjects, generating a sensitivity of 96.4% at 100% specificity [79]. In 2004 the same group identified BCLA-4 as a member of the ETS transcription family [80] and then expanded their study to 70 cancer subjects and 147 controls (89% sensitivity and 95% specificity) [81]. However, both of these papers have now been retracted [82, 83]. Despite this, BCLA-4 has been widely reported as a ‘promising biomarker’ in previous reviews and a further study in China using antibodies raised against the EISQLNAG sequence reported very high sensitivity and specificity [84]. Thus, BCLA-4 has a chequered history and although BCLA-4 ELISAs appear to detect bladder cancer better than other urinary markers, BCLA-4 has neither gained regulatory approval or been widely adopted as a urinary biomarker for bladder cancer detection.
Apolipoproteins A1 (APOA1) and E (APOE) and have been investigated in 5 and 6 equivocal studies respectively. The 5 APOA1 papers are from 2 research groups with both reporting high sensitivity and specificity (89.2% and 84.6% [85–87], and 94.6% and 92.0% [38, 88], respectively). Apolipoproteins are abundant in plasma and hence urinary concentrations will be influenced by haematuria. The study by Chen et al. [88] included 13 control subjects with haematuria and their urinary APOA1 was slightly elevated; the authors concluded that urinary APOA1 might need to be interpreted with reference to haematuria. As a urinary biomarker, APOE appears less useful than APOA1 [36–38, 48].
We were unable to find any consistent evidence of high sensitivity or specificity for any of the remaining proteins mentioned in 5 unequivocal studies (ICAM-1, β-gonadotropin, E-cadherin, carbonic anyhdrase IX), although we note that carbonic anhydrase IX urinary mRNA has recently been reported as a potentially useful biomarker [89].
In summary, none of the biomarkers investigated in ≥5 equivocal studies has the sensitivity and specificity required to act as a standalone biomarker for detecting bladder cancer. Finally, in our search for unproven but possible biomarkers we manually searched the reports for the proteins presented in <5 equivocal studies for those the highest sensitivity and specificity. MMP3 and TIMP2 showed high sensitivity and specificity in studies carried out in Egypt [49, 51]. These results should be treated with caution as in Egypt many cases of bladder cancer are bilharzial SCC. Additionally, TIMP2 has been reported to have lower sensitivity and specificity in other studies. A small study by Gecks et al. [90] suggested that Tenascin-C can be used to detect recurrent bladder cancer with 91% sensitivity at 80% specificity. In a study using urine from 68 cases (mixed stage and grade), 68 healthy controls and 16 patients with cystitis Lorenzi et al. [91] reported that measuring the serine peptidase HTRA-1 gave 92.7% sensitivity and 95.6% specificity for bladder cancer detection. At the time of writing no further studies have corroborated (or refuted) the biomarker potential of HTRA1-1.
Prognostic urinary biomarkers
Urinary biomarkers have the potential to inform not only on the presence or absence of bladder cancer, but also to provide prognostic information. Such a biomarker would provide information on outcome and could guide choices between conservative and radical treatment regimens. The word ‘prognostic’ has been applied variably to urinary biomarkers. For example, high levels of biomarker post resection are often reported as a poor prognostic indicator, but are most likely just indicative of residual disease. A truly prognostic indicator should indicate outcome in patients with tumours; not versus those without tumours. The majority of urinary protein biomarkers considered in this review increase in concentration with both stage and grade of disease and could therefore be considered as prognostic indicators. However, very few studies have directly investigated the association between urinary biomarker levels at presentation and outcome, and even fewer have investigated whether urinary biomarkers can provide prognostic information over and above that provided by standard clinicopathological factors (Table 2). Indeed, at the time of writing, only BTA, CEA, MMP9, tenascin-C, cystatin-B and the soluble extracellular domains of EGFR and EpCAM have been reported as independent prognostic indicators (Table 2) and these data require independent validation. The two NMP22 prognostic studies listed in Table 2 confuse disease detection and prognosis, although further literature searching identified a large study by Shariat et al. [92] which found that including pre-treatment urinary NMP22 levels slightly improved the ability of nomograms to predict later recurrence.
Biology of urinary biomarkers
The proteins that have been shown to be increased in concentration in the urine of bladder cancer patients are highly diverse in terms of their biological activities, the pathways that they are involved with and their cellular compartmentalisation. They include, amongst others, proteases, lipid binding/transport proteins, cytoskeletal components and cytokines. The most significantly over represented biological processes include “regulation of cell migration”, “response to wounding”, “regulation of apoptosis” and “inflammatory response” [93]. Eight of the proteins are in the KEGG_PATHWAY “Pathways in Cancer” (survivin, E-cadherin, fibronectin, IL6, MMP2, MMP9, PDGFR and VEGF). Other proteins are less obviously mechanistically linked to cancer with Apo-A1, apo-A4, apoE, clusterin, fibrinogen, fibronectin, thrombin and α1-antitrypsin all classed as plasma proteins and with α1-antitrypsin, thrombin, IL6 and fibronectin also being classed as acute phase proteins. Over half of the proteins are bona fide secreted proteins, but there are also 7 cytoplasmic/cytoskeletal and 6 plasma membrane proteins and 2 nuclear proteins (NMP22 and EN2) (Fig. 4).
DISCUSSION
We have systematically reviewed the literature concerning urinary proteins as biomarkers for bladder cancer. We focussed solely on proteins which are measured in solution in urine, rather than proteins present in cancer cells in the cell pellet or DNA, RNA or metabolite biomarkers. Thus, all of the biomarkers discussed above can be measured by immunoassay in a single urine sample which may enable biomarker multiplexing and point-of-care testing in the future.
We found that the majority of urine biomarker studies use patient populations enriched for high-grade and high-stage disease which is likely to inflate estimates of sensitivity and specificity and/or do not provide sufficient information to be thoroughly evaluated. It is clear that the urinary concentrations of the vast majority of proteins investigated as biomarkers positively correlate with both stage and grade of disease. Thus, low stage and grade disease is not easily detected. As low and high-grade bladder cancer can be considered separate entities at the genomic level [94], it might be expected that different urinary biomarkers would be required to detect each. By definition, the cancer cells are still relatively normal, both genomically and phenotypically, in low-grade disease so that although some alterations in gene expression have been noted [95], the processes involved in releasing proteins into the urine may be essentially normal. With high-grade and invasive disease it is likely that many of these processes are unregulated such that proteins released directly from the cancer cells, as breakdown products from the extracellular matrix, as a result of inflammatory responses or plasma proteins all find their way into the urine. Thus, although high-grade bladder cancer is a heterogeneous disease, it is perhaps not surprising that many of the urinary proteins reviewed can detect high-grade and stage disease with high sensitivity. A panel of protein markers, carefully selected on the basis of close associations with the various molecular sub-types of bladder cancer might offer the way forward in facile non-invasive detection of bladder cancer.
Our review shows that the use of pre-treatment urinary biomarker levels for prognostication has been addressed in very few studies compared with disease detection. This may be due to the long-term follow up of patients required for such studies, the lack of a perceived need for such biomarkers or the fact that in patients with a confirmed tumour molecular markers can be measured directly in the tumour rather than in urine. It is however possible that processes which release proteins from tumours (secretion, leakage, shedding) and degradation of the surrounding tissues are important prognostic indicators and are more effectively measured in urine than in the tumour itself.
In accord with others, we find that the “validated detection markers” (NMP22, BTA, UBC, Cyfra 21-1) do not rival flexible cystoscopy in terms of sensitivity and specificity [96]. We also find that many other biomarkers with are often reported as “promising” are not genuinely promising as their sensitivity or specificity is too low, or the evidence supporting their utility is equivocal. Finally, we have highlighted a small number of proteins that might warrant further validation and hopefully be confirmed as clinically useful biomarkers.
There are several limitations related to the review methodology. Firstly we may have introduced publication bias as we couldn’t access all of the publications online, however, as most were published before the year 2000 we reasoned that if that particular biomarker was significant, then the publication trail would have continued beyond 2000. Secondly, the review may be skewed to more positive data due to publication bias present in the literature i.e. only positive results get published. Thirdly, as we only included studies which reported both sensitivity and specificity, we could have been affected by outcome reporting bias i.e. where studies report selected outcomes usually those that are positive [97]. Despite these limitations we believe this review presents a comprehensive summary of the literature and a searchable database of proteins that have been investigated to date (Table S1), which will aid researchers searching for better urinary biomarkers. We conclude that:
• The majority of urine biomarker studies contain bias or are insufficiently reported.
• The urinary concentrations of a large number of proteins are increased by the presence of bladder cancer, but most proteins are not increased in all cases and are not specific to bladder cancer.
• NMP22, BTA, UBC and Cyfra 21-1 are the only well-validated urinary protein biomarkers and their sensitivity and specificity are well below those of cystoscopy.
• Fibronectin, clusterin, CEACAM1, apolipoprotein A4, calprotectin, CD147, coronin-1A, DJ-1, reg-1, stathmin-1, and γ-synuclein may be considered as possible biomarkers.
• Biomarkers supported by multiple “equivocal” studies include CEA, MMP9, VEGF, TPA, survivin, CA19-9, APOA1 and BCLA-4. Of these, only BLCA-4 reportedly has high-sensitivity, but is mired in controversy.
• Biomarkers supported by a single “equivocal” report of high sensitivity and specificity include MMP3 and HTRA.
None of the urinary protein biomarkers investigated to date can be used for accurate non-invasive detection of bladder cancer. Current efforts to combine protein biomarkers to improve test accuracy also fail to reach clinically useful sensitivity and specificity [48, 98]. DNA/RNA-based markers may supersede protein biomarkers in the near future [99–103]; however, with currently-available technology, these are more complex, expensive and time-consuming to measure than protein markers with little potential for point-of-care testing in the immediate future. Notwiths tanding, we hope that the increasing understanding of bladder cancer at the molecular-genomic level may enable selection of the correct cancer-specific proteins (or variants of proteins) to underlie a clinically applicable biomarker panel.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Appendices
The supplementary material is available in the electronic version of this article: http://dx.doi.org/10.3233/BLC-160054.
REFERENCES
[1] | Babjuk M , Oosterlinck W , Sylvester R , Kaasinen E , Böhle A , Palou-Redorta J , et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder, the 2011 update. Eur Urol (2011) ;59: (6):997–1008. |
[2] | www.nice.org.uk/guidance/ng2/chapter/1-recommendations#diagnosing-and-staging-bladder-cancer-2 . |
[3] | Lotan Y , Roehrborn C . Sensitivity and specificity of commonly available bladder tumor markers versus cytology: Results of a comprehensive literature review and meta-analyses. Urology (2003) ;61: :109–18. |
[4] | van Rhijn B , van der Poel H , van der Kwast T . Urine markers for bladder cancer surveillance: A systematic review. Eur Urol (2005) ;47: (6):736–48. |
[5] | Bryan R , Wei W , Shimwell N , Collins S , Hussain S , Billingham L , et al. Assessment of high-throughput high-resolution MALDI-TOF-MS of urinary peptides for the detection of muscle-invasive bladder cancer. Proteomics Clin Appl (2011) ;5: (9-10):493–503. |
[6] | Chou R , Gore J , Buckley D , Fu R , Gustafson K , Griffin J , et al. Urinary Biomarkers for Diagnosis of Bladder Cancer: A Systematic Review and Meta-analysis. Ann Intern Med (2015) ;163: (12):922–31. |
[7] | Tilki D , Burger M , Dalbagni G , Grossman H , Hakenberg O , Palou J , et al. Urine markers for detection and surveillance of non-muscle-invasive bladder cancer. Eur Urol (2011) ;60: (3):484–92. |
[8] | Boustead G , Fowler S , Swamy R , Kocklebergh R , Hounsome L , Section of Oncology B Stage, grade and pathological characteristics of bladder cancer in the UK: British Association of Urological Surgeons (BAUS) urological tumour registry. BJU Int (2014) ;113: (6):924–30. |
[9] | Zheng C , Lv Y , Zhong Q , Wang R , Jiang Q . Narrow band imaging diagnosis of bladder cancer: Systematic review and meta-analysis. BJU IntE (2012) ;110: (11):680–7. |
[10] | Kinders R , Jones T , Root R , Bruce C , Murchison H , Corey M , et al. Complement factor H or a related protein is a marker for transitional cell cancer of the bladder. Clin Cancer Res (1998) ;4: (10):2511–20. |
[11] | Giannopoulos A , Manousakas T , Gounari A , Constantinides C , Choremi-Papadopoulou H , Dimopoulos C . Comparative evaluation of the diagnostic performance of the BTA stat test, NMP22 and urinary bladder cancer antigen for primary and recurrent bladder tumors. J Urol (2001) ;166: (2):470–5. |
[12] | Boman H , Hedelin H , Holmäng S . Four bladder tumor markers have a disappointingly low sensitivity for small size and low grade recurrence. J Urol (2002) ;167: (1):80–3. |
[13] | Sánchez-Carbayo M , Urrutia M , Silva J , Romaní R , De Buitrago J , Navajo J . Comparative predictive values of urinary cytology, urinary bladder cancer antigen, CYFRA 21-1 and NMP22 for evaluating symptomatic patients at risk for bladder cancer. J Urol (2001) ;165: (5):1462–7. |
[14] | Li LY , Yang M , Z H.B. , Su XK , Xu WF , Chen Y , et al. Urinary fibronectin as a predictor of a residual tumour load after transurethral resection of bladder transitional cell carcinoma. BJU Int (2008) ;102: (5):566–71. |
[15] | Mutlu N , Turkeri L , Emerk K . Analytical and clinical evaluation of a new urinary tumor marker: Bladder tumor fibronectin in diagnosis and follow-up of bladder cancer. Clin Chem Lab Med (2003) ;41: (8):1069–74. |
[16] | Eissa S , Zohny SF , Zekri AR , El-Zayat TM , Maher AM . Diagnostic value of fibronectin and mutant p53 in the urine of patients with bladder cancer: Impact on clinicopathological features and disease recurrence. Med Oncol (2010) ;27: (4):1286–94. |
[17] | Eissa S , Swellam M , Sadek M , Mourad MS , El Ahmady O , Khalifa A . Comparative evaluation of the nuclear matrix protein, fibronectin, urinary bladder cancer antigen and voided urine cytology in the detection of bladder tumors. J Urol (2002) ;168: (2):465–9. |
[18] | Wunderlich H , Reichelt O , Zermann D , Schubert J , Berndt A , Kosmehl H Fetal fibronectin: A new screening-marker for bladder cancer? Oncol Rep (2001) ;8: (3):669–72. |
[19] | Menéndez V , Fernández-Suárez A , Galán J , Pérez M , García-López F . Diagnosis of bladder cancer by analysis of urinary fibronectin. Urology (2005) ;65: (2):284–9. |
[20] | Sánchez-Carbayo M , Urrutia M , González de Buitrago J , Navajo J . Evaluation of two new urinary tumor markers: Bladder tumor fibronectin and cytokeratin 18 for the diagnosis of bladder cancer. Clin Cancer Res (2000) ;6: (9):3585–94. |
[21] | Alías-Melgar A , Neave-Sánchez E , Suárez-Cuenca JA , Morales-Covarrubias J . Association of urine oncofetal fibronectin levels with urology’s most common disorders. Ann Clin Lab Sci (2013) ;43: (4):420–3. |
[22] | Hazzaa S , Elashry O , Afifi I . Clusterin as a diagnostic and prognostic marker for transitional cell carcinoma of the bladder. Pathol Oncol Res (2010) ;16: (1):101–9. |
[23] | Shabayek M , Sayed O , Attaia H , Awida H , Abozeed H . Diagnostic evaluation of urinary angiogenin (ANG) and clusterin (CLU) as biomarker for bladder cancer. Pathol Oncol Res (2014) ;20: (4):859–66. |
[24] | Stejskal D , Fiala R . Evaluation of serum and urine clusterin as a potential tumor marker for urinary bladder cancer. Neoplasma (2006) ;53: (4):343–6. |
[25] | Koltai T . Clusterin: A key player in cancer chemoresistance and its inhibition. Onco Targets Ther (2014) ;7: :447–56. |
[26] | Jenne D , Tschopp J . Clusterin: The intriguing guises of a widely expressed glycoprotein. Trends Biochem Sci (1992) ;17: (4):154–9. |
[27] | Tilki D , Singer BB , Shariat SF , Behrend A , Fernando M , Irmak S , et al. CEACAM A novel urinary marker for bladder cancer detection. Eur Urol (2010) ;57: (4):648–54. |
[28] | Ebbing J , Mathia S , Seibert FS , Pagonas N , Bauer F , Erber B , et al. Urinary calprotectin: A new diagnostic marker in urothelial carcinoma of the bladder. World J Urol (2014) ;32: (6):1485–92. |
[29] | Minami S , Sato Y , Matsumoto T , Kageyama T , Kawashima Y , Yoshio K , et al. Proteomic study of sera from patients with bladder cancer: Usefulness of S100A8 and S100A9 proteins. Cancer Genomics Proteomics (2010) ;7: (4):181–9. |
[30] | Tolson J , Flad T , Gnau V , Dihazi H , Hennenlotter J , Beck A , et al. Differential detection of S100A8 in transitional cell carcinoma of the bladder by pair wise tissue proteomic and immunohistochemical analysis. Proteomics (2006) ;6: (2):697–708. |
[31] | Bhagirath D , Abrol N , Khan R , Sharma M , Seth A , Sharma A . Expression of CD147, BIGH3 and Stathmin and their potential role as diagnostic marker in patients with urothelial carcinoma of the bladder. Clin Chim Acta (2012) ;413: (19-20):1641–6. |
[32] | Hemdan T , Lindén M , Lind S , Namuduri A , Sjöstedt E , de Ståhl T , et al. The prognostic value and therapeutic target role of stathmin-1 in urinary bladder cancer. Br J Cancer (2014) ;111: (6):1180–7. |
[33] | Afonso J , Longatto-Filho A , Baltazar F , Sousa N , Costa F , Morais A , et al. CD147 overexpression allows an accurate discrimination of bladder cancer patients’ prognosis. Eur J Surg Oncol (2011) ;37: (9):811–7. |
[34] | Kumar P , Nandi S , Tan TZ , Ler SG , Chia KS , Lim WY , et al. Highly sensitive and specific novel biomarkers for the diagnosis of transitional bladder carcinoma. Oncotarget (2015) ;6: (15):13539–49. |
[35] | Chen YT , Chen HW , Domanski D , Smith DS , Liang KH , Wu CC , et al. Multiplexed quantification of 63 proteins in human urine by multiple reaction monitoring-based mass spectrometry for discovery of potential bladder cancer biomarkers. J Proteomics (2012) ;75: (12):3529–45. |
[36] | Lindén M , Lind S , Mayrhofer C , Segersten U , Wester K , Lyutvinskiy Y , et al. Proteomic analysis of urinary biomarker candidates for nonmuscle invasive bladder cancer. Proteomics (2012) ;12: (1):135–44. |
[37] | Urquidi V , Goodison S , Ross S , Chang M , Dai Y , Rosser CJ . Diagnostic potential of urinary α1-antitrypsin and apolipoprotein E in the detection of bladder cancer. J Urol (2012) ;188: (6):2377–83. |
[38] | Chen C , Lin T , Tsai C , Wu C , Chung T , Chien K , et al. Identification of potential bladder cancer markers in urine by abundant-protein depletion coupled with quantitative proteomics. J Proteomics (2013) ;85: :28–43. |
[39] | Iwaki H , Kageyama S , Isono T , Wakabayashi Y , Okada Y , Yoshimura K , et al. Diagnostic potential in bladder cancer of a panel of tumor markers (calreticulin, gamma -synuclein, and catechol-o-methyltransferase) identified by proteomic analysis. Cancer Sci (2004) ;95: (12):955–61. |
[40] | Lee H , Choi S , Ro J . Overexpression of DJ-1 and HSP90α, and loss of PTEN associated with invasive urothelial carcinoma of urinary bladder: Possible prognostic markers. Oncol Lett (2012) ;3: (3):507–12. |
[41] | Orenes-Piñero E , Cortón M , González-Peramato P , Algaba F , Casal I , Serrano A , et al. Searching urinary tumor markers for bladder cancer using a two-dimensional differential gel electrophoresis (2D-DIGE) approach. J Proteome Res (2007) ;6: :4440–8. |
[42] | Tailly G , Cornelissen M , Vereecken RL , Verduyn H , Devos P , De Roo M . Urinary carcinoembryogenic antigen (CEA) in the diagnosis and follow-up of bladder carcinoma. Br J Urol (1983) ;55: (5):501–7. |
[43] | Neville AM , Nery R , Hall RR , Turberville C , Laurence DJ . Aspects of the structure and clinical role of the carcinoembryonic antigen (CEA) and related macromolecules with particular reference to urothelial carcinoma. Br J Cancer Suppl (1973) ;1: :198–207. |
[44] | Wahren B , Nilsson B , Zimmerman R . Urinary CEA for prediction of survival time and recurrence in bladder cancer. Cancer (1982) ;50: (1):139–45. |
[45] | Saied GM , El-Metenawy WH , Elwan MS , Dessouki NR . Urine carcinoembryonic antigen levels are more useful than serum levels for early detection of Bilharzial and non-Bilharzial urinary bladder carcinoma: Observations of 43 Egyptian cases. World J Surg Oncol (2007) ;5: :4. |
[46] | Halim AB , el-Ahmady O , Hamza S , Aboul-Ela M , Oehr P . Simultaneous determination of urinary CEA, ferritin and TPA in Egyptian bladder cancer patients. Int J Biol Markers (1992) ;7: (4):234–9. |
[47] | Zimmerman R , Wahren B , Edsmyr F . Assessment of serial CEA determinations in urine of patients with bladder carcinoma. Cancer (1980) ;46: (8):1802–9. |
[48] | Chen LM , Chang M , Dai Y , Chai KX , Dyrskjøt L , Sanchez-Carbayo M , et al. External validation of a multiplex urinary protein panel for the detection of bladder cancer in a multicenter cohort. Cancer Epidemiol Biomarkers Prev (2014) ;23: (9):1804–12. |
[49] | Eissa S , Ali-Labib R , Swellam M , Bassiony M , Tash F , El-Zayat T . Noninvasive diagnosis of bladder cancer by detection of matrix metalloproteinases (MMP-2 and MMP-9) and their inhibitor (TIMP-2) in urine. Eur Urol (2007) ;52: (5):1388–96. |
[50] | Eissa S , Labib RA , Mourad MS , Kamel K , El-Ahmady O . Comparison of telomerase activity and matrix metalloproteinase-9 in voided urine and bladder wash samples as a useful diagnostic tool for bladder cancer. Eur Urol (2003) ;44: (6):687–94. |
[51] | El-Sharkawi F , El Sabah M , Hassan Z , Khaled H . The biochemical value of urinary metalloproteinases 3 and 9 in diagnosis and prognosis of bladder cancer in Egypt. J Biomed Sci (2014) );21: (72). DOI: 10.1186/s12929-014-0072-4 |
[52] | Fernández CA , Wszolek MF , Loughlin KR , Libertino JA , Summerhayes IC , Shuber AP . A novel approach to using matrix metalloproteinases for bladder cancer. J Urol (2009) ;182: (5):2188–94. |
[53] | Mohammed MA , Seleim MF , Abdalla MS , Sharada HM , Abdel WAH . Urinary high molecular weight matrix metalloproteinases as non-invasive biomarker for detection of bladder cancer. BMC Urol (2013) ;13: (25). DOI: 10.1186/1471-2490-13-25 |
[54] | Rosser CJ , Dai Y , Miyake M , Zhang G , Goodison S . Simultaneous multi-analyte urinary protein assay for bladder cancer detection. BMC Biotechnol (2014) ;14: (24). DOI: 10.1186/1472-6750-14-24 |
[55] | Urquidi V , Kim J , Chang M , Dai Y , Rosser CJ , Goodison S . CCL18 in a multiplex urine-based assay for the detection of bladder cancer. PLoS One (2012) ;7: (5). DOI: 10.1371/journal.pone.0037797 |
[56] | Abogunrin F , O’Kane HF , Ruddock MW , Stevenson M , Reid CN , O’Sullivan JM , et al. The impact of biomarkers in multivariate algorithms for bladder cancer diagnosis in patients with hematuria. Cancer (2012) ;118: (10):2641–50. |
[57] | Bian W , Xu Z . Combined assay of CYFRA21-1, telomerase and vascular endothelial growth factor in the detection of bladder transitional cell carcinoma. Int J Urol (2007) ;14: (2):108–11. |
[58] | Sun Y , He D , Ma Q , Wan X , Zhu G , Li L , et al. Comparison of seven screening methods in the diagnosis of bladder cancer. Chin Med J (Engl) (2006) ;119: (21):1763–71. |
[59] | Goodison S , Chang M , Dai Y , Urquidi V , Rosser CJ . A multi-analyte assay for the non-invasive detection of bladder cancer. PLoS One (2012) ;7: (10). DOI: 10.1371/journal.pone.0047469 |
[60] | Ecke TH , Schlechte HH , Schulze G , Lenk SV , Loening SA . Four tumour markers for urinary bladder cancer–tissue polypeptide antigen (TPA), HER-2/neu (ERB B2), urokinase-type plasminogen activator receptor (uPAR) and TP53 mutation. Anticancer Res (2005) ;25: (1B):635–41. |
[61] | Sánchez-Carbayo M , Herrero E , Megías J , Mira A , Soria F . Comparative sensitivity of urinary CYFRA 21-1, urinary bladder cancer antigen, tissue polypeptide antigen, tissue polypeptide antigen and NMP22 to detect bladder cancer. J Urol (1999) ;162: (6):1951–6. |
[62] | Stefanović V , Mitić-Zlatković M , Ignjatović I , Vlajković M , Sćepović Z . Tissue polypeptide antigen and carcinoembryonic antigen lack diagnostic accuracy in urothelial carcinoma. Int Urol Nephrol (1999) ;31: (4):443–9. |
[63] | Carbin BE , Ekman P , Eneroth P , Nilsson B . Urine-TPA (tissue polypeptide antigen), flow cytometry and cytology as markers for tumor invasiveness in urinary bladder carcinoma. Urol Res (1989) ;17: (5):269–72. |
[64] | Li X , Wang Y , Xu J , Zhang Q . Sandwich ELISA for detecting urinary Survivin in bladder cancer. Chin J Cancer Res (2013) ;25: (4):375–81. |
[65] | Ohsawa I , Nishimura T , Kondo Y , Kimura G , Satoh M , Matsuzawa I , et al. Detection of urine survivin in 40 patients with bladder cancer. J Nippon Med Sch (2004) ;71: (6):379–83. |
[66] | Shariat S , Casella R , Khoddami S , Hernandez G , Sulser T , Gasser T , et al. Urine detection of survivin is a sensitive marker for the noninvasive diagnosis of bladder cancer. J Urol (2004) ;171: (2):626–30. |
[67] | Srivastava AK , Singh PK , Srivastava K , Singh D , Dalela D , Rath SK , et al. Diagnostic role of survivin in urinary bladder cancer. Asian Pac J Cancer Prev (2013) ;14: (1):81–5. |
[68] | Hausladen DA , Wheeler MA , Altieri DC , Colberg JW , Weiss RM . Effect of intravesical treatment of transitional cell carcinoma with bacillus Calmette-Guerin and mitomycin C on urinary survivin levels and outcome. J Urol (2003) ;170: (1):230–4. |
[69] | Roy R , Louis G , Loughlin KR , Wiederschain D , Kilroy SM , Lamb CC , et al. Tumor-specific urinary matrix metalloproteinase fingerprinting: Identification of high molecular weight urinary matrix metalloproteinase species. Clin Cancer Res (2008) ;14: (20):6610–7. |
[70] | Sheryka E , Wheeler MA , Hausladen DA , Weiss RM . Urinary interleukin-8 levels are elevated in subjects with transitional cell carcinoma. Urology (2003) ;62: (1):162–6. |
[71] | Urquidi V , Chang M , Dai Y , Kim J , Wolfson ED , Goodison S , et al. IL-8 as a urinary biomarker for the detection of bladder cancer. BMC Urol (2012) ;12: (12). DOI: 10.1186/1471-2490-12-12 |
[72] | Liu X , Dowell A , Patel P , Viney R , Foster M , Porfiri E , et al. Cytokines as effectors and predictors of responses in the treatment of bladder cancer by bacillus Calmette-Guérin. Future Oncol (2014) ;10: (8):1443–56. |
[73] | Chuang C , Liao S . Evaluation of CA19-9 as a tumor marker in urothelial malignancy. Scand J Urol Nephrol (2004) ;38: (5):359–65. |
[74] | Tizzani A , Cassetta G , Cicigoi A , Piana P , Cerchier A , Pecchio F , et al. Tumor markers (CEA, TPA and CA 19-9) in urine of bladder cancer patients. Int J Biol Markers (1987) ;2: (2):121–4. |
[75] | Yue T , Partyka K , Maupin K , Hurley M , Andrews P , Kaul K , et al. Identification of blood-protein carriers of the CA 19-9 antigen and characterization of prevalence in pancreatic diseases. Proteomics (2011) ;11: (19):3665–74. |
[76] | Nagao K , Itoh Y , Fujita K , Fujime M . Evaluation of urinary CA19-9 levels in bladder cancer patients classified according to the combinations of Lewis and Secretor blood group genotypes. Int J Urol (2007) ;14: (9):795–59. |
[77] | Vestergaard E , Wolf H , Orntoft T . Increased concentrations of genotype-interpreted Ca 19-9 in urine of bladder cancer patients mark diffuse atypia of the urothelium. Clin Chem (1998) ;44: (2):197–204. |
[78] | Getzenberg R , Konety B , Oeler T , Quigley M , Hakam A , Becich M , et al. Bladder cancer-associated nuclear matrix proteins. Cancer Res (1996) ;56: (7):1690–4. |
[79] | Konety B , Nguyen T , Dhir R , Day R , Becich M , Stadler W , et al. Detection of bladder cancer using a novel nuclear matrix protein, BLCA-4. Clin Cancer Res (2000) ;6: (7):2618–25. |
[80] | Van Le T , Myers J , Konety B , Barder T , Getzenberg R . Functional characterization of the bladder cancer marker, BLCA-4. Clin Cancer Res (2004) ;10: (4):1384–91. |
[81] | Van Le T , Miller R , Barder T , Babjuk M , Potter D , Getzenberg R . Highly specific urine-based marker of bladder cancer. Urology (2005) ;66: (6):1256–60. |
[82] | Van Le T , Myers J , Getzenberg R , Konety B , Barder T . Retraction: Functional characterization of the bladder cancer marker, BLCA-4. Clin Cancer Res (2013) ;19: (12):3327. |
[83] | Van Le T , Miller R , Barder T , Babjuk M , Potter D , Getzenberg R . Retraction notice to Highly specific urine-based marker of bladder cancer Urology 66 -Urology (2014) ;83: (6):1448. |
[84] | Feng CC , Wang PH , Guan M , Jiang HW , Wen H , Ding Q , et al. Urinary BLCA-4 is highly specific for detection of bladder cancer in Chinese Han population and is related to tumour invasiveness. Folia Biol (Praha) (2011) ;57: (6):242–7. |
[85] | Li C , Li H , Zhang T , Li J , Liu L , Chang J . Discovery of Apo-A1 as a potential bladder cancer biomarker by urine proteomics and analysis. Biochem Biophys Res Commun (2014) ;446: (4):1047–52. |
[86] | Li CY , Li HJ , Zhang T , Gao HS , C J.W. , Men XL , et al. [Significance of apolipoprotein A1 as biomarker for early diagnosis and classification of bladder urothelial carcinoma]. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi (2013) ;31: (4):266–70. |
[87] | Li H , Li C , Wu H , Zhang T , Wang J , Wang S , et al. Identification of Apo-A1 as a biomarker for early diagnosis of bladder transitional cell carcinoma. Proteome Sci (2011) ;9: (1). DOI: 10.1186/1477-5956-9-21 |
[88] | Chen YT , Chen CL , Chen HW , Chung T , Wu CC , Chen CD , et al. Discovery of novel bladder cancer biomarkers by comparative urine proteomics using iTRAQ technology. J Proteome Res (2010) ;9: (11):5803–15. |
[89] | de Martino M , Lucca I , Mbeutcha A , Wiener H , Haitel A , Susani M , et al. Carbonic Anhydrase IX as a Diagnostic Urinary Marker for Urothelial Bladder Cancer. Eur Urol (2015) ;68: (4):552–4. |
[90] | Gecks T , Junker K , Franz M , Richter P , Walther M , Voigt A , et al. B domain containing Tenascin-C: A new urine marker for surveillance of patients with urothelial carcinoma of the urinary bladder? Clin Chim Acta (2011) ;412: (21-22):1931–6. |
[91] | Lorenzi T , Lorenzi M , Altobelli E , Marzioni D , Mensà E , Quaranta A , et al. HtrA1 in human urothelial bladder cancer: A secreted protein and a potential novel biomarker. International Journal Of Cancer (2013) ;133: (11):2650–61. |
[92] | Shariat SF , Zippe C , Lüdecke G , Boman H , Sanchez-Carbayo M , Casella R , et al. Nomograms including nuclear matrix protein 22 for prediction of disease recurrence and progression in patients with Ta, T1 or CIS transitional cell carcinoma of the bladder. J Urol (2005) ;173: (5):1518–25. |
[93] | Huang D , Sherman B , Lempicki R . Systematic and integrative analysis of large gene lists using DAVID Bioinformatics Resources. Nature Protoc (2009) ;4: (1):44–57. |
[94] | Knowles M , Hurst C . Molecular biology of bladder cancer: New insights into pathogenesis and clinical diversity. Nat Rev Cancer (2015) ;15: (1):25–41. |
[95] | Aaboe M , Marcussen N , Jensen K , Thykjaer T , Dyrskjøt L , Orntoft T . Gene expression profiling of noninvasive primary urothelial tumours using microarrays. Br J Cancer (2005) ;93: (10):1182–90. |
[96] | Schmitz-Dräger B , Droller M , Lokeshwar V , Lotan Y , Hudson M , van Rhijn B , et al. Molecular markers for bladder cancer screening, early diagnosis, and surveillance: The WHO/ICUD consensus. Urol Int (2015) ;94: (1):1–24. |
[97] | Cochrane Handbook for Systematic Reviews of Interventions 1st ed. http://handbook.cochrane.org. Higgins J, Green S, editors. Chichester: Wiley-Blackwell; (2008) . |
[98] | Jeong S , Park Y , Cho Y , Kim YR , Kim HS . Diagnostic values of urine CYFRA21-1, NMP22, UBC, and FDP for the detection of bladder cancer. Clin Chim Acta (2012) ;414: :93–100. |
[99] | Kandimalla R , Masius R , Beukers W , Bangma C , Orntoft T , Dyrskjot L , et al. A 3-plex methylation assay combined with the FGFR3 mutation assay sensitively detects recurrent bladder cancer in voided urine. Clin Cancer Res (2013) ;19: (17):4760–9. |
[100] | Reinert T , Borre M , Christiansen A , Hermann G , Ørntoft T , Dyrskjøt L . Diagnosis of bladder cancer recurrence based on urinary levels of EOMES, HOXA9, POU4F2, TWIST1, VIM, and ZNF154 hypermethylation. PLoS One (2012) ; 7: (10). DOI: 10.1371/journal.pone.0046297 |
[101] | Tognieri F , Ward D , Foster J , Devall A , Wojtowicz P , Alyas S , et al. Genomic complexity of urothelial bladder cancer revealed in urinary cfDNA. European Journal of Human Genetics (2016) ;1–8. |
[102] | Mengual L , Burset M , Ribal M , Ars E , Marín-Aguilera M , Fernández M , et al. Gene expression signature in urine for diagnosing and assessing aggressiveness of bladder urothelial carcinoma. Clin Cancer Res (2010) ;16: (9):26242633. |
[103] | Ward D , Baxter L , Gordon N , Ott S , Savage R , Beggs A , et al. Multiplex PCR and next generation sequencing for the non-invasive detection of bladder cancer PLOS ONE (2016) ;11: (2). DOI: 10.1371/journal.pone.0149756 |
[104] | Miyake M , Ross S , Lawton A , Chang M , Dai Y , Mengual L , et al. Investigation of CCL18 and A1AT as potential urinary biomarkers for bladder cancer detection. BMC Urology (2013) ;13: (42). DOI: 10.1186/1471-2490-13-42 |
[105] | Korman HJ , Peabody JO , Cerny JC , Farah RN , Yao J , Raz A . Autocrine motility factor receptor as a possible urine marker for transitional cell carcinoma of the bladder. J Urol (1996) ;155: (1):347–9. |
[106] | Babjuk M , Kostírová M , Mudra K , Pecher S , Smolová H , Pecen L , et al. Qualitative and quantitative detection of urinary human complement factor H-related protein (BTA stat and BTA TRAK) and fragments of cytokeratins 8, 18 (UBC rapid and UBC IRMA) as markers for transitional cell carcinoma of the bladder. Eur Urol (2002) ;41: (1):34–9. |
[107] | Ramakumar S , Bhuiyan J , Besse J , Roberts S , Wollan P , Blute M , et al. Comparison of screening methods in the detection of bladder cancer. J Urol (1999) ;161: (2):388–94. |
[108] | Gutiérrez Baños JL , Rebollo Rodrigo MH , Antolín Juárez FM , Martín García B . NMP 22, BTA stat test and cytology in the diagnosis of bladder cancer: A comparative study. Urol Int (2001) ;66: (4):185–90. |
[109] | Poulakis V , Witzsch U , De Vries R , Altmannsberger HM , Manyak MJ , Becht E . A comparison of urinary nuclear matrix protein-22 and bladder tumour antigen tests with voided urinary cytology in detecting and following bladder cancer: The prognostic value of false-positive results. BJU Int (2001) ;88: (7):692–701. |
[110] | Saad A , Hanbury DC , McNicholas TA , Boustead GB , Morgan S , Woodman AC . A study comparing various noninvasive methods of detecting bladder cancer in urine. BJU Int (2002) ;89: (4):369–73. |
[111] | Wiener H , Mian C , Haitel A , Pycha A , Schatzl G , Marberger M . Can urine bound diagnostic tests replace cystoscopy in the management of bladder cancer? J Urol (1998) ;159: (6):1876–80. |
[112] | Boman H , Hedelin H , Jacobsson S , Holmäng S . Newly diagnosed bladder cancer: The relationship of initial symptoms, degree of microhematuria and tumor marker status. J Urol (2002) ;168: (5):1955–9. |
[113] | Mian C , Lodde M , Haitel A , Egarter Vigl E , Marberger M , Pycha A . Comparison of two qualitative assays, the UBC rapid test and the BTA stat test, in the diagnosis of urothelial cell carcinoma of the bladder. Urology (2000) ;56: (2):228–31. |
[114] | Halling KC , King W , Sokolova IA , Karnes RJ , Meyer RG , Powell EL , et al. A comparison of BTA stat, hemoglobin dipstick, telomerase and Vysis UroVysion assays for the detection of urothelial carcinoma in urine. J Urol (2002) ;167: (5):2001–6. |
[115] | Pode D , Shapiro A , Wald M , Nativ O , Laufer M , Kaver I . Noninvasive detection of bladder cancer with the BTA stat test. J Urol (1999) ;161: (2):443–6. |
[116] | Gibanel R , Ribal M , Filella X , Ballesta A , Molina R , Alcaraz A , et al. BTA TRAK urine test increases the efficacy of cytology in the diagnosis of low-grade transitional cell carcinoma of the bladder. Anticancer Res (2002) ;22: (2B):1157–60. |
[117] | Hatzichristodoulou G , Kübler H , Schwaibold H , Wagenpfeil S , Eibauer C , Hofer C , et al. Nuclear matrix protein 22 for bladder cancer detection: Comparative analysis of the BladderChek® and ELISA. Anticancer Research (2012) ;32: (11):5093–7. |
[118] | Mahnert B , Tauber S , Kriegmair M , Nagel D , Holdenrieder S , Hofmann K , et al. Measurements of complement factor H-related protein (BTA-TRAK assay) and nuclear matrix protein (NMP22 assay)–useful diagnostic tools in the diagnosis of urinary bladder cancer? Clin Chem Lab Med (2003) ;41: (1):104–10. |
[119] | Miyake M , Goodison S , Rizwani W , Ross S , Bart Grossman H , Rosser CJ . Urinary BTA: Indicator of bladder cancer or of hematuria. World J Urol (2012) ;30: (6):869–73. |
[120] | Giannopoulos A , Manousakas T , Mitropoulos D , Botsoli-Stergiou E , Constantinides C , Giannopoulou M , et al. Comparative evaluation of the BTAstat test, NMP22, and voided urine cytology in the detection of primary and recurrent bladder tumors. Urology (2000) ;55: (6):871–5. |
[121] | Lokeshwar VB , Schroeder GL , Selzer MG , Hautmann SH , Posey JT , Duncan RC , et al. Bladder tumor markers for monitoring recurrence and screening comparison of hyaluronic acid-hyaluronidase and BTA-Stat tests. Cancer (2002) ;95: (1):61–72. |
[122] | Raitanen MP , Grou F . The role of BTA stat Test in follow-up of patients with bladder cancer: Results from FinnBladder studies. World J Urol (2008) ;26: (1):45–50. |
[123] | Del Nero A , Esposito N , Currò A , Biasoni D , Montanari E , Mangiarotti B , et al. Evaluation of urinary level of NMP22 as a diagnostic marker for stage pTa-pT1 bladder cancer: Comparison with urinary cytology and BTA test. Eur Urol (1999) ;35: (2):93–7. |
[124] | Thomas L , Leyh H , Marberger M , Bombardieri E , Bassi P , Pagano F , et al. Multicenter trial of the quantitative BTA TRAK assay in the detection of bladder cancer. Clin Chem (1999) ;45: (4):472–7. |
[125] | Leyh H , Marberger M , Conort P , Sternberg C , Pansadoro V , Pagano F , et al. Comparison of the BTA stat test with voided urine cytology and bladder wash cytology in the diagnosis and monitoring of bladder cancer. Eur Urol (1999) ;35: (1):52–6. |
[126] | Svatek RS , Karam J , Karakiewicz PI , Gallina A , Casella R , Roehrborn CG , et al. Role of urinary cathepsin B and L in the detection of bladder urothelial cell carcinoma. J Urol (2008) ;179: (2):478–84. |
[127] | Nisman B , Barak V , Shapiro A , Golijanin D , Peretz , Pode D . Evaluation of urine CYFRA 21-1 for the detection of primary and recurrent bladder carcinoma. Cancer (2002) ;94: (11):2914–22. |
[128] | Sánchez-Carbayo M , Espasa A , Chinchilla V , Herrero E , Megías J , Mira A , et al. New electrochemiluminescent immunoassay for the determination of CYFRA 21- Analytical evaluation and clinical diagnostic performance in urine samples of patients with bladder cancer. Clin Chem (1999) ;45: (11):1944–53. |
[129] | Fernandez-Gomez J , Rodríguez-Martínez JJ , B S.E. , García Rodríguez J , Allende DM , Jalon A , et al. Urinary CYFRA 21.1 is not a useful marker for the detection of recurrences in the follow-up of superficial bladder cancer. Eur Urol (2007) ;51: (5):1267–74. |
[130] | Morgan R , Bryan RT , Javed S , Launchbury F , Zeegers MP , Cheng KK , et al. Expression of Engrailed-2 (EN2) protein in bladder cancer and its potential utility as a urinary diagnostic biomarker. European Journal of Cancer (2013) ;49: (9):2214–22. |
[131] | Mian C , Lodde M , Haitel A , Vigl E , Marberger M , Pycha A . Comparison of the monoclonal UBC-ELISA test and the NMP22 ELISA test for the detection of urothelial cell carcinoma of the bladder. Urology (2000) ;55: (2):223–6. |
[132] | Grossman HB , Messing E , Soloway M , Tomera K , Katz G , Berger Y , et al. Detection of bladder cancer using a point-of-care proteomic assay. JAMA (2005) ;293: (7):810–6. |
[133] | Jamshidian H , Kor K , Djalali M . Urine concentration of nuclear matrix protein 22 for diagnosis of transitional cell carcinoma of bladder. Urol J (2008) ;5: (4):243–7. |
[134] | Lee K . Evaluation of the NMP22 test and comparison with voided urine cytology in the detection of bladder cancer. Yonsei Med J (2001) ;42: (1):14–8. |
[135] | Shariat SF , Marberger MJ , Lotan Y , Sanchez-Carbayo M , Zippe C , Lüdecke G , et al. Variability in the performance of nuclear matrix protein 22 for the detection of bladder cancer. J Urol (2006) ;176: (3):919–26. |
[136] | Hosseini J , Golshan AR , Mazloomfard MM , Mehrsai AR , Zargar MA , Ayati M , et al. Detection of recurrent bladder cancer: NMP22 test or urine cytology? Urol J (2012) ;9: (1):367–72. |
[137] | Grossman HB , Soloway M , Messing E , Katz G , Stein B , Kassabian V , et al. Surveillance for recurrent bladder cancer using a point-of-care proteomic assay. JAMA (2006) ;295: (3):299–305. |
[138] | Horstmann M , Patschan O , Hennenlotter J , Senger E , Feil G , Stenzl A . Combinations of urine-based tumour markers in bladder cancer surveillance. Scand J Urol Nephrol (2009) ;43: (6):461–6. |
[139] | Todenhöfer T , Hennenlotter J , Aufderklamm S , Kühs U , Gakis G , Germann M , et al. Individual risk assessment in bladder cancer patients based on a multi-marker panel. J Cancer Res Clin Oncol (2013) ;139: (1):49–56. |
[140] | Kumar A , Kumar R , Gupta NP . Comparison of NMP22 BladderChek test and urine cytology for the detection of recurrent bladder cancer. Jpn J Clin Oncol (2006) ;36: (3):172–5. Epub Epub 2006 Mar 6. |
[141] | Casella R , Huber P , Blöchlinger A , Stoffel F , Dalquen P , Gasser T , et al. Urinary level of nuclear matrix protein 22 in the diagnosis of bladder cancer: Experience with 130 patients with biopsy confirmed tumor. J Urol (2000) ;164: (6):1926–68. |
[142] | Feng J , He W , Song Y , Wang Y , Simpson RJ , Zhang X , et al. Platelet-derived growth factor receptor beta: A novel urinary biomarker for recurrence of non-muscle-invasive bladder cancer. PLoS One (2014) ;9: (5):e96671. |
[143] | Chen Y , Chen H , Domanski D , Smith D , Liang K , Wu C , et al. Multiplexed quantification of 63 proteins in human urine by multiple reaction monitoring-based mass spectrometry for discovery of potential bladder cancer biomarkers. J Proteomics (2012) ;75: (12):3529–45. |
[144] | Hakenberg OW , Fuessel S , Richter K , Froehner M , Oehlschlaeger S , Rathert P , et al. Qualitative and quantitative assessment of urinary cytokeratin 8 and 18 fragments compared with voided urine cytology in diagnosis of bladder carcinoma. Urology (2004) ;64: (6):1121–6. |
[145] | May M , Hakenberg O , Gunia S , Pohling P , Helke C , Lübbe L , et al. Comparative diagnostic value of urine cytology, UBC-ELISA, and fluorescence in situ hybridization for detection of transitional cell carcinoma of urinary bladder in routine clinical practice. Urology (2007) ;70: (3):449–53. |
[146] | Ecke TH , Arndt C , Stephan C , Hallmann S , Lux O , Otto T , et al. Preliminary Results of a Multicentre Study of the UBC Rapid Test for Detection of Urinary Bladder Cancer. Anticancer Res (2015) ;35: (5):2651–5. |
[147] | Mungan N , Vriesema J , Thomas C , Kiemeney L , Witjes J . Urinary bladder cancer test: A new urinary tumor marker in the follow-up of superficial bladder cancer. Urology (2000) ;56: (5):787–92. |
[148] | Sánchez-Carbayo M , Herrero E , Megías J , Mira A , Soria F . Initial evaluation of the new urinary bladder cancer rapid test in the detection of transitional cell carcinoma of the bladder. Urology (1999) ;54: (4):656–61. |
[149] | Raitanen MP , Kaasinen E , Rintala E , Hansson E , Nieminen P , Aine R , et al. Prognostic utility of human complement factor H related protein test (the BTA stat Test). Br J Cancer (2001) ;85: (4):552–6. |
[150] | Nilsson B , Wahren B , Esposti PL , Edsmyr F . Prediction of survival and recurrence in bladder carcinoma. Urol Res (1982) ;10: (3):109–13. |
[151] | Iles RK , Persad R , Trivedi M , Sharma KB , Dickinson A , Smith P , et al. Urinary concentration of human chorionic gonadotrophin and its fragments as a prognostic marker in bladder cancer. Br J Urol (1996) ;77: (1):61–9. |
[152] | Bryan R , Regan H , Pirrie S , Devall A , Cheng K , Zeegers M , et al. Protein shedding in urothelial bladder cancer: Rognostic implications of soluble urinary EGFR and EpCAM. Br J Cancer (2015) ;112: :1052–8. |
[153] | Offersen BV , Knap MM , Horsman MR , Verheijen J , Hanemaaijer R , Overgaard J . Matrix metalloproteinase-9 measured in urine from bladder cancer patients is an independent prognostic marker of poor survival. Acta Oncol (2010) ;49: (8):1283–7. Epub Epub 2010 Sep 15. |
[154] | Kibar Y , Goktas S , Kilic S , Yaman H , Onguru O , Peker AF . Prognostic value of cytology, nuclear matrix protein 22 (NMP22) test, and urinary bladder cancer II (UBC II) test in early recurrent transitional cell carcinoma of the bladder. Ann Clin Lab Sci (2006) ;36: (1):31–8. |
[155] | Becker M , Szarvas T , Wittschier M , vom Dorp F , Tötsch M , Schmid KW , et al. Prognostic impact of plasminogen activator inhibitor type 1 expression in bladder cancer. Cancer (2010) ;116: (19):4502–12. |
[156] | Guan Z , Zeng J , Wang Z , Xie H , Lv C , Ma Z , et al. Urine tenascin-C is an independent risk factor for bladder cancer patients. Mol Med Rep (2014) ;9: (3):961–6. |
[157] | Costello CB , Kumar S . Prognostic value of tissue polypeptide antigen in urological neoplasia. J R Soc Med (1985) ;78: (3):207–10. |
[158] | Yang H , Li H , Wang Z , Gao J , Guo Y . Is urinary soluble Fas an independent predictor of non-muscle-invasive bladder cancer? A prospective chart study. Urol Int (2013) ;91: (4):456–61. 2013 Aug 14. |
[159] | Kelloniemi E , Rintala E , Finne P , Stenman UH , Group. F . Tumor-associated trypsin inhibitor as a prognostic factor during follow-up of bladder cancer. Urology (2003) ;62: (2):249–53. |
[160] | Feldman A , Banyard J , Wu C , McDougal W , Zetter B . Cystatin B as a tissue and urinary biomarker of bladder cancer recurrence and disease progression. Clin Cancer Res (2009) ;15: (3):1024–31. |
Figures and Tables
Fig.1
Search strategy outline and results.
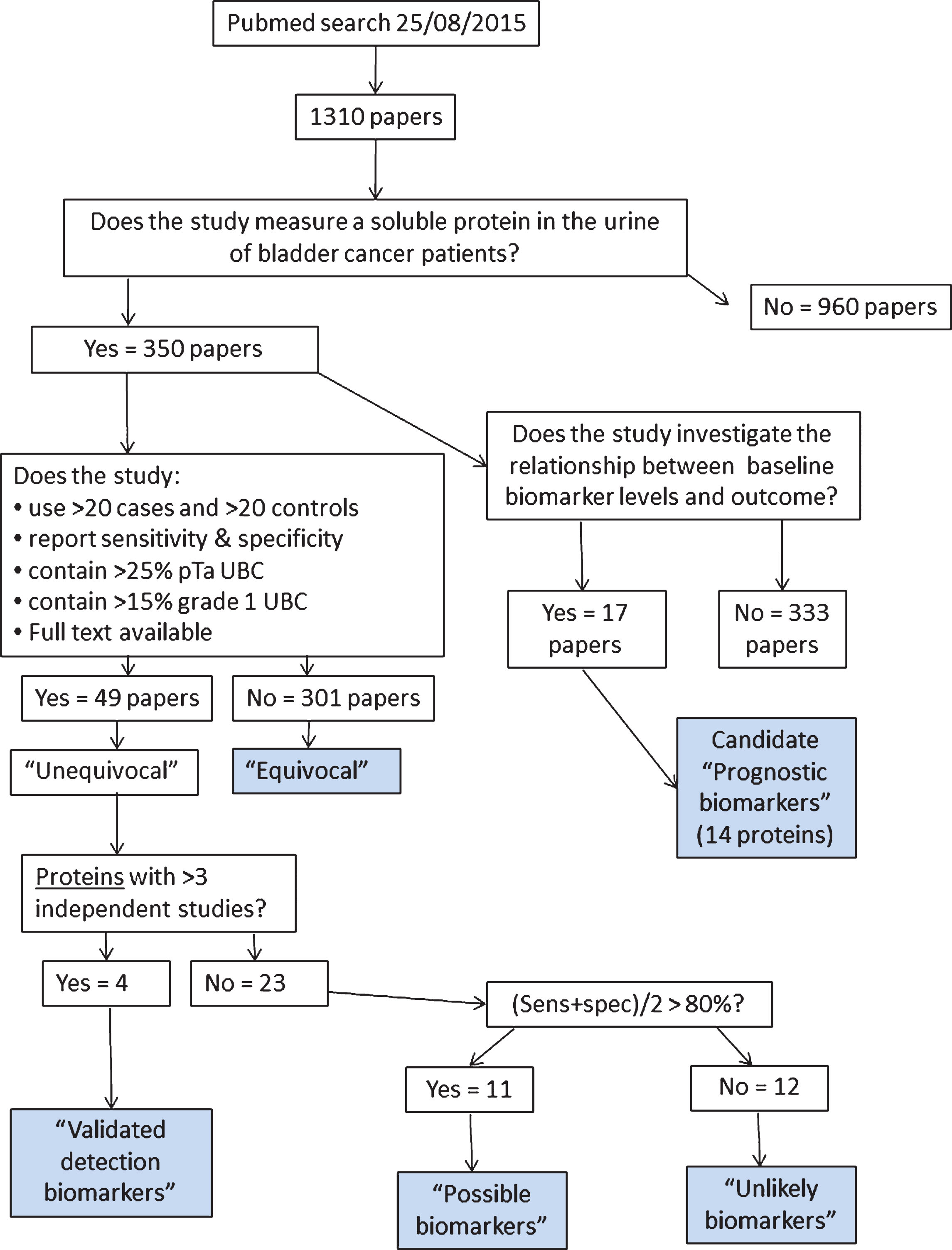
Fig.2
Numbers of publications for the most commonly investigated urinary protein biomarkers. Papers providing measurement data included, reviews excluded.
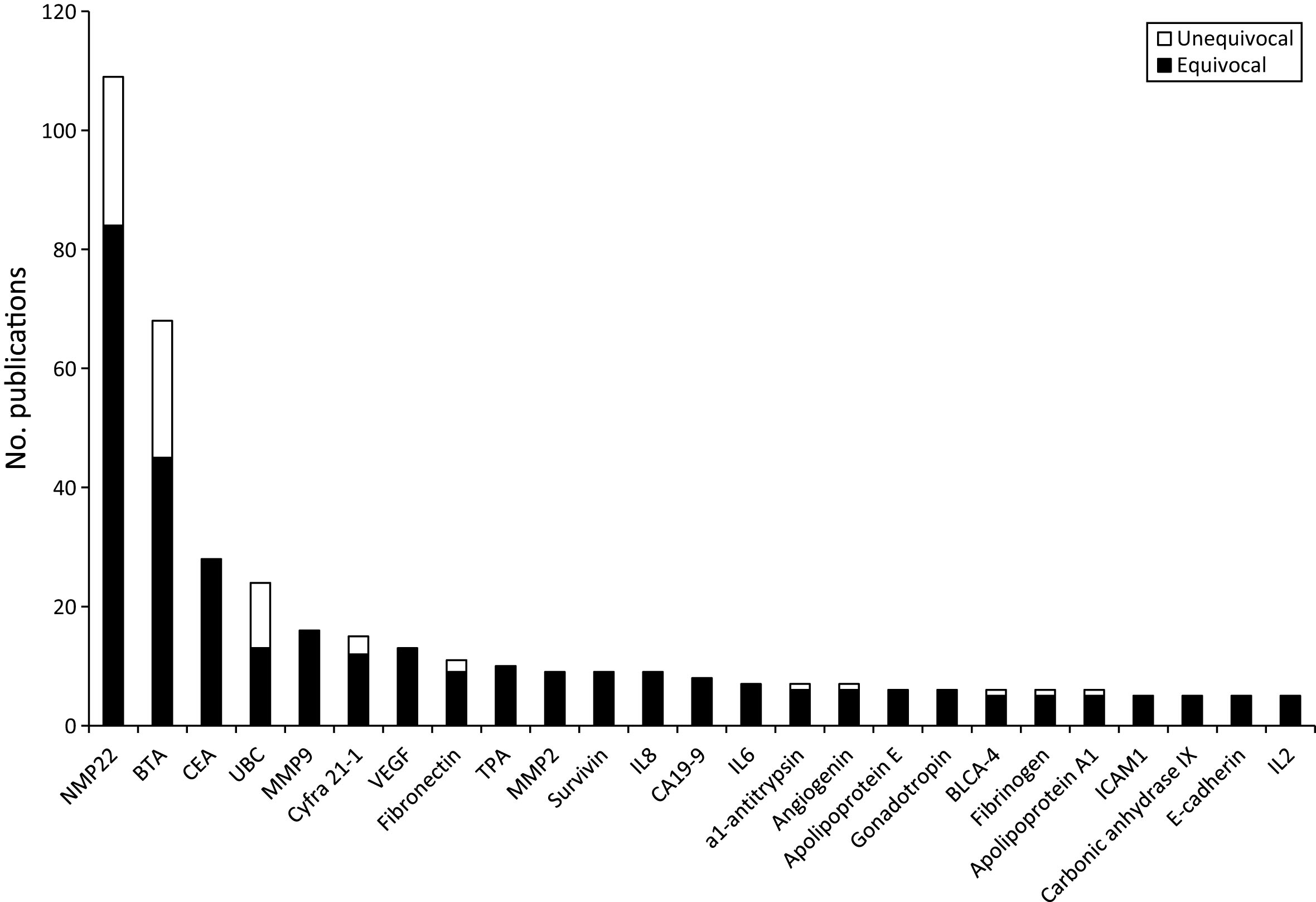
Fig.3
The history and lifecycle of bladder cancer biomarkers. The number of publications for each biomarker with >10 publications in total is shown for each half-decade from 1971. CEA peaks in the 1970s and TPA in the 1980s. BTA and cyfra 21-1 peak in 1996–2000 whilst fibronectin, NMP22 and UBC peak in 2001–2005. The rate of publication of all of these biomarkers are now declining whereas MMP9 and VEGF continue to rise.
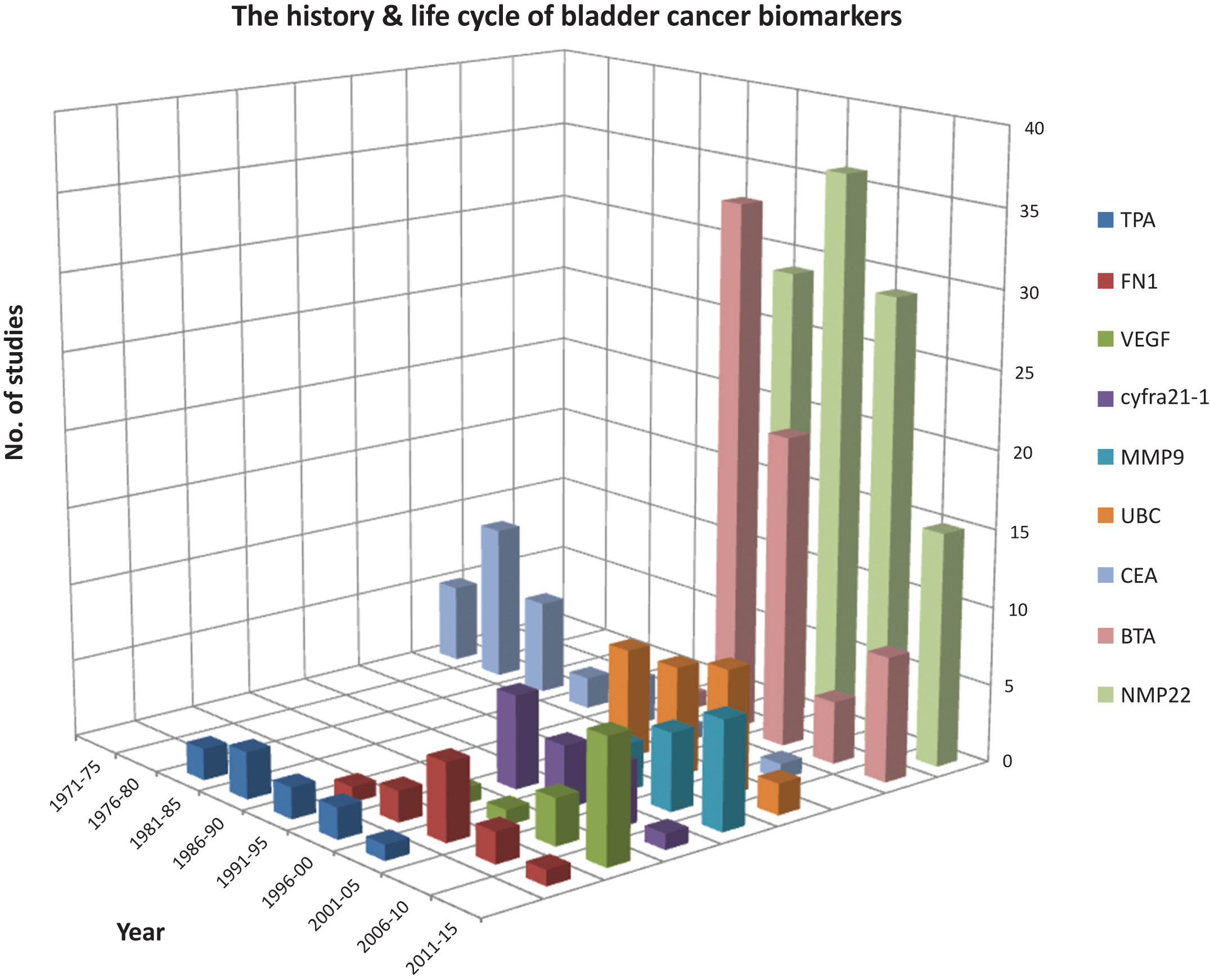
Fig.4
Cellular compartmentalisation of protein biomarkers reported≥1 unequivocal or≥5 unequivocal biomarker studies.
Table 1
Summary of unequivocal biomarker studies. For proteins with multiple studies, sensitivity and specificity are presented as means weighted according to sample size in each study. #indicates potentially several genes. NMP22, BTA and UBC data include studies using quantitative and point-of-care versions of the assay
| Protein name | Gene symbol | Sensitivity (%) | Specificity (%) | Cancers (n) | Controls (n) | Refs |
| Alpha-1-anti-trypsin | SERPINA1 | 70.6 | 71.8 | 206 | 102 | [104] |
| Angiogenin | ANG | 66 | 75 | 50 | 20 | [23] |
| Apolipoprotein A4 | APOA4 | 79.2 | 100 | 110 | 66 | [34] |
| Autocrine motility factor receptor | AMFR | 84 | 75 | 45 | 62 | [105] |
| BIGH3 | TGFBI | 70 | 80 | 30 | 30 | [31] |
| Bladder tumour antigen (BTA) | BTA# | 64 | 76.6 | 2258 | 2994 | [11, 12, 55, 106–125] |
| Calprotectin | S100A8 &S100A9 | 80.4 | 92.5 | 46 | 135 | [28] |
| Cathepsin B | CTSB | 55.7 | 56.1 | 122 | 107 | [126] |
| Cathepsin L | CTSL | 71.3 | 74.8 | 122 | 107 | [126] |
| CCL18 | CCL18 | 70.4 | 67.7 | 206 | 102 | [104] |
| CD147 (EMMPRIN) | BSG | 96.7 | 100 | 30 | 30 | [31] |
| CEACAM1 | CEACAM1 | 74 | 95 | 93 | 82 | [27] |
| Clusterin, | CLU | 76.3 | 86.5 | 168 | 151 | [22, 23] |
| Coronin-1A | CORO1A | 66.7 | 100 | 110 | 66 | [34] |
| CYFRA21-1 | KRT19 | 64.4 | 85.5 | 293 | 331 | [127–129] |
| DJ-1 | PARK7 | 83.3 | 100 | 110 | 66 | [34] |
| EN2 | EN2 | 82 | 75 | 466 | 52 | [130] |
| FDP | FGA &FGB | 52 | 91 | 57 | 139 | [107] |
| Fibronectin | FN1 | 89 | 85.6 | 126 | 41 | [14, 15] |
| NMP22 | NUMA1 | 61.8 | 80.3 | 4528 | 7728 | [11, 12, 107–112, 116–118, 120, 123, 126, 131–141] |
| PDGFRβ | PDGFRB | 70.6 | 81.2 | 117 | 68 | [142] |
| Prothrombin | F2 | 71.1 | 75.0 | 76 | 80 | [143] |
| Reg-1 | REG1A | 81.3 | 81.2 | 32 | 48 | [41] |
| Semenogelin-2 | SEMG2 | 66.7 | 80 | 110 | 66 | [34] |
| Stathmin-1 | STMN1 | 90.0 | 86.7 | 30 | 30 | [31] |
| Urinary bladder carcinoma antigen (UBC) | KRT8 &KRT18 | 64.4 | 80.3 | 753 | 1072 | [11, 12, 106, 112, 113, 131, 144–148] |
| γ-synuclein, | SNCG | 87.5 | 90.0 | 110 | 66 | [34] |
Table 2
Prognostic urinary biomarkers for bladder cancer
| Marker | No. of studies | No. of patients | Comments | Refs |
| BTA | 1 | 97 | Independent prognostic indicator | [149] |
| Carcinoembryonic antigen (CEA) | 3 | 425 | Independent prognostic indicator | [44, 47, 150] |
| β-HCG | 1 | 52 | Prognostic in MIBC | [151] |
| EGFR | 1 | 436 | Independent prognostic indicator | [152] |
| EpCAM | 1 | 607 | Independent prognostic indicator | [152] |
| MMP9 | 1 | 188 | Independent prognostic indicator | [153] |
| NMP22 | 2 | 333 | Detection-prognosis | [109, 154] |
| Plasminogen Activator Inhibitor type I (PAI-1) | 1 | 244 | Not prognostic | [155] |
| PDGFRβ | 1 | 185 | Predicts recurrence in NMIBC | [142] |
| Tenascin-C | 1 | 66 | Independent prognostic indicator | [156] |
| Tissue polypeptide antigen (TPA) | 1 | 97 | Prognostic | [157] |
| Urinary sFas | 1 | 128 | Predicts recurrence in NMIBC | [158] |
| Urine tumour-associated trypsin inhibitor (TATI) | 1 | 157 | Not prognostic | [159] |
| Cystatin-B | 1 | 47 | Independent prognostic indicator | [160] |




